Abstract
Introduction
Olfactory dysfunction, partial or complete loss of sense of smell, is a common condition that can significantly impact a person’s quality of life and is often difficult to treat. Various factors influence the ability to smell, such as physical activity, genetics, nutrition, smoking, head trauma, medical treatments, and virus exposure. 1 Impairment of the sense of smell, known as hyposmia or anosmia, is prevalent among 3%-20% of the population. 1 The negative impact of impaired olfaction on quality of life is significant, affecting the enjoyment of food, personal hygiene, and social relationships while also contributing to more significant depressive symptoms and overall physical and mental well-being. 1
In addition, decreased smell function can impair the ability to sense warning odors, increasing the risk of danger from fires, environmental toxins, natural gas leaks, and spoiled food. 1 It is concerning that less than a quarter of individuals with olfactory dysfunction are aware of their problem, and among adults over 70 years old, misidentification rates for warning odors are alarmingly high, highlighting a significant public health concern. 1
As people age, their risk of experiencing olfactory dysfunction increases, which can be caused by various factors, including acute and chronic sino-nasal disease, upper respiratory infections, exposure to toxic chemicals, and degenerative diseases. 1 Around 10% of individuals aged 65 years or older experience some form of olfactory dysfunction, ranging from mild loss to complete anosmia. 1 The relationship between olfactory dysfunction and neurodegenerative disorders has gained recognition recently due to its high prevalence, early onset, persistence throughout disease progression, and ease of olfactory testing. 1
During the COVID-19 outbreak, anosmia has risen among many patients. 2 It is considered one of the characteristic symptoms of COVID-19 infection. 2 A meta-analysis has estimated that the prevalence of olfactory dysfunction among COVID-19 patients is approximately 52.73%. 3 In addition, 1 study reported that nearly 73% of COVID-19 patients experienced anosmia, which occurred before their diagnosis in many cases. Furthermore, anosmia was the initial symptom in over 26.6% of these patients. 3
Several medications have been proposed as potential treatments for anosmia. 4 Olfactory training has been previously recommended as a safe and effective way to address olfactory dysfunction, but currently, no medications are approved for treating this condition. 4 Corticosteroids have been extensively studied for COVID-19-related olfactory and gustatory dysfunctions. 4 Still, their use may come with additional risks and can even reduce the clearance of the virus from the body. 4 Some patients with olfactory disorders can be resolved with surgical therapy. 5
Insulin can be administered intranasally and reach the brain through the olfactory bulb. 6 Recent studies hypothesized that intranasal insulin effectively treats olfactory dysfunction.7,8 It has already shown improvement in some cases; meanwhile, it had the opposite effect on normosmic subjects. 9
Due to the controversy of evidence, this systematic review aims to comprehensively evaluate the evidence regarding the effectiveness and safety of topical insulin use in olfactory dysfunction patients. The results of this review can inform clinical practice guidelines and improve patient outcomes following management with insulin.
Materials and Methods
This study is a systematic review of studies discussing the outcomes and safety of intranasal insulin in the management of smell loss. PubMed, Google Scholar, and CENTRAL databases were searched using the following words in the title or abstracts: “Intranasal insulin AND (Smell OR Olfactory).” The outputs of electronic database searches were transferred to Rayyan Software for screening and selection. 10
All study titles with the abstracts from this search were screened systematically. Later, the results were filtered to include all original research articles.
Then, the subsequent step was determining and applying the inclusion criteria to select the studies that will be considered in the review. Abstracts were checked manually to choose the appropriate ones to be included.
The titles, abstracts, and full text were independently reviewed carefully for eligibility by the first and second authors. A third senior author resolved any disagreement between the 2 authors.
Inclusion Criteria
The included studies were published without time frame limitations, mentioning intranasal insulin’s effect in managing olfactory dysfunction in adults and relevant outcomes to the research question. Studies that reported the number of patients who underwent rhinoplasty with and without the use of external nasal splinting were included.
