Abstract
Pleomorphic adenoma is a common tumor of the salivary gland tumor, but it is rare in the ear. To the authors’ knowledge, this patient is the first reported case of pleomorphic adenoma arising in the internal auditory canal. There have been few documents reporting the manifestations and the therapy strategy of this disease. This study clearly demonstrated the experience in treating pleomorphic adenoma, including the characteristics of the clinical manifestation, the key procedures of surgery, and key points of the diagnosis. The translabyrinthine approach under the microscope performed to remove this tumor is a good option for resecting this type of tumor.
Keywords
Introduction
Pleomorphic adenoma (PA) is one of the common tumors that arise from the salivary glands, also known as “mixed tumor”. It often occurs in the major salivary glands, including the parotid, submandibular and sublingual glands, and rarely affects in the minor salivary glands and respiratory tract. 1 Ectopic PA is a type of mixed tumors with malignant potential that occurs outside of the salivary glands. Between 21% and 64% of all PAs are malignant. However, ectopic PA in the internal auditory canal (IAC) has not been reported. In this report, we present an extremely rare case of PA in the IAC and describe the resection using the translabyrinthine approach.
Case Report
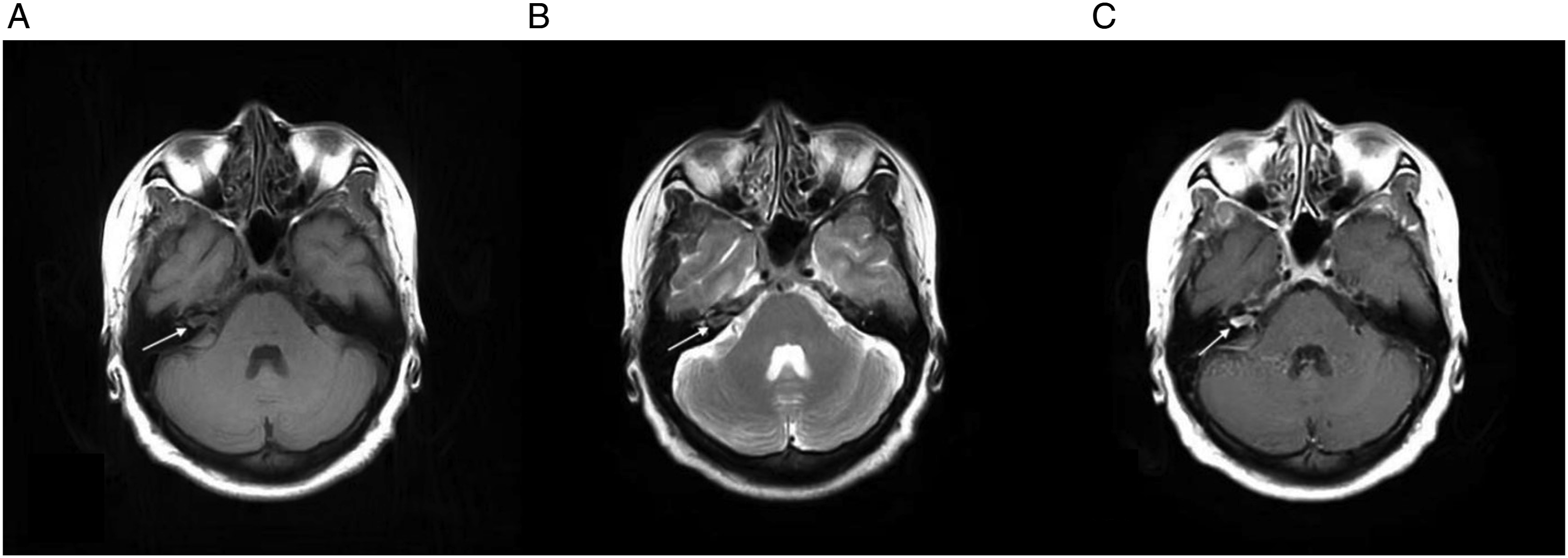
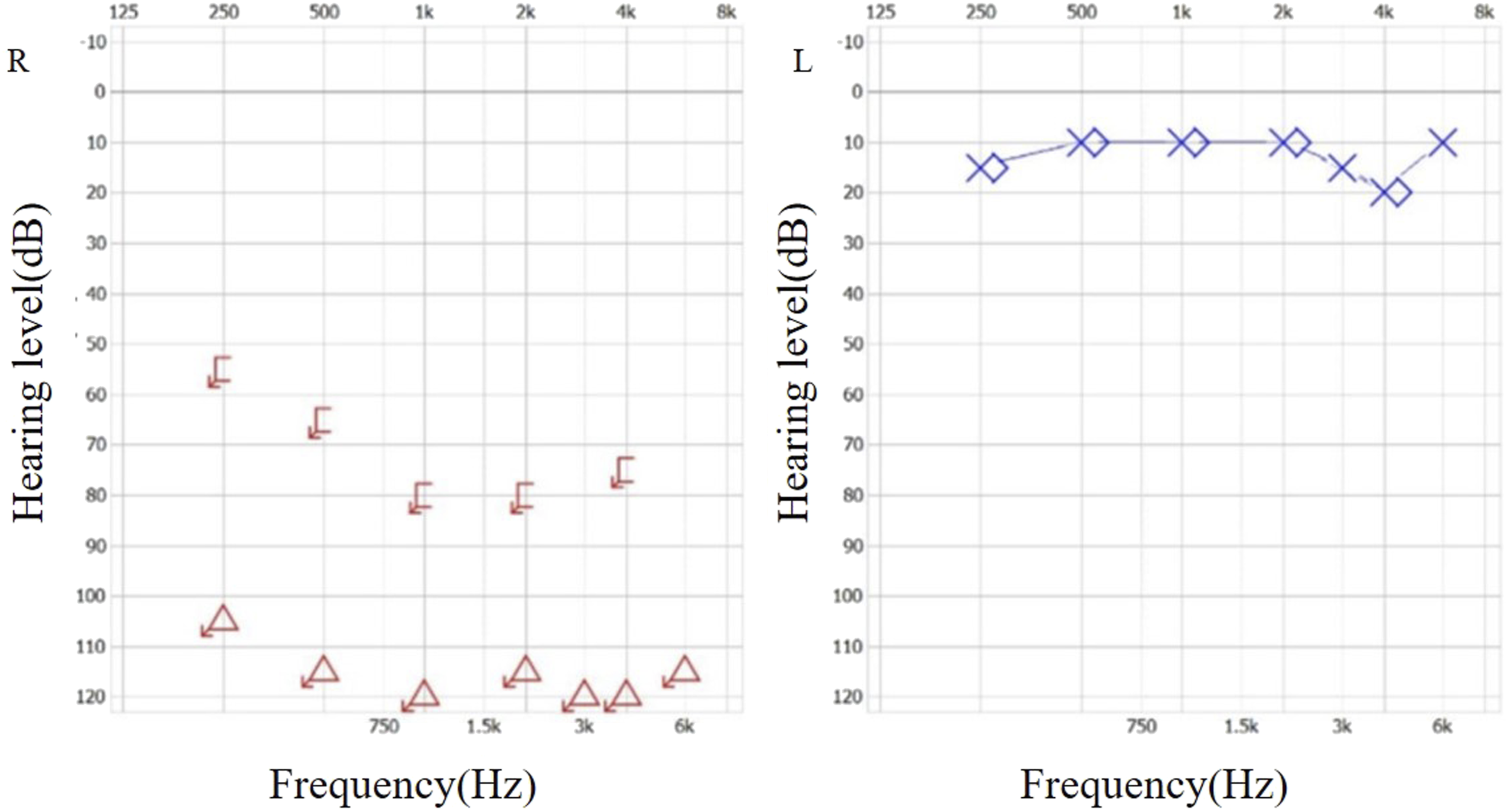
The patient was a 52-year-old female. She had experienced progressive sensorineural hearing loss in her right ear for 2 years, but had not received treatment. Six months ago, she developed symptoms of right facial spasms, beginning with involuntary spasms of the right eyelid. After undergoing magnetic resonance imaging (MRI) at a local hospital, an acoustic neuroma was diagnosed in her right IAC. Further radiological examinations were performed after referral to our hospital, including high-resolution computed tomography (CT) of the IAC, MRI of the brain, and computed tomography angiography (CTA)/computed tomography venography (CTV) of the intracranial artery. The high-resolution CT of her right IAC showed enlargement and irregular erosion of the temporal bone (Figure 1). The contrast-enhanced MRI of the brain showed thickening of the nerve and widening of the right IAC, with avid enhancement (Figure 2). The CTA/CTV of the intracranial artery showed slight enlargement of the right IAC. A pure tone audiology test