Abstract
Introduction
Allergic rhinitis (AR) is defined as systemic sensitization to allergens, with an allergic response in the nasal cavity upon exposure to causative allergens. 1 Sensitization to the allergen and reexposure to the sensitized nasal passage initiate immunoglobulin E (IgE)-mediated inflammatory reactions and induce symptoms of rhinorrhea, sneezing, nasal pruritus, and congestion. 2 The diagnosis of AR is often made on the basis of clinical characteristics, evidence of sensitization, and response to pharmacotherapy. 3 Evidence of sensitization to a known allergen usually involves a combination of skin or blood testing, and a skin prick test and measurement of serum-specific IgE have been regarded as standardized tests for the diagnosis of AR. 4
The nasal provocation test (NPT) is another tool that can be used for the diagnosis of AR. In an NPT, a causative antigen is directly administrated into the nasal cavity, and if the patient has been sensitized to the allergen, the administered antigen can cause immediate and late-phase allergic inflammation, including subjective nasal or ocular discomfort, and objective changes such as a decrease in the cross-sectional area of the nasal cavity or peak nasal inspiratory flow (PNIF) in AR patients. 5 Skin sensitization to allergens demonstrated through the skin prick test is not directly associated with the severity of AR. 6 The presence of serum-specific IgE (sIgE) alone does not necessarily imply the existence of allergic symptoms, and there are a number of patients who are sensitized but are not clinically allergic.6,7 The purpose of NPT is to reproduce the reaction occurring during natural exposure to the causative allergen. 7 Therefore, NPT demonstrates both the presence of allergen sIgE and the causal role of allergens in the nasal cavity. 8
Studies have evaluated the relationship between tests for the diagnosis of AR. Chusakul et al 9 reported optimal cutoff values of symptoms score and PNIF after allergen exposure as a predictor of AR and evaluated the relationship between changes in NPT and the wheal size of the skin prick test. Kim and Jang 5 evaluated the relationship between NPT and skin prick test by analyzing changes in acoustic rhinometry measurement outcomes after nasal provocation in AR patients who were diagnosed based on the skin prick test. The relationship between rhinometric responses to nasal allergen provocation and skin prick test outcomes has also been evaluated in children. 10 However, compared with the skin prick test, studies that have evaluated the relationship between serum sIgE and NPT have not been thoroughly reported, and the relationship between the multiple allergen simultaneous test (MAST) and the NPT has not yet been reported in South Korea.
This study aimed to review the clinical data of participants who underwent both MAST and NPT and to evaluate the relationship between MAST and NPT outcomes.
Materials and Methods
Ethical Considerations
This study was approved by the institutional review board of Chung-Ang University Hospital (2204-013-19414), and the review board waived the requirement for informed consent.
Participants
This was a retrospective, single-tertiary-center-based study. The medical records of participants who underwent both MAST and NPT at Chung-Ang University Hospital between January 2020 and February 2022 were reviewed. House dust mites are one of the most common antigens in AR, and the European house dust mite [Dermatophagoides pteronyssinus (DP)] and American house dust mite [Dermatophagoides farinae (DF)] are regarded as the most important species in terms of allergic reactions. 11 Therefore, we performed NPT against DP and DF. Positive MAST for DP and DF was diagnosed when patients fulfilled the following criteria: high titer (≥2 positivity or ≥0.70 IU/ml) of IgE antibodies specific for DF and DP in the MAST. 12 Eosinophil count and eosinophil cationic protein (ECP) levels were simultaneously measured with MAST. All participants completed a questionnaire for their rhinitis symptoms, including nasal obstruction, rhinorrhea, sneezing, and itching, at their first visit before being tested. We used a standardized 10 cm visual analog scale (VAS) ruler, wherein the patient was asked to indicate the severity of symptoms from 0 cm (no symptoms) to 10 cm (worst troublesome) for each of these symptoms. The sum of the rhinitis symptom scores ranged from 0 to 40.
We did not perform NPT in patients who had used anti-allergy medications, such as antihistamines or vasoconstrictors within the previous 7 days, intranasal steroids within a month, and systemic corticosteroids within the last 3 months. 1 Patients who had contraindications to the use of epinephrine in case of an emergency, those who had undergone any nasal surgery within the last 3 months, and pregnant or lactating females did not perform NPT either.