Exclusion Criteria
The excluded studies were (1) studies with no full-access link; (2) studies in languages other than English; (3) study types other than original research articles such as case reports, letters, review articles, and systematic review articles; (4) studies that discuss supplements and traditional medicine; and (5) studies performed on laboratory scale/animal models.
Essential data points were extracted from the final record of included studies to be summarized. Furthermore, references of chosen studies were manually revised to define similar studies.
Screening and Data Extraction
First, screening of the title and abstract was performed by the reviewers. Eligible articles were imported for review and assessment for inclusion. The data were extracted using an Excel sheet. We extracted the following information: (1) basic information (the first author’s surname, publication year, and sample size), (2) the study objectives, (3) the study outcomes, (4) gender, (5) type of olfactory dysfunction (anosmia or hyposmia), (6) causes of dysfunction, (7) type of insulin, (8) duration, (9) frequency of administration, and (10) any complications.
Results
A total of 66 references were identified by looking up the literature on 3 databases. A total of 33 were obtained from PubMed, 21 articles from Google Scholar, and 12 from CENTRAL databases. A total of 21 references were removed as duplicates, so the number of studies was 45 articles. By reviewing the articles, 6 articles followed the inclusion criteria and were selected to be part of the systematic review, and 177 subjects were included (Figure 1).
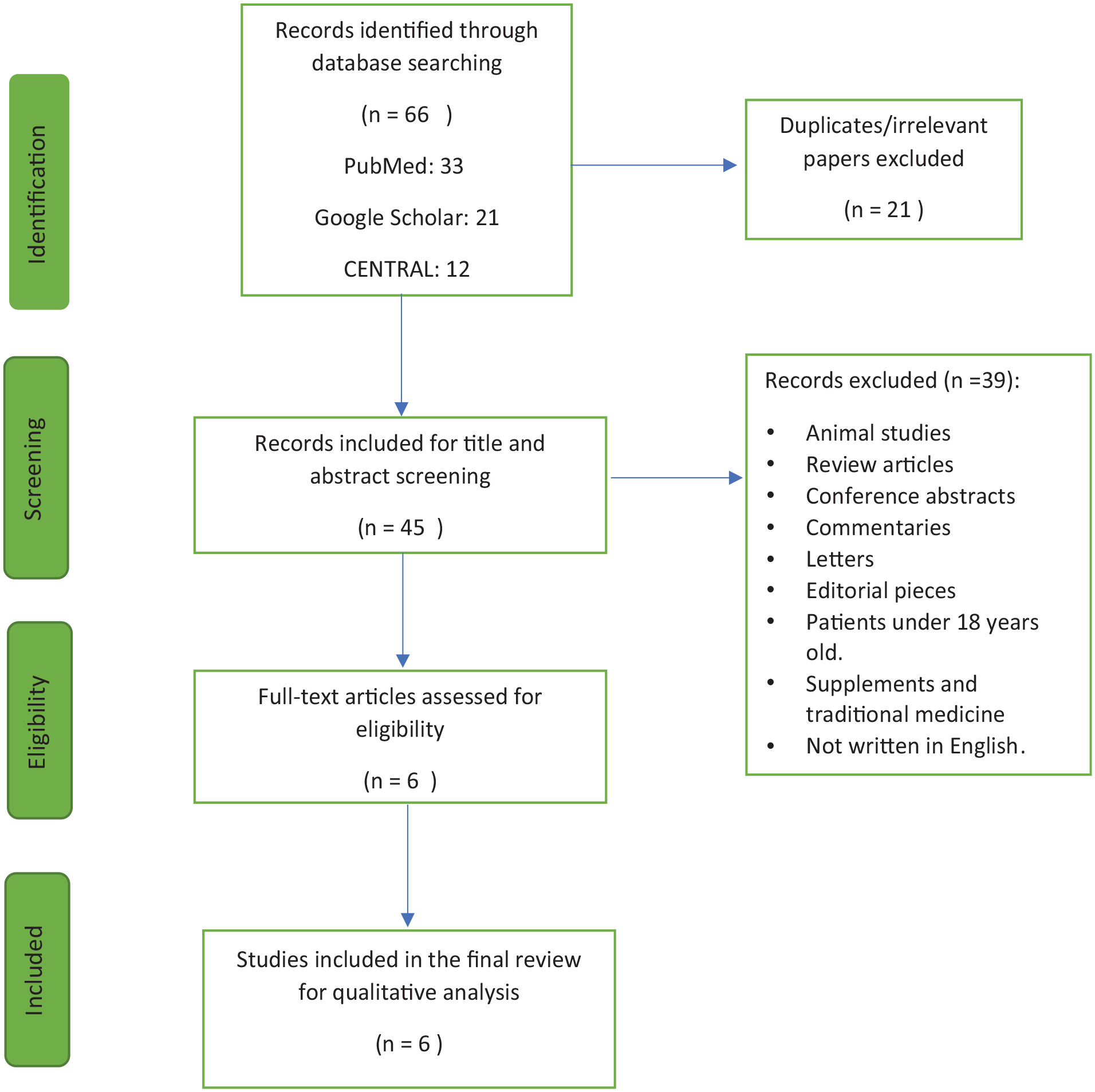
PRISMA flow diagram.
