Abstract
A 72-year-old Japanese man with a 4-month history of hoarseness and 1-week history of difficulty breathing was admitted to our department. He underwent right total nephrectomy for primary clear cell-type renal cell carcinoma (RCC) 6 years ago and left partial nephrectomy for the metastasis 4 years ago. Flexible laryngeal fiberscope examination revealed bilateral subglottic stenosis without obvious mucosal lesions. Enhanced computerized tomography (CT) scan of the neck revealed that the cricoid cartilage had become bilaterally expansive and tumorous lesion exhibiting enhancement. We performed tracheostomy on the appointed day and biopsied the tumor in the cricoid cartilage via the skin incision. Results of histologic and immunohistologic examinations for AE1/AE3, CD10, and vimentin positivity were consistent with clear cell-type RCC. Chest and abdomen CT scans revealed a few tiny metastases in the upper lobe of the left lung but no recurrence in the abdomen. At 2 weeks from the day of tracheostomy, total laryngectomy was performed. Postoperatively, the patient was treated transorally with axitinib (10 mg/day) and as of 12 months he remains alive with unchanging lung metastasis. Next-generation sequencing of targeted regions using a surgical specimen from the tumor revealed a frameshift mutation in the von Hippel-Lindau gene (p.T124Hfs*35) and a missense mutation in the TP53 gene (p.H193R).
Introduction
Renal cell carcinoma (RCC) accounts for 3% of adult carcinomas and 85% of primary kidney tumors 1 and occurs frequently in older adults. 2 Clear cell-type RCC is diagnosed in 75 to 85% of RCC patients. 3 Between 20 and 30% of RCC patients reportedly have a metastatic lesion at the time of the initial diagnosis, and 20 to 30% of patients undergoing nephrectomy for localized RCC develop metastatic disease. 4 Common metastatic sites of RCC include the lungs (45.2%), bone (29.5%), lymph nodes (21.8%), liver (20.3%), adrenal glands (8.9%), and brain (8.1%). 5 Metastasis of RCC to the head and neck is rare, occurring in only 3.6 to 7.6% of the patients with RCC, with 3% of these metastases involving the thyroid.6,7 Laryngeal tumors metastasizing from other malignant tumors generally comprise 0.09 to 0.4% of all larynx tumors, including cutaneous malignant melanoma, lung carcinoma, colon adenocarcinoma, and RCC. 8 Metastasis of RCC to the larynx is extremely rare, with only 13 cases reported in the English-language literature since 1987.9 -20 Here, we report an unusual case of metastasis to the cricoid cartilage from RCC presenting with subglottic stenosis, necessitating total laryngectomy.
Case Report
A 72-year-old Japanese man with a 4-month history of hoarseness and 1-week history of difficulty breathing was admitted to our department. He underwent right total nephrectomy for primary clear cell-type RCC 6 years ago and left partial nephrectomy for the metastasis 4 years ago. The patient was regarded as completely cured and followed with no adjuvant treatment for RCC. Flexible laryngeal fiberscope examination revealed bilateral subglottic stenosis without obvious mucosal lesion (Figure 1A). Bilateral vocal cord mobility was conserved but restricted. Enhanced computerized tomography (CT) scan of the neck revealed that the cricoid cartilage had become a bilaterally expansive and tumorous lesion exhibiting enhancement with a left-right diameter of 35 mm (Figure 1B and C). The thyroid gland and thyroid cartilage were not affected by the tumor. We performed tracheostomy on the appointed day and biopsied the tumor in the cricoid cartilage via the skin incision. The tumor was highly hemorrhagic, which caused difficulty in hemostasis with electrocoagulation; bleeding was finally controlled by gauze compression.