Protocol for NPT
Laboratory setup and materials for provocation
We followed the protocol for NPT as described in a previous study. 13 Patients were first acclimatized for 15 minutes in a room that maintained constant temperature and humidity (20 ± 1.5°C, relative humidity 40%-60%). 14 We purchased 5000 AU/mL stock DP and DF solution (#6691, HollisterStier Allergy, Spokane, WA, USA), and the stock was diluted 1:10 with normal saline. Saline was used to evaluate nonspecific hyperreactivity. Using a metered dose pump sprayer, 100 μL of saline or DP/DF solution was sprayed onto both nostrils.
Subjective and objective measurements after nasal challenge
Subjective symptoms, including nasal obstruction, rhinorrhea, sneezing, nasal itching, and ocular symptoms, were measured using the VAS. We used a standardized 100 mm VAS ruler, and the patient was asked to indicate the severity of symptoms from 0 mm (no symptoms) to 100 mm (worst troublesome). The total nasal symptom score (TNSS) was calculated as the summation of all subjective symptoms, wherein the summation of the symptom scores ranged from 0 to 500. PNIF was used to assess objective measurements using an inspiratory flow meter (Clement Clarke International, Harlow, UK). The patient’s nose and mouth were completely covered with a mask connected to a flow meter. Baseline scores for the subjective and objective measurements were obtained before the challenge. To detect nonspecific hyperreactivity, normal saline solution was applied to both nostrils, and after 10 minutes, subjective and objective measurements were obtained. Next, DP and DF solutions were applied to both nostrils, and subjective and objective measurements were obtained again after 15 minutes. The subjective measurement (change in VAS score) was calculated as [(post-challenge VAS) − (baseline VAS)]. The objective measurement (change in PNIF) was calculated as [(baseline PNIF) − (post-challenge PNIF)]/(baseline PNIF) × 100 (%). If the VAS change was ≥55 mm and/or the PNIF change was ≥40% at 15 minutes after the challenge, it was determined as a positive response. 1
Statistical analysis
R version 3.6.2 was used for statistical analysis, and the mean and standard deviation were described for the variables. Data frequency was counted for participants, sex variables, and nonspecific hyperreactivity. A two-sample t-test was used to test for the difference in the sex ratio, and the chi-square test was used to test the difference in nonspecific hyperreactivity between the groups. Intergroup differences in descriptive data were assessed using the Wilcoxon rank-sum test. Receiver operating characteristic (ROC) curves were used to compare the models, and the areas under the curve (AUC) were also calculated. If AUC > 0.5, the MAST result and the test are statistically significantly associated, which is consistent with the Wilcoxon rank-sum test result.15,16 Thus, the Wilcoxon rank-sum test was used to determine the association between the MAST results and the tests. DeLong’s test was performed to compare the 2 ROC curves. 17 The level of significance was set at 0.05.
Results
Participants
After reviewing the medical records of 116 patients, 20 were excluded because of incomplete tests or missing data, and 96 were enrolled in the analysis. Among the 96 participants, 26 were classified as MAST positive and 70 were grouped as MAST negative. In the MAST-positive group, 13 were male and 13 were female, whereas in the MAST-negative group, 37 were male and 33 were female. The mean age was 33.81 ± 14.55 years in the MAST-positive group, and 46.26 ± 18.18 years in the MAST-negative group. Among rhinitis symptoms, VAS scores for nasal obstruction, rhinitis, and sneezing were different between the MAST-positive and MAST-negative groups. The mean ECP level was 20.75 ± 20.36 (μg/L) in the MAST-positive group and 16.82 ± 15.18 (μg/L) in the MAST-negative group, and the difference was not statistically significant. The eosinophil count was 333.96 ± 265.05 (cells/μL) in the MAST-positive group and 218.69 ± 156.73 (cells/μL) in the MAST-negative group, and the difference was not statistically significant. The percentage of nonspecific hyperreactivity between the MAST-positive and negative groups was not statistically significant (Table 1).
Participant Characteristics.

Abbreviations: ECP, eosinophil cationic protein; MAST, multiple allergen simultaneous test; VAS, visual analog scale.
P < 0.05.