Several studies have investigated the efficacy and safety of intranasal insulin therapy for the treatment of olfactory dysfunction. The 6 included studies consisted of 1 pilot study, 2 placebo-controlled, double-blind settings in a within-subject design, 1 single-blinded, placebo-controlled, crossover study, 1 randomized clinical trial, and 1 in vitro characterization and clinical evaluation clinical study.
Mohamad et al found that intranasal insulin fast-dissolving film significantly improved olfactory detection scores and discrimination values in patients with post-COVID-19 anosmia. Thanarajah et al demonstrated that intranasal insulin improved olfactory threshold performance in healthy male participants, and the effect was dose dependent. Rodriguez-Raecke et al showed that intranasal insulin reduced olfactory sensitivity for n-butanol in female subjects but not in males and had no effect on peanut odorants. Rezaeian et al found that intranasal insulin administration increased odor identification in hyposmic patients, with a significant positive development observed in patients treated with 40 I.U. of insulin. Schöpf et al demonstrated that intranasal insulin improved olfactory threshold and subjective intensity ratings of odors in post-infectious anosmic patients. Brünner et al showed that intranasal insulin decreased olfactory sensitivity but did not affect olfactory discrimination ability in healthy normosmic participants. Overall, these studies suggest that intranasal insulin may be a promising approach for the treatment of olfactory dysfunction, but further research is needed to determine optimal dosing and safety profiles. The characteristics of the studies, including the sample size, study population, type of olfactory dysfunction, causes of dysfunction, type of insulin, duration, frequency of administration, study objectives, and study outcomes, are summarized in Table 1.
Summary of Studies Investigating Olfactory Dysfunction and Insulin Intervention.
