Abstract
Introduction
Amyloidosis is a heterogenous disease characterized by the extracellular accumulation of insoluble misfolded fibrillary protein. Amyloid deposition appears as an amorphous and eosinophilic material on hematoxylin and eosin (H&E)-stained slides. Diagnosis is confirmed by positive Congo red staining and birefringence under polarized light. Current classification system uses the schema of letter A standing for amyloid followed by a suffix that designates the parent or precursor protein name. 1 For example, immunoglobulin light chain-derived amyloid fibril protein is AL (amyloid light chain) and the associated disease is designated as AL amyloidosis. ATTR stands for amyloid transthyretin and further specifications can be added after the protein name, for example, ATTRwt (wild type) or ATTRv (variant). Based on the clinical presentation and pathogenesis, amyloidosis is further classified as systemic versus localized form, and acquired versus hereditary type. 2 Amyloid subtyping should be defined early and clearly because it guides clinical management and informs prognosis.
Amyloid deposition in the upper aerodigestive tract is relatively uncommon. The most often involved site is the larynx, followed by the tongue, oral mucosa, and pharynx. Laryngeal amyloidosis accounts for only 0.2% to 1.2% of all laryngeal benign lesions. 3 Patients often presented with dysphonia, cough, and/or sense of foreign material. 4 It is usually treated under microlaryngoscopy via laser or microsurgical excision. 5 Oral tongue is the most common site for amyloid deposition in oral cavity and majority of patients present with macroglossia. Pharyngeal amyloidosis is extremely rare with less than 50 cases reported to date. Although there are previously published small case series, many cases did not have complete laboratory or pathologic workup. Here, we reviewed the clinical information, laboratory results, and pathologic findings from 28 cases of amyloid deposition involving the larynx, oral cavity, or pharynx in our institute, and compared our studies with the published case series in the English literature including at least 3 cases.
Materials and Methods
The study protocol was approved by the Institutional Review Board, and it was conducted at an academic teaching hospital. The institutional database from 2000 to 2020 was retrospectively reviewed and a total of 39 specimens from 28 patients were identified with amyloid deposition in the larynx, oral cavity, or pharynx. Congo red stain was manually performed on all cases to confirm the diagnosis by following standard protocol. Kappa and lambda in situ hybridization (ISH) were performed on Ventana BenchMark ULTRA (Roche, Basel, Switzerland) by using ISH staining module. Slides from selected cases were also reviewed by TH and AAS. A Chi-squared test was performed by using the Windows Excel software. (Microsoft Corporation, Redmond, WA) Literature search was performed by using the combination of keywords “amyloid,” “larynx,” “oral cavity,” “tongue,” or “pharynx” in PubMed databases. Case series that included at least 3 cases were included. Studies focused on treatment with very limited clinical information or diagnostic workups were excluded.
Results
A total of 39 specimens from 28 patients (17 female, 11 male) were retrieved with a mean age of 62.1 years (range 17-91 years). H&E sections showed amorphous and waxy material accumulated predominantly in the lamina propria (Figure 1A and D). Congo red demonstrated salmon-pink staining in the areas with amyloid deposition (Figure 1B and E) and apple green birefringence under polarized light (Figure 1C and F).