revealed severe sensorineural hearing loss in the right ear (Figure 3). Based on these findings, an acoustic neuroma was suspected in the right IAC. Preoperative axial CT scan: The right IAC was slightly enlarged and irregular in shape. Preoperative axial MRI scans and gadolinium-enhanced sequence: the right IAC was enlarged and the right auditory nerve was thickened, with avid enhancement after injection of the contrast agent. A right acoustic neuroma was suspected (indicated by arrows). (A) Iso-hypointense signals on T1-weighted MRI sequences. (B) Iso-hyperintense signals on T2-weighted MRI sequences. (C) Heterogeneous enhancement post gadolinium. Pure-tone audiometry: Normal hearing in the left ear; profound severe sensorineural hearing loss in the right ear.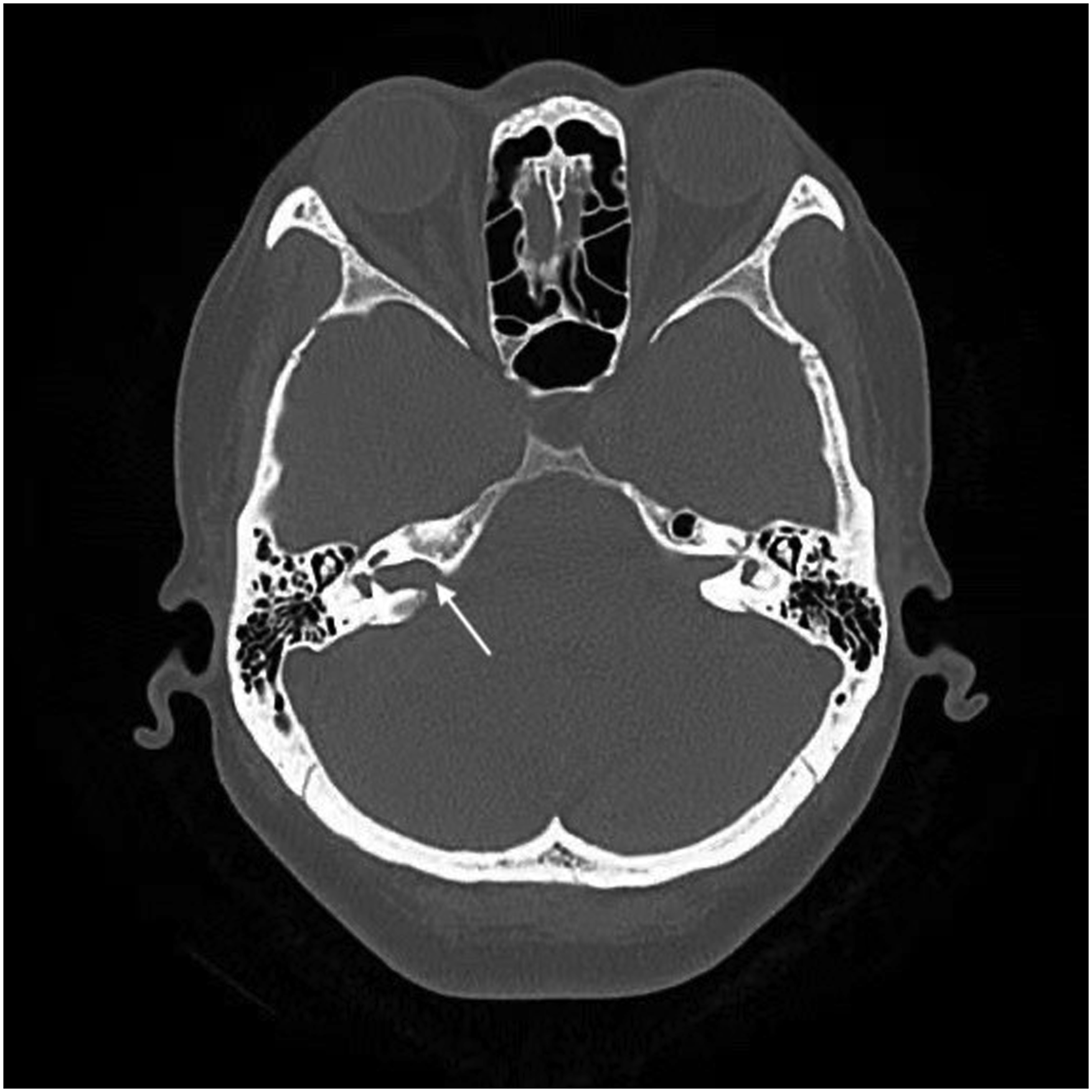
The translabyrinthine approach under the microscope was performed to remove the IAC tumor under general anesthesia. During the operation, an arc postauricular incision was made to prepare the musculoskeletal flap. The sigmoid sinus, which protruded from the mastoid cortex, was exposed after removing the mastoid air cells. The sigmoid sinus was pressed backward and downward with bone wax to locate the meninges. Then the high-riding