Abstract
Objectives:
The aim of this study is to determine whether thrombin time (TT) could be used as diagnostic biomarkers and predict the prognosis for sudden sensorineural hearing loss (SSNHL).
Methods:
Sixty-one patients diagnosed with SSNHL and 65 people who underwent physical examination were recruited. Data on the patient’s background, clinical course, and laboratory findings were collected. SSNHL patients were divided into the effective and ineffective groups according to the hearing recovery from the treatment and were assessed by binary logistic regression. Receiver-operating characteristic (ROC) analysis was carried out for the best discriminating cutoff value of the biomarker with the corresponding sensitivity and specificity was calculated.
Results:
The SSNHL group exhibited prolonged TT (19.11 ± 1.12 seconds) compared to the control group (17.58 ± 2.18 seconds, P < .001). Binary logistic regression analysis found a significant positive association between TT and SSNHL and was observed with an odds ratio (OR) 1.769 [95% confidence interval (CI) 1.344-2.330, P < .001] in the unadjusted model. Even after adjustment using the variables included in the multivariate models, TT was significantly predictive of SSNHL. A TT cutoff value of 17.65 seconds provides optimal separation between patients with SSNHL and controls in the ROC analysis [Area Under the Curve (AUC) 0.773, 95% CI 0.689-0.856; sensitivity, 0.918; and specificity, 0.569]. TT in the effective group of SSNHL patients was shorter (18.76 ± 1.06 seconds) than that in the ineffective group (19.43 ± 1.09 seconds, P = .018). The cutoff value of TT as progress predictors was 19.85 seconds. The TT < 19.85 seconds showed an effective rate 59.09% (26/44) higher than 17.65% (3/17) of TT ≥ 19.85 seconds.
Conclusions:
TT is a potential biomarker of SSNHL and is independently associated with the prognosis of patients with SSNHL.
Keywords
Introduction
Sudden sensorineural hearing loss (SSNHL) is characterized by rapid loss of hearing (3 days), almost always unilateral, that presents with varying levels of severity from mild to profound. The incidence of sudden deafness is 5 to 27/100,000/year, and there are 5% to 30% of patients who cannot recover hearing after treatment,1,2 bringing serious trouble to the patient’s life. The common clinical manifestations of SSNHL are hearing loss, tinnitus, fullness, and may accompany dizziness or vertigo. The researchers summarized that the etiology of SSNHL may be bacterial or viral infection, vascular lesions, and vestibular disfunction at present. 2 The SSNHL treatments include systemic steroid, 1 intratympanic steroid administration, 3 hyperbaric oxygen therapy, 4 vasodilators, 5 anticoagulants combined with systemic steroid therapy. 6
The prognosis of hearing recovery from SSNHL depends on many factors, including the severity of hearing loss, age, presence of vertigo, and the shape of audiogram. 1 A report suggested that the time from onset to treatment is an important factor that affects the prognosis of patients with refractory SSNHL, 7 and there are studies that have shown that metabolic syndrome 8 can affect the prognosis of SSNHL. Retrospective studies have also found that hematologic parameters, such as the neutrophil/lymphocyte ratio and the platelet/lymphocyte ratio were associated with prognosis of SSNHL. 9
The timely diagnosis and treatment of SSNHL will result in a better prognosis. 10 Therefore, it is particularly important to find an easily accessible indicator for the diagnosis of SSNHL. The convention coagulation test (CCT) was found to be disordered in many diseases. It can be used to predict disease and prognosis, such as in COVID-19 infection, 11 polycystic ovary syndrome, 12 breast cancer. 13 Studies about coagulation dysfunction in SSNHL were mostly focused on serum fibrinogen, 14 whether the other coagulation function could be a diagnostic and prognostic biomarker of SSNHL is still controversial.15,16
In this retrospective study, we balanced the main factors that affect the prognosis between SSNHL and the control, explored in the CCT of prothrombin time, activated partial thromboplastin (APTT), international normalized ratio (INR), thrombin time (TT), and fibrinogen (FIB) as a diagnostic and prognostic indicator of SSNHL with the statistical analysis method.
Patients and Methods
Participants
SSNHL refers to the sudden sensorineural hearing loss of unknown etiology within 72 hours with at least 3 consecutive frequency lost 20 dB. 1 Sixty-one patients diagnosed as SSNHL were enrolled from January 2018 to December 2021. All patients aged 18 to 79 years had no history of familial deafness, cancer, or previous sudden deafness. All patients took CCT before therapy. The day interval from the onset of the symptom to the start of treatment was within 20 days. We excluded patients who were pregnant and those taking an anticoagulant drug, as well as those with blood disease, cancer, Ménière’s disease, herpes zoster oticus, or any other known cause of hearing loss disease. At the same time, 65 adults with no SSNHL who took CCT on physical examination were enrolled as a control. The exclusion criteria were the same as those of the SSNHL group.
