Abstract
Introduction
Endoscopic sinus surgery (ESS) is the most common and effective surgical method for the clinical treatment of nasal diseases.1,2 Ensuring optimal visualization of the surgical field poses a common challenge for both anesthesiologists and surgeons. While anesthesiologists aim to minimize intraoperative blood loss through hypotensive anesthesia while maintaining stable cardiovascular and respiratory functions, surgeons employ various techniques, such as intranasal application of topical vasoconstrictors, to limit bleeding and improve visibility.3,4 Epinephrine, a widely used vasoconstrictor in ESS, is often infiltrated into cotton pledgets at high concentrations (1:1000) for intraoperative hemostasis. Studies have shown that this improves surgical field visualization.5,6 However, low concentrations of epinephrine are often injected submucosally to reduce the amount of bleeding in the surgical field, and the most popular injected epinephrine concentration is 1:100,000 (68 respondents, 71%), followed by 1:200,000 (16 respondents, 17%). 7 Ahmed et al showed that injectable epinephrine at either 1:200,000 or 1:100,000 could achieve more vasoconstriction and less bleeding without increasing hemodynamic effects. 8 Infiltration of the pterygopalatine fossa with low-concentration epinephrine is an effective technique for reducing surgical field bleeding during ESS. 9 A meta-analysis demonstrated that greater palatine canal injections of local anesthesia with 1:80,000 epinephrine for ESS effectively reduced intraoperative bleeding. Additionally, this procedure did not show significant adverse effects. 10 In fact, it has been shown that local infiltration of low concentrations of epinephrine (1:400,000) can also cause temporary significant hemodynamic changes. 11 However, Valdes et al suggested that nasal submucosal injection of low-concentration epinephrine during ESS did not affect the quality of surgical field visualization. 12 This leads us to question whether nasal submucosal injection of low-concentration epinephrine can exert a good hemostatic effect in ESS. At present, there is no medical evidence-based to resolve this disagreement. Therefore, this article focuses on low concentrations of epinephrine to systematically review the literature on the clinical effects of nasal submucosal injection of epinephrine and to determine whether it can provide surgeons with good visibility during sinus surgery.
Methods
This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines and carried out the study based on a prospective protocol (PROSPERO CRD42023439122) according to the recommendations of the Cochrane Collaboration.
Information Sources and Search Strategy
We conducted a systematic literature search of PubMed, Scopus, Cochrane Library, Web of science, China National Knowledge Infrastructure, and Wanfang Database without language restriction for studies including combined key terms and expanded Medical Subject Heading (MeSH) of the terms topical. In addition to online searches, we also identified studies through manual searches to ensure the complete inclusion of eligible articles. References were uploaded to EndNote (Clarivate Analytics) and filtered for relevance. We completed the search on January 10, 2023. Taking PubMed as an example, the specific search strategy is shown in Table 1.
Search Strategy.

Abbreviations: MeSH, Medical Subject Heading.
Inclusion and Exclusion Criteria
The inclusion criteria used for study selection were the following:
(1) Population: patients undergoing endoscopic nasal surgery, American Society of Anesthesiologists I or II, without cardiovascular, and other basic diseases.
(2) Intervention: intranasal application of the surgical site with low-concentration epinephrine, with or without local anesthetics such as lidocaine. The low concentrations of epinephrine were defined as concentrations ranging from 1:200,000 to 1:80,000. 8 If epinephrine was administered both by irrigation or soaking cotton balls and by injection at the same time in the study, the effect of injection was considered to be predominant.
(3) Comparison: local anesthetics or saline without epinephrine was used as a control group.
(4) Outcome: for eligibility, a study had to report on at least one of the following parameters: the primary outcomes included surgical field visual bleeding grade and total bleeding loss, the secondary outcomes included heart rate and mean arterial pressure (MAP).
(5) Study design: The study design was required to be a randomized controlled trial (RCT) or a crossover study with a randomized, double-blind design.
The exclusion criteria were defined as follows:
(1) Studies that did not meet the inclusion criteria;
(2) Repetitively published articles;
(3) Studies applied to animals; and
(4) Case report, reviews, meta-analysis, or correspondence.
Study Selection
Two reviewers separately screened all articles, any disagreements were resolved through negotiation. They screened all studies based on titles and abstracts and excluded references that obviously did not adhere to our inclusion criteria. The full reports were selected for final inclusion by 2 reviewers.
