Abstract
Introduction
Laryngeal cancer is the second most common malignant tumor of the head and neck. According to the Global Cancer Observatory estimations, more than 160,000 new cases of laryngeal cancer occurred in men and more than 24,000 in women in 2020 worldwide. 1 The 5-year relative survival rate for laryngeal cancer in Germany is 64% in men and 63% in women. 2
Total laryngectomy (TLE), with an incidence of approximately 3000 procedures annually in Germany, 3 remains the treatment of choice for recurrent and advanced stages of laryngeal and hypopharyngeal tumors.4,5 TLE, often accompanied by adjuvant radiotherapy or radiochemotherapy, has a negative impact on quality of life, particularly with regard to speech and swallowing function, as well as pulmonary rehabilitation.6 -9
In this regard, the oncological curative aspects are no longer the sole point of interest, but also the postoperative rehabilitation to improve the quality of life of laryngectomized patients. 10 The insertion of a voice valve at the time of surgery or shortly after is regarded as the method of choice for speech rehabilitation. However, this procedure is not without complications.6,7,11 -13
The creation of a permanent tracheostoma changes the pulmonary situation, mainly due to the loss of the conditioning and filtering functions of the upper airways. These functions include warming, humidification, filtering of the inspired air, as well as adapting the breathing dynamics. For these reasons, the so-called “heat moisture exchangers” (HMEs) are indispensable, primarily due to their significant role in partially restoring the physiological environment in the lower airways, which helps prevent metaplastic changes in the bronchial mucosa. However, it is important to note that these beneficial effects necessitate high patient compliance.14 -23
Despite these rehabilitative measures, laryngectomized patients may experience repeated hospitalizations due to inflammation of the tracheal mucosa associated with bleeding and crust formation.7,8,17 Therefore, improving the pulmonary situation is essential to improve the results after TLE and, consequently, to improve the patient’s quality of life.
This study aims to evaluate the effects of the new generation of heat moisture exchangers (NG-HMEs) on pulmonary rehabilitation, quality of life, and patient satisfaction among HME-experienced laryngectomized patients. The primary goals of this study are defined as improvement in the pulmonary situation measured through a decrease in the frequency, intensity, and impact of cough and mucus production in daily life. In addition, the impact on quality of life and patient satisfaction will be evaluated,24,25 while patient compliance and usage patterns will serve as secondary outcome measures.
Materials and Methods
Study Design
In this prospective study, laryngectomized patients with prior HME experience were recruited at the Department of Otorhinolaryngology, Head and Neck Surgery of the University Hospital Erlangen in the period between June 1, 2021 and November 30, 2021. The study was approved by the ethics committee of the University of Erlangen-Nuremberg (244_21 B). All patients provided their written informed consent to participate in the study. The inclusion and exclusion criteria were defined as follows:
Inclusion criteria:
- Laryngectomized patients who are followed up in the ENT Department of the University Hospital Erlangen.
- TLE was performed at least 4 months ago.
- Minimum age 18 years.
- Sufficient cognitive and linguistic abilities to understand the contents of the questionnaire and the written consent after detailed information about the study was obtained.
- Post-TLE care with ATOS Medical® assistive devices.
Exclusion criteria:
- Difficulty in understanding or communicating or cognitive impairment.
- Unfinished oncological treatment.
- Post-TLE care through other manufacturers.
During regular outpatient follow-up visits, patients were interviewed about their current post-TLE care, this point of time is referred to as “baseline.” Following this, all patients were instructed to use the NG-HMEs. After 6 weeks (follow-up 1) and 12 weeks (follow-up 2) of using the NG-HMEs, a new questionnaire was administered. All questionnaires were completed by the patients themselves. The survey was conducted using validated questionnaires, such as the cough and sputum assessment questionnaire (CASA-Q), the European Quality of Life 5 Dimensions Index Score (EQ-5D Index Score), and the European Quality of Life 5 Dimensions Visual Analog Scale (EQ-5D-VAS). Furthermore, study-specific questionnaires were used to report usage patterns, the number of dry coughs, and expectorations per day.24,26,27
The study flow is shown in the CONSORT flow diagram (Figure 1).
