Abstract
Introduction
Laryngopharyngeal reflux (LPR) is a general term for the reflux of gastroduodenal contents into the laryngopharynx, oropharynx, and even the nasopharynx, causing a variety of signs and symptoms, and is an inflammatory response of the upper airways involving gas and/or fluid reflux of gastroduodenal contents into the upper airway mucosa. 1,2 The inflammatory response is initially caused by direct stimulation of pepsin, trypsin, and other enzymatic molecules, and the gastroduodenal contents may also irritate the lower esophagus, stimulating chemoreceptors and causing reflex coughing and hypersecretion of the laryngopharyngeal mucosa, but the exact mechanism requires more evidence. 3 -6 There is no gold standard for the diagnosis and treatment of LPR, and its pathogenesis and treatment are still controversial. 7 -10 LPR has a large number of nonspecific clinical manifestations, such as chronic cough, excessive mucus production, dysphonia, dysphagia, and throat clearing. 11 -13 It has been suggested that the development of LPR may be associated with a high-fat, low-protein diet, stress or anxiety, and tobacco. 14 -17 An epidemiologic study in China reported 9182 out of 90,440 ear–nose–throat (ENT) patients with LPR (10.15%). 18 According to a Brazilian survey, the prevalence of LPR was estimated to be 26.8% of ENT visits. 19
Given the complexity of the incidence and symptoms of LPR, our review will attempt to focus on the current treatment of LPR through a simple algorithm and suggest several possible future directions.
Materials and Methods
A review was performed utilizing PubMed Databases to identify original articles about LPR between the years 2000 and 2023. The search was performed using the search string: “Reflux, Laryngopharyngeal or Regurgitation, Gastric or Gastric Regurgitation or Supraesophageal Gastric Reflux or Gastric Reflux, Supraesophageal or Reflux, Supraesophageal Gastric or Supraesophageal Gastric Reflux (SEGR) or Gastric Reflux, Supraesophageal (SEGR) or Reflux, Supraesophageal Gastric (SEGR) or Laryngopharyngeal Reflux.”
Results
Health Education
Health education for LPR is essential for the prevention and treatment of this condition. Such education can help patients gain an understanding of LPR, enhance patient compliance, and improve the efficacy of treatment. A study by Pisegna et al 20 completed a face-to-face, semi-structured interview, a questionnaire, and the Reflux Symptom Index (RSI) with 51 patients and found that 62.7% (n = 32/51) of patients did not take their medication as prescribed in the absence of health education. This shows that proper health education plays a key role in the prevention and treatment of LPR. 20
Diet and Lifestyle
Diet and lifestyle changes are often used as an important part of comprehensive LPR treatment. Given that the development of LPR may be associated with a high-fat, low-protein diet, stress or anxiety, and tobacco, and that obesity may lead to more severe LPR disease, lifestyle and dietary changes are critical. 21 According to Lechien et al 22 gastroduodenal enzyme reflux could alter the laryngopharyngeal and oral microbiome, impair recovery, and affect mucosal maintenance. They suggested that the patient’s diet is critical, as it might influence the composition of the microbiome. 22
Currently, it is recommended that all patients be placed on an anti-reflux diet and reassessed after 3 months. 23,24 A low-fat, low-quick-release sugar, high-protein, alkaline, and plant-based diet is a cost-effective treatment approach. 25 There is a need for more studies on how different types of diets affect LPR patients. Meanwhile, one study reviewed four patients with persistent reflux symptoms and found that eliminating allergenic foods from the diet appeared to be effective in treating LPR, which raises food sensitivity as a possible factor in patients with intractable LPR. 26 It is worth noting that motivating patients to change their lifestyle and dietary habits is a more demanding task, and much depends on the quality of health education provided by the physician, thus placing higher demands on their abilities. Overall, it is worthwhile to incorporate lifestyle and dietary changes into the treatment protocol to maximize symptom improvement.
Pharmacotherapy
Pharmacotherapy is the mainstay of treatment for LPR and includes common proton pump inhibitor (PPI), potassium competitive acid blocker (P-CAB), H2 receptor antagonist (H2RA), alginate, and prokinetic drugs. In addition, potential drugs include baclofen. The details of these drugs are shown in Table 1.
Pharmacotherapy Options for LPR.
