Abstract
Introduction
Voice and swallowing issues affect approximately 1% of children in the United States 1 and are increasing in both incidence and prevalence yearly, 2 possibly due to the improved survival of medically complex children and premature infants in the past 2 decades. 2 Identifying and addressing dysphagia at an early stage is crucial to setting pediatric patients up for success; children with either short-term or chronic swallowing problems can experience interrupted development and long-term health consequences. 3 The ability to take an age-appropriate diet can also influence the development of social skills and healthy eating patterns. 4 Further, dysphagia can lead to nutritional deficits, failure to thrive, aspiration, and patient/family distress, among other harmful sequelae. It is, therefore, essential to identify children with dysphagia and intervene appropriately.
The videofluoroscopic swallow study (VFSS), also known as the modified barium swallow study, is 1 of the 2 primary instruments used to assess pediatric dysphagia. 3 VFSS is superior to the bedside evaluation for assessing aspiration events, particularly for solid foods, and can definitively demonstrate the presence or absence of silent aspiration. 5 Despite its advantages, VFSS does have its limitations: it necessitates radiation exposure, is time-intensive for speech-language pathologists and fluoroscopy technicians, and requires patient cooperation. This balance of considerations calls for the early identification of pediatric patients, who will not benefit from a VFSS. Anxiety, combativeness, food refusal, or other complicating factors can lead to inconclusive studies that are uninterpretable by the provider and unactionable to the patient. To minimize radiation exposure, excess costs, patient inconvenience, and burden to the healthcare system; it is critical to avoid ordering VFSS in patients who are highly unlikely to participate and provide clinically useful results.
While standardized procedures for VFSS have the potential to decrease study time and radiation dose, 6 there are no corresponding broadly implemented guidelines for pretest patient selection, which could spare pediatric patients from unhelpful studies. Clear guidelines for ordering a VFSS have been proposed. 7 The literature unanimously recommends that pediatric patients be assessed by a speech-language pathologist prior to referral for VFSS. 8 While in-person assessment by a speech-language pathologist before a VFSS could lower study failure rates, it requires cooperation, time, and extra financial burden for families, especially because it may require a separate appointment, leading to increased travel and missed work. Less burdensome measures, including simple screening questions pre-study optimization of diet, might aid study efficiency. 8 Information about whether a pediatric patient can tolerate an oral intake of at least 0.5 oz at home might be helpful to identify patients who are unlikely to produce a diagnostically useful VFSS. Hence, the purpose of this study is to determine whether this oral intake of at least 0.5 oz is associated with VFSS success in pediatric patients.
Methods
This study received Institutional Review Board exemption from Duke University. A REDcap (version 12.0.25) database9,10 of pediatric patients of ages 0 to 18 years presenting to Duke University Health System’s Children’s Health Center for VFSS between 2012 and 2021 was built; REDcap is an application created at Vanderbilt University to capture clinical data in a centralized format. These patients were identified through a query of the electronic health record using CPT codes 92611 and 74230 (codes for the radiology and speech language pathology portions of the VFSS, respectively). Patients who did not meet these criteria or whose VFSS encounters were canceled or “no-showed” were excluded. In addition, VFSS records from before 2014 were ultimately excluded, because these notes were not in a standard format and did not consistently capture the same information as later notes. Patient charts were examined by 3 reviewers and assessed for a variety of quantitative and qualitative metrics, as listed below, with discrepancies discussed until a consensus could be reached. These were linked to patient identification numbers and securely stored in a protected virtual network space.
Outcome
The main aim was whether a study was deemed to be “limited,” which was defined as (1) overt refusal of all food or drink, (2) an amount of oral intake so small that no useful observations could be made, or (3) so few captured swallows that no certain conclusions could be drawn. A VFSS was considered to be “diagnostic” if clinically useful information could be obtained. For example, frank aspiration captured on VFSS was defined here as a “diagnostic” study, while overt refusal to participate was considered “non-diagnostic.” All studies that included the word “limited” or “low participation” in the VFSS note were flagged and discussed by 2 reviewers to reach a consensus on their diagnostic utility.
Additional Variables of Interest
Other variables collected included home feeding (whether the patient could take ≥ 0.5 oz of oral intake at home based on chart information), patient demographics, indication for study, patient comorbidities, and specialty of ordering physician.