Nasal Responsiveness to NPT
The baseline PNIF was 108.65 ± 26.37 in the MAST-positive group and 117.71 ± 48.57 in the MAST-negative group, and the difference was not statistically significant. The change in PNIF between pre- and post-allergen nasal challenge was 27.12 ± 31.20 (%) in the MAST-positive group and −6.41 ± 97.54 (%) in the MAST-negative group, and the difference was statistically significant (P < 0.05). The baseline subjective symptom score was 177.69 ± 97.54 (mm) in the MAST-positive group and 145.01 ± 82.92 (mm) in the MAST-negative group, and the difference was not statistically significant. The change in the summation of subjective symptoms between pre- and post-allergen nasal challenge was 64.42 ± 80.44 (mm) in the MAST-positive group and 11.30 ± 48.69 (mm) in the MAST-negative group, and the difference was statistically significant (P < 0.05). Among the subjective symptoms, nasal obstruction, rhinorrhea, sneezing, itching, and ocular discomfort scores were statistically significant between the MAST-positive and negative groups (P < 0.05) (Table 2).
Results of the Nasal Provocation Test.
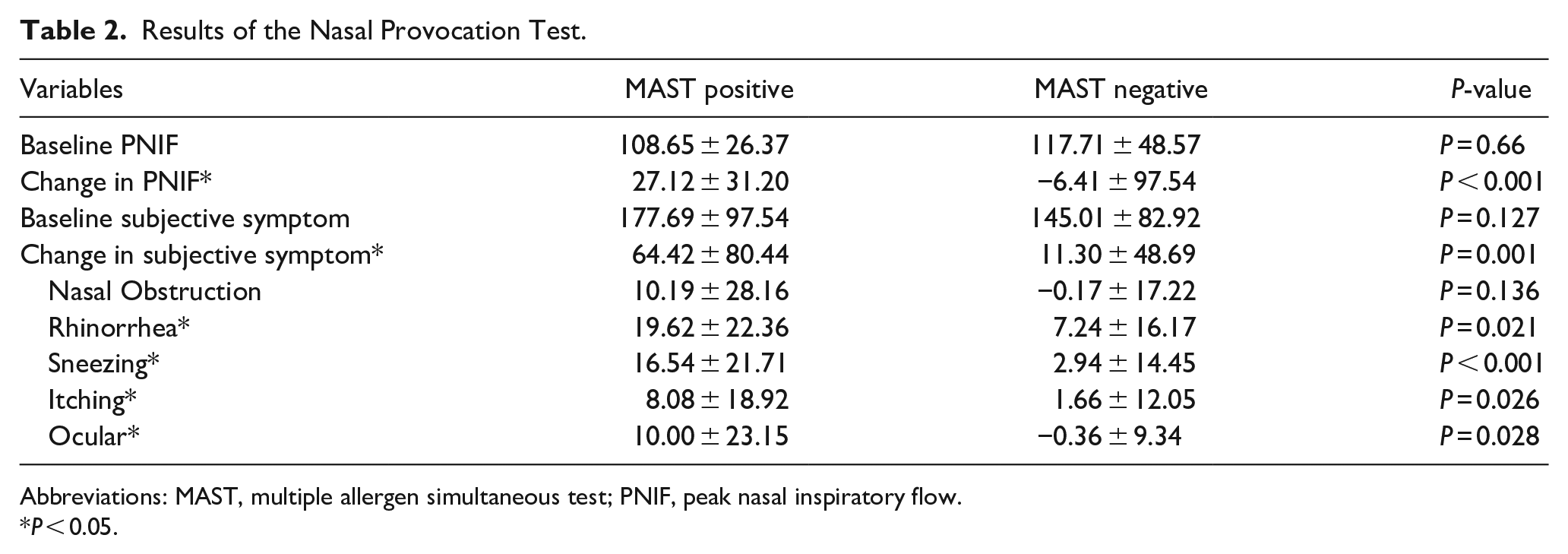
Abbreviations: MAST, multiple allergen simultaneous test; PNIF, peak nasal inspiratory flow.
P < 0.05.