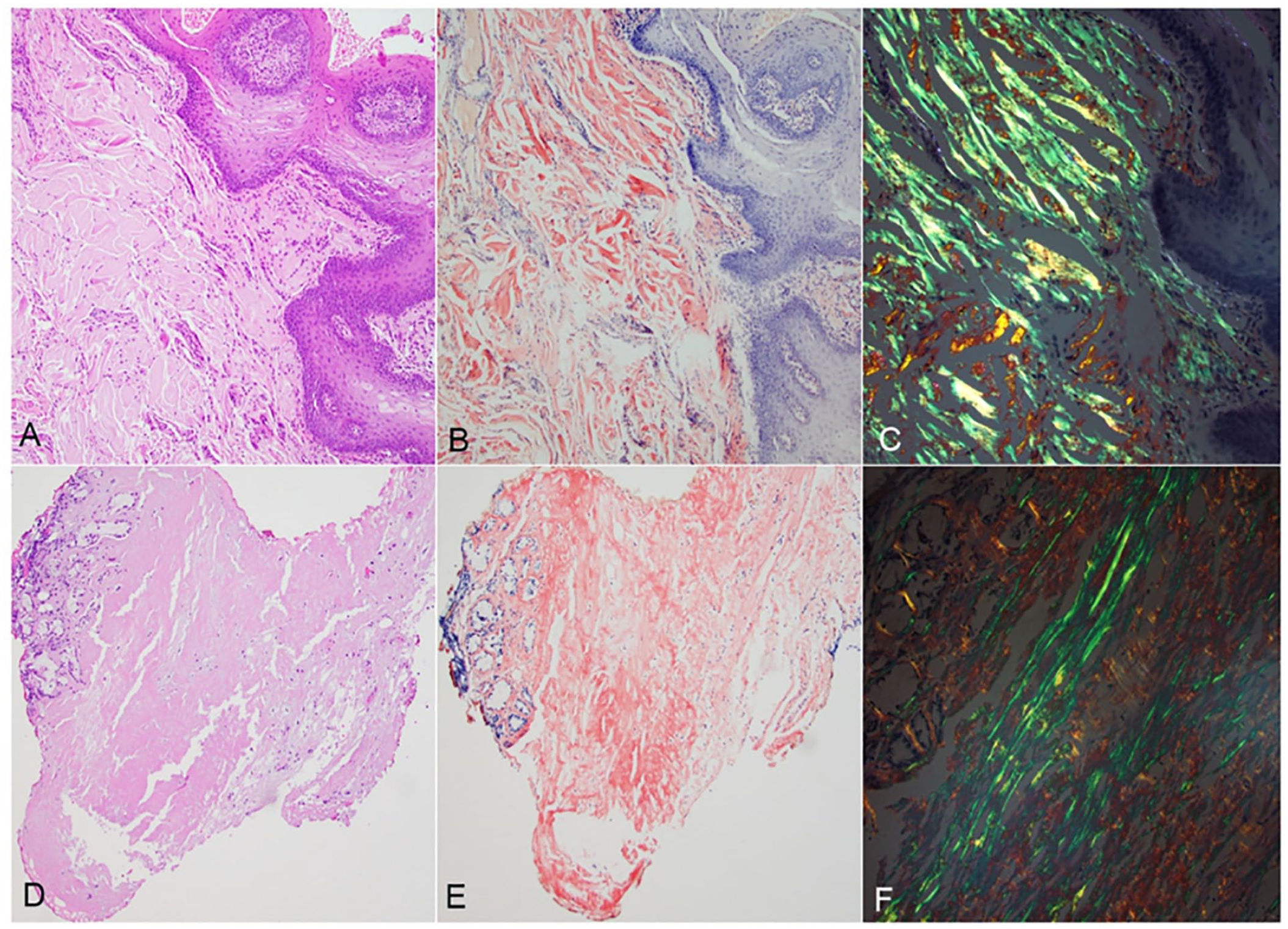
Representative pictures of amyloid deposition in tongue (A-C) and larynx (D-F) biopsy. (A and D) H&E sections showed amorphous material in lamina propria; (B and E) Congo red stains highlighted amyloid in salmon-pink color; (C and F) Congo red stain observed under polarized light showed a characteristic “apple green” birefringence. H&E, hematoxylin and eosin.
Amyloid Deposition in Larynx
Seventeen specimens collected from 12 patients were from the larynx (Table 1) including 7 in the true vocal cord, 2 in the supraglottis, and 3 in both locations. Three patients had multifocal disease involving larynx and nasopharynx (2 patients) or larynx and tongue (1 patient). All patients presented with dysphonia and had biopsy-confirmed disease under laryngoscopy. Five patients had head and neck computed tomography (CT) imaging in our electronic medical system. CT demonstrated a laryngeal mass in 3 patients (ranging from 1.5 to 3.5 cm in greatest dimension) (Figure 2A and B) and soft tissue thickening in the other 2 patients. Figure 2B demonstrates a large supraglottic mass causing airway obstruction in a 17-year-old male. No underlying hematopoietic disorder or monoclonal protein was detected in this patient. The disease recurred 5 years after resection. He received a second laser resection and has been stable for 6 years.