Data Collection
Detailed medical history was obtained from all participants, including details of baseline characteristics (age, sex, hypertension, diabetes, hypertriglyceridemia, hypercholesterolemia, and autoimmune disease), clinical characteristics [affected side, accompanying symptoms (such as tinnitus, vertigo, ear fullness, headache, dizziness), time to treatment and hearing level at admission, frequency classification], and treatment regimen (steroids, batroxobin or vasodilators with or without physiotherapy, and tympanic injection of steroids).
Clinical and Other Measurements
Hearing loss was determined by pure-tone audiometry (Conera, Denmark) before and after treatment. Pure-tone average (PTA) was measured as the average of thresholds (dB HL) at 7 frequencies of 0.125, 0.25, 0.5, 1, 2, 4, and 8 kHz. According to hearing curves, patients were classified into a low-frequency group (frequencies ≤ 1 kHz, average threshold ≥ 20 dB HL at 0.25 and 0.5 kHz at least), a high-frequency group (frequencies ≥ 2 kHz, average threshold ≥ 20 dB HL at 4 and 8 kHz at least), a flat-type group (all frequencies, average threshold ≤ 80 dB HL at 0.25, 0.5, 1, 2, 4, and 8 kHz at least), or a total deafness group (all frequencies, average threshold > 80 dB HL at 0.25, 0.5, 1, 2, 4, and 8 kHz at least). 17 Based on the audiogram, PTA classified the severity of hearing loss as mild (26-40 dB), moderate (41-55 dB), moderately severe (56-70 dB), severe (71-90 dB), or profound (>90 dB). The patients were divided into mild and moderate degrees, moderately severe and severe degrees, or profound degrees. Siegel’s criteria 18 were used to assess the hearing recovery according to the hearing gain average at all the frequencies (0.125, 0.25, 0.5, 1, 2, 4, and 8 kHz).
The parameters of the coagulation function were measured by an automatic coagulation analyzer (Sysmex CS5100 or CS2000i, Sysmex, Kobe, Japan) before therapy.
Treatment and Evaluation
The basic principles of treatment were according to the Chinese guideline of SSHNL. 17 The dosage of the medication was adjusted based on the drug label and the patient’s underlying condition. Patients underwent comprehensive treatment, including treatment with steroids and vasodilators, with or without physiotherapy or tympanic injection of glucocorticoids. All patients were treated with systemic glucocorticoid (prednisone 120 mg/day in 53 patients and 80 mg/day in other 8 patients for 3 days and then reduced 40 mg for another 3 days) and intravenous vasodilators (nicotinic acid injection 40 mg/day or cinepazide maleate injection 320 mg/day). Intravenous batroxobin (10 U batroxobin for the first time and then reduced to 5 U, once or twice in total when the fibrinogen > 1 g/L) was given to patients whose hearing curves were of flat type and had total deafness. Lidocaine (0.1-0.2 mg/day) was intravenously injected for patients with tinnitus. The patients were categorized into 2 groups according to hearing recovery after treatment. The hearing gain average at all frequencies of at least 15 dB was divided into an effectual group, and less than 15 dB was divided into ineffectual group. 18
This study was reviewed and approved by the institutional review board and ethics committee of Fujian Geriatric Hospital (Ethics Committee No. 2020-03-01). All the methods were performed in accordance with the relevant guidelines and regulations. Informed consent was obtained from all participants.
Statistical Analysis
Post hoc power analysis was performed to assess the power on the basis of our sample size by using PASS version 11 (NCSS, Kaysville, UT, USA). The sample size was calculated based on the area under the receiver operating characteristic (ROC) curve (AUC). A sample of 61 patients and 65 healthy controls would provide a power >99% at α level 0·05 to detect a difference of 0.273 between the alternative hypothesis and the null hypothesis (AUC = 0.500).
Data were analyzed with SPSS version 17.0 (IBM Corp., New York, NY, USA). Continuous variable data were analyzed with the Student’s t test. Categorical variable data were analyzed with the chi-square test or Fisher’s exact test. Binary logistic regression analysis was carried out to verify diagnostic biomarkers and prognostic factors for SSNHL. The ROC curve analysis was subjected to the selection of cutoff points for diagnostic and prognosis with the corresponding sensitivity and specificity was calculated. P < .05 was considered to indicate statistical significance.