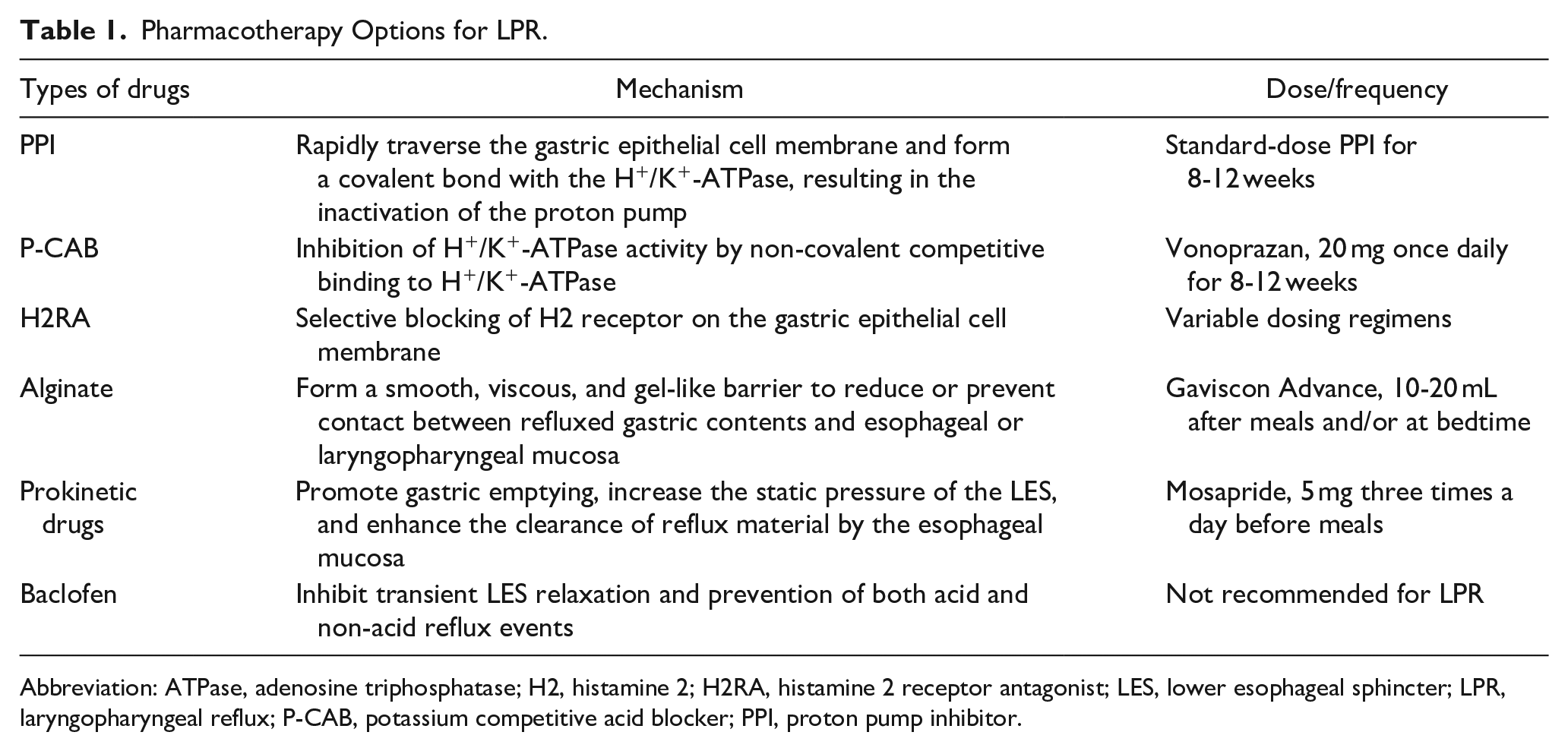
Abbreviation: ATPase, adenosine triphosphatase; H2, histamine 2; H2RA, histamine 2 receptor antagonist; LES, lower esophageal sphincter; LPR, laryngopharyngeal reflux; P-CAB, potassium competitive acid blocker; PPI, proton pump inhibitor.