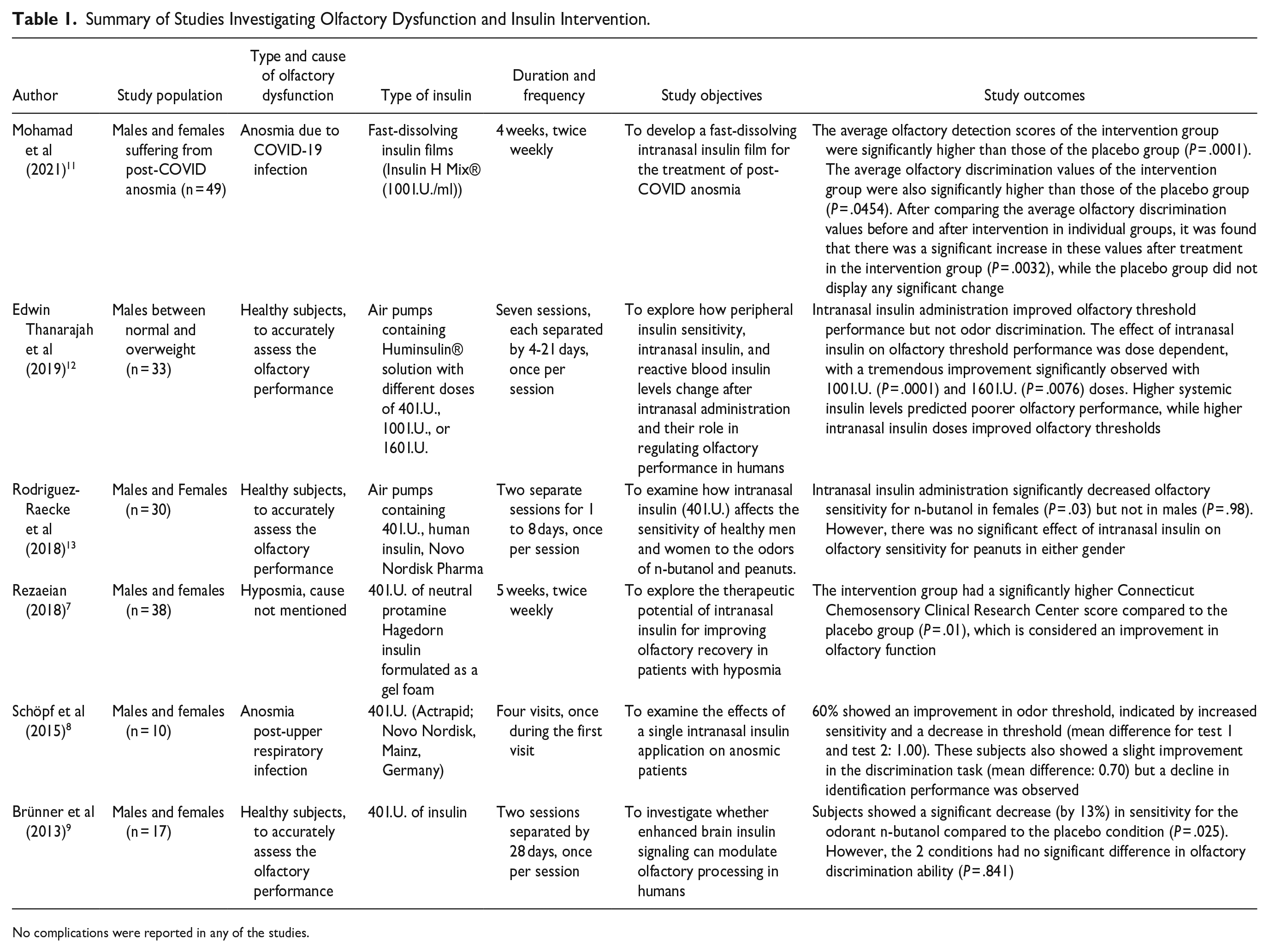
No complications were reported in any of the studies.
The findings of the included studies demonstrated the potential benefits of intranasal insulin therapy for olfactory dysfunction. The results suggest that intranasal insulin may hold promise as a treatment. The efficacy of intranasal insulin varied depending on the dose administered, with different studies testing doses ranging from 40 I.U. to 160 I.U. The gender differences were also evident, with some studies including only men or accounting for hormonal influences in women. The average follow-up period in the studies was 30 weeks, and no complications were reported, indicating the safety of intranasal insulin administration. Also, the findings suggest that intranasal insulin can be safely administered at various doses and formulations.