Results
Clinical Characteristics and Pretreatment Blood CCT Parameters of Participants
A total of 126 participants were included in this study. The clinical characteristics and CCT parameters are detailed in Table 1. The SSNHL group comprised 61 patients (29 females and 32 males) with average age of 52.33 years. The control group included 65 persons (22 females and 43 males) with average age of 49.98 years. Both groups were comparable in terms of age, gender, hypertension, diabetes, hypertriglyceridemia, hypercholesterolemia, and autoimmune disease. The probability of occurrence of headache and dizziness in the SSNHL group was similar to that of the control group, but other ear-related symptoms such as vertigo, tinnitus, and fullness were significantly different from the control group (all P < .001). Five coagulation functions were within the normal range in both control and SSNHL patients. The SSNHL group exhibited a significantly prolonged TT (19.11 ± 1.12 seconds) compared to the control group (17.58 ± 2.18 seconds, P < .001). Serum fibrinogen (FIB) in SSNHL (2.57 ± 0.53 g/L) was lower but did not show statistical difference from that of the control group (2.76 ± 0.58 g/L, P = .060).
Clinical Characteristics and CCT Parameters of Controls and SSNHLs.
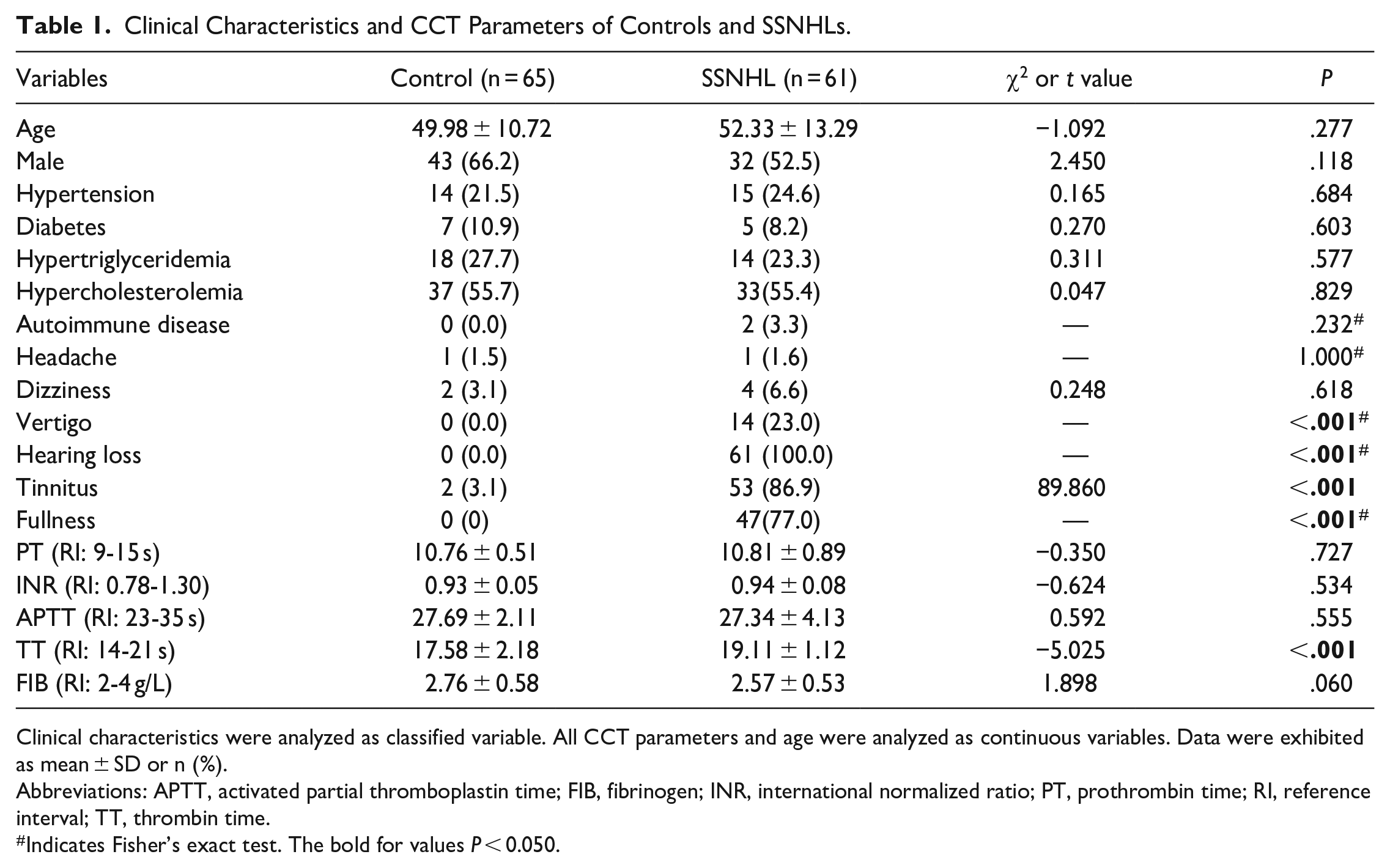
Clinical characteristics were analyzed as classified variable. All CCT parameters and age were analyzed as continuous variables. Data were exhibited as mean ± SD or n (%).