Correlation of MAST and NPT
ROC curve analysis for change in PNIF between pre- and post-allergen nasal challenge had an AUC value of 0.722 (P < 0.05), and changes in subjective symptoms between pre- and post-allergen nasal challenge score had an AUC value of 0.713 to the result of MAST (P < 0.05) (Figure 1). The ECP level had an AUC value of 0.516 (P > 0.05) and the eosinophil count had an AUC value of 0.624 (P > 0.05). Among the subjective symptoms, changes in nasal obstruction between pre- and post-allergen nasal challenge had an AUC value of 0.596 (P > 0.05), changes in rhinorrhea had an AUC value of 0.650 (P < 0.05), sneezing had an AUC value of 0.728 (P < 0.05), itching had an AUC value of 0.622 (P < 0.05), and ocular discomfort had an AUC value of 0.600 (P < 0.05) (Figure 2).
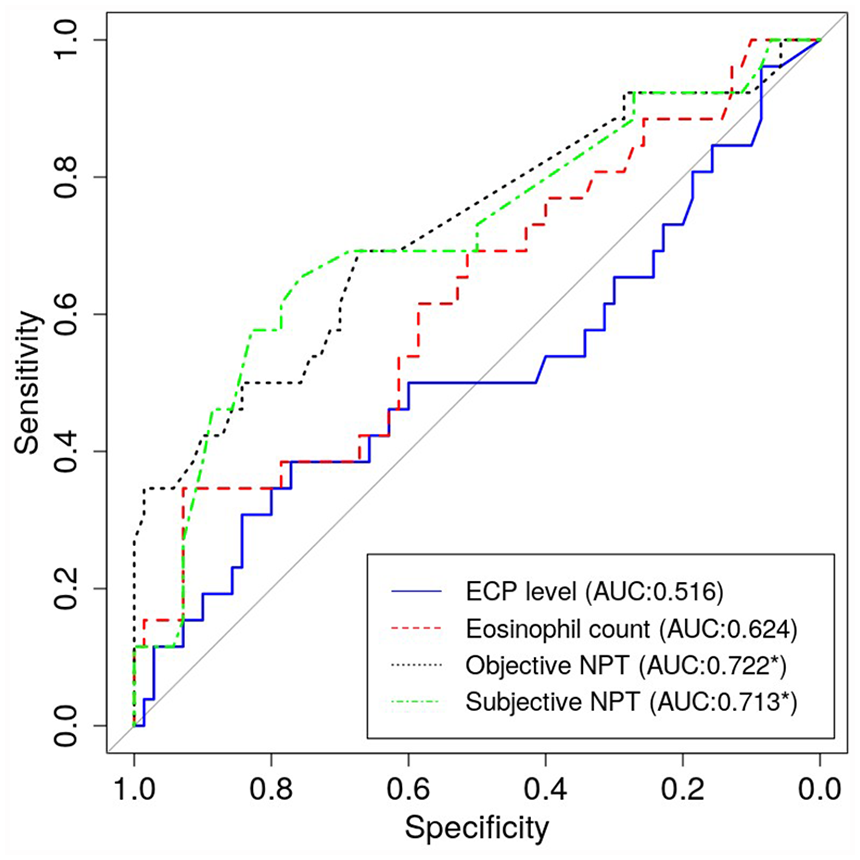
Receiver operating characteristic curve analysis for NPT, ECP level, and eosinophil count. The AUC was obtained and referenced against the result of multiple allergen simultaneous tests.