Summary of Clinical and Pathologic Information From All Patients.
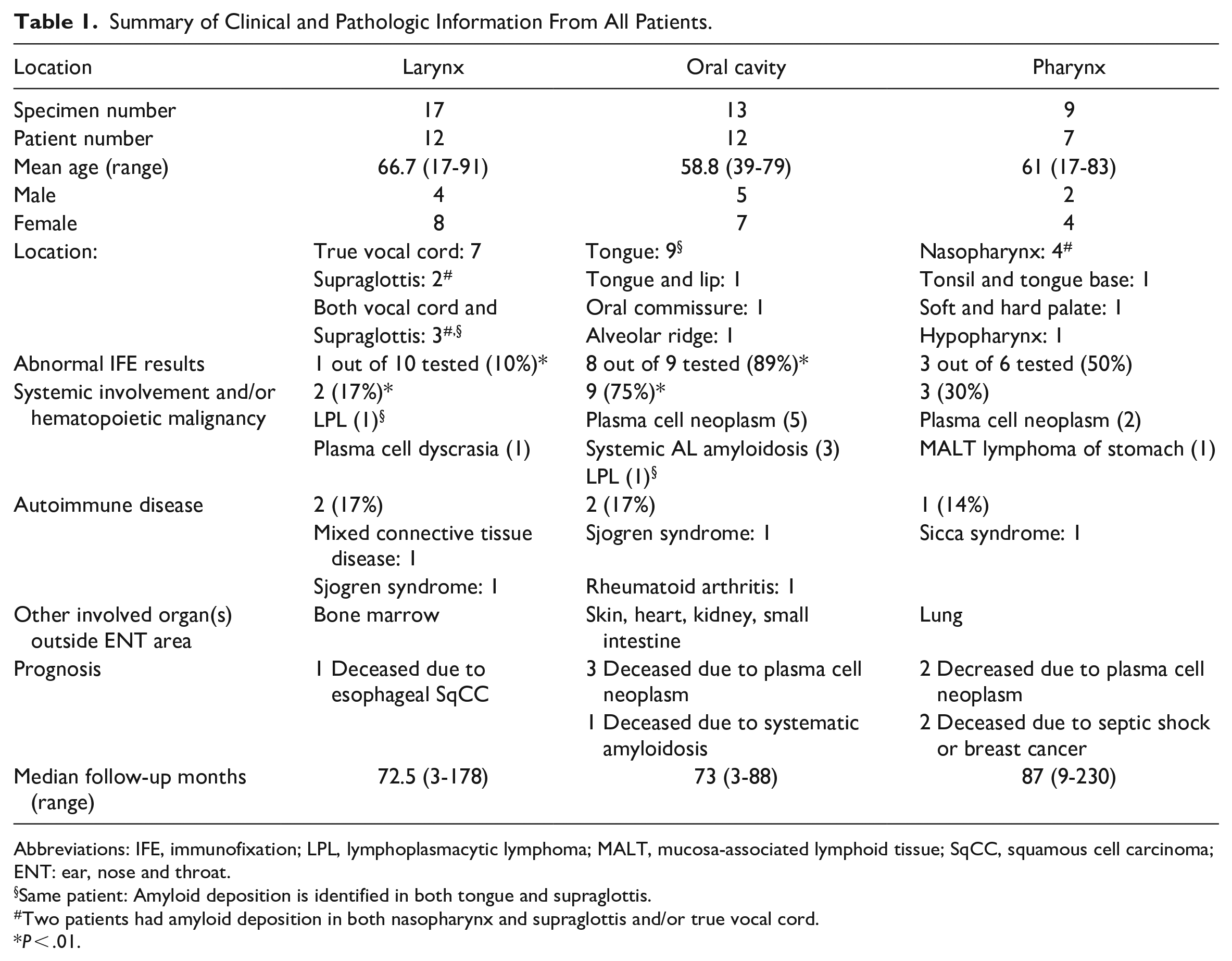
Abbreviations: IFE, immunofixation; LPL, lymphoplasmacytic lymphoma; MALT, mucosa-associated lymphoid tissue; SqCC, squamous cell carcinoma; ENT: ear, nose and throat.