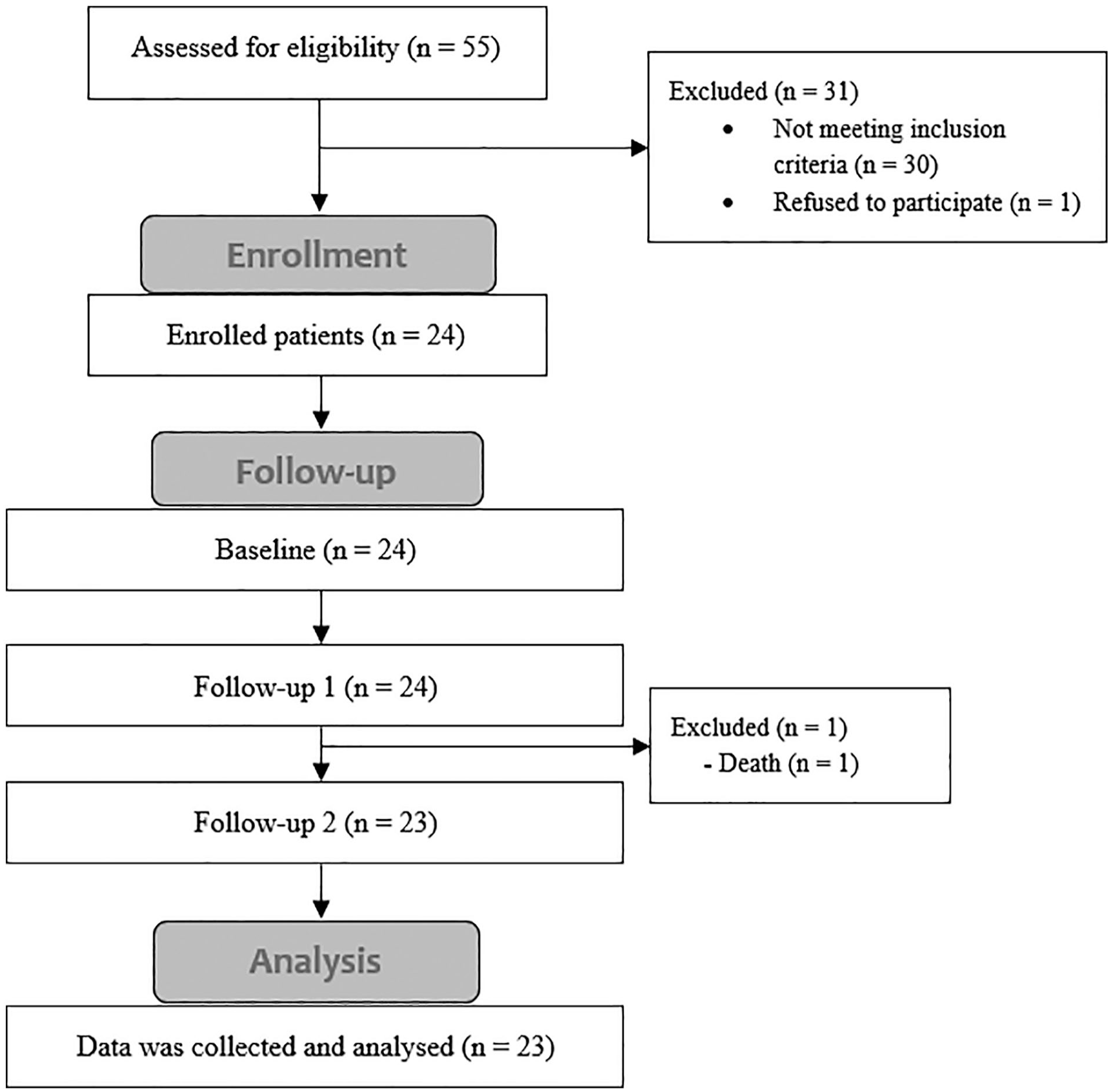
CONSORT flow diagram of the study.
Patients would report their HME usage patterns each time using study-specific questionnaires. These would include the following questions: How many hours a day would they use the HMEs, how often and why do they change their HME, and whether they use NG-HMEs situationally? In addition, patients would receive a series of questions about the frequency of dry cough and expectorations in the past 24 hours, peristomal skin irritation, tracheal irritation, physical activity, doctor visits, and shortness of breath. This questionnaire was developed by Hilgers et al. 27 and is used in many studies investigating the effects of HMEs.18,19,28 -30
In addition, patients would report their pulmonary complaints using the CASA-Q. CASA-Q is a validated questionnaire for cough and sputum symptoms and their impact on daily life in patients with chronic obstructive pulmonary disease (COPD). 24 The CASA-Q includes 20 questions divided into 4 domains: cough symptoms (COUS), cough impact (COUI), sputum symptoms (SPUS), and sputum impact (SPUI). Through these questions, patients would report their pulmonary symptoms, such as cough frequency, intensity, the occurrence of cough attacks, mucus production and consistency, and the impact of these complaints on their daily life in the last 7 days. Subsequently, a score ranging from 0 to 100 would be calculated for each domain. The higher the score, the less troublesome the symptoms are.
Quality of life would be reported using the EQ-5D index score and the EQ-5D-VAS. 25 The EQ-5D index score is a validated instrument in which 5D of health (mobility, self-care, daily activities, pain/discomfort, and anxiety/depression) and their impact on overall quality of life are evaluated. The EQ-5D-VAS enables patients to subjectively rate their overall health status on a scale from 0 to 100 points, where 0 points represent the worst possible health status, and 100 points represent the best possible health status.
Finally, patients were asked to rate their overall experience with NG-HMEs on a scale of 1 to 5 (1 point very poor; 5 points very good) and indicate if they would prefer to use them after the conclusion of the study.