jugular bulb was exposed. The surface bone of the jugular bulb was removed and sealed with gelatin sponges and bone wax. The sigmoid sinus-meningeal angle was fully exposed after this. To observe the facial nerve, the bone overlying the vertical segment of the facial nerve was removed. After resection of the semicircular canals under the facial nerve, the IAC was exposed by 180 degrees. There was an obvious enlargement of the IAC, and a tumor, measuring 1.5 × 1.5cm, was observed under the microscope (Figures 4A and 4B). To expose the region of the cerebellopontine angle, the IAC bone was resected backwards. After intracapsular excision of the main mass, the tumor appeared to be close to the superior vestibular nerve, with posterior of the tumor protruding into the cerebellopontine angle. The total tumor was resected block by block, under the facial nerve monitor. The aditus ad antrum and vestibule were sealed with bone wax. (A) Intraoperative view of the tumor mass filling the IAC. (B) The IAC was exposed after complete tumor resection. (C) Mild facial paralysis after surgery. (D) Facial paralysis disappeared 4 months after the operation.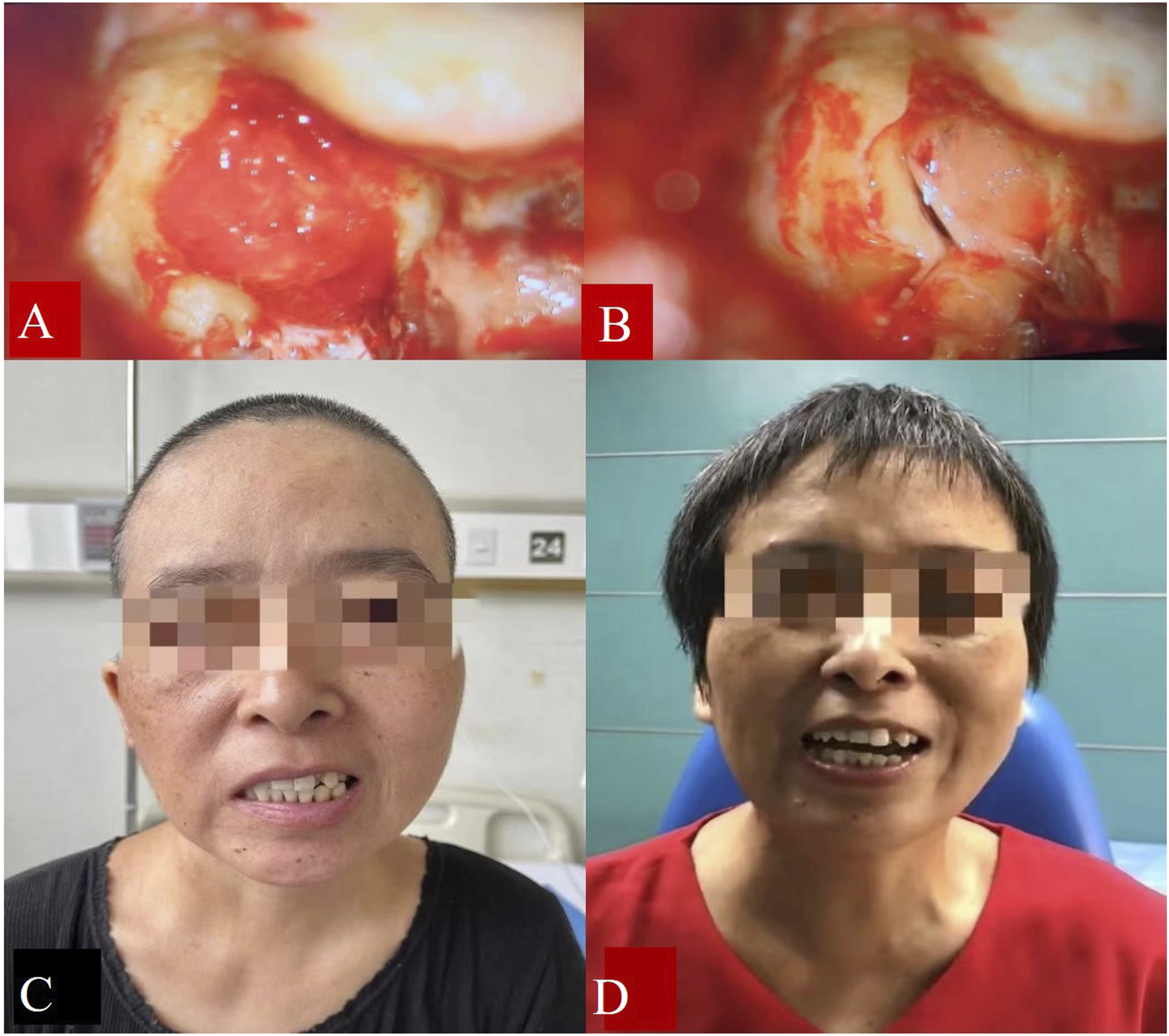