Same patient: Amyloid deposition is identified in both tongue and supraglottis.
Two patients had amyloid deposition in both nasopharynx and supraglottis and/or true vocal cord.
P < .01.
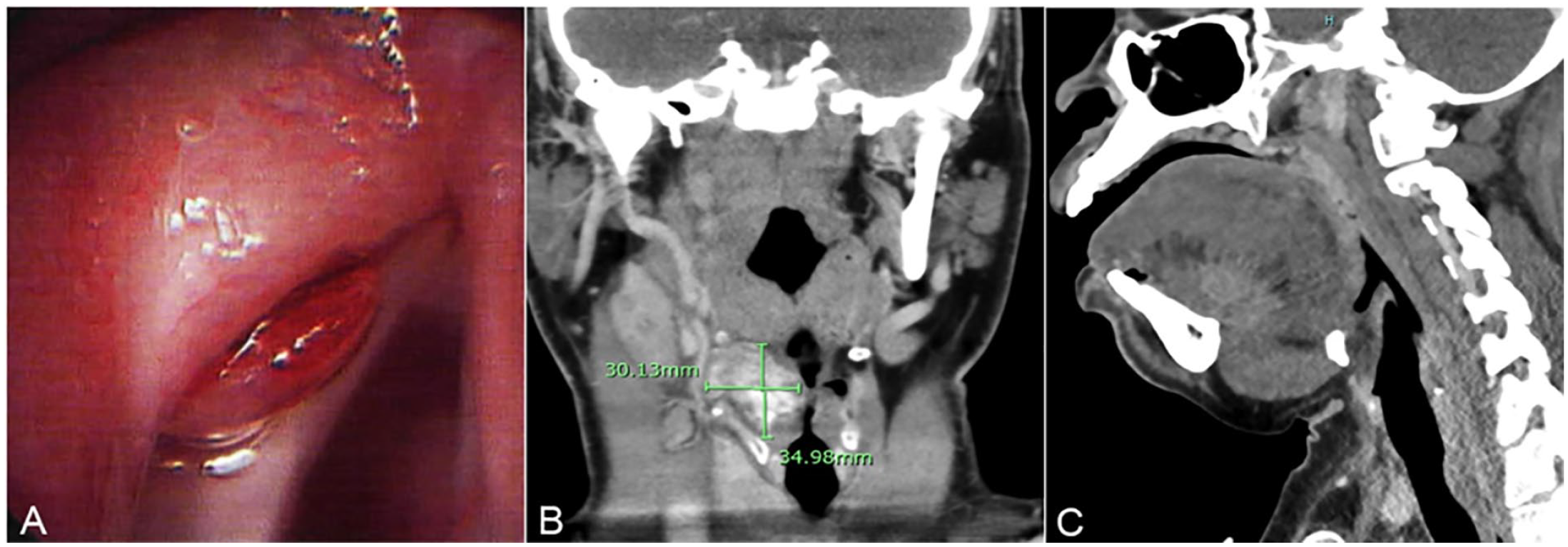
(A) Laryngoscopy image of left false vocal cord mass with cystic-like submucosal fullness in a patient with isolated laryngeal amyloid deposition. (B) CT scan revealed a large mass centered within the right vocal cord and laryngeal ventricle with areas of heterogeneous hyperdensity in a patient with isolated larynx amyloidosis. The lesion caused narrowing of the glottic and subglottic larynx. (C) A patient with systemic amyloidosis presented with macroglossia. CT scan showed diffuse enlargement of the oral tongue without discrete mass. The degree of tongue enlargement compromised the oral airway.