PPI. Standard-dose PPI is now widely used as the recommended first-line treatment for LPR and should also be used empirically in patients with suspected LPR. The results of a meta-analysis involving 13 randomized controlled trials (including 831 patients with LPR) revealed that patients who received PPI experienced a significant improvement in their total RSI. 27 However, it has been reported in the literature that up to 40% of patients with LPR do not experience symptomatic relief with PPIs. 28 In some studies, PPIs were not superior to placebo in the treatment of individuals with suspicion of LPR. 29,30 According to Brazilian otolaryngologists 19 and members of the American Broncho-Esophageal Association (ABEA), 31 twice-daily PPI use for 2-3 months is the most effective treatment. European general practitioners prefer once-daily PPIs. 32 East Asian otolaryngologists are more likely to use once-daily PPIs, while West Asian otolaryngologists prefer twice-daily PPIs. 13 In terms of precautions for use, long-term use of PPIs may increase the risk of gastric tumors and acute nephritis. 33 The use of PPIs should be carefully considered in the elderly, as drug–drug interactions and adverse reactions are more common and the risk of medication use is greater. 34 It is common to recommend an increase in dose or a change in the type of PPI if the significant symptomatic improvement is not achieved with standard doses. Therefore, it is clear that the effectiveness of PPI acid suppression in the treatment of LPRD remains controversial. However, PPI is still the treatment of choice for LPR due to its easy availability, non-invasiveness, and affordability.
P-CAB. P-CAB is a novel gastric acid inhibitor whose mechanism of action differs from that of PPIs in that it inhibits H+/K+-ATPase activity by non-covalently (hydrogen bonding and ionic robustness) competitively binding to H+/K+-ATPase with K+. It has a strong inhibitory effect on gastric acid production because it has a long plasma half-life, is stable in an acidic environment, and can remain in the secretory tubules for a long time; it can inhibit both resting and activated proton pumps. Several studies have demonstrated that P-CAB does not perform worse than PPI in terms of the rate of esophagitis mucosal healing and the relief of reflux symptoms. 35 -37 One study showed that 20 mg vonoprazan once daily had a more potent acid-suppressive effect than 20 mg or 40 mg rabeprazole twice daily and maintained intragastric pH > 4 throughout the day. 38 These results suggest that P-CAB has the potential to take the place of PPI. The recommended dose of P-CAB is 20 mg once daily, independent of meals, which makes it easier to use than PPIs, improves patient compliance, and reduces the risk of patients not taking adequate doses. However, clinical evidence for the use of P-CAB in the treatment of LPR is still sparse, and studies in children, the elderly, and pregnant and breastfeeding women are lacking, so P-CAB should be used with great caution.
H2RA. H2RA is the earliest acid-suppressing drug, but it has been used as a second-line treatment because of its shorter duration of action (4-8 hours) and weaker acid-suppressing effect than PPI. 39 However, it has been found in some studies that adding H2RA for nighttime to twice-daily PPI therapy could improve acid control by reducing nocturnal acid breakthroughs. 40 -42 It also found that H2RA tolerance can occur in a very short period of time, which diminishes the effect of H2RA. 41,42 In summary, adding H2RA for nighttime to twice-daily PPI therapy may be considered for patients with nocturnal reflux symptoms.
Alginate. By forming a smooth, viscous, and gel-like barrier, alginate reduces or prevents contact between refluxed gastric contents and esophageal or laryngopharyngeal mucosa. 43 Pepsin activity is significantly inhibited by alginate as well. 43 It is an effective tool for non-acid reflux and mixed reflux that does not respond well to PPIs. One study included 100 outpatients with LPR. 12 Each patient received magnesium alginate–simethicone (20 mL/three times daily) for 2 months. 12 The patient’s symptoms were assessed at baseline and 15, 30, and 60 days after treatment using a visual analog scale. 12 Finally, alginate was found to significantly relieve patients’ dysphonia, dysphagia, and cough, and no clinically relevant adverse events were reported, suggesting that alginate is effective and safe in the treatment of LPR. 12 There were 50 patients with LPR randomized into two treatment groups in a non-inferiority randomized controlled trial: (a) alginate suspension (Gastrotuss® 20 mL three times a day) and (b) PPI group (omeprazole 20 mg once daily). 44 After 2 months of treatment, both groups experienced a significant decrease in mean RSI and reflux finding score (RFS), and there was no significant difference in RSI change or RFS change between the groups, indicating that alginate is not less effective than PPI. 44 Alginate is fast-acting, long-lasting, inexpensive, and has no significant adverse effects, making it a backup option when PPI treatment is unsatisfactory. When used for LPR, it is recommended that Gaviscon Advance be taken 10 to 20 mL after meals and/or at bedtime. However, in certain countries, such as China, alginates have not yet entered the market. Alginates are not readily available in these countries.