Data Extraction and Management
Two reviewers independently extracted the details of study population, intervention, and outcomes. The data extraction tables included the following items:
(1) General information: author, publication year, study design, age, sex, locally infiltrated solution components, and type of surgery.
(2) Information on main results: visual bleeding grade in the surgical field (a 6-subscale to estimate the intensity of wild bleeding in a single operation), total bleeding loss, heart rate, and MAP.
Risk of Bias in Individual Studies
For each included study, 2 authors independently reviewed the risk of bias, and any disagreements were resolved by discussion between these authors. According to the Cochrane Handbook, the Cochrane Risk of Bias Tool was used to assess the risk of bias assessment of included RCTs in 7 ways, including: (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of outcome assessment, (5) incomplete outcome data, (6) selective reporting, and (7) other bias. Review Manager (RevMan version 5.3; Cochrane Collaboration) is used to show the risk of bias of individual studies. Authors’ assessments about the risk of bias manifested as percentages across the entire body of included studies. Red, yellow, and green indicate high, unclear, and low risk of bias, respectively. A trial would be considered “low risk” of bias if all 7 domains were judged as “low risk,” “moderate risk” if any of the 7 items were judged as “unclear risk,” or “high risk” if any of the items were judged as “high risk.”
Statistical Analyzes
Meta-analyses were performed using Review Manager (RevMan version 5.3; the Cochrane Collaboration). Data not directly supplied from the standard deviation were estimated using the range and sample size. The heterogeneity among the included studies was quantified by calculating I2 statistic. P < 0.05 or I2 ≥ 50% were considered statistically significant. When heterogeneity is significant, the random effects model was used to pool data, otherwise fixed effects model will be used. Continuous variable data was expressed as mean difference (MD) and 95% confidence interval (CI), and count data were expressed as odds ratio (OR) and 95% CI. We used funnel plots to visually assess the risk of publication bias.
Results
Literature Search and Trial Selection
A total of 692 studies were identified by database search, of which 54 studies were eligible for the full text search. In total, 11 studies met the eligibility criteria and were included for analysis. Figure 1 shows the flow chart of article inclusion and exclusion.
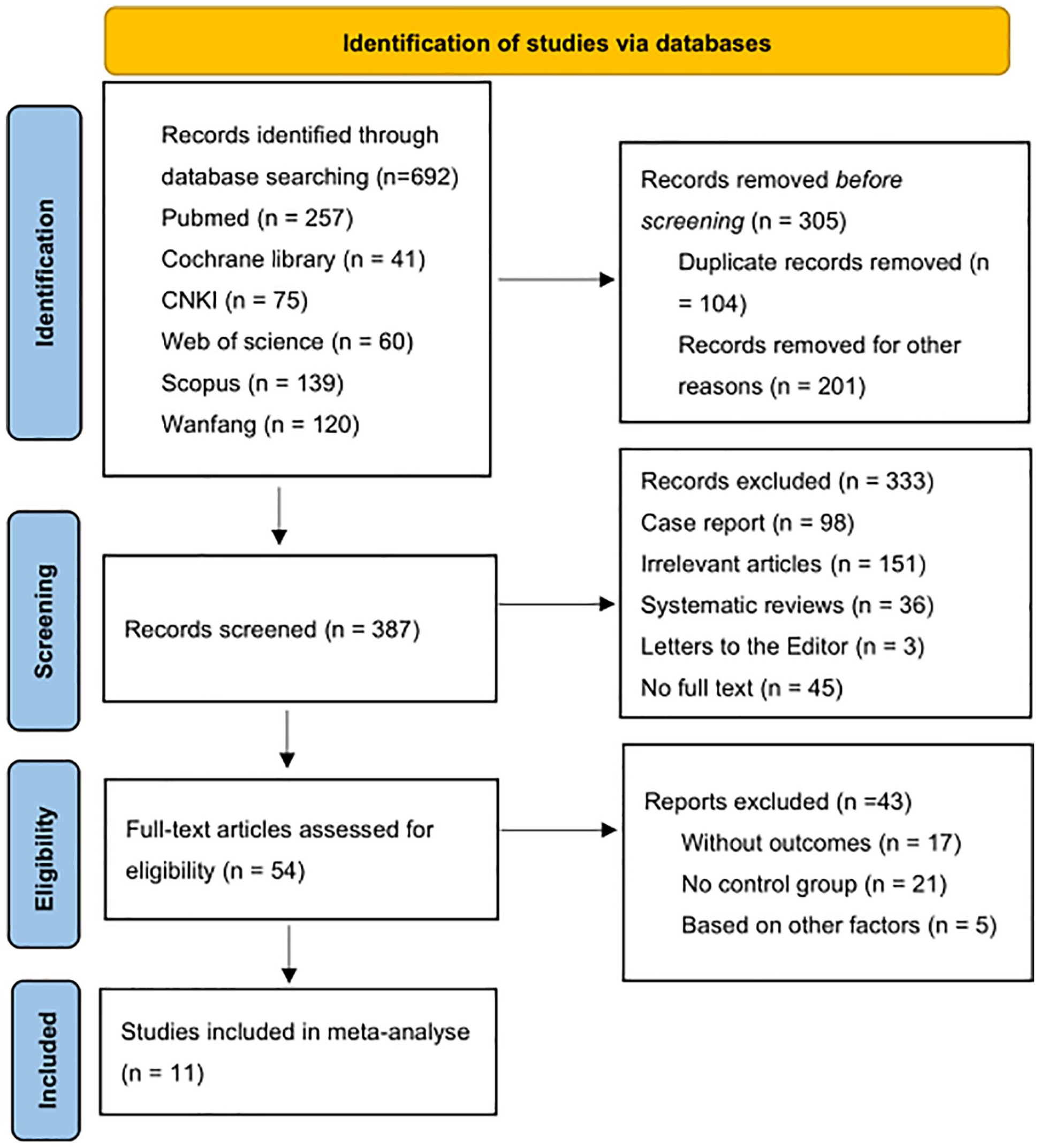
Study flow diagram.
Trial Characteristics