Serum/urine protein electrophoresis (SPEP/UPEP) and immunofixation (IFE) were tested in 10 out of 12 patients and only 1 patient (10%) was found to have monoclonal protein of kappa light chain. Her subsequent bone marrow biopsy revealed plasma cell dyscrasia and amyloid deposition in marrow space. Another 61-year-old female patient with history of lymphoplasmacytic lymphoma (LPL) in remission presented with laryngeal amyloid deposition. Liquid chromatography tandem mass spectrometry (LC-MS/MS) confirmed AL kappa type. Her subsequent bone marrow biopsy and flow cytometry were both negative for active disease.
LC-MS/MS from 1 patient detected a peptide profile consistent with ATTR with a low level of amino acid sequence abnormality in Thr60Ala (p.Thr80Ala). This finding raised the possibility of hereditary ATTR amyloidosis; however, the patient did not have any known family history. Echocardiogram demonstrated stage I diastolic dysfunction with normal left ventricular ejection fraction. Her kidney and pulmonary functions were both normal. Patient was referred to genetic counseling and sequencing of TTR genes is currently pending.
Three patients had underlying autoimmune diseases including 1 patient with mixed connective tissue disease, and 1 with Sjogren syndrome. Seven patients (58%) had multiple local recurrences that required repeated laser resections. During follow-up, only 1 patient died from esophageal cancer. The median follow-up was 72.5 months (range 3-178 months).
Amyloid Deposition in Oral Cavity
Thirteen specimens from 12 patients (7 female, 5 male, with a mean age of 58.8 years, ranging 39–79 years) showed amyloidosis in the oral cavity and 9 (75%) were in the tongue, 1 in both tongue and lip, 1 in the oral commissure, and 1 in the maxillary alveolus (Table 1). Eight (67%) patients presented with macroglossia, 2 of them with a firm tongue nodule, and 2 with tender spots in the oral cavity. In one of the patients with macroglossia, this was the initial symptom of multiple myeloma. Figure 2C depicts a CT scan from a 39-year-old female with significant macroglossia causing airway compromise that required urgent tracheostomy. This patient presented with rapidly progressive systemic amyloidosis involving the tongue, kidney, and small intestine that caused acute kidney failure and ischemic bowel disease that required surgical intervention. She received chemotherapy with bortezomib and cyclophosphamide but passed away 3 months after the initial diagnosis.
SPEP/UPEP and IFE were tested in 9 patients and monoclonal protein was detected in 8 (89%) cases. All 9 patients had systematic involvement (9/12, 75%), which was significantly higher than that of laryngeal amyloidosis (P < .01). Six patients had hematopoietic neoplasms including 5 with plasma cell neoplasm and the previously mentioned patient with LPL. Three patients had biopsy-confirmed systemic AL amyloidosis (2 lambda and 1 kappa) that involved the skin, heart, kidney, and/or small intestine. Bone marrow biopsies were performed in 2 out of 3 cases and revealed 5% to 10% plasma cells. Two patients had a history of autoimmune disease including 1 Sjogren syndrome and 1 rheumatoid arthritis case. Upon follow-up, 3 patients died of their plasma cell neoplasm, and 1 died due to systemic amyloidosis (mentioned above). The median follow-up time was 73 months (range 3–88 months).
Amyloid Deposition in Pharynx
There were 9 specimens with pharyngeal amyloidosis (Table 1) that were collected from 7 patients, including 4 in the nasopharynx, 1 in the oropharynx (tonsil and tongue base), 1 involving the soft and hard palate, and 1 in the hypopharynx. SPEP/UPEP was performed in 6 patients and 3 of them had monoclonal protein (50%). All 3 of them had biopsy-confirmed hematopoietic malignancy including 1 with extramedullary plasmacytoma of the tonsil and base of tongue, 1 with multiple myeloma and 1 with a gastric mucosa-associated marginal zone lymphoma (MALT). Figure 3 shows an extramedullary plasmacytoma of tonsil from a 79-year-old male with abundant amyloid deposition (Figure 3A and 3B). ISH revealed kappa monoclonal plasma cells (Figure 3C and D). The patient with MALT lymphoma also had biopsy-confirmed lung amyloidosis and was subsequently diagnosed with esophageal squamous cell carcinoma. He died of septic shock and multiple organ failure. Finally, 1 patient had a history of Crohn disease and Sicca syndrome. This patient died of breast cancer. The median follow-up time was 87 months (range 9–230 months).
