Abstract

Significance Statement
External auditory canal (EAC) reconstruction poses a challenge, as patency should be maintained to properly convey sound and aid hearing, whereas EAC stenosis leads to wax accumulation, recurrent infections, and subsequent hearing loss. Several factors should be considered during the surgical decision-making, involving the EAC defect size, location, additional auricular defect, and exposure of other tissues. Individual assessment of composite defects can lead to innovative, safe, and efficient reconstructive techniques in a single-stage manner.
Reconstruction of EAC defects can be challenging due to the anatomy, defect characteristics, and spatial confinements. 1 Addressing each auricular defect in a systematic and logical manner enables a satisfactory functional and aesthetic outcome.2-4 We hereby present a novel reconstructive technique for extended auricular defects involving the EAC.
This technique was applied in an 83-year-old female presented with an advanced auricular skin cancer involving the posterior EAC wall, diagnosed as T2N0M0, based on the Pittsburgh classification.
5
The lesion was resected under local tumescent anesthesia, removing the pinna and 270° of the cartilaginous EAC. The temporal bone, following fresh frozen sections, was deemed clear, and thus, it was preserved. The resulting defect included three-dimensional loss of the cartilaginous EAC along with retroauricular skin and soft tissue, exposing the temporal bone (Figure 1(A)). (A) Extended defect of the external auditory canal and the retroauricular area of the right ear following complete ear resection. (B) Temporalis myofascial flap harvest.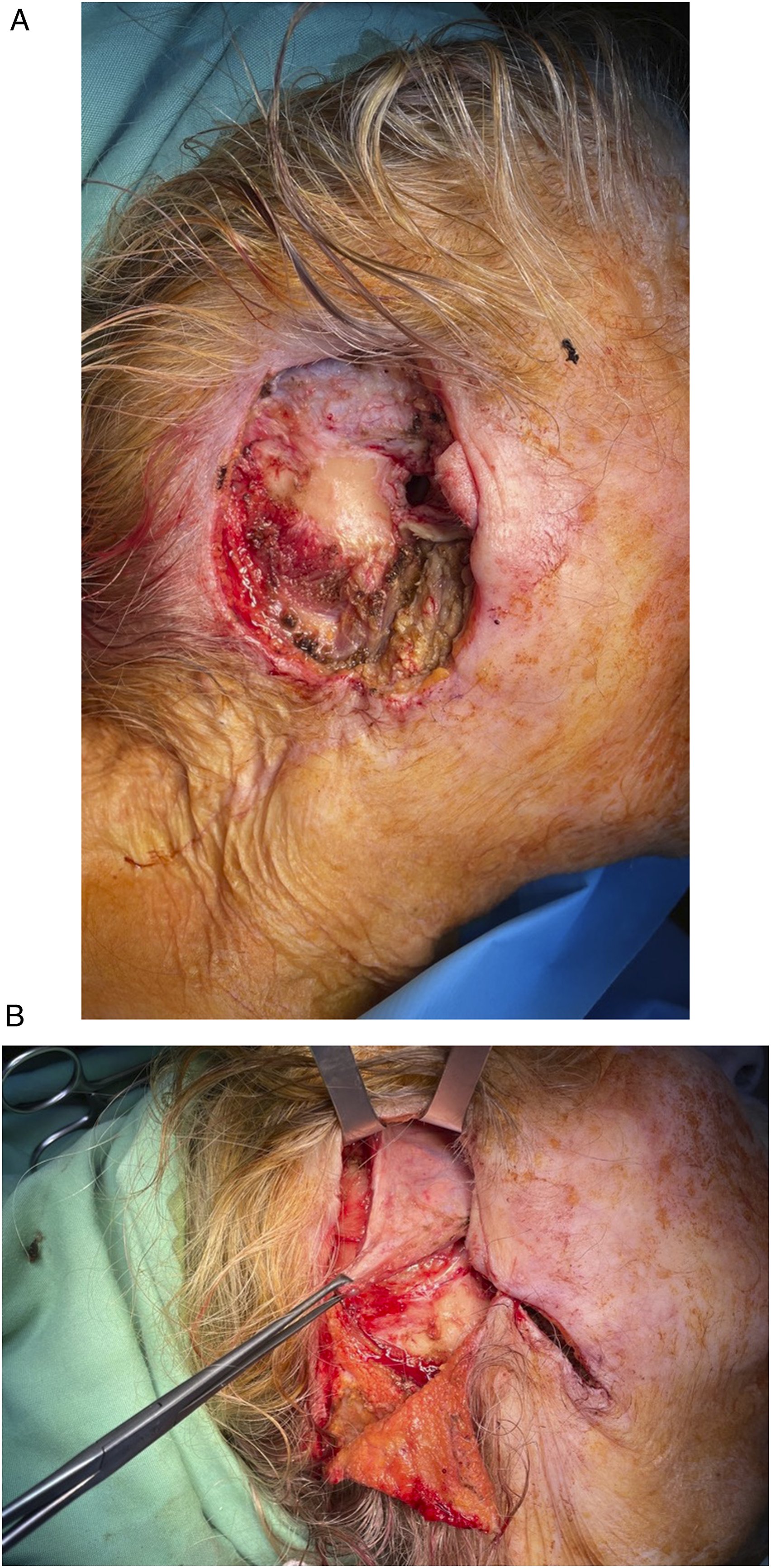
Without further incision, the deep temporal fascial plane was dissected superior to the defect. An anteriorly based rectangular myofascial flap was incised, incorporating the inferior part of the temporalis muscle and overlying deep temporal fascia, and elevated, dissecting deep to the muscle. The length of the temporalis myofascial flap (TMFF) was estimated to reach the inferiormost point of the