The NG-HMEs
The NG-HMEs—commercially known as the Provox® Life HME series—consist of 6 different HMEs adapted to various daily situations and activities. These are the life HME night, go, home, energy, protect, and freehands. The NG-HMEs offer different values of breathing resistance and humidification capacity, offering the patients HMEs better adapted to their daily routine. The values for breathing resistance and humidification capacity are shown in Table 1.
Values of Moisture Loss and Pressure Drops for the Provox XtraMoist, Provox XtraFlow, and NG-HMEs as Provided by the Manufacturer ATOS Medical, Malmö Sweden.
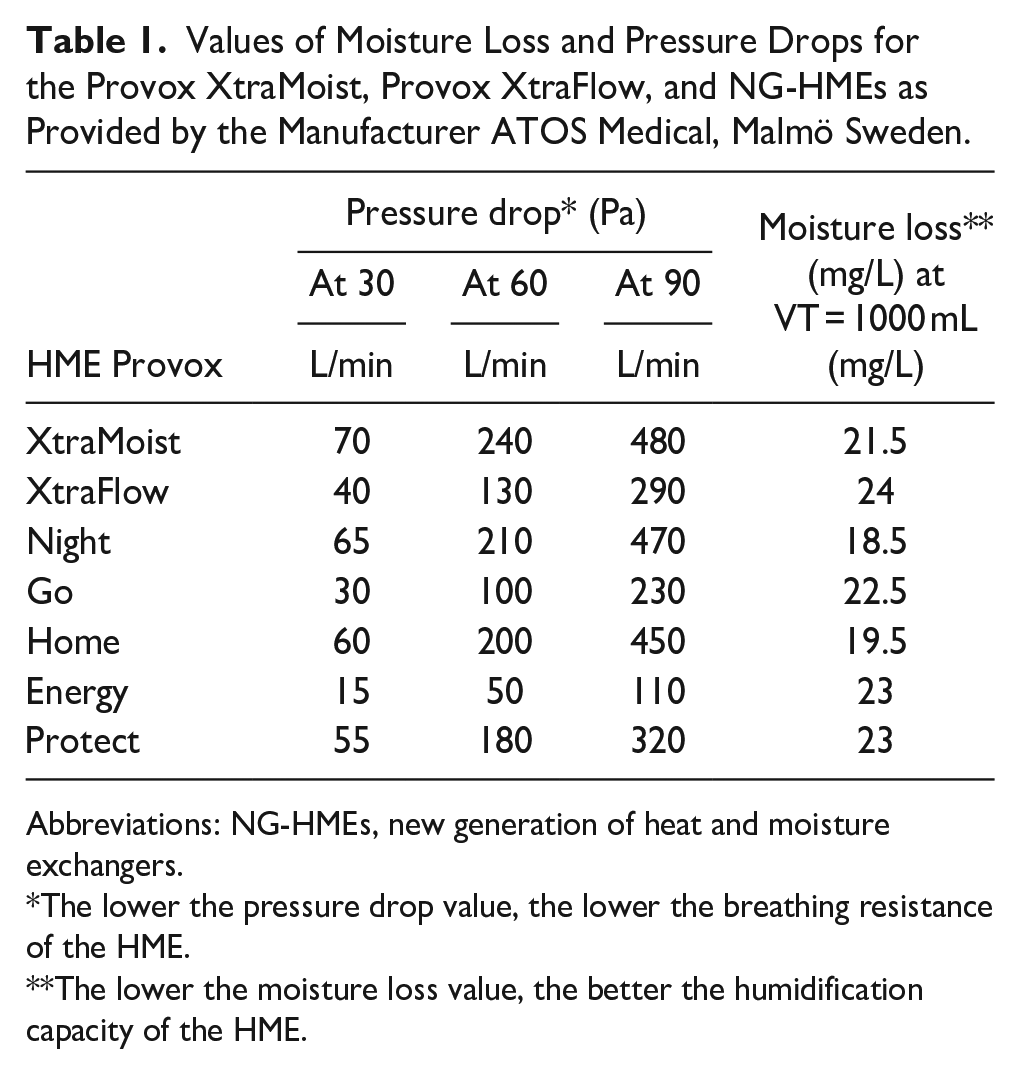
Abbreviations: NG-HMEs, new generation of heat and moisture exchangers.
The lower the pressure drop value, the lower the breathing resistance of the HME.
The lower the moisture loss value, the better the humidification capacity of the HME.
Statistical Evaluation
The metric variables were tested for normal distribution using histograms, the Q-Q plot, and the Kolmogorov-Smirnov test. Metric variables were presented as mean with standard deviation (mean ± SD), as well as median and the 25th; 75th percentiles. In addition, the minimum and maximum were reported (min-max). Comparisons between baseline and follow-up 2 were performed by paired t-test (for normally distributed data) or by Wilcoxon signed-rank test (for non-normally distributed data). The effect size was reported with r, where r = 0.1 corresponded to a small effect, r = 0.3 to a medium effect, and r = 0.5 to a large effect. Differences in nominal variables between baseline and follow-up 2 were evaluated using cross-tabulations and the Mc-Nemar test. A P-value of <0.05 was defined as significant. All data were analyzed using SPSS 29.0 (SPSS Inc., Chicago, IL, USA).