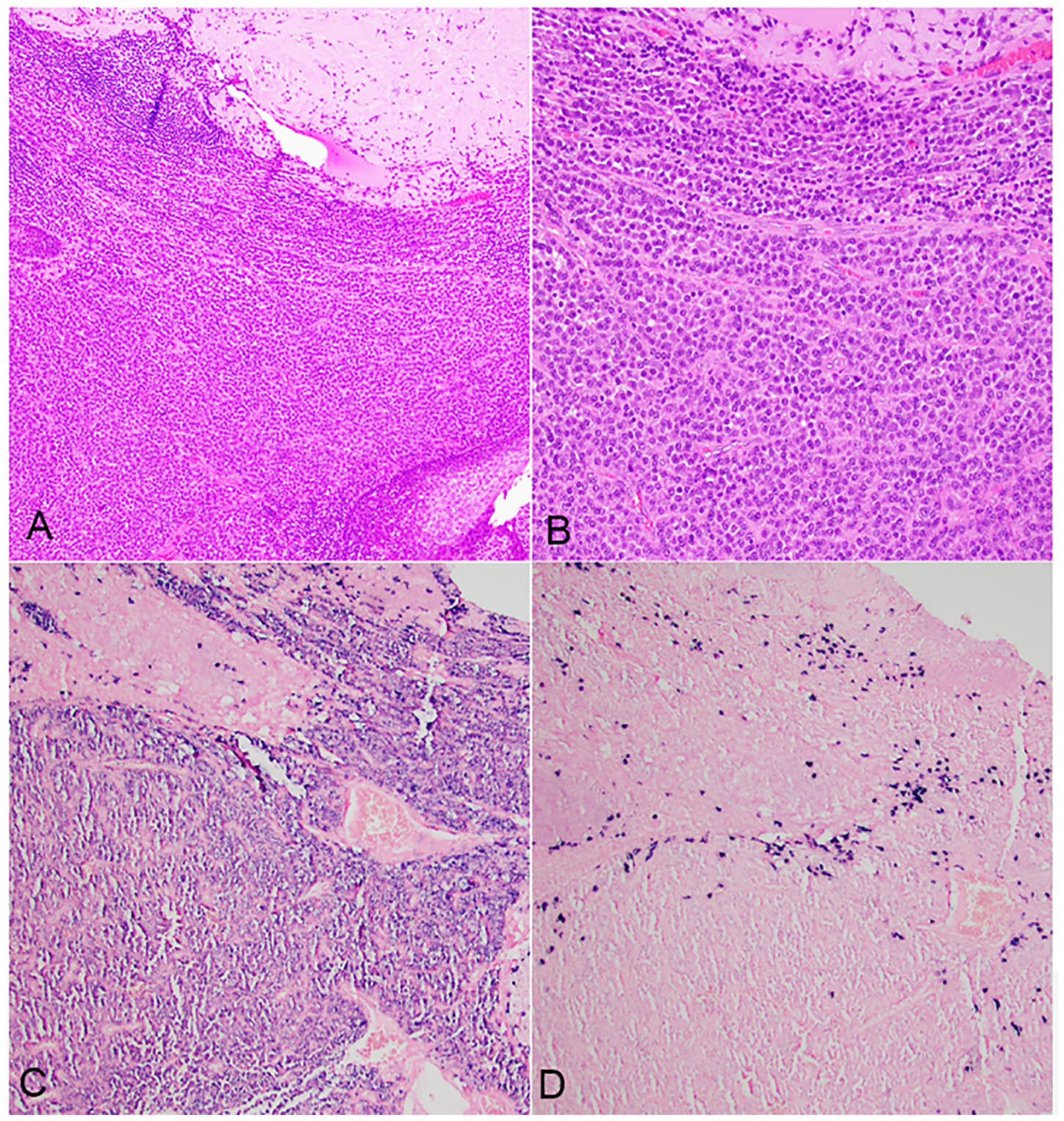
Patient with extramedullary plasmacytoma involving tonsil. (A and B) Sheets of plasma cell infiltration (lower) and abundant amyloid deposition (upper) were identified in tonsil. A small focus of squamous mucosa is present at right lower corner of A. (C and D) Kappa and lambda in situ hybridization demonstrate monoclonality with kappa restriction.