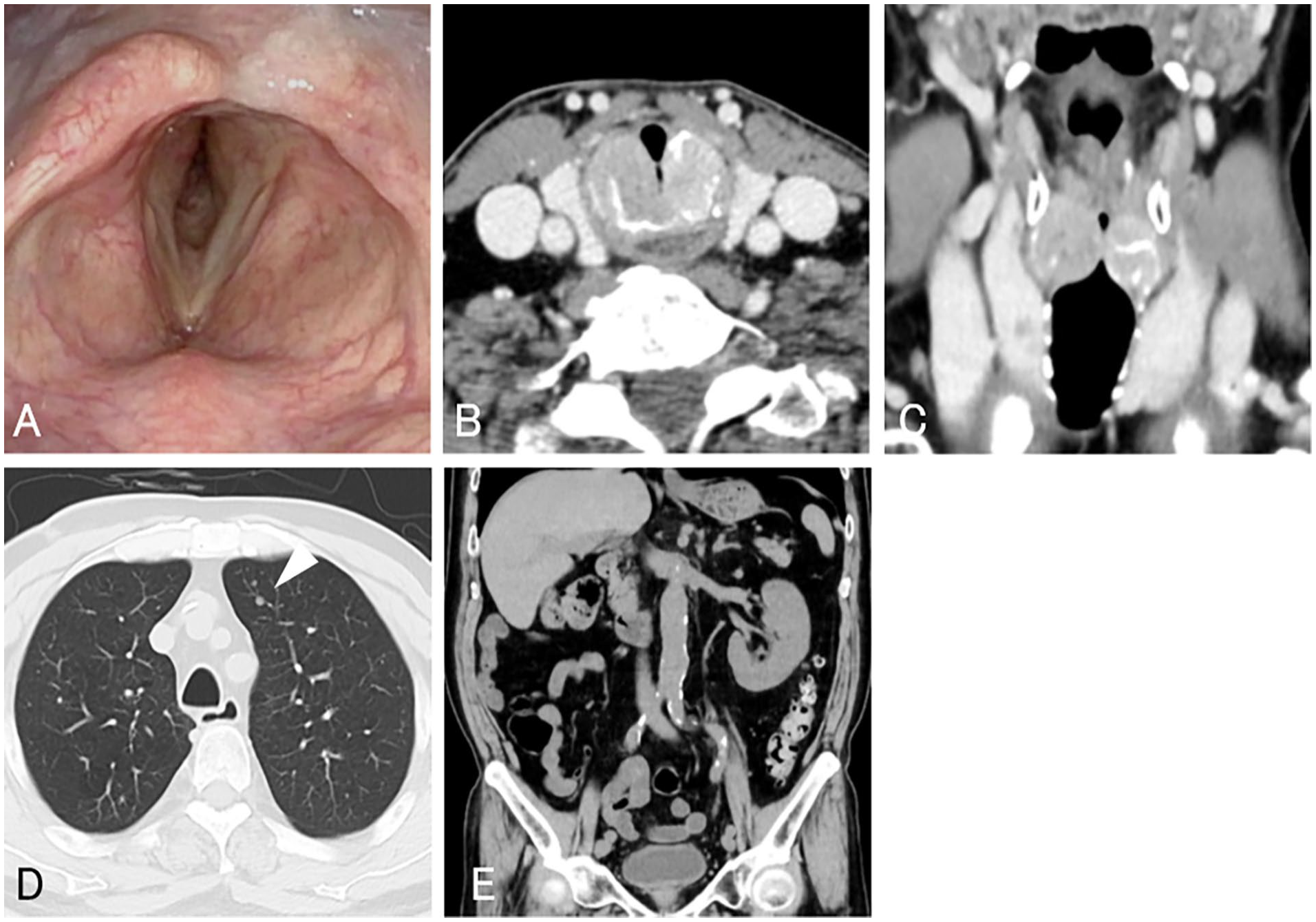
Laryngeal fiberscope examination revealed bilateral subglottic stenosis without obvious mucosal lesion (A). Enhanced CT scan of the neck revealed that the cricoid cartilage had become a bilaterally expansive and tumorous lesion exhibiting enhancement with a left-right diameter of 35 mm in the axial (B) and coronal (C) sections. Chest CT scan revealed a 3-mm nodule in the upper lobe of the left lung (triangle, D). Abdomen CT scan revealed the right kidney was absent and no lymph node swelling was observed in the abdomen (E). CT, computerized tomography.
Histologically, hematoxylin and eosin staining demonstrated the presence of neoplastic cells with clear cytoplasm that proliferated in an alveolar and trabecular pattern (Figure 2A). Tumor cells were positive for AE1/AE3 (Figure 2B), CD10 (Figure 2C), vimentin (Figure 2D), and epithelial membrane antigen (EMA). Staining of tumor cells for cytokeratin (CK)7, CK20, and p40 was negative. The results of histologic and immunohistologic examinations were consistent with clear cell-type RCC.