The indication of “inadequate intake by mouth” was defined as an ordering indication of G-tube/NG dependence and/or failure to thrive/growth concerns. An individual was said to have a “cardiopulmonary comorbidity” if they had at least one of the following comorbidities: lung disease, heart disease, airway abnormality, ventilator dependence, or tracheostomy dependence. “Neuromuscular comorbidities” were defined as having at least one of the following comorbidities: seizure disorder, cerebral palsy (CP), hypoxic-ischemic encephalopathy (HIE), hypotonia, neuromuscular disorder, or other neurologic disorder. An individual had a comorbidity of “GI disorders” if they had at least one of the following comorbidities: gastroesophageal reflux disease, eosinophilic esophagitis, or other gastrointestinal disorder. The “behavioral” comorbidity was present if an individual had autism or documented oral aversion.
Statistical Analysis
Univariate comparisons of VFSS success with categorical variables were examined using chi-squared tests. All analyses were performed via repeated measures logistic regression modeling using generalized estimating equations with an exchangeable working correlation matrix and empirical standard errors. The outcome was defined as diagnostic or nondiagnostic VFSS. In the unadjusted model, the only predictor used was the ability of the patient to take ≥0.5 oz by mouth routinely. The adjusted model included this same predictor as well as the following: age group, sex, race, ethnicity, an indication of “inadequate intake by mouth,” and the comorbidities of G-tube dependence, cardiopulmonary disease, neuromuscular, GI disorders, behavioral issues, or developmental delay. These comorbidity variables were of interest because of their potential effects on swallowing ability or dysfunction. The initial adjusted model included age as a continuous variable, along with cardiopulmonary and secondary reason indications, while cardiopulmonary comorbidity was not included. Age was converted into a categorical variable in the final adjusted model to examine the difference in diagnostic VFSS by age group (infants, toddlers, and children/adolescents), and the 2 included indications (cardiopulmonary and secondary reason) for ordering VFSS were removed because comorbidities were thought to be more informative. Finally, an investigation of whether the effect of minimal PO (by mouth) intake at home differed by age group with regard to diagnostic VFSS was performed by including an interaction term between these 2 predictors.
Analyses were performed in SAS 9.4 (SAS Institute Inc., Cary, NC).
Results
Demographic and Clinical Characteristics
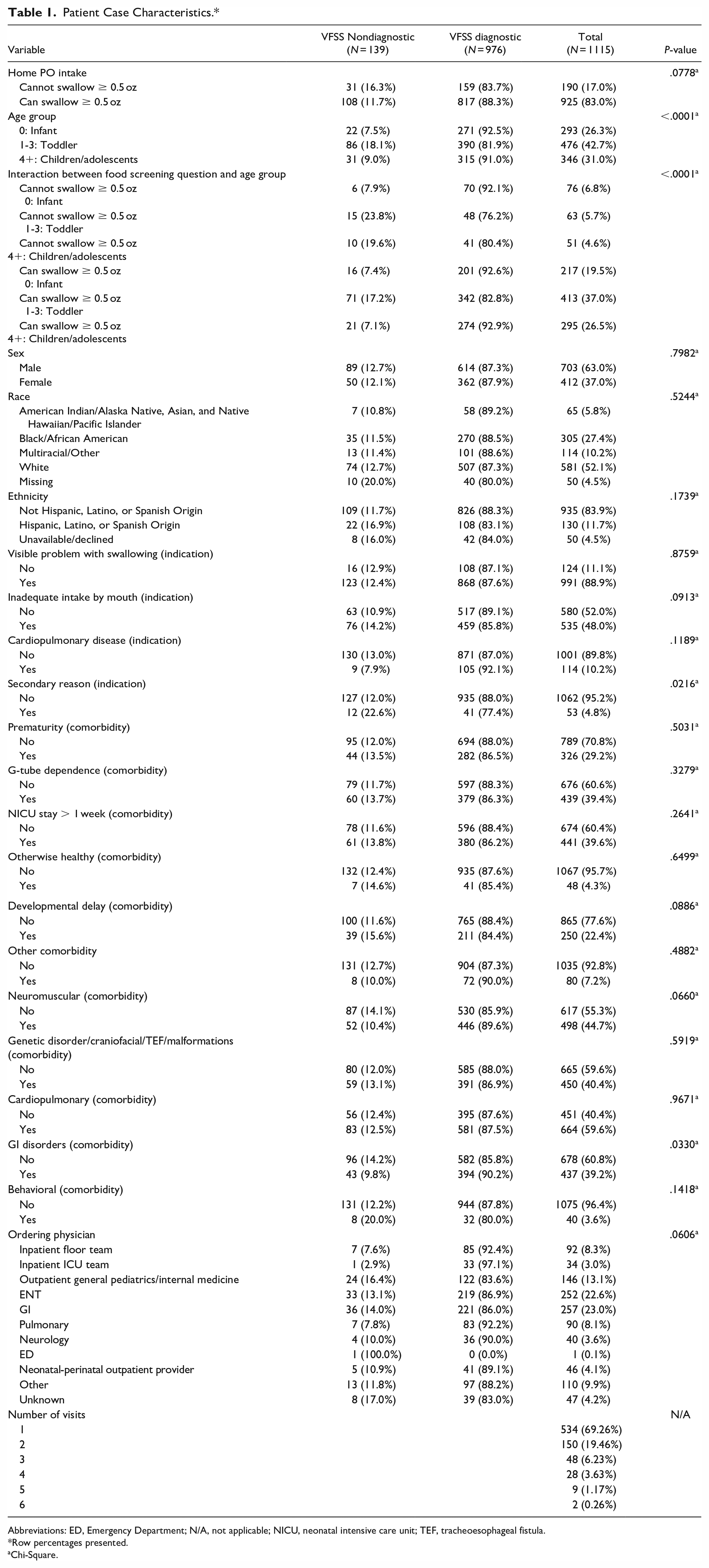