Results
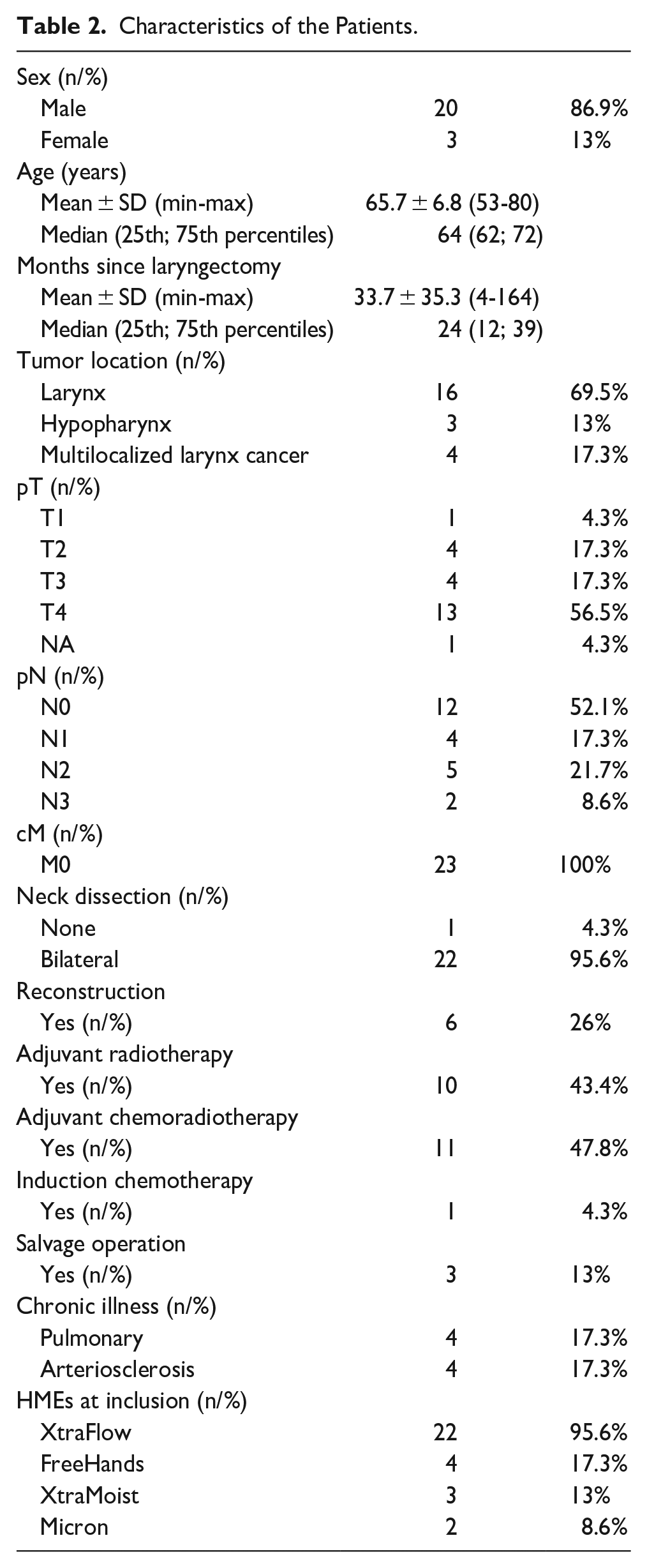
Except 2 individuals, all participants (n = 23) in our study had undergone TLE at our department, on average the TLE occurred before 33.7 ± 35.3 (4-164) months. The average age of patients was 65.7 ± 6.8 (53-80) years at the time of inclusion in the study, and the male gender represented 87% (n = 20). Reconstruction surgery was performed on 6 patients. In all, 21 patients received adjuvant therapy, 10 patients received radiotherapy, and 11 received adjuvant radiochemotherapy. Four patients had chronic lung disease. Two patients had COPD, 1 patient had bronchial asthma, and 1 patient had pulmonary Langerhans cell histiocytosis. The patient characteristics are shown in Table 2.
Characteristics of the Patients.
Pulmonary Rehabilitation
The results of the CASA-Q domains after 12 weeks using NG-HMEs are presented in Table 3.
Results of the Parameters of Pulmonary Rehabilitation.