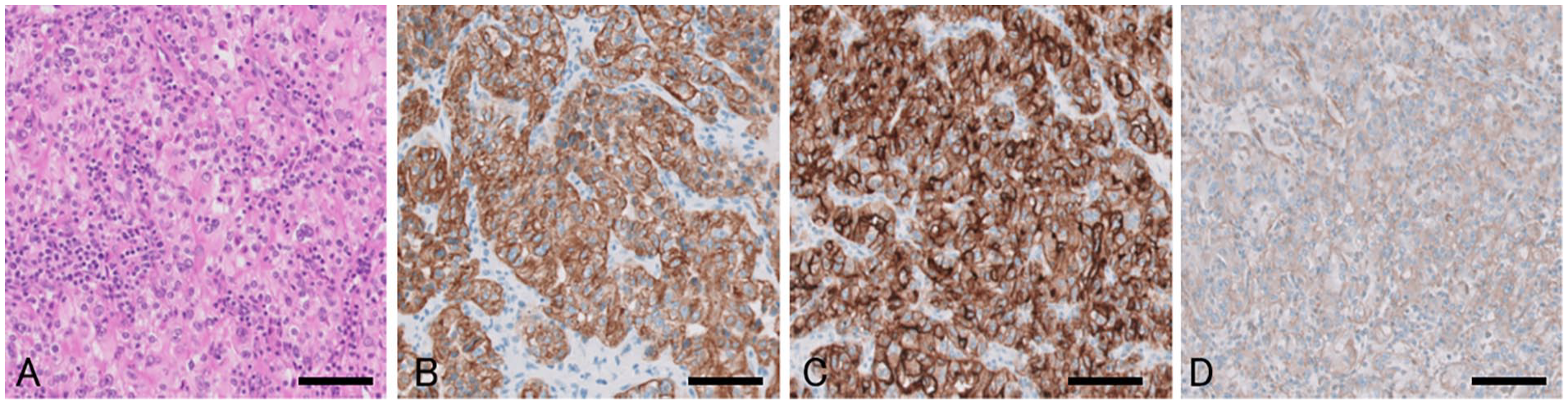
Hematoxylin and eosin staining demonstrated the presence of neoplastic cells with clear cytoplasm proliferating with an alveolar and trabecular pattern (A). Tumor cells were positive for AE1/AE3 (B), CD10 (C), and vimentin (D). All scale bars: 100 μm.
Chest CT scan revealed two 3-mm nodules in the upper lobe of the left lung, which were diagnosed as lung metastases (Figure 1D). Abdomen CT scan revealed that the right kidney was absent, and no lymph node swelling was noted in the abdomen (Figure 1E). Fluorodeoxyglucose (FDG)‑positron emission tomography (PET)/CT revealed uptake in the cricoid cartilage and no uptake in the kidney or abdomen.
After the tracheostomy and biopsy, bleeding from the tumor in the cricoid cartilage recurred, and decannulation could not be performed. Therefore, at 2 weeks after the day of tracheostomy, total laryngectomy with total thyroidectomy was performed under general anesthesia. The patient’s clinical course proved uneventful thereafter. Postoperatively, the patient was treated transorally with axitinib (10 mg/day) and as of 12 months he remains alive without local recurrence of the tracheostoma and with unchanging tiny lung metastases. Next-generation sequencing (NGS) panel testing of targeted regions in 160 cancer-related genes using a surgical specimen from the tumor in the cricoid cartilage was performed as previously described 21 and it revealed a frameshift mutation in the von Hippel-Lindau (VHL) gene (p.T124Hfs*35) and a missense mutation in the TP53 gene (p.H193R).
Discussion
RCC can metastasize to the head and neck regions, including the larynx and thyroid, bypassing the lungs via the valveless paravertebral venous plexus of Batson. 22 Recurrence may develop several years after nephrectomy of the primary RCC without specific signs or symptoms.
We searched for the cases of RCC metastasis to the larynx, including the cricoid cartilage, reported in the English-language literature since 1987. To our knowledge, only 13 such cases have been reported, and these cases are summarized in Table 1.9 -20 The median age of these patients was 57 years (range, 48-83 years). All the patients were male. Seven patients had already undergone nephrectomy for primary RCC, with a median duration from nephrectomy to onset of laryngeal symptoms of 7 years (range, 3.5-14 years). The other 6 patients were diagnosed with RCC for the first time by histologic diagnosis of a laryngeal tumor. A variety of laryngeal symptoms were described for the 13 patients and included hoarseness in 8 patients (62%), dyspnea in 2 (15%), hemoptysis in 2 (15%), dysphonia in 2 (15%), foreign body sensation in 2 (15%), cough in 2 (15%), and pain in 2 patients (15%), with a median duration from onset of laryngeal symptoms to hospital consultation of 2 months (range, 0.5-12 months). Location of the metastatic laryngeal tumor from RCC was described and included vocal cord in 3 patients (23%), subglottis in 3 patients (23%), thyroid cartilage in 2 patients (15%), and cricoid cartilage in 2 patients (15%). The median size of the metastatic laryngeal tumor from RCC was 15 mm (range, 4-57 mm).