Discussion
Insulin is a hormone primarily involved in regulating glucose metabolism and energy homeostasis in the body. Recent studies have suggested that insulin may also affect olfactory function. The mechanisms underlying this effect have yet to be fully understood, but several potential hypotheses have been proposed. One proposed mechanism is that insulin may modulate the activity of olfactory receptors, which are specialized proteins located on the cilia of olfactory sensory neurons. These receptors detect and transduce odorant molecules into electrical signals that the brain can interpret. Insulin may interact with these receptors directly or indirectly through downstream signaling pathways, thereby influencing their sensitivity and responsiveness to different odors. 14 Another potential mechanism is that insulin may affect the brain’s neural processing of olfactory information. The olfactory system is complex and involves multiple brain regions, including the olfactory bulb, piriform cortex, and amygdala. Insulin may influence the activity and connectivity of neurons within these regions, altering the processing and integration of olfactory information and ultimately affecting olfactory function. 15
Insulin therapy is a potential management option for olfactory dysfunction or smell loss. Compared to other management methods for smell loss, such as olfactory training and drug therapy, insulin therapy is a promising approach that warrants further investigation. 3 Olfactory training involves repeated exposure to specific odors to improve olfactory function and has shown some success in clinical studies. Drug therapy, such as corticosteroids, has also been used to treat olfactory dysfunction, but its efficacy remains variable, and it may have side effects. By contrast, insulin therapy has shown potential in preclinical studies and case reports and has the advantage of being a naturally occurring hormone with a well-established safety profile.16,17
While concerns have been raised about the safety of insulin therapy for olfactory dysfunction, current evidence suggests that it is generally safe when administered appropriately and at low doses. 18 However, careful monitoring and appropriate medical management are essential to ensure the safe use of insulin in the management of smell loss.
The ability to smell is influenced by several factors, including physical activity, genetics, nutrition, smoking, head trauma, medical treatments, and virus exposure. 1 Olfactory dysfunctions known as hyposmia and anosmia were widely spread after the COVID-19 infection. 2 Recent studies have suggested that intranasal insulin could be an effective treatment for olfactory dysfunction and has shown improvement in some cases.6,8
In this review, 2 studies have tested the intranasal insulin effect on patients suffering from anosmia.8,11 Mohamad et al evaluated the effectiveness of a fast-dissolving intranasal insulin film for treating post-COVID anosmia twice a week for 4 weeks. 11 The study found that the average olfactory detection and discrimination scores were significantly higher in the intervention group than in the placebo group after 4 weeks of treatment (P = .0001 and P = .0454, respectively), which refers to the effectiveness of insulin films in enhancing olfactory dysfunction. 11
On the other hand, Schöpf et al have administered only a single dose of air pump intranasal insulin. 8 The study found an improvement in odor threshold, indicating increased sensitivity and a decrease in threshold. 8 These subjects also showed a slight improvement in the discrimination task, but a decline in identification performance was observed. 8 Therefore, the results of the 2 studies suggest that the frequency and dosage form of insulin may enhance the treatment outcomes.
Other studies have examined the effects of intranasal insulin on olfactory performance in healthy subjects.9,12,13 Thanarajah et al found that intranasal insulin administration improved olfactory threshold performance without effect on odor discrimination. 12 On the contrary, the study by Rodriguez-Raecke et al tested the sensitivity to the odors of n-butanol and peanuts. It showed that intranasal insulin administration led to a significant decrease in olfactory sensitivity for n-butanol in females but not males. However, there was no significant effect of intranasal insulin on olfactory sensitivity for peanuts in all subjects. 13
In consistency with Rodriguez-Raecke et al’s study, Brünner et al have investigated whether enhanced brain insulin signaling can modulate olfactory processing in humans. 9 They found that subjects showed a significant decrease (by 13%) in sensitivity for the odorant n-butanol compared to the placebo condition (P = .025). 9 However, the 2 conditions had no significant difference in olfactory discrimination ability. 9
Brünner et al have explained the change in olfactory function and the effect of insulin by which insulin signaling affects the olfactory threshold in normal humans. 9 In addition, impaired olfactory sensitivity occurred for a non-food odorant, whereas no impairment was found for olfactory discrimination. 9 Moreover, olfactory sensitivity is controlled by the peripheral olfactory system. Meanwhile, odor discrimination and identification are controlled by higher-order brain areas, which suggests that intranasal insulin does not reach it. 9
In the study by Mohamad et al, the average olfactory discrimination values were tested before and after insulin administration. 11 It was found that there was a significant increase in these values after treatment in the intervention group (P = .0032), while the placebo group did not display any significant change. 11 This controversy of olfactory discrimination results maybe due to the difference in doses as that was the only study that tested 100 I.U. of insulin; the usual dose tested in the other studies was 40 I.U.