Abbreviations: APTT, activated partial thromboplastin time; FIB, fibrinogen; INR, international normalized ratio; PT, prothrombin time; RI, reference interval; TT, thrombin time.
Indicates Fisher’s exact test. The bold for values P < 0.050.
TT was Assessed as Diagnostic Factors for SSNHL
A significant positive association between TT and SSNHL with an odds ratio (OR) 1.769 [95% confidence interval (CI): 1.344-2.330] was showed in the unadjusted model (Table 2). Even after adjustment using variables included in the multivariate models, TT was significantly associated with SSNHL [model 2: adjusted OR, 1.964 (95% CI: 1.416-2.725) and model 3: adjusted OR, 2.064 (95% CI: 1.442-2.953)]. The ROC result showed that TT could significantly distinguish the SSNHL from the control group (Figure 1A). The AUC was 0.773 (95% CI: 0.689-0.856), and the TT cutoff value was 17.65 seconds, with 0.918 sensitivity and 0.569 specificity.
Different Logistic Regression Models for Assessing the Diagnostic Factors of SSNHL.
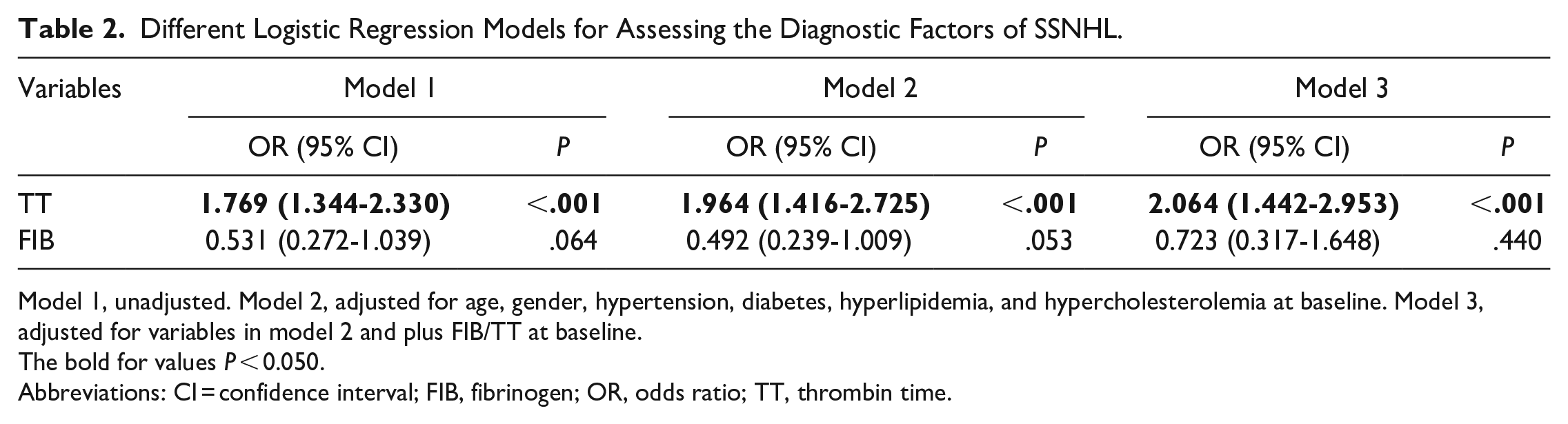
Model 1, unadjusted. Model 2, adjusted for age, gender, hypertension, diabetes, hyperlipidemia, and hypercholesterolemia at baseline. Model 3, adjusted for variables in model 2 and plus FIB/TT at baseline.
The bold for values P < 0.050.
Abbreviations: CI = confidence interval; FIB, fibrinogen; OR, odds ratio; TT, thrombin time.