Clinical characteristics of 13 patients with renal cell carcinoma metastasizing to the larynx.
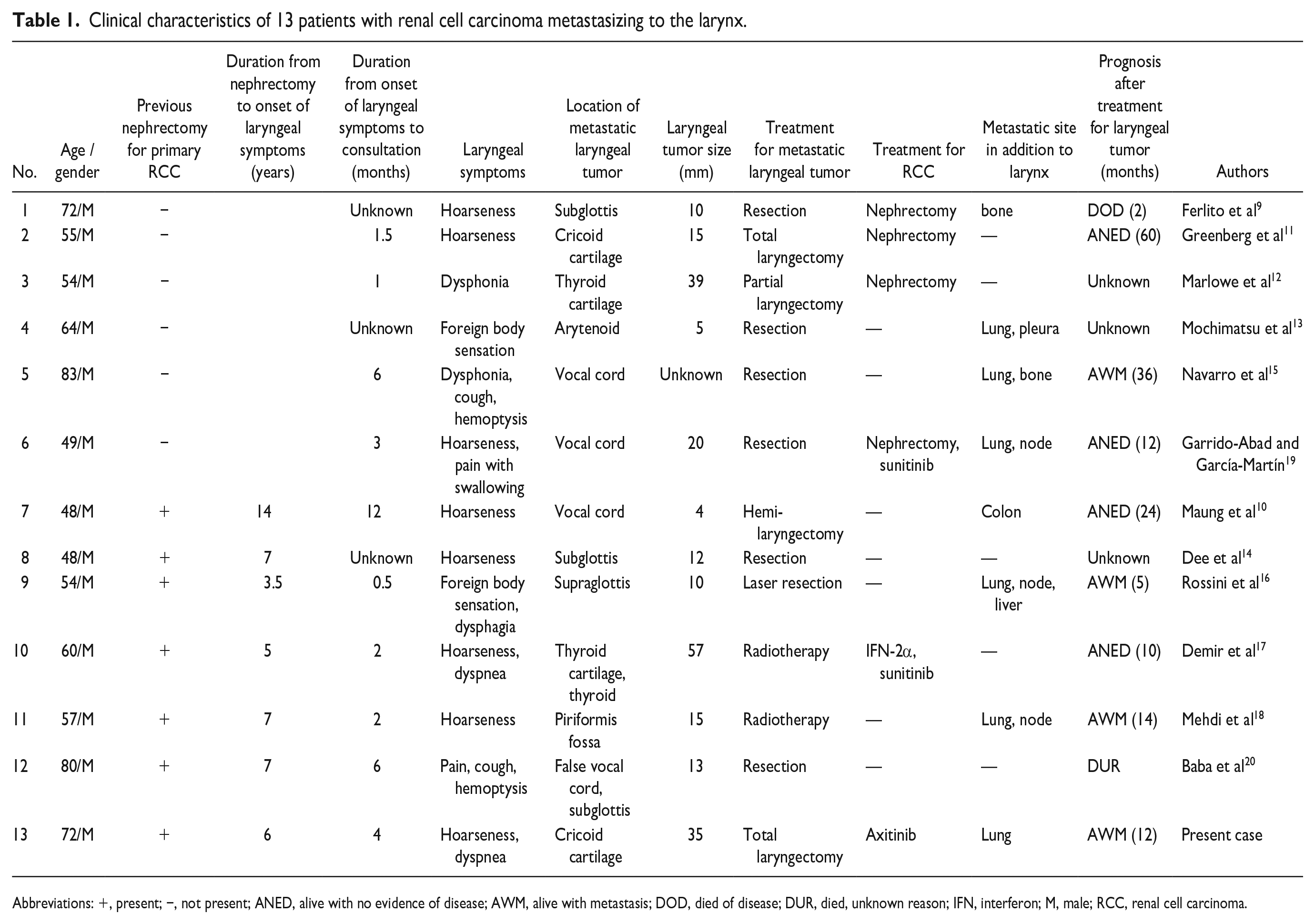
Abbreviations: +, present; −, not present; ANED, alive with no evidence of disease; AWM, alive with metastasis; DOD, died of disease; DUR, died, unknown reason; IFN, interferon; M, male; RCC, renal cell carcinoma.