A final number of 11 included articles were used in the meta-analysis, which were all RCTs. In terms of the epinephrine technique, all studies used submucosal injection. The local infiltration of designated sites such as nasal septum, middle meatus, inferior turbinate, and sphenopalatine foramen was performed under general anesthesia before surgery. The volume varied from 2 ml to 10 ml, and the injection time was controlled at 15 to 30 s. The studies were generally small, with a sample size of 10 to 45, and the proportion of males was 43%. Additional information, including the solution composition of the epinephrine group and the control group and the type of surgery were detailed in Table 2.
Characteristics of Included Studies.
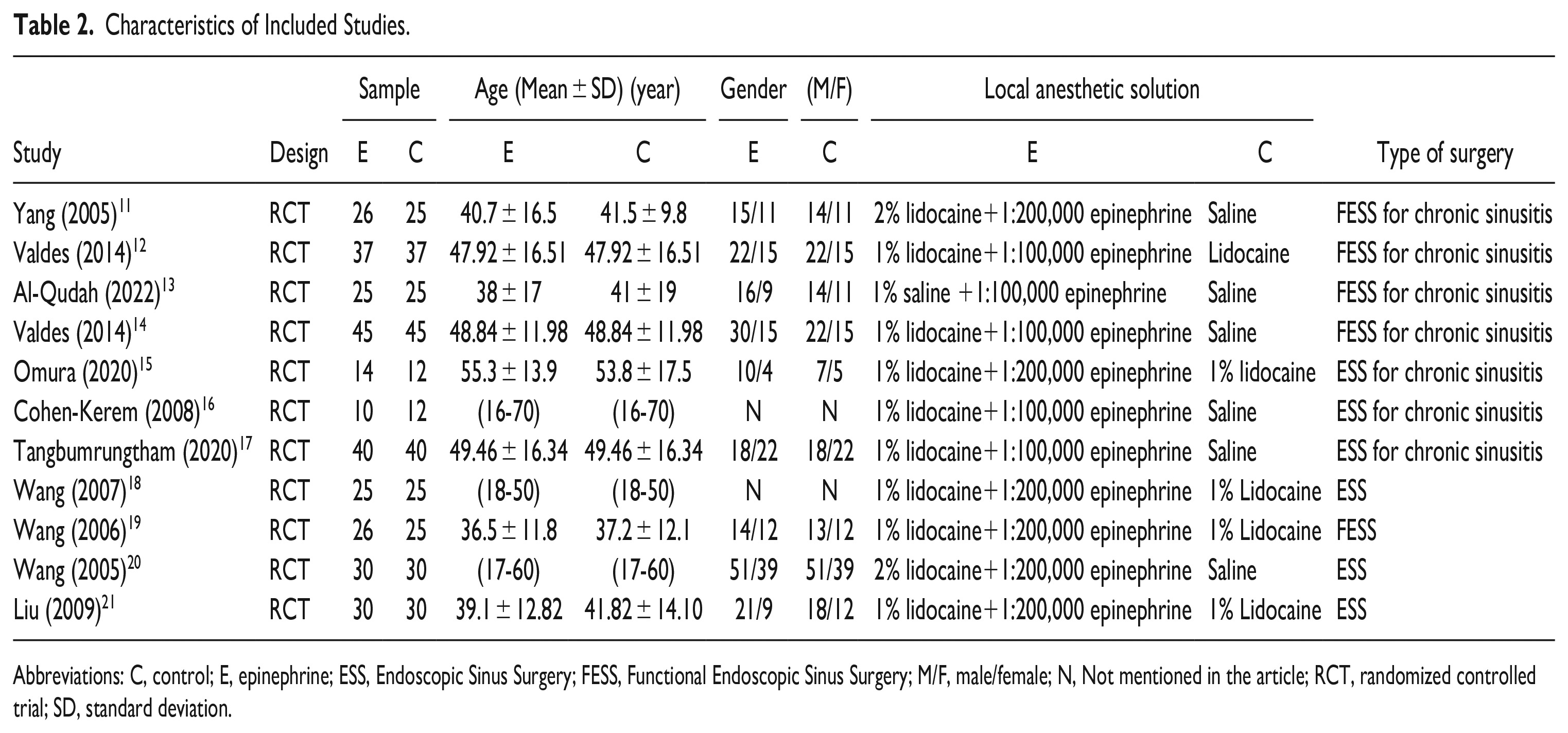
Abbreviations: C, control; E, epinephrine; ESS, Endoscopic Sinus Surgery; FESS, Functional Endoscopic Sinus Surgery; M/F, male/female; N, Not mentioned in the article; RCT, randomized controlled trial; SD, standard deviation.
Risk of Bias
The quality of methodology for the included studies was based on the Cochrane Risk of Bias Tool. The summary of the authors’ judgments about each domain of risk of bias is shown in Figure 2A. Among the included studies, 4 had a low risk of bias, 4 had a moderate risk of bias, and 3 had a high risk of bias. The results of the sensitivity analysis showed