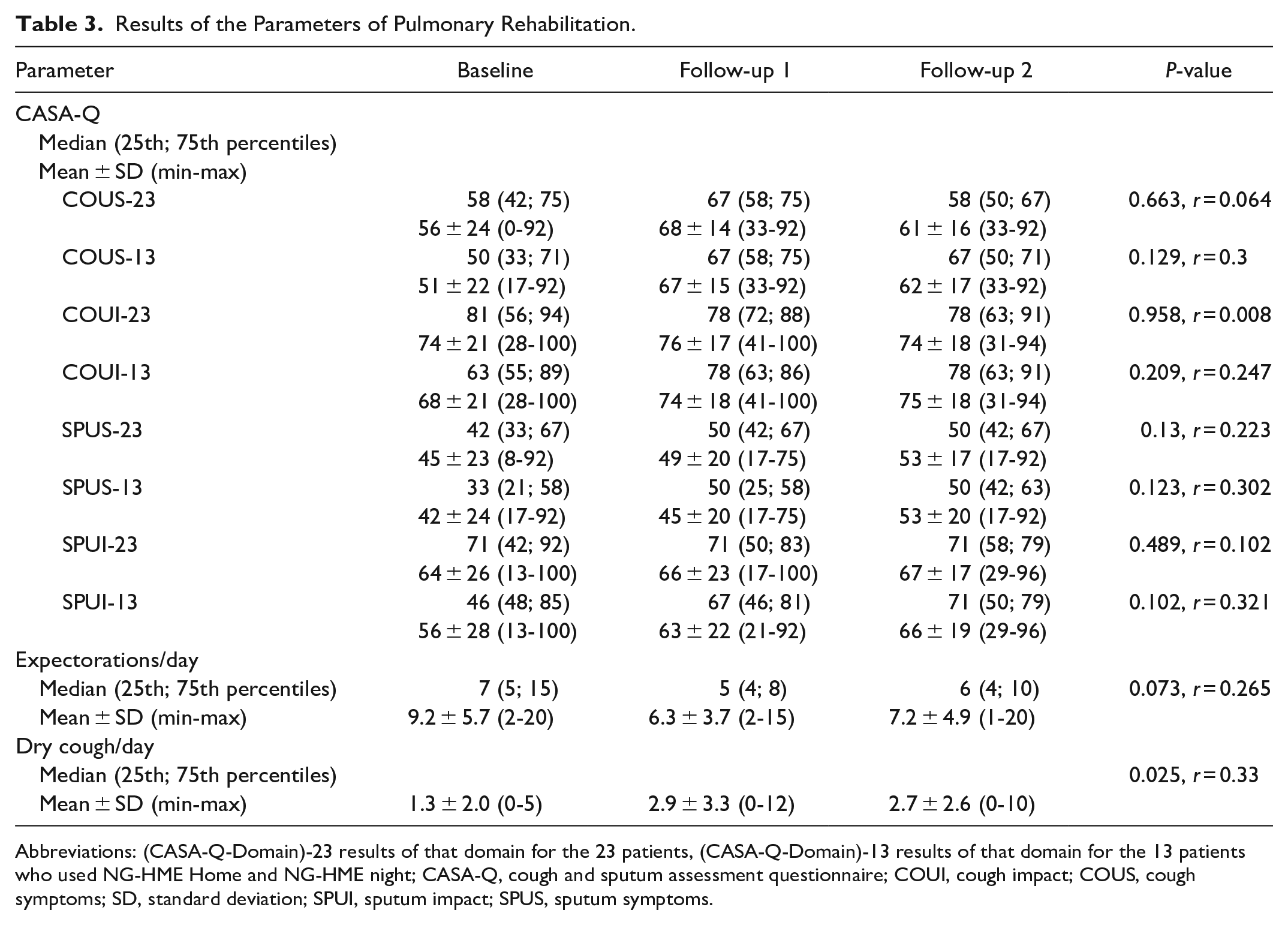
Abbreviations: (CASA-Q-Domain)-23 results of that domain for the 23 patients, (CASA-Q-Domain)-13 results of that domain for the 13 patients who used NG-HME Home and NG-HME night; CASA-Q, cough and sputum assessment questionnaire; COUI, cough impact; COUS, cough symptoms; SD, standard deviation; SPUI, sputum impact; SPUS, sputum symptoms.
A total of 13 (57%) of the 23 patients examined used NG-HMEs as instructed (NG-HME Home at home and NG-HME Night at night), resulting in an average duration of use of 23 ± 3.32 (12-24), 24 (24; 24) hours per day. These patients reported the following changes (Baseline vs follow-up 2) in the CASA-Q domains: COUS + 11 (P = .129, r = 0.3), COUI + 7 (P = .209, r = 0.247), SPUS + 11 (P = .123, r = 0.302), and SPUI + 10 (P = .102, r = 0.321). Mean values for the CASA-Q domains for these patients in comparison to the means of the entire sample are shown in Figure 2.