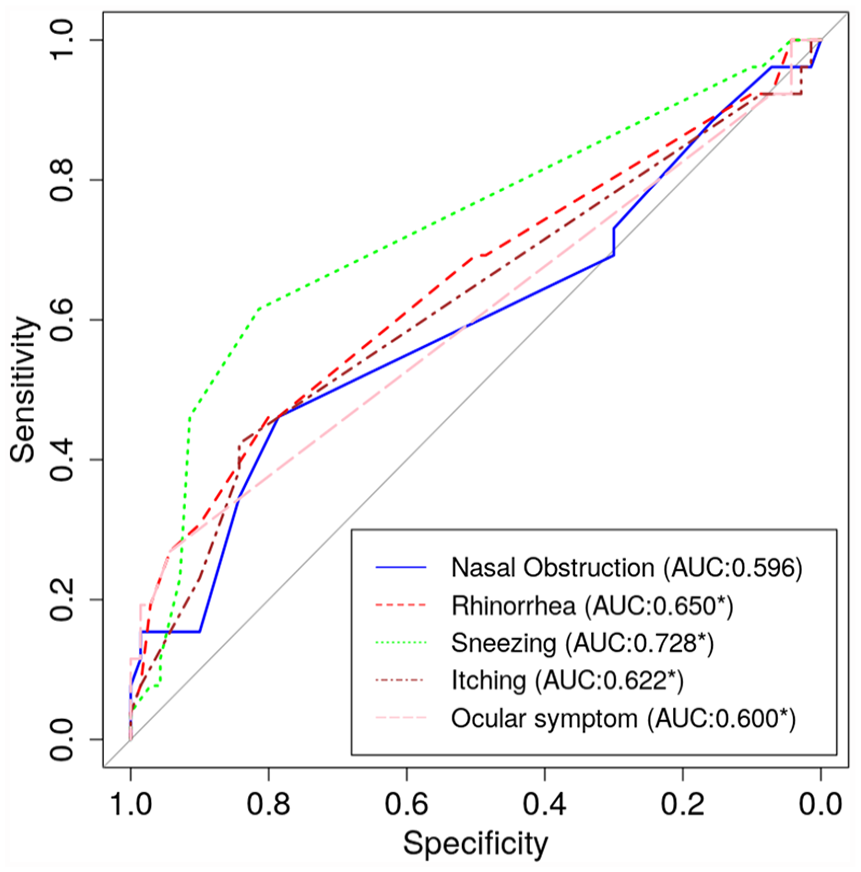
Receiver operating characteristic curve analysis for subjective symptom score of NPT. The AUC was obtained and referenced against the result of multiple allergen simultaneous tests.
Discussion
The diagnosis of AR is based on a typical history of allergic symptoms and diagnostic tests. The first step in diagnostic testing is nasal examination, followed by in vivo or in vitro tests. In vivo test includes a skin test such as skin prick test or allergen NPT, and in vitro test includes the determination of allergen-specific IgE in serum and inflammatory mediators at the local level (in the nasal mucosa or nasal lavage fluid). 18 As with a skin prick test, detection of allergen-specific IgE in serum is thought to be an indicator of the degree of IgE-mediated sensitivity to a specific allergen, 19 and many studies have proved that both of these tests have good sensitivity and specificity.20,21
Allergen NPT is a medical procedure aimed at reproducing the inflammatory reaction induced by allergens in the nasal mucosa. 18 The procedure of NPT involves the intranasal application of specific doses of allergens after exclusion of nasal hyperreactivity. 18 It is known to be a safe and reproducible test, and is considered to be useful in the identification of specific allergens that are responsible for rhinitis symptoms.18,22
Studies regarding the correlation between the skin prick test and NPT have been reported. Changes in subjective symptoms and objectively measured outcomes after allergen application were significantly correlated with the results of the skin prick test.4,6,23 It has been reported that changes in rhinitis symptoms such as nasal obstruction, rhinorrhea, itching and change of total nasal volume, and minimal cross-sectional area after nasal allergen provocation were significant indicators of skin test-based diagnosis of AR. 6 Rhinorrhea, sneezing, and itching symptoms with acoustic rhinometry parameters after a house dust mite extract intranasal challenge were significantly different between AR and healthy volunteers, who were grouped based on the skin prick test. 14 Furthermore, the degree of change after NPT was significantly associated with the degree of allergic skin test reaction. 24 Compared with the skin prick test, the relationship between the presence or the level of sIgE in serum and NPT was heterogeneously reported. Haxel et al 25 reported that a positive sIgE result was verified by the NPT in 69% of cases in DP and 70% in DF, and concluded that the level of sIgE was significantly associated with the result of NPT. The positive NPT result correlated with the presence of rhinitis symptoms and sIgE in serum. 26 However, in another study, NPT showed no significant correlation with serum total and sIgE levels to DP. 11
In this study, using the results of MAST as a reference standard, the AUC value of subjective symptom change after nasal allergen challenge was 0.713 and objective PNIF change after nasal allergen challenge was 0.722 for DP/DF (Figure 1). We found that a cutoff value of a subjective TNSS change of more than 17.5 had a sensitivity of 68.6%, and a specificity of 69.2%, while a cutoff value of an objective PNIF change of more than 6.51 had a sensitivity of 67.1% and a specificity of 69.2%. These findings suggest that the outcome of NPT is closely associated with MAST.