that the combined effect value of the meta-analysis did not change significantly, indicating that the results of the meta-analysis were relatively stable, and the bias caused by a single study was small. A funnel plot was used to analyze the publication bias and the results showed that the distribution of the funnel plot was basically symmetric (Figure 2B), which means little publication bias.
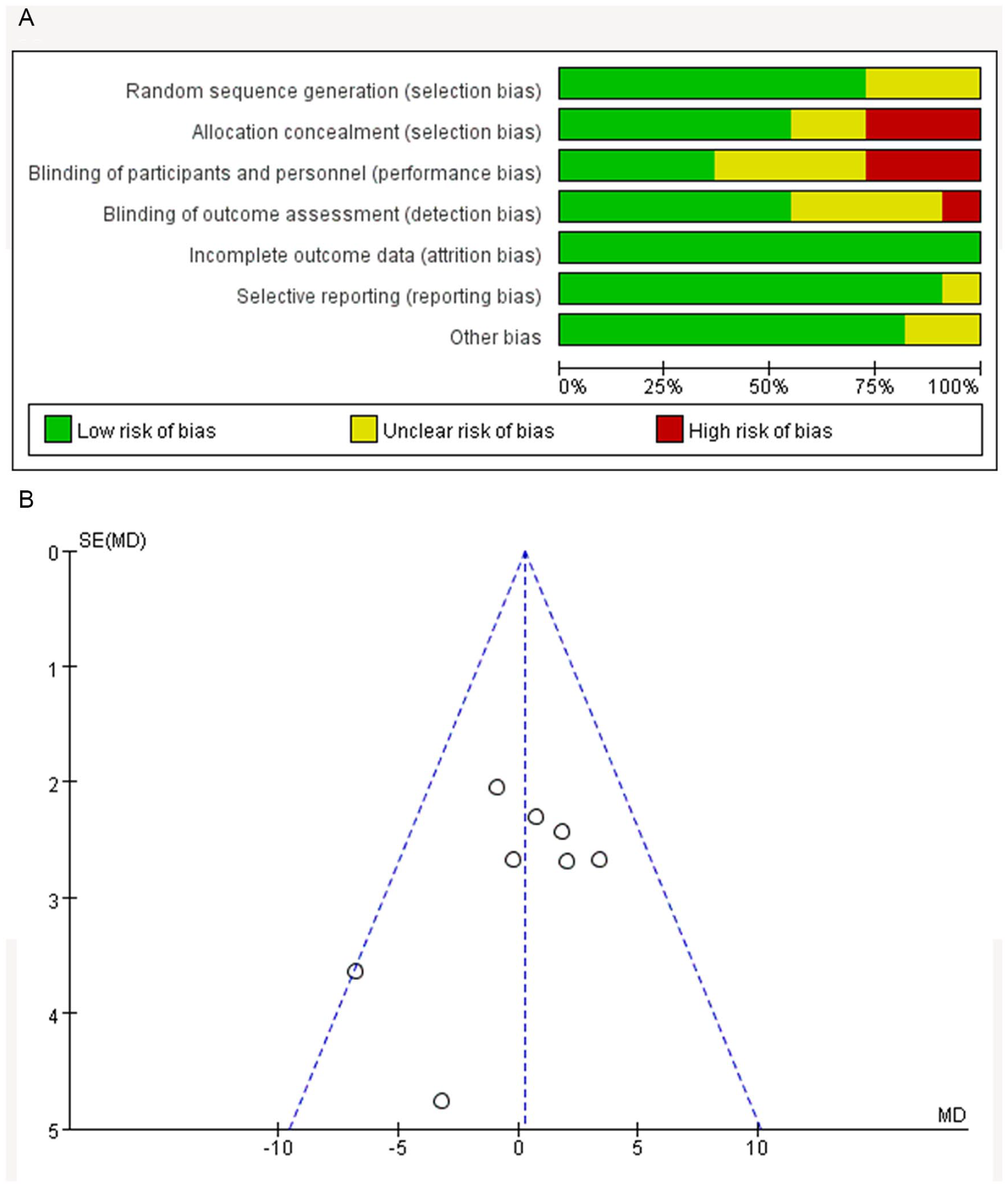
(A) Risk of bias: review authors’ judgments about each risk of bias item presented as percentages across all included studies. Green, low risk of bias; yellow, unclear risk of bias; and red, high risk of bias. (B) Funnel plot to assess the publication bias.
Surgical Field Visual Bleeding Grade
Five studies (n = 239, 121 in the epinephrine group and 118 in the control group) reported the grade of visual bleeding in the surgical field. There was low heterogeneity between studies (P = 0.24, I2 = 27%). The outcome of the meta-analysis did not show statistical significance between the 2 groups (MD −0.19; 95% CI −0.40 to 0.01; P = 0.07, Figure 3A) in the surgical field visual bleeding grade.
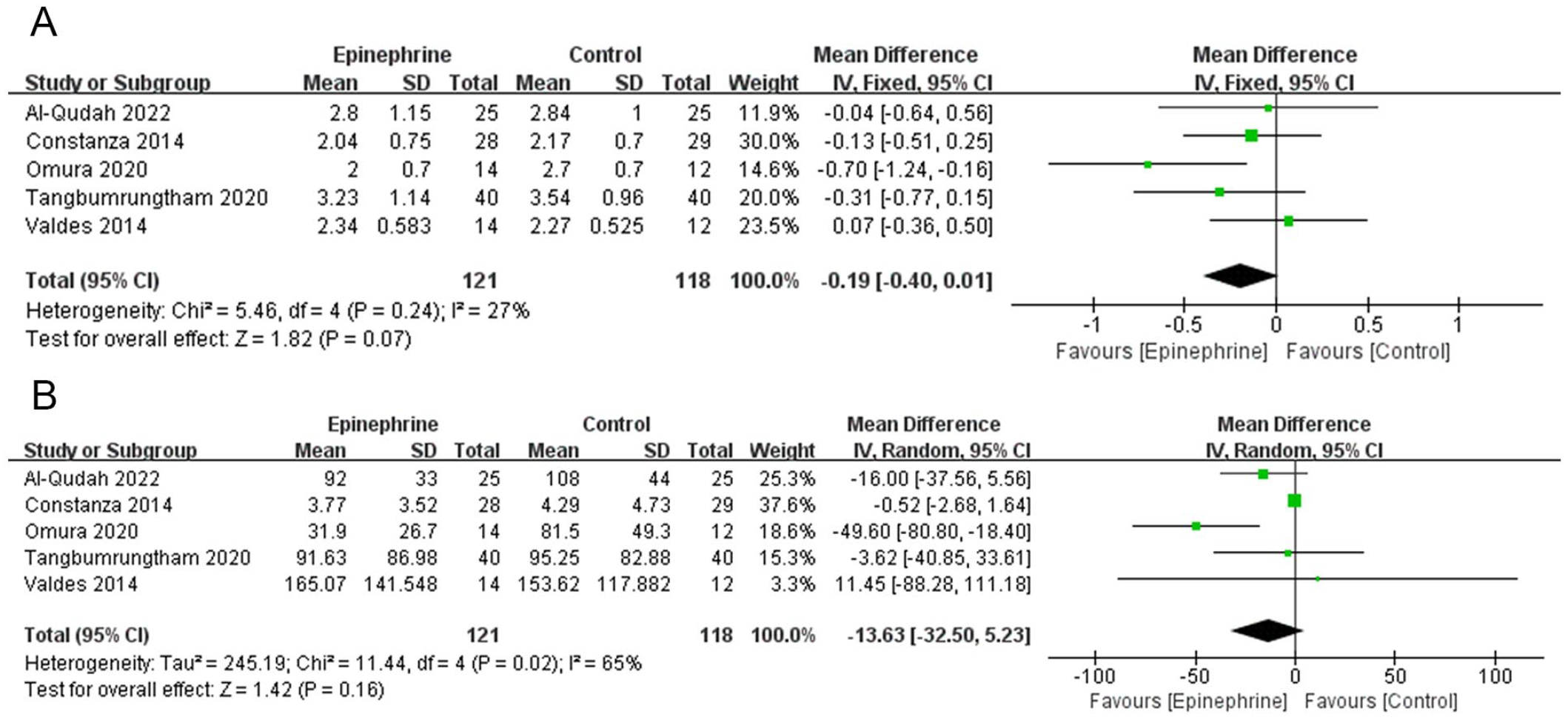
(A) Mean difference of surgical field visual bleeding grade between the epinephrine group and the control group. (B) Mean difference of total blood loss between the epinephrine group and the control group. CI, confidence interval; IV, inverse variance; SD, standard deviation.
Total Blood Loss
Five studies (n = 239, 121 in the epinephrine group and 118 in the control group) compared total blood loss between the 2 groups. Significant heterogeneity was calculated (P = 0.02, I2 = 65%), and the random effects model was used to combine the data. Additionally, there was no statistical significance between the 2 groups (MD −13.63; 95% CI −32.5 to 5.23; P = 0.16, Figure 3B) in total blood loss.
Heart Rate
Eight studies (n = 370, 186 in the epinephrine group and 184 in the control group) compared the preoperative baseline heart rate. We balanced the heterogeneity between studies using a randomized model (P = 0.03, I2 = 54%). The results showed no statistical differences in baseline heart rate between the 2 groups (MD −0.84; 95% CI −3.78 to 2.10; P = 0.58, Figure 4A). Nine studies (n = 427, 214 in the epinephrine group and 213 in the control group) compared intraoperative heart rate between the 2 groups. There was a high heterogeneity between the studies (P < 0.01, I2 = 92%) and the random effects model was used to combine the data. In comparison with the control group, the epinephrine group did not show statistical differences in intraoperative heart rate (MD 2.62; 95% CI −2.01 to 7.26; P = 0.27, Figure 4B).