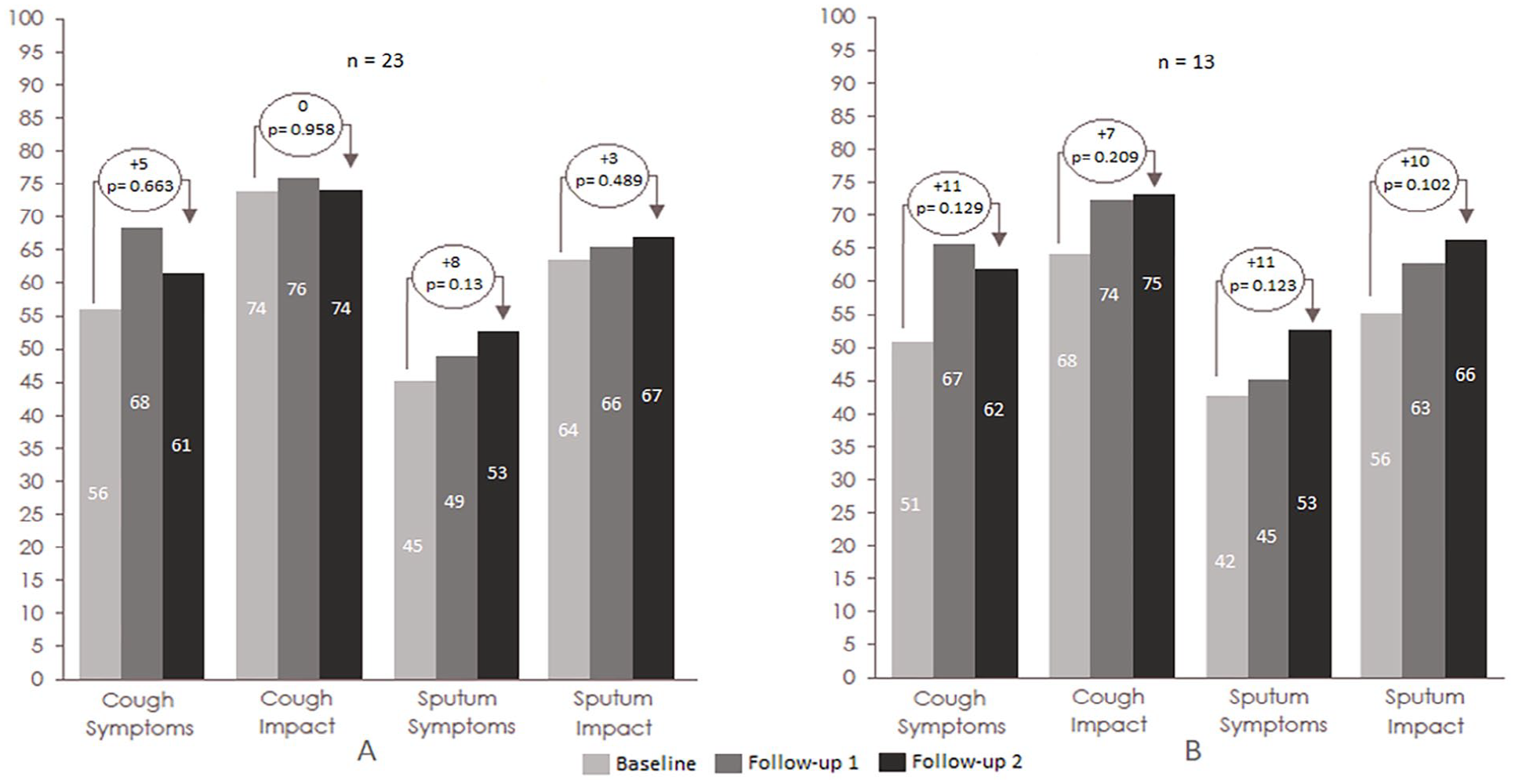
Mean values for CASA-Q domains: (A) for all patients and (B) for the patients who used NG-HME Home and NG-HME Night.
In all, 18 patients (78.2%) reported a usage duration of 24, 24 (24; 24) hours per day, which was equivalent to the duration of use at baseline. For this group, the CASA-Q domains changed as follows: COUS + 11 (P = .604, r = 0.087), COUI + 1 (P = .776, r = 0.017), SPUS + 9 (P = .106, r = 0.269), and SPUI + 2 (P = .798, r = 0.043).
In all, 17 patients (73.9%) used NG-HME Night every night. These patients reported the following changes: COUS + 11 (P = .138, r = 0.254), COUI + 2 (P = .712, r = 0.063), SPUS + 10 (P = .074, r = 0.307), and SPUI + 4 (P = .396, r = 0.146).
It should be noted that 6 patients (26%) reported an increase in COUS of ≥10.6, 5 patients (21.7%) reported an increase in COUI of ≥10.1, 9 patients (39.1%) reported an increase in SPUS of ≥9.5, and 9 patients (39.1%) reported an increase in SPUI of ≥7.6, which according to Rebelo et al. 31 defined as clinically important differences.