missing EAC. Consequently, a myofascial sling was transposed and inset to reline the EAC (Figure 1(B)). A retroauricular rotation flap with its pedicle strategically placed inferiorly was elevated and transposed to cover the TMFF. Although a skin graft could have been used on the medial side of the TMFF, this was avoided for several reasons. Instead, a silastic sheet was fashioned into a cone and placed within the canal, and Betadine-impregnated gauze packing was inserted as a bolster. The postoperative period was uneventful. Rapid epithelization of the medial TMFF portion was encountered, preserving the reconstructed canal widely patent on follow-up at 2 and 8 months (Figure 2). Postoperative outcome at 8 months: (A) Macroscopic and (B) EAC endoscopic result.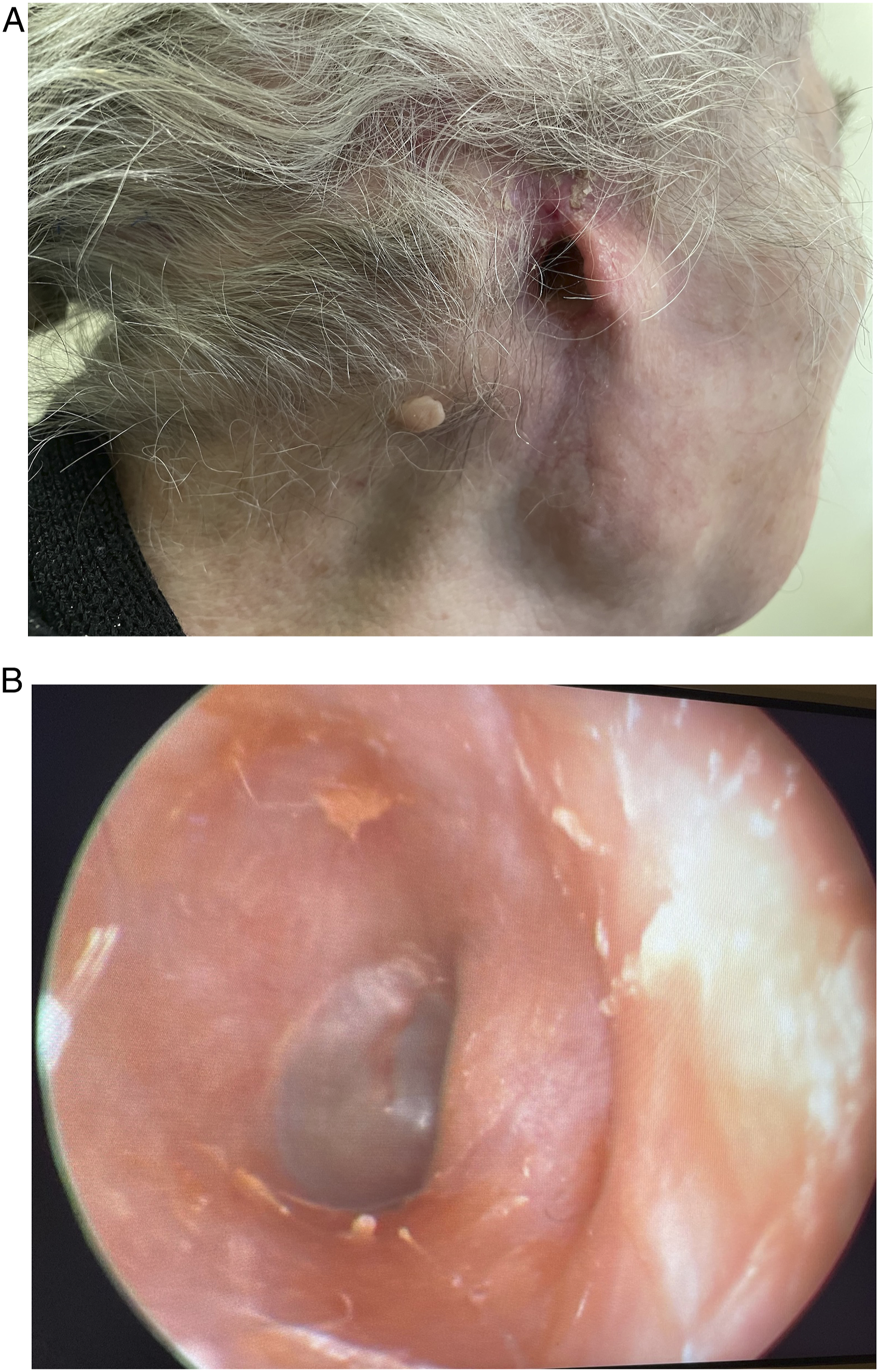
The structural deficit of the EAC necessitates work on a three-dimensional level to restore the framework initially and then provide skin of appropriate thickness and texture. An algorithm suggests that deficiencies covering less than 180° of the total EAC circumference are best treated using skin grafts, composite grafts, or local advancement flaps, while those more than 180° with a loco-regional flap. 1
The temporal area, with its unique anatomy compared to the remaining scalp, and multiple sources of vascular supply, enables multiple vascular territories to be used as pedicled flaps to reconstruct periauricular and temporal bone defects. 1 A flap from the temporoparietal, the superficial temporal, the deep temporal fascia, or the temporalis muscle can be used, based on the defect characteristics.1,6 Free flaps are also worthwhile in such extensive defect reconstruction, presumed ideal for filling dead space. Common donor areas include the anterolateral thigh and the abdomen, necessitating adequate expertise and postoperative care though to minimize morbidity. 7