Interestingly, it has been reported that the relationship between sIgE and NPT outcome was different between younger and older adults. 27 The positive predictive value of NPT for sIgE was lower in older adults than in younger adults, and the negative predictive values of NPT for sIgE were higher in older adults. 27 In our study, we did not evaluate and compare the relationship between sIgE and NPT according to age; thus, the differential relationship between sIgE and NPT according to age should be further evaluated in future studies.
Among the subjective rhinitis symptoms, the change in sneezing symptoms after the allergen challenge showed the highest AUC value for the MAST reference result. In contrast to our findings, nasal obstruction and rhinorrhea remained worse in the AR group after NPT, and a similar but less marked difference was observed for sneezing and itching, which was performed in the same country against the same allergen (house dust mite). 28 The difference between this previous study and the current study is the concentration of allergen. We applied a lower concentration of the allergen stock solution than that used in the previous study. Therefore, it is possible that the change in specific nasal symptoms could be influenced by the concentration of the allergen. As both these studies were based on a relatively small number of enrolled participants, a large population-based study might be helpful in assessing the relationship between specific nasal symptoms or changes in PNIF and allergen concentration.
Nonspecific hyperreactivity was assessed by measuring the PNIF response after the saline challenge. As a result, a total of 9 patients were defined as nonspecific hyperreactivity cases in PNIF and rhinitis symptoms after the saline challenge, 4 were included in the MAST-positive group, and 5 were included in the MAST-negative group. Nonspecific hyperreactivity refers to the aggravation of nasal symptoms after irritation by nonspecific, nonallergenic stimuli, such as sudden changes in temperature, cold air, and perfume. 29 Nonspecific hyperreactivity is known to be a common feature in patients suffering from both allergic and nonallergic rhinitis, and the mechanisms have not been clearly proved. 30 So far, limited efforts have been invested into gaining better insight into the underlying pathophysiology of nonspecific hyperreactivity. As a consequence of the neglect of nonspecific hyperreactivity, patients with nonspecific hyperreactivity might be suboptimally controlled and/or dissatisfied with the current treatment. As nonspecific hyperactivity can be measured using NPT, we suggest that NPT should be more actively utilized in the clinical field.
Our study had several limitations. First, we classified the patients into MAST-positive or negative groups, and we did not consider discerning nonallergic rhinitis such as local AR. In the current study, we have found that some patients with positive NPT showed low level of sIgE. In those cases, local AR could be considered as one of potential diagnosis. Additional diagnostic tests such as detecting specific IgE, ECP, and histamine levels in nasal secretions could be helpful in diagnosis of local AR. Furthermore, we did not evaluate the different relationship between NPT and MAST according to the specific IgE levels. In this study, we found that all MAST-positive patients did not show the PNIF change ≥40% at 15 minutes after the challenge (Table 2), which means that MAST positivity does not directly guarantee the positive result of NPT. Further, large population-based study needs to compensate these limitations. Second, we enrolled participants who were subjectively sensitized to DP or DF only and did not consider other allergens. As we only applied DP and DF mixtures to the nasal cavity during NPT, we enrolled participants who were seropositive for IgE against DP and DF. It is possible that MAST and NPT may show different correlations depending on the type of allergen.
Conclusion
In conclusion, this study is the first to evaluate the relationship between MAST and NPT using house dust mite antigen extracts in a Korean population. We found that the MAST results were significantly associated with subjective and objective changes after the allergen nasal challenge. Future studies regarding antigen type- or antigen concentration-specific responses after NPT are warranted.
Footnotes
Acknowledgements
IYK helped us in performing the statistical analysis of this study.
Author Contributions
Concept and design: H.J. Min; Acquisition, analysis, or interpretation of data: H.J. Min; Drafting of the article: H.J. Min, K.S. Kim; Critical revision of the article for important intellectual content: K.S. Kim; Statistical analysis: H.J. Min; andSupervision: K.S. Kim.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (2022R1F1A1063720). This research was supported by the Chung-Ang University Research Grants in 2022.
Ethical Considerations
This study was approved by the institutional review board of Chung-Ang University Hospital (2204-013-19414), and the review board waived the requirement for informed consent.
Data Availability Statements
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