As shown in Table 3, the use of NG-HMEs for 12 weeks resulted in a decrease in the number of expectorations per day (P = .073, r = 0.265). However, there was an increase in the number of daily episodes of dry cough (P = .025, r = 0.33).
Quality of Life
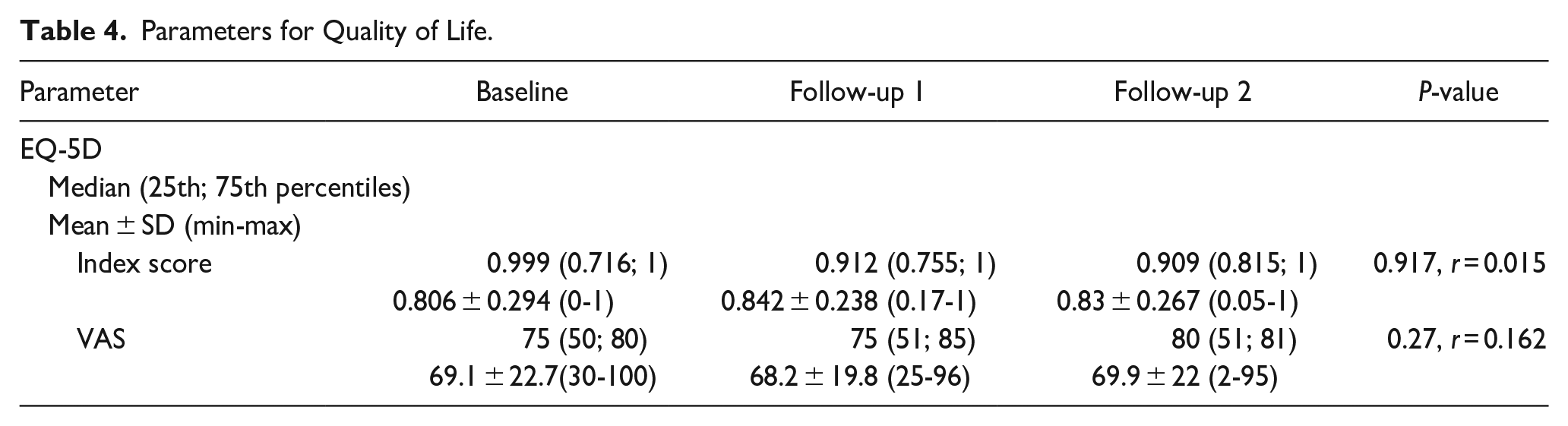
Table 4 shows that the use of NG-HMEs for 12 weeks did not result in any statistically significant changes in patient quality of life. The EQ-5D index score was 0.806 ± 0.294 at baseline and 0.830 ± 0.267 at follow-up 2 (P = .917). Similarly, the EQ-5D-VAS scores at baseline and follow-up 2 were not significantly different 69.1 ± 22.7 versus 69.9 ± 22, respectively (P = .27).
Parameters for Quality of Life.
Patient Usage Patterns and Satisfaction
After 12 weeks, the mean duration of NG-HME use decreased compared to previous HME use, with patients using them for an average of 21.87 ± 4.64 (12-24), 24 (24; 24) hours per day compared to 24, 24 (24; 24) hours per day at baseline (P = .034). Two patients (8.6%) reported difficulty selecting which NG-HME to use for which daily situation.
The number of HMEs used per day decreased significantly over time from baseline: 3 (2; 5) (1-20) to follow-up 2: 3 (2; 3) (1-6) HMEs per day.
The NG-HMEs Home, Go, and Night were the most frequently used by patients, with 56.5% (n = 13), 65.2% (n = 15), and 56.5% (n = 13), respectively, using them daily. In addition, 56.5% of the patients (n = 13) reported using the NG-HMEs as instructed for each situation.
The change reason of HMEs reported by the patients differed when comparing between baseline and follow-up 2 (P = .051). The number of patients who changed HME to adapt to daily situations increased from 2 (8.6%) at baseline to 11 (47.8%) at follow-up 2 with the NG-HMEs (P = .012). On the contrary, the number of patients who changed HME due to blockage with secretion decreased from 17 (73.9%) at baseline to 11 (47.8%) patients (P = .146).
Patient Satisfaction
Patient satisfaction with NG-HMEs was generally high. All patients reported a rating of 3 or more out of 5 (n = 23), with 34.7% of the patients (n = 8) rated their experience with the NG-HMEs as 5 out of 5, 47.8% (n = 11) rated it as 4 out of 5, and only 17.4% (n = 3) rated it as 3 out of 5. In all, 21 patients (91.3%) expressed their intention to continue using NG-HMEs beyond completion of the study.