Discussion
Laryngeal Amyloidosis
Laryngeal amyloidosis is an uncommon event. Wierzbicka et al 5 reported it was only seen in 0.52% of patients encountered for microlaryngoscopic procedures in a decade. It affects patients with a wide range of ages, but it is rare in the pediatric group.6-8 Most patients presented with dysphonia. Rare cases, including one of our patients, presented with large obstructive masses causing airway compromise9,10 that required urgent tracheostomy. Localized laryngeal amyloidosis is treated by sharp microsurgical excision or laser excision under microlaryngoscopy. No significant difference in recurrence has been reported between these 2 methods.11,12 In our study, 58% of patients had recurrent disease that required additional surgical intervention. The recurrence rate ranges from 25% to 100% in previous studies (Supplemental Table 1).3,13-18 Wu et al 12 showed young age and long interval from the onset to treatment increased the risk for recurrence. Local recurrence after 16 years of treatment has been reported, suggesting long term follow-up is necessary in these patients. 16
The mechanism for localized laryngeal amyloidosis is unknown. AL has been confirmed as the most common source of laryngeal amyloidosis by both immunohistochemical staining8,13 and recent LC-MS/MS study. 19 Lambda light chain has been reported more frequently than kappa.8,13 The second most common type is reactive amyloidosis (Serum Amyloid A, AA) 20 which is associated with systemic inflammatory disease. This may explain the finding that a subset of patients had autoimmune diseases.
Our study confirmed the previous finding that most laryngeal amyloidosis is of localized form. A small subset of patients has systemic involvement and/or underlying hematopoietic disorder. The percentage of these patients is difficult to estimate because of limited case numbers and lack of comprehensive workup in many published cases. In our study, 2 (17%) patients had hematopoietic disorders. Systemic involvement has been observed in 0% to 36% in the previous studies and the rate of underlying hematopoietic disorder ranges from 0% to 17%3,7,13-15,17-19 (Supplemental Table 1). Three patients including one of our cases had hereditary ATTR amyloidosis7,19 and 2 of these 3 cases were identified by amyloid typing utilizing mass spectrometry. Laser microdissection and mass spectrometry-based amyloid proteomic analysis was developed by Mayo Clinic in 2009. 21 It has been demonstrated as a single assay that effectively subtypes amyloid protein with high sensitivity and specificity. 22 In our opinion, all patients with laryngeal amyloidosis should be screened with mass spectrometry-based amyloid subtyping and protein electrophoresis to avoid misdiagnosis of rare cases with hematopoietic disorder or hereditary type amyloidosis. If the screening tests are negative, patients can be followed up for potential local reoccurrence.
Oral Amyloidosis
There has been a total of 11 case series about oral cavity amyloidosis with more than 3 patients17,23-32 in the English literature (Supplemental Table 2). When we combined our cases with previous 11 case series, the most common location was tongue (68%), followed by multifocal involvement in the oral cavity (15%), oral mucosa including lip (13%), and palate (4%). In contrast to laryngeal amyloidosis, amyloid deposition in tongue is highly associated with underlying hematopoietic disorder and systemic involvement, especially plasma cell neoplasms, and detectable paraproteinemia. In our study, 6 (50%) patients had hematopoietic disorders, 89% had a monoclonal protein spike and 3 cases (25%) had systemic AL amyloidosis. In the previous studies with more than 10 cases,17,25,26,30 50% to 100% of patients had hematopoietic disorder, including multiple myeloma (71%), monoclonal gammopathy of undetermined significance (14%), plasma cell dyscrasia (9%) and non-Hodgkin lymphoma (6%) (Supplemental Table 2). Macroglossia is the most common clinical presentation of oral tongue amyloidosis. In one of our patients and in a few case reports,33,34 macroglossia was the initial manifestation for multiple myeloma. Because of high risk for underlying hematopoietic disorder and systemic disease, comprehensive workup including serum free light chains, protein electrophoresis, and/or bone marrow biopsy are essential for these patients.