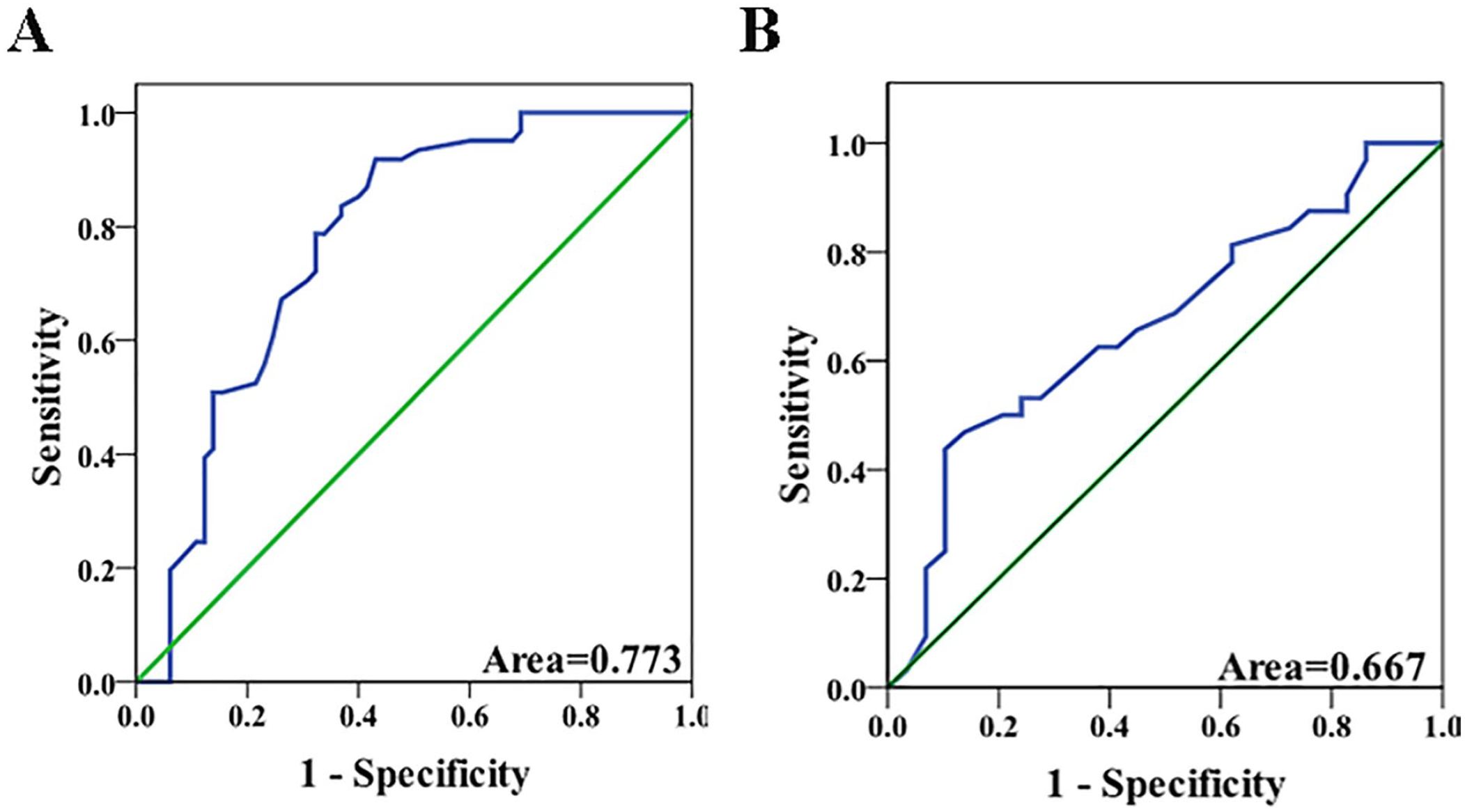
(A) Receiver operating characteristic (ROC) curves for thrombin time (TT) as predictors of SSNHL. (B) ROC curves for TT as progress predictors of SSNHL.
Clinical Characteristics and CCT Parameters of SSNHL With the Outcome of Treatment
The 61 patients with SSNHL were divided into 2 groups (the ineffective group and the effective group) according to the hearing recovery of the treatment. The ineffective group comprised 32 patients (15 women and 17 men) with average age of 55.16 years. The effectual group consisted of 29 patients (14 women and 15 men) with average age of 49.21 years. The clinical characteristics and blood CCT parameters of the 2 groups are shown in Table 3. Briefly, the younger age, shorter TT, lower frequency, or no physiotherapy prompted a more effective treatment outcome.
Clinical Characteristics and CCT Parameters of SSNHL With the Outcome of Treatment.