Table 1 displays the demographics of this sample of 1115 total cases from 771 unique patients. Most participants (69.3%) had only 1 visit, but some individuals had multiple visits ranging from 2 to 6 total visits. More cases were performed in male patients (63.0%) than in female patients (37.0%). The race breakdown for all cases is as follows: White (52.1%), Black/African American (27.4%), Multiracial/Other (10.2%), American Indian/Alaska Native or Asian or Native Hawaiian/Pacific Islander (5.8%), and missing (4.5%). The categories of American Indian/Alaska Native, Asian, and Native Hawaiian/Pacific Islander were combined due to the small number of patients in each of these categories. A minority (11.7%) of studies were performed on people who identified as Hispanic, Latino, or Spanish origin. Studies were most frequently ordered by ENT (22.6%), GI (23.0%), or outpatient general pediatrics/internal medicine (13.1%), but several other specialties also ordered VFSS.
Patient Case Characteristics.*
Abbreviations: ED, Emergency Department; N/A, not applicable; NICU, neonatal intensive care unit; TEF, tracheoesophageal fistula.
Row percentages presented.
Chi-Square.
Among those who could swallow ≥0.5 oz, the minority of cases (11.7%) had a nondiagnostic VFSS, while 16.3% of individuals who could not swallow ≥0.5 oz had a nondiagnostic VFSS. Across all age groups, the minority had nondiagnostic VFSS; the percentage of nondiagnostic studies for each age group was 7.5% for infants, 18.1% for toddlers, and 9.0% for children/adolescents. About half of the nondiagnostic VFSS cases came from toddlers who could swallow at least 0.5 oz.
Most patients had clinically overt dysphagia (88.9%). Cases were about evenly split between those who were (52.0%) and were not (48.0%) taking enough calories by mouth. Only 10.2% of cases were performed for a cardiopulmonary indication, and 4.8% were performed for secondary reasons such as the establishment of baseline swallow function or clearance for speech therapy in patients without clinical dysphagia.
The majority of cases had comorbid conditions with only 4.3% described as “otherwise healthy.” Just under half (44.7%) of cases had neuromuscular comorbidities, while just over half (59.6%) of cases had cardiopulmonary comorbidities. For other comorbidities, 29.2% were born premature, 39.6% had a NICU stay >1 week, 22.4% had a developmental delay, 40.4% had a genetic disorder, 3.6% had a behavioral disorder, 39.4% were G-tube dependent, 39.2% had GI disorders, and 7.2% had another comorbidity. The distributions and frequencies of most variables were similar across cases that were categorized as nondiagnostic VFSS and those categorized as diagnostic VFSS, except for age group, interaction between minimal PO intake at home and age group, the secondary reason indication, and the GI comorbidity.