Prokinetic drugs. Prokinetic drugs such as mosapride can promote gastric emptying, increase the resting pressure of the lower esophageal sphincter (LES), and enhance the clearance of reflux material by the esophageal mucosa. The combination of PPIs and prokinetic drugs can improve the therapeutic effect. Some studies found that the combination of drugs can effectively improve symptoms such as hiccups, postprandial bloating, and nausea. 45,46 Clinicians can use prokinetic drugs in combination with PPIs as a widely available measure. However, there is a limitation of mosapride in practice. Although patients take PPIs twice a day before meals, patients should take mosapride three times a day before meals, which can lead to low adherence problems due to confusion.
Baclofen. Baclofen inhibits transient LES relaxation and may prevent both acid and non-acid reflux events. 47 Based on results of a meta-analysis involving a total of 283 subjects across nine studies, baclofen significantly reduced the morbidity of gastroesophageal reflux disease (GERD), the duration of reflux episodes, and the incidence of transient LES relaxation compared with placebo in the treatment of GERD. 48 In another study, 32 subjects treated with a combination of PPI and baclofen for 1 month showed significant improvement in throat discomfort, globus, and total RSI score. 47 In comparison to baseline scores, there were notable improvements in voice, throat, coughing while lying down, dyspnea, globus, heartburn, and total RSI score after 3 months of treatment. 47 However, there was no substantial improvement in postnasal drip, dysphagia, or cough. 47 This suggests that baclofen may also have some potential for use in the treatment of LPR. Given the paucity of research and clinical practice on the use of baclofen for LPR, however, it should not be considered for LPR at this time because of its uncertain efficacy.
External upper esophageal sphincter compression device
The external upper esophageal sphincter (UES) compression device is a relatively new LPR treatment available. It generates 20-30 mmHg of intraluminal esophageal pressure by applying cricoid cartilage pressure and strengthens the UES to reduce reflux. As an adjunct to PPI therapy, a recent study assessed the effectiveness of an external UES compression device in patients with LPR symptoms. 49 The study was split into two phases: the first used a double-dose PPI for 4 weeks, and the second used an external UES compression device and PPI for the same period of time. 49 Efficacy was assessed at the end of each phase using the RSI and GERD questionnaire. 49 The results demonstrated that the combination of an external UES compression device and PPI provided better symptomatic relief in patients with LPR compared to PPI alone. 49 The external UES compression device is recommended for nighttime sleep. Limited clinical trial data exist for this device, but it has great potential for future use because it is a well-tolerated, non-invasive device that can be effectively combined with drug therapy. Unfortunately, as a novel therapy at this stage, the device is not yet widely available, which means that it is not easily accessible to many LPR patients.
Surgery
Surgical treatment is not a stable option for LPR and its role in LPR is unclear. The nine trials included in a review analysis all demonstrated that anti-reflux surgery could be considered for individuals who have not responded to PPI therapy and laparoscopic fundoplication is the preferred surgical treatment for treating LPR symptoms. 50 Based on a review of 27 observational studies, researchers found that anti-reflux surgery for LPR can vary greatly in effectiveness, ranging from 10% to 93%, 51 and some researchers also found that fundoplication may not be effective for LPR owing to variations in diagnostic methods and metrics used to evaluate its effectiveness. 52 On the other hand, there is a dearth of literature comparing the differences in the efficacy of surgical and pharmacological treatments. Therefore, surgical treatment should not be considered as a routine option for LPR at this time, and factors influencing the efficacy of surgery should be carefully explored in the future.
Discussion
LPR, a prevalent disorder in otolaryngology, poses a challenge to both clinicians and patients. The precise mechanism of LPR pathology is unknown and accurate diagnosis remains elusive. There are numerous treatment options for LPR, such as health education, lifestyle, and dietary modifications, pharmacotherapy, external UES compression devices, and even surgery. As illustrated in Figure 1, we attempt to propose a simple diagnostic and treatment algorithm for patients with suspected LPR based on recent research, which may be helpful to clinicians. PPI remains the simple first-line treatment for LPR and a clinician’s first choice. However, we also face the challenge that many patients do not experience an effective improvement in their symptoms. And there is a lack of effective options for drug-tolerant LPR patients and elderly LPR patients. Therefore, more evidence is needed to discover effective therapies. P-CAB and alginate could be considered as options when PPI therapy is unsatisfactory and external UES compression devices have sufficient potential value to be given appropriate attention. We should also have a better understanding of the pathogenesis and diagnosis of LPR, which may help us to propose more effective and simpler treatment options.