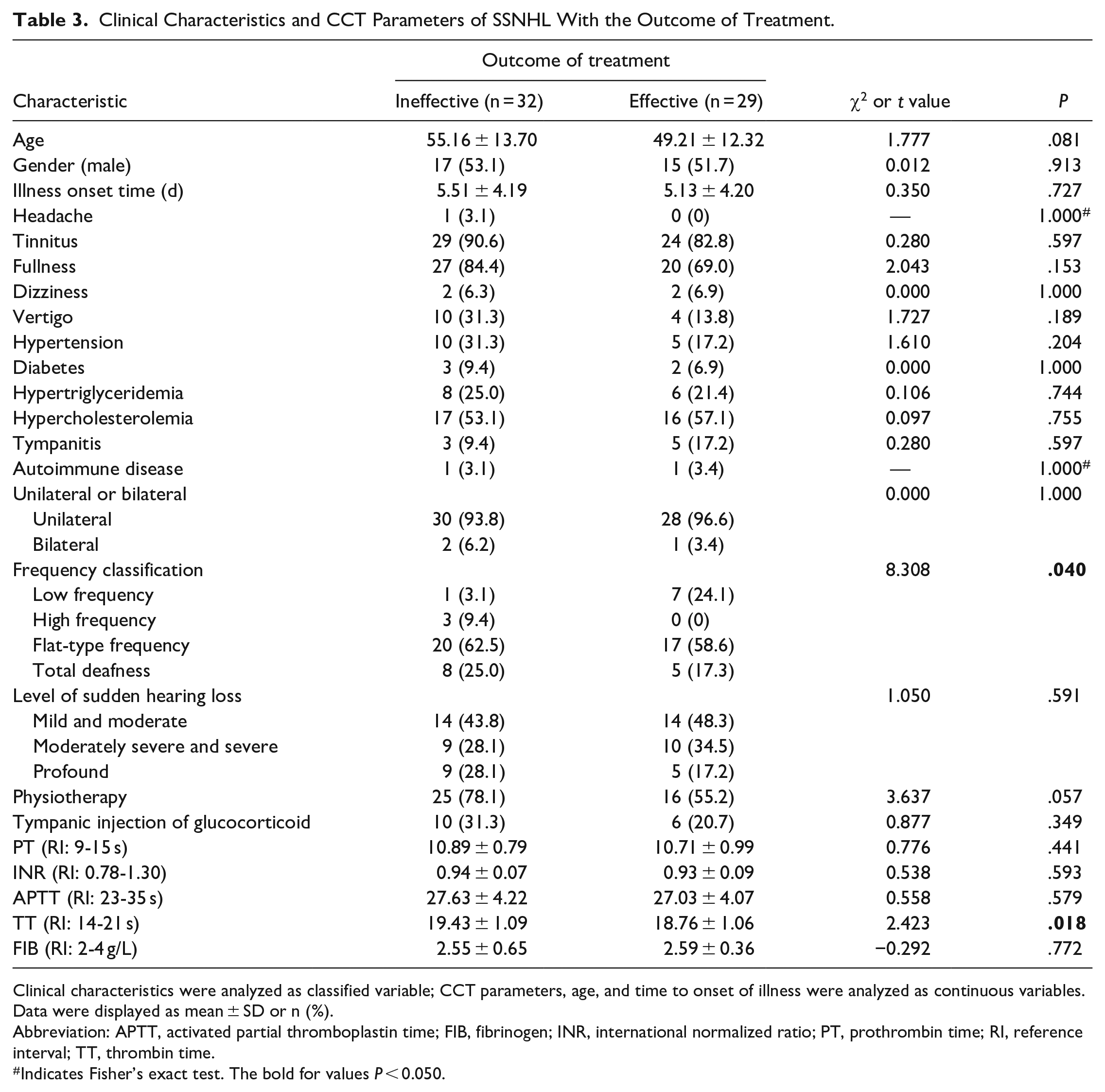
Clinical characteristics were analyzed as classified variable; CCT parameters, age, and time to onset of illness were analyzed as continuous variables. Data were displayed as mean ± SD or n (%).
Abbreviation: APTT, activated partial thromboplastin time; FIB, fibrinogen; INR, international normalized ratio; PT, prothrombin time; RI, reference interval; TT, thrombin time.
Indicates Fisher’s exact test. The bold for values P < 0.050.
TT was Assessed as Prognostic Factors for SSNHL
A significant negative association was observed between the efficacy of TT and SSNHL with an OR 0.560 (95% CI 0.340-0.923) in the unadjusted model (Table 4). This association remained significant after adjusting for potential confounders [model 2: adjusted OR, 0.518 (95% CI: 0.286-0.936) and model 3: adjusted OR, 0.509 (95% CI: 0.276-0.936)]. The TT cutoff value (19.85 seconds), determined by ROC curve analysis (Figure 1B), was then used to divide the 61 SSNHL patients into 2 groups to validate the ability to predict the prognosis. We calculated the effect of the treatment outcome and found that the effective rate in TT < 19.85 seconds group was 59.09% (26/44), significantly higher than 17.65% (3/17) in the TT 19.85 seconds group (P = .004, Figure 2). This means that TT was significantly associated with the prognosis of SSNHL. The lower the TT, the better the outcome of the treatment.