Statistical Analysis
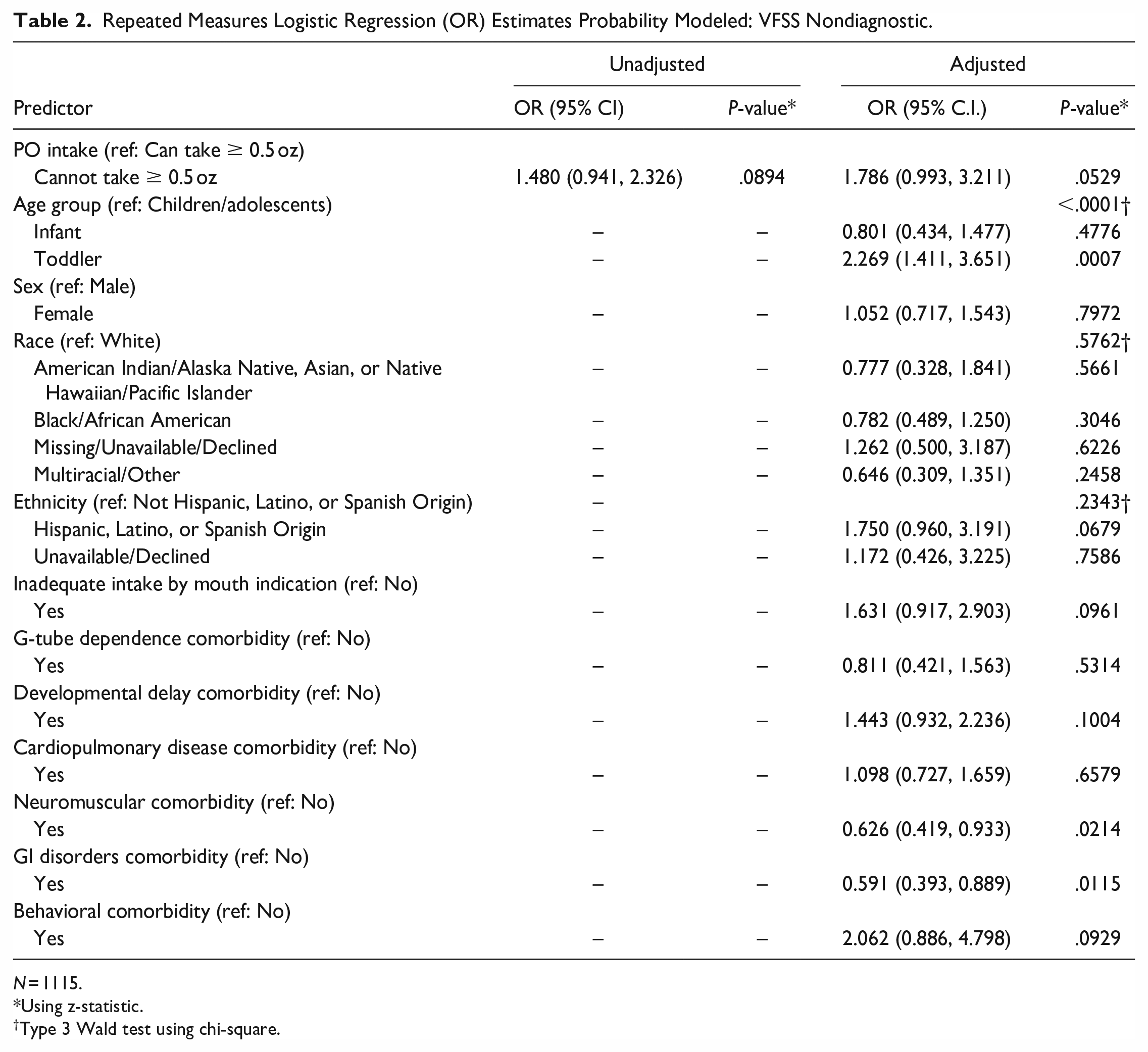
Table 2 displays the results of the repeated measures logistic regression model using generalized estimating equations with an exchangeable working correlation matrix and empirical standard errors (unadjusted and adjusted). The only variable included in the model for the unadjusted analysis was the question of whether the patient could take ≥0.5 oz by mouth routinely. In the unadjusted model, patients who could not take ≥0.5 oz by mouth had higher odds of nondiagnostic VFSS compared to those who could take ≥0.5 oz by mouth (unadjusted odds ratio 1.480, CI: 0.941-2.326, P = .0894), though this was not significant. These results remained the same in the adjusted analyses with patients who could not take ≥0.5 oz by mouth having higher likelihoods of nondiagnostic studies, with an adjusted odds ratio of 1.786 (CI: 0.993-3.211, P = .0529), though this was not statistically significant at the 0.05 level. Age group was associated with diagnostic VFSS (Type 3 Wald test P-value of <.0001). Compared to children and adolescents, toddlers had significantly higher odds of nondiagnostic VFSS (OR 2.269, CI 1.411-3.651, P = .0077), while there was no evidence of a significant difference in the comparison between infants and children/adolescents. Sex was not found to be associated with diagnostic VFSS. Overall, race and ethnicity were also not found to be associated with diagnostic VFSS (Type 3 Wald test P-values of .5762 and .2343, respectively). While the indication of “inadequate intake by mouth” was not found to be associated with diagnostic VFSS, those who had this indication had higher odds of nondiagnostic VFSS compared to those who did not have this indication (OR 1.631, CI 0.917-2.903, P = .0961). Each of the comorbidities—G-tube dependence, cardiopulmonary, developmental delay, and behavioral issues—was not found to be associated with diagnostic VFSS. In contrast, comorbidities of neuromuscular disorders and GI disorders were each associated with diagnostic VFSS: patients with neuromuscular or GI diagnoses had lower odds of a nondiagnostic VFSS compared to those who did not have neuromuscular or GI comorbidities, respectively (i.e., these patients had higher odds of a diagnostic VFSS; neuromuscular OR 0.626, CI 0.419-0.933, P = .0214; GI OR 0.591, CI 0.393-0.889, P = .0115). Among those with diagnostic VFSS studies, 359 individuals (80.5%) with a neuromuscular comorbidity had the ability to consume at least 0.5 oz. Similarly, 320 individuals (81.2%) with a GI comorbidity had the ability to consume at least 0.5 oz and produced a diagnostic VFSS. These findings are shown in Supplemental Table 1. Among those with a diagnostic VFSS, 46.4% (207 individuals) of those with a neuromuscular comorbidity were children/adolescents. There were 236 toddlers (44.5%) without a neuromuscular comorbidity who had a diagnostic VFSS. There were 