Discussion
This study examines the effects of NG-HMEs on pulmonary rehabilitation among laryngectomized patients aiming to explore their effects in HME-experienced patients, who had reported high compliance in the use of HMEs. Regarding pulmonary rehabilitation, our data show that NG-HMEs produce a clinically significant change in pulmonary symptoms depending on the duration of usage and the situation-specific application of NG-HMEs. The results also demonstrate that the NG-HMEs have no impact on the patient’s quality of life. Furthermore, the use of NG-HMEs results in a lower number of daily changes in HMEs. This is particularly noteworthy because patients are instructed to adapt the use of NG-HMEs to their daily situations, rather than having to replace them routinely or due to blockage with secretions. In general, patient satisfaction is high among the studied patients, and the majority of patients would prefer to use the NG-HMEs in the future beyond the completion of the study.
The results of our study are relatively consistent with another cross-over study on NG-HMEs conducted by Longobardi et al. 28 In a cohort of patients comparable to ours, Longobardi et al. 28 reported clinically and statistically significant improvements in the parameters of pulmonary rehabilitation, mainly in the CASA-Q, as well as unchanged quality of life in both the EQ-5D index score and the EQ-5D-VAS after 6 weeks of use. Of particular importance is the fact that the duration of use of NG-HMEs in the Longobardi et al. 28 study was reported to be 23.8 ± 0.7 hours per day and that all patients used the NG-HMEs adapted to daily situations as instructed, that is, all patients used NG-HME Home during the day and NG-HME Night at night. Our study included 13 patients with relatively similar usage patterns to those of patients from Longobardi et al., 28 however, using the NG-HMEs for an average of 23 ± 3.32 (12-24), 24 (24; 24) hours per day. Those patients also reported more profound improvements in the mean scores of CASA-Q domains than the entire cohort of patients.
Interestingly, despite the positive impact reported of NG-HMEs, our patients reported a decrease in average daily use, which was lower than that reported by Longobardi et al., 28 who, unlike our study, contacted the patients by phone once a week. One possible explanation for the decrease in usage may be attributed to the fact that the XtraFlow HME, which has a low breathing resistance, 32 was the most used HME before the change to NG-HMEs. Generally, laryngectomized patients tend to prefer HMEs with lower breathing resistance.29,33 In our study, the patients also preferred the NG-HMEs with the lowest breathing resistance.
This highlights the importance of consistent use of NG-HMEs and their potential to improve pulmonary rehabilitation, even in HME-adherent laryngectomized patients.
It should be noted that the patients studied here reported an increase in the number of episodes of dry cough. Macri et al. 34 reported that patients may experience increased cough episodes when they start using HMEs. The change to NG-HMEs may have contributed to this observed effect in combination with the predominantly used HME, which has the lowest breathing resistance and the lowest humidification capacity, leading to increased tracheal irritation. Interestingly, these results did not negatively impact patient satisfaction with NG-HMEs.
Cost-effectiveness and cost savings also play an important role in acceptance and application, which must be considered on a country-specific basis according to the respective healthcare system. For patients and insurance providers in Germany, the cost of NG-HMEs is similar to that of previously used HMEs.
Our study has several limitations. First, there is a lack of randomization and blinding of patients with respect to the HMEs used. Second, patient education and instruction on the proper use of NG-HMEs occurred only at the beginning of the study, and there was no regular short-term follow-up to check if patients applied NG-HMEs as instructed. Therefore, there may have been a deficit in patient information during the study. In addition, the questionnaires utilized in our study may have limitations. While some of the questionnaires are validated, they were developed to evaluate and follow pulmonary complaints and their impact on daily life in COPD patients; therefore, they are not specific for laryngectomized patients. Other questionnaires were developed and modified by researchers such as Hilgers et al. 27 for the specific reason of investigating the effects of HMEs on pulmonary rehabilitation after TLE. However, this limitation is common in the literature on this topic.
It should be emphasized that the study was limited to adherent patients who have previous experience with HMEs and have reported effective pulmonary care and a high quality of life at the time of inclusion in the study. Clinically significant results, as per Rebelo et al., 31 testify to the potential of the NG-HMEs to improve the pulmonary situation of laryngectomized patients.
In this context, prospective multicenter studies with a long follow-up period with an increased number of evaluated patients are required, in combination with regular and consistent patient education and follow-up to fully assess the value and usefulness of the NG-HMEs and thus increase the validity and evidence of the results presented.
Conclusion
The results show that NG-HMEs could have a clinically significant positive effect on pulmonary rehabilitation after TLE. Our results do not show a decline in quality of life after using the NG-HMEs. The possible effects of NG-HMEs require further evaluation in long-term studies with larger cohorts of patients to fully assess their efficacy.
Footnotes
Acknowledgements
The present work was performed in (partial) fulfillment of the requirements for obtaining the degree “Dr. med.”
Data Availability
The utilized questionnaires and data that support the findings of this study are available from the corresponding author, Omar Almajali, upon reasonable request.
Remarks
This study was conducted using Provox® Life™ HMEs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this research was obtained from the ethics committee of the University of Erlangen-Nuremberg (244_21 B).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the ethics committee of the University of Erlangen-Nuremberg (244_21 B) approved protocols and in accordance with the principles of the Declaration of Helsinki.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