The Association Between Hearing Recovery and TT was Assessed Using Binary Logistic Regression.

Model 1, unadjusted. Model 2, adjusted for age, gender, illness onset time, vertigo, and frequency classification at baseline. Model 3, adjusted for variables in model 2 and plus physiotherapy at baseline.
The bold for values P < 0.050.
Abbreviations: CI, confidence interval; OR, odds ratio; TT, thrombin time.
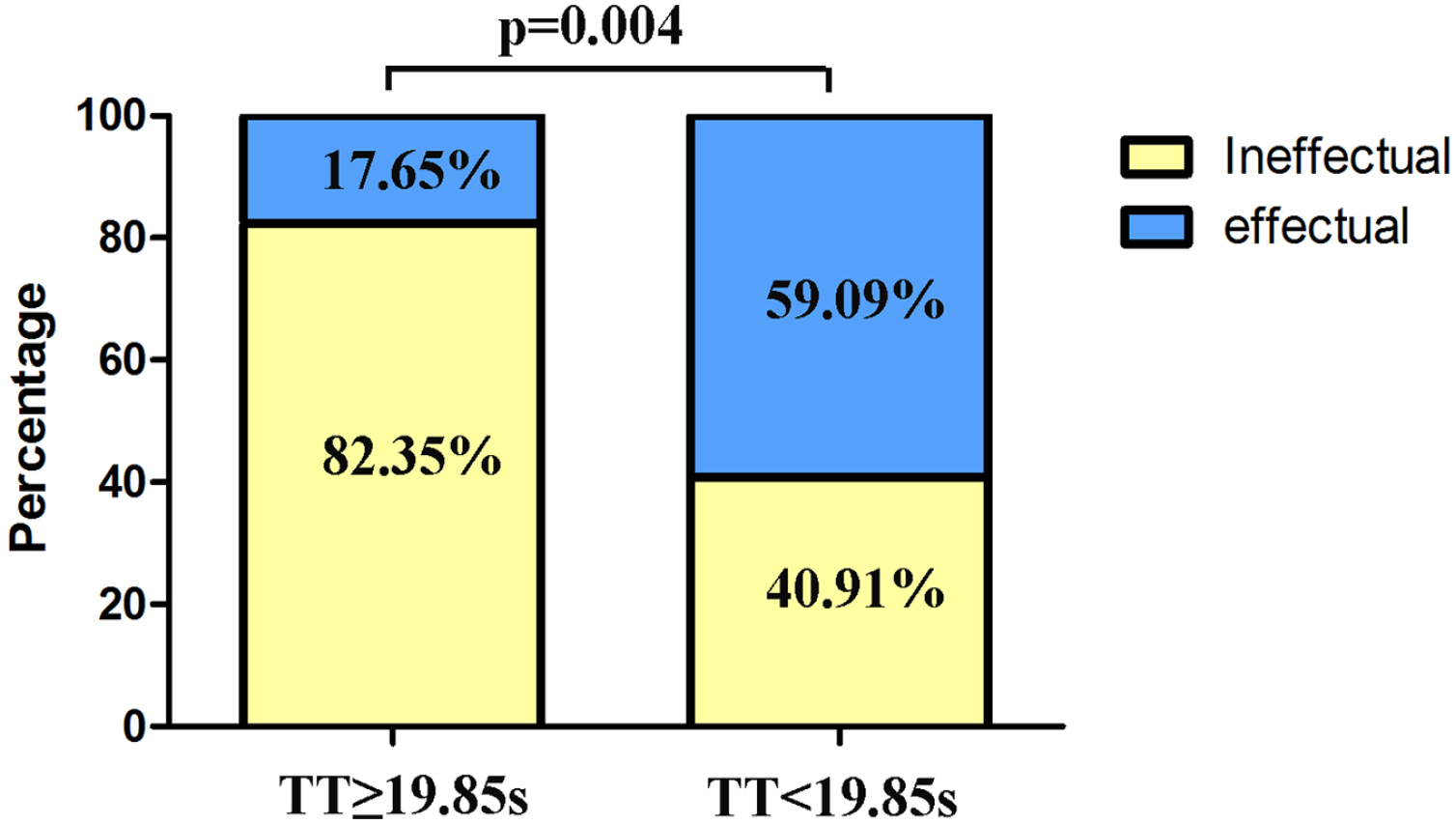
The treatment outcome of 2 groups divided by the TT cutoff value: 61 patients were divided into 2 groups by the TT cutoff value (19.85 seconds) determined by the ROC curve.