Considering an extended EAC and retroauricular area defect, as well as the usual comorbidities of this elderly group, a reconstructive plan to be executed safely under local anesthesia is favored. The proposed technique takes advantage of the contiguous, already exposed posterior temporal area, which facilitates the rapid dissection of a tailor-made pedicled TMFF. Based on the vascular supply of the middle temporal artery, TMFF is a robust flap with a good arc of rotation, which is limited though by the muscle contraction. However, incorporating temporalis muscle to the TMFF provides firmness, ensuring patency of EAC, and thus tackling postoperative stenosis and hearing impairment. The TMFF, fixed to the lowermost point of the remaining EAC, reconstructs the structural deficit completely, permitting the reconstruction of the retroauricular skin and soft tissue defects with a flap, a typical rotational flap in this case.
Reviewing the pertinent literature, such a technique of myofascial sling for three-dimensional EAC reconstruction has not been described. Yu used a rather small pedicled temporalis myofascial (PTM) along with pedicled postauricular periosteal flap and intact skin for the reconstruction of the deficient posterior EAC wall, treating chronic suppurative otitis media. 8 PTM stabilized the cartilaginous EAC instead of reconstructing the EAC. The technique presented herein obviates further skin incisions, potential pedicle injury, and prolonged surgery. Strengths also include the single-stage reconstruction, and the surgery performance under local anesthesia, which both mitigate the periprocedural complication risks and shorten the length of stay. These surgical attributes are highly desirable in this COVID-19 pandemic era. 9
In conclusion, extended composite EAC defects can be reconstructed safely and efficiently in a straightforward manner, should the fundamental reconstructive principles be followed, and modern techniques applied.
Footnotes
Author Confirmation Statement
KS: study conception and design, data collection, interpretation of results, and draft manuscript preparation; NB: interpretation of results and draft manuscript preparation; GT and EGL: draft manuscript preparation. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The patient has given written informed consent for this study.