Moreover, Thanarajah et al reported that the effect of intranasal insulin on olfactory threshold performance was controlled by dose—the most significant improvements were observed with 100 I.U. and 160 I.U. doses. 12 In the study by Rezaeian, odor identification was enhanced on a dose of 40 I.U. insulin foam gel. 7 He found that the intervention group had a significantly higher Connecticut Chemosensory Clinical Research Center score than the placebo group (P = .01), indicating improved olfactory function. 7 Thus, it is recommended to apply further studies regarding doses, odor discrimination, and odor identification.
Regarding gender, Thanarajah et al have included only men in their study to avoid the effect of hormonal changes on the olfactory function that occurs in women. 12 Moreover, the study by Brünner et al included women, but they separated sessions by 28 days according to the menstrual cycle to overcome the effect of hormonal changes. 9 Rodriguez-Raecke et al have included both males and females and reported a difference between them in study outcomes. By contrast, females had a significant decrease in olfactory sensitivity for n-butanol (P = .03). Meanwhile, males did not (P = .98) 13 . They dated the difference back to the difference in insulin and leptin signaling regulation between men and women. 13
The average follow-up on patients in all 6 studies was 30 weeks. No complications were reported in studies suggesting that intranasal insulin can be safely administered at different doses and in various formulations.7 -9,11 -13
Several clinical trials are currently underway. One of the ongoing clinical trials investigating the potential use of insulin therapy for olfactory dysfunction is the Clinical Assessment of Insulin Fast Dissolving Film in Treatment of Post Infection Anosmia study (NCT04893339) conducted in India. This randomized, double-blind, placebo-controlled trial aims to evaluate the efficacy of a fast-dissolving insulin film for the treatment of post-infection anosmia. It provides valuable information about the potential role of insulin therapy in the management of post-infection anosmia. 19 Another clinical trial investigating the potential use of insulin therapy for olfactory dysfunction is the Intranasal Insulin for COVID-19-related Smell Loss study (NCT04910962) conducted in the United States. This randomized, double-blind, placebo-controlled trial aims to evaluate the efficacy of intranasal insulin for treating smell loss in patients with COVID-19. The results of this study, expected to be available by April 2024, will provide important information about the potential use of insulin therapy for managing olfactory dysfunction in patients with COVID-19. 20
Overall, while insulin therapy for managing olfactory dysfunction is a promising approach, further research is needed to fully understand its efficacy and safety profile. Future studies should investigate the optimal dose and duration of insulin therapy for the management of smell loss, as well as the long-term safety and potential side effects of insulin therapy. In addition, future studies should investigate the mechanisms underlying the therapeutic effects of insulin on olfactory dysfunction, which may provide insights into the pathophysiology of olfactory dysfunction and may lead to the development of more targeted and effective therapies. 21
Conclusion
In conclusion, intranasal insulin has been suggested as a potential treatment for olfactory dysfunction, including post-COVID anosmia. The reviewed studies have shown promising results regarding the ability of intranasal insulin to improve olfactory function, particularly the olfactory threshold, with some variations in the effect on discrimination and identification ability. The frequency and dosage form of insulin may enhance the treatment outcomes. However, further studies are needed to investigate the optimal doses and the effect on different types of odors and to explore the potential gender differences in response to intranasal insulin. Furthermore, the reviewed studies suggest that intranasal insulin can be safely administered and may offer a promising avenue for treating olfactory dysfunction.
Footnotes
Authors’ Note
This work has not been previously presented.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