In the present case, we knew of the patient’s history of nephrectomy for RCC at the first visit to the Department of Otolaryngology but did not consider the cricoid cartilage tumor as a metastasis from RCC. Histologic findings from the biopsy specimens of the tumor and immunohistologically positive staining for various markers, such as AE1/AE3, CD10, vimentin, and EMA, proved metastasis from clear cell-type RCC.
With regard to treatment of the metastatic laryngeal tumor from RCC in the cases summarized in Table 1, surgical resection using direct laryngoscopy and some kinds of laryngectomy was performed in 7 patients (54%) and 4 patients (31%), respectively. In the present case, we selected total laryngectomy as the treatment. Metastatic tumors from RCC are reportedly often hypervascularized and tend to be highly hemorrhagic.18,23 We could not perform decannulation after biopsy due to repeated bleeding from the tumor in the cricoid cartilage. We thought that the tumor had to be resected to stop the bleeding. Chest CT scan revealed a few tiny lung metastases. FDG-PET/CT scan revealed no uptake in the right residual kidney or the abdomen lymph nodes. The National Comprehensive Cancer Network (NCCN) guidelines for kidney cancer recommended that candidates for surgical metastasectomy should include patients who develop oligometastases in the lung, bone, and brain after a prolonged disease-free interval from nephrectomy, as long-term relapse-free survival has been reported in some patients. 24 We regarded the metastatic tumor in the cricoid cartilage as an oligometastasis, which is defined by a state of limited systemic metastatic tumors for which local ablative therapy could be curative. Therefore, our patient was expected to survive over the long-term following total laryngectomy with subsequent chemotherapy, even if the patient lost his voice.
Among the cases summarized in Table 1, molecular-targeted drugs such as sunitinib and axitinib were administered to 2 patients and 1 patient, respectively. The VHL gene, which is encoded on chromosome 3p25, is reportedly mutated or silenced in >50% of sporadic clear cell-type RCC cases. 25 The mutation identified by NGS panel testing using DNA obtained from the tumor in the cricoid cartilage was VHL p.T124Hfs*35. Mutations in VHL lead to the accumulation of hypoxia-inducible factors in cells and activation of many downstream hypoxia-driven genes, including vascular endothelial growth factor (VEGF) and other genes involved in angiogenesis, cell growth, or survival. 26 VEGF receptor (VEGFR) tyrosine kinase inhibitors (TKIs) target and deactivate these downstream pathways. 27 The NCCN guidelines recommend systemic chemotherapy with TKIs targeting the VEGFR, such as axitinib, cabozantinib, and lenvatinib, in combination with immune checkpoint inhibitors such as pembrolizumab or nivolumab for patients with relapse and/or metastatic clear cell-type RCC. 24
Among the cases summarized in Table 1, 4 of 6 patients who were diagnosed with RCC based on histologic findings from the laryngeal tumor underwent nephrectomy and the other 2 patients did not undergo nephrectomy because they did not meet the surgical indications. Of the 13 patients summarized in Table 1, lung and bone metastases from RCC, in addition to laryngeal metastasis, were identified through the clinical course in 6 (46%) and 2 (15%) patients, respectively. The median duration of follow-up for the 9 patients was 12 months (range, 2-60 months) (Table 1). One patient died of RCC, and 8 patients with RCC were still alive at the time of report publication.
Conclusions
We reported a rare case of RCC metastasizing to the cricoid cartilage presenting with subglottic stenosis. Total laryngectomy contributed to the patient’s longer survival. In patients who undergo nephrectomy for RCC, clinicians should be aware of the slight possibility of metastasis to the larynx years later.
Footnotes
Acknowledgements
Authors’ Note
This material has not been published elsewhere, either in whole or in part. All authors have been personally and actively involved in the substantive work of preparing the manuscript and are jointly and individually responsible for its content.
Author Contributions
NB, AK, RT, SS, and TG performed surgery and provided bedside care. SB, NH, YK, and HN performed histologic and mutational investigations. NB drafted the manuscript. All authors approved the final version of the manuscript.
Data Availability Statements
The data that support the findings of this study are available from the corresponding author, NB, upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed on patient tumor samples in this study were carried out in accordance with the ethical standards of the Institute Ethics Committee and the Declaration of Helsinki of 1964 and its subsequent amendments or comparable ethical standards.
Informed Consent
Written informed consent was obtained from the patient and his family for the publication of clinical details and images.