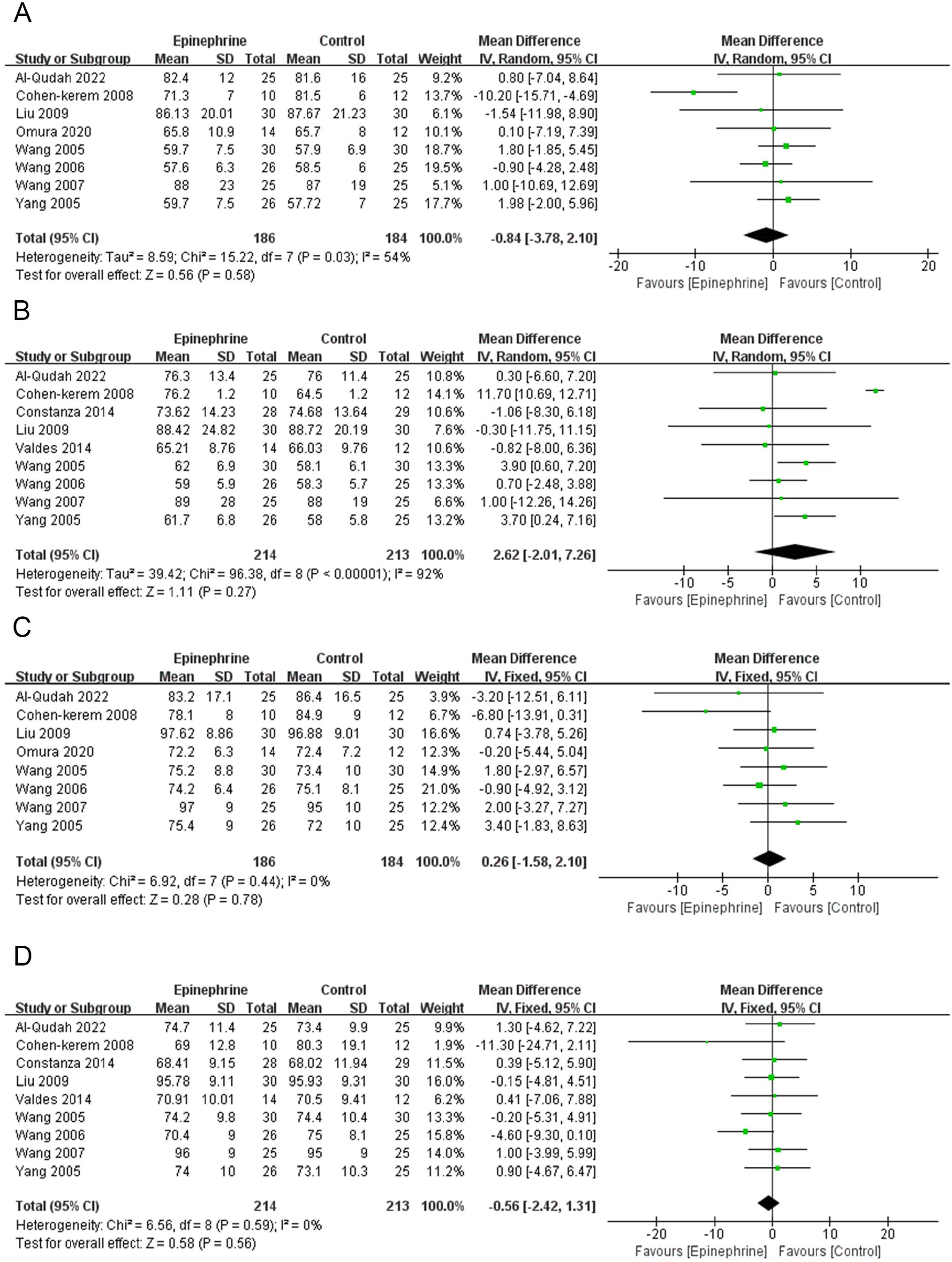
(A) Mean difference of baseline heart rate between the epinephrine group and the control group. (B) Mean difference of intraoperative heart rate between the epinephrine group and the control group. (C) Mean difference of baseline MAP between the epinephrine group and the control group. (D) Mean difference of intraoperative MAP between the 2 groups.
Map
Eight studies (n = 370, 186 in the epinephrine group and 184 in the control group) compared the preoperative baseline MAP. There was low heterogeneity between studies (P = 0.44, I2 = 0%). In comparison with the control group, the epinephrine group did not show statistical differences in baseline MAP (MD 0.26; 95% CI −1.58 to 2.10; P = 0.78, Figure 4C). Nine studies (n = 427, 214 in the epinephrine group and 213 in the control group) compared intraoperative MAP between the 2 groups and did not show no statistical significance (MD −0.56; 95% CI −2.42 to 1.31; P = 0.56). There was also low heterogeneity between the studies (P = 0.59, I2 = 0%, Figure 4D).