169 individuals (42.9%) with a GI comorbidity and a diagnostic VFSS who were toddlers; 38% of toddlers without a GI comorbidity produced a diagnostic VFSS. These findings are shown in Supplemental Table 2. The above model, with the addition of an interaction between intake of ≥ 0.5 oz and age group (divided into subgroups of infants, toddlers, and children/adolescents), had no significant interaction (P-value = .3003), indicating there was no evidence of a difference in effect of intake of at least 0.5 oz by age group on diagnostic VFSS.
Repeated Measures Logistic Regression (OR) Estimates Probability Modeled: VFSS Nondiagnostic.
N = 1115.
Using z-statistic.
Type 3 Wald test using chi-square.
Discussion
Individuals who were unable to take at least 0.5 oz by mouth did have a higher likelihood of a nondiagnostic VFSS compared to those who were able to consume this amount, but this was not found to be associated with diagnostic VFSS at the 0.05 significance level. Age was associated with diagnostic VFSS, with toddlers having higher likelihoods of a nondiagnostic VFSS compared to children and adolescents. However, the overall interaction between age and the ability to take at least 0.5 oz by mouth routinely was not associated with diagnostic VFSS, indicating that there is no evidence that the 0.5-oz screening question has a differential effect on VFSS by age group. This analysis also found that the comorbidities of neuromuscular disorders and GI disorders were each associated with producing more clinically useful VFSS compared to those who did not have each of these comorbidities, respectively. Taken together, the present findings show that children with GI and neuromuscular comorbidities are more likely to have a diagnostic VFSS than those without these comorbidities, while toddlers are less likely to have a diagnostic VFSS than children and adolescents.
When interpreting these results, it is important to remember that patients were categorized by comorbid diagnosis, not by symptom. Many of the comorbidities that were queried have a wide range of manifestations and disease severities, which might lead to variable impacts on swallowing function, and therefore there is no significant link between a single diagnosis and diagnostic VFSS. For example, some patients with G-tubes might take some food by mouth regularly, while others depend entirely on their G-tubes for nutrition. Similarly, some patients with developmental delay might swallow normally, while others have swallowing difficulties or refuse to swallow entirely. These variations in presentation could explain why some of the comorbidities here were not found to be associated with diagnostic VFSS. Future research might include a measure of disease severity rather than simply the presence or absence of a single given diagnosis to characterize the specific signs and symptoms that best predict diagnostic or nondiagnostic VFSS.