Pharyngeal Amyloidosis
Amyloidosis involving the pharynx is extremely rare and mainly documented in case reports. The most commonly involved site is the nasopharynx followed by the oropharynx, especially the palatine tonsil, and the hypopharynx. The most common clinical manifestation of nasopharyngeal amyloidosis is nasal obstruction with or without hearing loss. Other reported symptoms were postnasal drip, otitis media, epistaxis, and Eustachian tube dysfunction. The largest case series of nasopharyngeal amyloidosis was reported by Sakagiannis et al. 35 and included 7 cases. All cases were localized forms and AA type. Although most cases were localized, rare case reports with hematopoietic disorders36,37 and/or systemic amyloidosis 38 have been reported. Patients with oropharyngeal or hypopharyngeal amyloidosis usually presented with dysphonia, throat pain, or dysphagia. A total of 12 cases of oropharyngeal amyloidosis have been reported in the English literature39-50 and 8 of them involved the palatine tonsil. Three cases were associated with plasma cell neoplasms41,44,45 and 1 patient was diagnosed with LPL/Waldenstrom macroglobulinemia. 47 Only 4 cases of hypopharyngeal amyloidosis, including one of our cases, have been reported.51-53 Three of 4 patients including our patient were diagnosed with multiple myeloma.51,53 These findings suggest that nasopharyngeal amyloidosis is mostly localized, while amyloid deposition in tonsil and hypopharynx tend to be associated with hematopoietic malignancy and systemic involvement. Further studies are needed due to limited case numbers.
Conclusions
Amyloid deposition involving different locations of the upper aerodigestive tract is associated with distinct clinical presentation, and variable risk for systemic involvement. Our study confirmed that laryngeal amyloidosis is mostly of localized form, but about 10% to 20% of patients are associated with hematologic malignancy or hereditary type amyloidosis. We recommended using mass spectrometry-based amyloid subtyping, free serum light chain assay, and protein electrophoresis as screening tests. Patients with negative screening tests may need a long time follow-up considering high local recurrence rate. Amyloid deposition in the oral cavity has significantly higher risk of underlying hematologic malignancy or systemic disease, which warrants a comprehensive work-up including bone scans and possible bone marrow biopsy. Our study also contributes to the literature by adding additional cases of pharynx amyloidosis. Based on the limited data, amyloid deposition in oropharynx and hypopharynx appears to be more likely associated with hematologic malignancy compared to oropharyngeal involvement.
Supplemental Material
sj-docx-1-ear-10.1177_01455613231189144 – Supplemental material for Amyloid Deposition in the Upper Aerodigestive Tract, A Single Institute Experience
Supplemental material, sj-docx-1-ear-10.1177_01455613231189144 for Amyloid Deposition in the Upper Aerodigestive Tract, A Single Institute Experience by Tieying Hou, Asma Abu-Salah, Oscar W. Cummings, Stacey Halum, Hector Mesa and Dongwei Zhang in Ear, Nose & Throat Journal
Supplemental Material
sj-docx-2-ear-10.1177_01455613231189144 – Supplemental material for Amyloid Deposition in the Upper Aerodigestive Tract, A Single Institute Experience
Supplemental material, sj-docx-2-ear-10.1177_01455613231189144 for Amyloid Deposition in the Upper Aerodigestive Tract, A Single Institute Experience by Tieying Hou, Asma Abu-Salah, Oscar W. Cummings, Stacey Halum, Hector Mesa and Dongwei Zhang in Ear, Nose & Throat Journal
Footnotes
Data Availability Statement
All data supporting the findings of this study are available within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
The study was performed according to the ethical standards of the human subjects and was approved by the Institutional Review Board at Indiana University.
Consent Statement
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