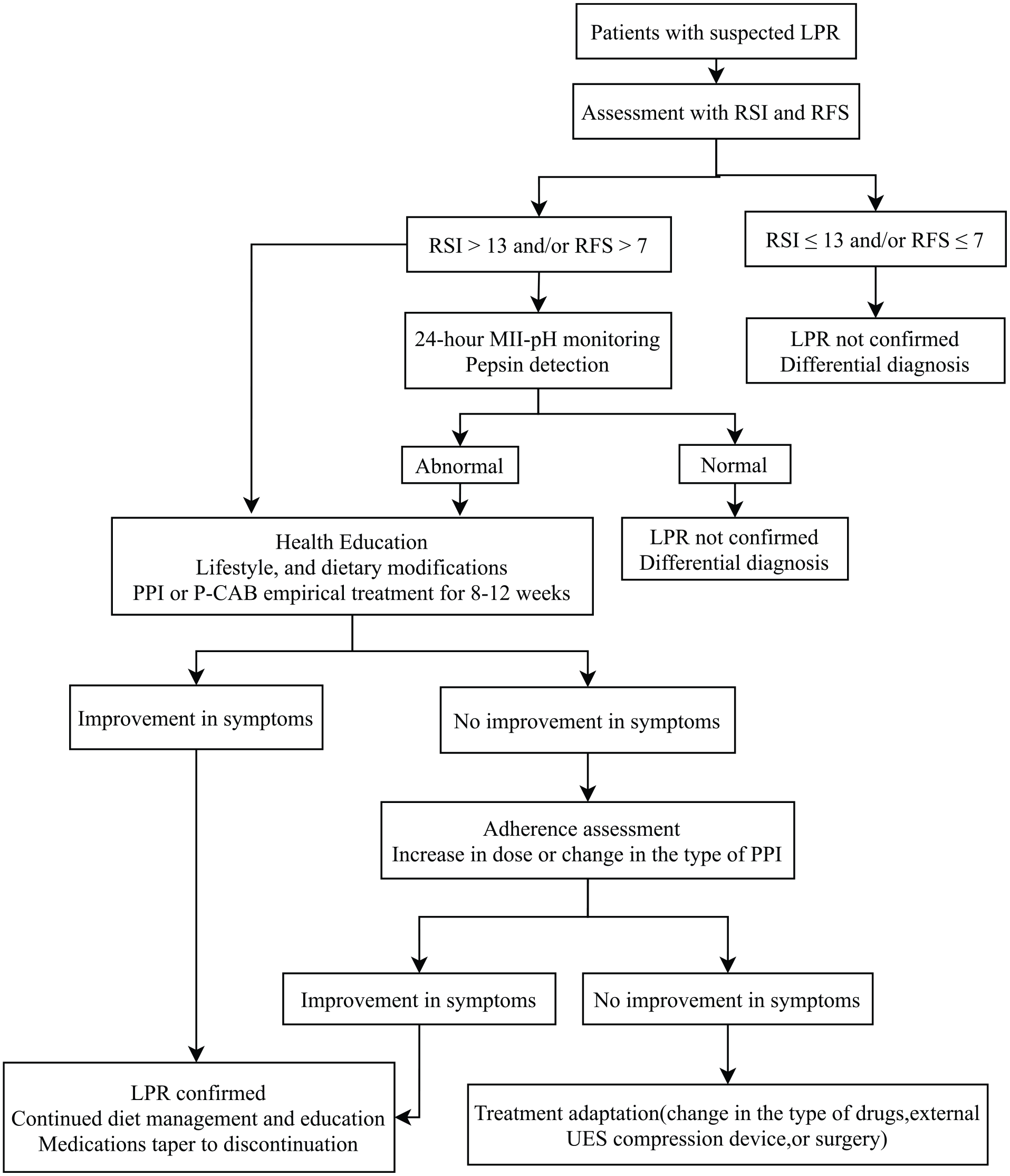
Proposed diagnostic and therapeutic algorithm of suspected laryngopharyngeal reflux.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