Further, it is important to remember that in general, pediatric patient behavior in the clinic does not always mimic at-home behavior. For example, children with significant comorbidities might be able to reach the threshold of at least 0.5 oz of intake by mouth at home in the setting of unlimited time and attempts. These patients might have more difficulty readily taking the same quantity in a limited amount of time in a regimented manner, especially because the number of attempts allowed during VFSS must be balanced with the need for radiation with each attempt. Similarly, children might find the foreign environment of a doctor’s office overwhelming and frightening, which would prevent them from readily consuming food or drink as they might in their own homes. These possible reasons are non-exhaustive; there remain several factors that might contribute to a discrepancy between home feeding behavior and feeding behavior in the clinic. The impact of the clinical environment might contribute to the differences in diagnostic VFSS by age group, in that younger children might be more anxious, and thus less likely to produce a useful study. A screening question about home oral intake might provide some insight into a pediatric patient’s clinical situation, but other questions about a child’s past behavior and level of acceptance of unfamiliar surroundings are likely necessary to provide a fuller picture.
While the screening question was not found to be associated at the 0.05 significance level with diagnostic VFSS, these results suggest that whether a child routinely consumes at least 0.5 oz by mouth still may help identify patients who might not produce a clinically useful swallow study. The 0.5 oz cutoff for the present analysis was chosen arbitrarily; future studies might focus on determining age- or size-appropriate threshold volumes of PO intake needed to predict a diagnostic VFSS. Instead of considering this 0.5-ounce screening question in isolation, accounting for a patient’s individual clinical picture, including behavior in clinic or eating patterns, might provide a more accurate representation of a patient’s ability to cooperate enough to produce a VFSS that can guide management. These findings can positively guide clinical decision-making; a better notion of how clinicians might distinguish poor candidates for VFSS can benefit children, caretakers, and providers alike by preventing nondiagnostic VFSS attempts. Avoiding an uninformative VFSS has various benefits, including sparing pediatric patients’ needless radiation exposure and stress, sparing caretakers the burdens of an extra appointment, and sparing clinicians and hospitals the costs and personnel requirements of these studies.
This study is not without limitations. The chart review component necessitated inference when details were not explicitly stated in a patient’s note; though every effort was made to standardize the responses in such situations, it is always possible that there remain some inconsistencies. In addition, the information about each study was gathered from a clinical note, which might not have provided accurate or adequate detail for the purposes of this study. One example of missing information is the textures of liquids being taken at home prior to VFSS. Finally, comorbidities and indications often encompassed a range of symptoms, and were grouped to aid analysis; this precluded investigation of different symptoms that might be predictive of VFSS difficulty. Future studies might collect data that allow for the stratification of patients based on signs and symptoms, instead of by a diagnosis of a specific comorbidity. Further investigation is also warranted to determine the association of certain comorbidities with diagnostic or nondiagnostic VFSS. Despite these limitations, the present findings have clinically significant implications that might guide future diagnostic steps as efficiently as possible.
Conclusion
There is no strong evidence of an association between the ability to take at least 0.5 oz by mouth and the production of a diagnostically useful VFSS, but age was associated with diagnostic VFSS. Toddlers had higher odds of a nondiagnostic VFSS compared to children and adolescents. Certain comorbidities were significantly associated with useful swallow studies: patients with GI and neuromuscular disorders were more likely to produce useful studies compared to those without these comorbidities.
Supplemental Material
sj-docx-1-ear-10.1177_01455613231205532 – Supplemental material for Predictive Factors in Identifying Pediatric Patients at Risk of Diagnostically Limited Videofluoroscopic Swallow Studies
Supplemental material, sj-docx-1-ear-10.1177_01455613231205532 for Predictive Factors in Identifying Pediatric Patients at Risk of Diagnostically Limited Videofluoroscopic Swallow Studies by Julia Canick, Dehra McGuire, Kayla W. Kilpatrick, Raquel Perry, Maragatha Kuchibhatla, Erika Juhlin and Janet W. Lee in Ear, Nose & Throat Journal
Footnotes
Author’s Note
This work was presented by Dr. Dehra McGuire at The Fall Voice Conference, San Francisco, October 6 to 8, 2022.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, J.C., upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Biostatistics, Epidemiology and Research Design (BERD) Methods Core funded through Grant Award Number UL1TR002553 from the National Center for Advancing Translational Sciences (NCATS), a component of the National Institutes of Health (NIH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Ethics and Consent Statements
The Ethics Committee of the Duke University Institutional Review Board waived the need for ethics approval and the need to obtain consent for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this non-interventional study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