Discussion
This study is a preliminary report and shows that TT can be used as a diagnostic indicator of SSNHL and predict the prognosis. TT is related to SSNHL; it must be further confirmed whether TT is the cause or the concomitant symptom of SSNHL. Five parameters of the coagulation function were within the normal range in both control and SSNHL patients in this study and other researches,19-21 which suggested that the range of conventional coagulation indicators for terminal vessels could be in a narrower window.
The timely diagnosis and treatment of SSNHL will result in a better prognosis. 10 Therefore, it is particularly important to find an easily accessible indicator for the diagnosis of SSNHL. TT is easily detected on blood tests. In this study, the ROC curve method was used to assess the diagnostic accuracy of the model. It showed that TT can be a good result in terms of sensitivity (0.918), albeit a poor result in terms of specificity (0.569). High sensitivity should be chosen for the current treatment method, which is reliable. The missed patients with SSNHL had a poor prognosis. Furthermore, the poor specificity of TT was acceptable for TT was not used to diagnose accuracy in the common population, but for the rapid diagnosis of SSNHL and the prediction of the outcome of the treatment. The gold standard for SSNHL diagnosis is audiometric testing. The CCT could be a routine adjunct test although at present there is no drug targeting TT. The doctors can pay more attention to TT and treat SSNHL as early as possible before the audiometric testing report comes out or with no baseline audiogram exists.
In this study, TT was associated with the prognosis of SSNHL. When TT > 17.65 seconds combined with the patient’s clinical characteristics, medical histories could suggest a better diagnosis of SSNHL. When TT > 19.85 seconds predicts a poor prognosis, it means that more attention should be paid to the SSNHL progression. More refined and individualize treatment plans should be provided for a better treatment. However, the same as the other reported factors, 22 TT alone cannot completely determine the recovery from SSNHL, suggesting the need for future basic and clinical studies. A multifactor prediction model according to these factors might enhance the predicted ability for the SSNHL prognosis.
Cochlear vascular microthrombosis has been hypothesized to be one of the pathogenic mechanisms of SSNHL that is refractory to regular management. 2 TT reflects the conversion of fibrinogen to fibrin after the addition of the thrombin reagent. Measurement of fibrinogen and TT allows detecting fibrinogen disorders that can lead to thrombotic or bleeding events. 23 Five parameters of the coagulation function have been found to be in the normal range in both control and SSNHL patients, and only TT, but not FIB, could be the diagnostic and prognostic indicator of SSNHL. This suggests that there is no disorder in the qualitative and quantitative aspects of FIB, but thrombin, coagulation factors, platelets, or other factors that prolong TT in SSNHL need further exploration and discussion.
FIB has been reported to be a predictor and prognosis indicator of SSNHL,24,25 defibrinogenation therapy benefited the SSNHL patients.24,26 As some studies1,27 have proved, in this study, FIB is neither a diagnostic indicator nor a prognostic predictor. It could be the reason why there is no consensus on the effectiveness of defibrinogenation therapy that the coagulation state was influenced by many factors.12,13,28 The statistical results are likely depending on the set of the control group. In this study, the basic characteristics including age, sex, hypertension, dysmetabolism of diet (diabetes, hypertriglyceridemia, hypercholesterolemia), and autoimmune disease were balanced. More research is needed to assess the benefit and risk of bleeding from defibrinogenation therapy.
This is a case–control retrospective study. The advantages are that it can be preliminarily used for exploring the etiology because it is more time and labor saving and easier to implement without damage. There are also some limitations. First, there was recall bias and the ability of demonstrating causality was not as strong as that of a prospective study. Secondly, no prior sample size calculation was performed, but the sample size power for this study reached >99% by post hoc sample size power assessment. Thirdly, the study was a retrospective study of a single center. More centers should be enrolled for internal and external verification in the future.
Conclusions
TT is a potential biomarker for the diagnosis of SSNHL and is independently associated with the prognosis of patients with SSNHL. This may facilitate patient counseling and individualized treatment.
Footnotes
Acknowledgements
We thank all the participants of the study.
Author Contributions
Data Availability Statement
Data is available on request from the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Found-ation of Fujian Province (grant no.: 2020J01097 and 2022J01215) and Research Project of the Treatment of Auditory System Dysfunction in Aging Population (grant no.: 2020YFC2005204).