Discussion
Effective endoscopic surgery relies on adequate identification of anatomical structures, and even a small amount of mucosal exudation can limit observation. Furthermore, given that mucosal bleeding originating from the sinus anatomy can result in a substantial volume of hemorrhage within a brief timeframe, prompt hemostasis is imperative to ensure the overall well-being and safety of the patient. 22 Therefore, epinephrine combined with local anesthetics is widely used by otolaryngologists to perform local application at the surgical site. This is because epinephrine can directly activate α-receptors on the vascular wall, inducing local vasoconstriction and reducing intraoperative blood loss, thereby promoting smooth progress of endoscopic surgery. At present, there is no standard operating procedure for vasoconstrictors, and several concentrations of epinephrine are administered, ranging from 1:200,000 to 1:1,000. 23 Studies have shown that concentrations of 1:200,000 to 1:80,000 epinephrine are appropriate and safe enough to excite α-receptors in blood vessel walls to constrict blood vessels while avoiding the risk of cardiac risks caused by the absorption of high concentrations of epinephrine into the blood.8,24 Additionally, the observation period for secondary parameters such as heart rate and MAP in this study was limited to only 5 min. Although the outcome parameters obtained in Shenoy’s study were measured at intervals of 15 min after a single injection of infiltration, they also believed that the effect of epinephrine lasted 2.5 h. 9 In fact, the half-life of locally infiltrated epinephrine in the nasal cavity has not been clearly reported, but it is known that the half-life of epinephrine by plasma infusion is only 5 or 10 min. 25 In addition, all the articles included in this study reported hemodynamic parameters measured in a short period of time after intranasal epinephrine. Therefore, a consequence of this unknown factor is a limitation of this study. These significant differences can be determined in the future with larger sample sizes and additional clinical trials.
There were no statistically significant differences between the 2 groups in the studied parameters: total blood loss, heart rate, and MAP. These results indicate that there is not enough evidence that intranasal administration of epinephrine affects hemodynamic fluctuations and total blood loss during surgery, at least at concentrations of 1:200,000 to 1:100,000. In fact, we considered in the analysis of the results that the bleeding grade of the visual field appeared to be significant when the low-concentration epinephrine administered by submucosal injection was compared with the control group. These significant differences can be determined in the future with larger sample sizes and additional clinical trials. In addition, there are many factors that influence the visual score of the surgical field, including tissue edema, mucosal hypertrophy, and polypoid lesions, which can reduce the free space in the nasal cavity, and a small amount of blood that can also limit the visual field and make the quality of the surgical field worse.26,27 Even more important, the measurement of this result comes from the subjective feeling of the surgeon. The included studies used the Boezaart Scale to evaluate the cleanliness of the surgical field. Although there may be various subjective factors, we firmly believe that the credibility of our results will remain intact. This is due to the fact that our scale has demonstrated a remarkable inter-rater reliability of 0.74 and an intra-rater reliability of 0.83. 28
Increased MAP is considered to be a key factor leading to increased surgical blood loss, so the application of controlled antihypertensive techniques is believed to reduce bleeding in the surgical field. 29 However, several studies have shown little correlation between MAP and surgical blood loss.30,31 Wormald et al 32 suggested that lowering MAP did not always improve the surgical field. This may be a side effect of drug use, such as rebound hypertension, reflex tachycardia, vasodilation, and myocardial depression. There are still some controversies on whether there is a correlation between MAP and blood loss in the surgical field. Furthermore, for the purposes of this study, those with cardiovascular comorbidities and unstable hypertension were excluded. However, in clinical practice, these patients still undergo surgery, so these results can only be temporarily extrapolated to patients without these comorbidities.
Limitations
This study has some limitations. First, the number of subjects included in this study is small and there is a lack of sufficiently powerful comparative studies. This is followed by potential confounders such as the effect of general anesthetics, the amount of epinephrine used and absorbed, the history of vasopressor use, and injection of concomitant local anesthetics. In addition, there is also heterogeneity between surgeons and institutions in the way surgery is performed and surgical field is evaluated, the types of surgeries performed, and the actual way epinephrine was administered probably differed between different studies, such as where epinephrine was injected into the nasal cavity, how much epinephrine was used for irrigation and so on. Additional studies may be necessary to investigate whether lidocaine is a confounding variable when assessing the hemostatic effects of intranasal epinephrine.
Conclusions
There is not enough evidence to support that nasal submucosal application of low-concentration epinephrine is more effective than non-administration in improving surgical field cleanliness and reducing overall blood loss. More research is needed to strengthen the evidence. It is suggested that the concentration of local epinephrine can be reasonably increased under the assumption of ensuring safety. These studies should be clearly reported with adequate sample size, low risk of bias, and provide data in a format that enables meta-analysis.
Footnotes
Authors’ Note
This study was a meta-analysis and did not involve with human participants performed by any of the authors.
Author Contributions
Data Availability
The research data used to support the findings of this study are included in the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethic/Consent Statement
All analyses were made based on previously published studies; therefore, no ethical approval or patient consent was required.