The final pathological result of the resected tissue was PA. The patient’s postoperative recovery was good, with spasms around her right eye disappearing. However, mild facial paralysis occurred, identified as House-Brackmann grade Ⅲ(Figure 4C). Postoperative follow-up was conducted through MRI review. Four months after the operation, the facial paralysis disappeared and facial muscle function was fully recovered (Figure 4D). Twenty months after the operation, no tumor recurrence was found (Figure 5). Postoperative axial MRI scans and gadolinium-enhanced sequences: (A) and (B) Patchy mass with short T1 and long T2 signals in the right mastoid (4 months post-operation). (C) No tumor was seen after enhancement in the right cerebellar-pontine region (4 months post-operation). (D) and (E) Patchy hyperintense signals in the right mastoid (1 year post-operation). (F) No tumor recurrence was observed post gadolinium enhancement (1 year post-operation).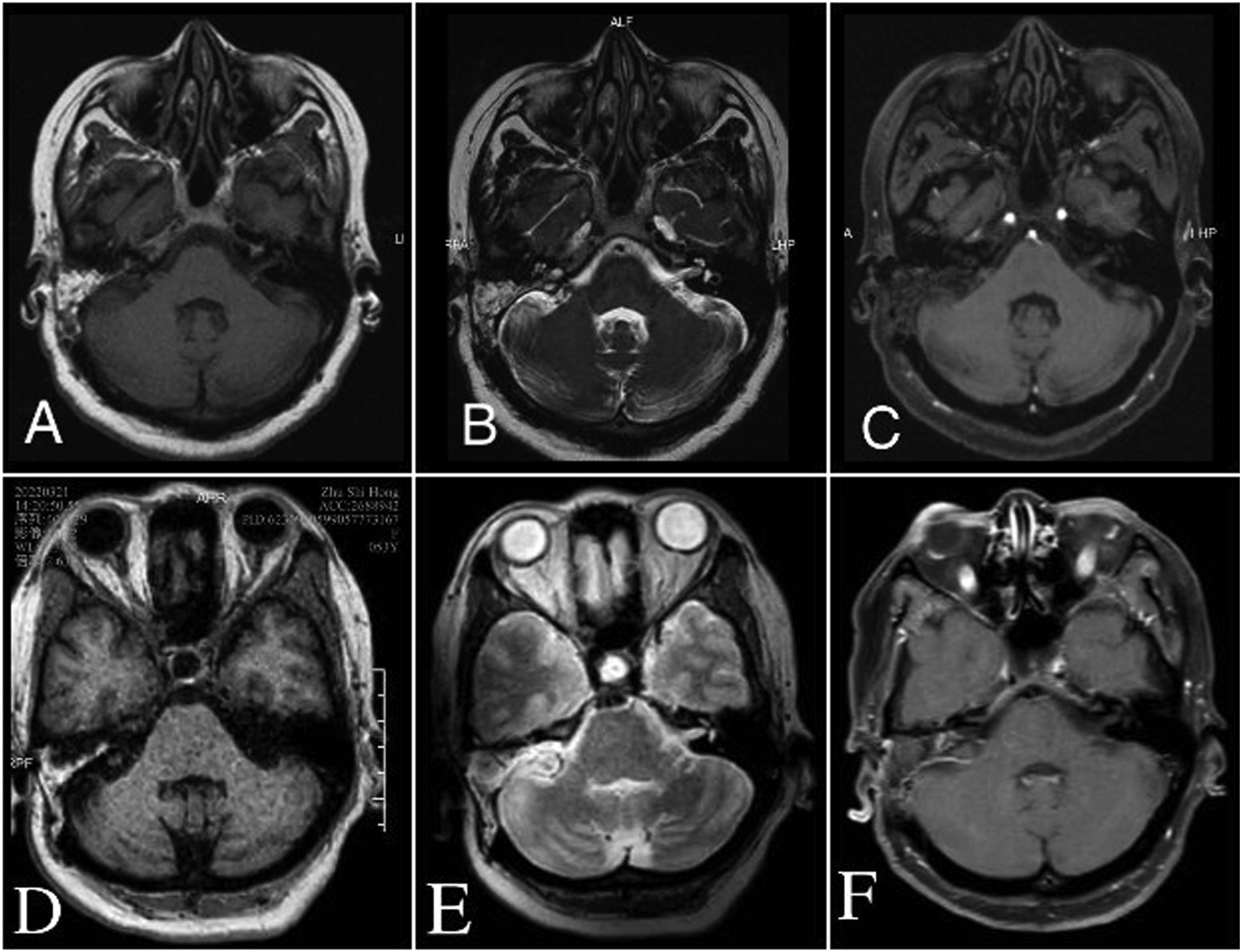
Discussion
PAs in the ear most commonly occur in the external auditory canal,2-5 where the common original tissues are the cerumen glands and the ectopic salivary glands.5-7 However, the origin of PAs in the middle ear is unknown and remains controversial. 8 Some researchers believe they are derived from ectopic salivary glands, 6 while others propose an epithelial origin, with multiple tissue components in the tumor derived from the same tissue of the germ layer.9,10
To diagnose tumors in the IAC, the clinical presentation, radiological evaluations, and histopathological examination should all be considered. First, the predominant symptom is compression due to tumor growth. As the nerves in the IAC are compressed, the dysfunction gradually worsens, including the auditory, vestibular, and facial nerves. As a result, it can be difficult to identify the origin and malignancy of the tumor based on clinical presentation alone.11,12 Second, radiology examinations, such as contrast-enhanced CT and MRI, can be used to identify the type of IAC tumor. Some types of IAC tumors, such as osteoma, lipoma, and hemangioma, can be diagnosed through radiology examinations. PAs in the IAC show hypointensity on T1-weighted imaging and iso-hyperintensity on T2-weighted imaging due to the myxoid stroma, which is the main component of the tumor and contains a high amount of water.13,14 Approximately 90.6% of PAs show progressive enhancement on MRI. 15 Acoustic neuromas, on the other hand, show hypointensity or iso-hyperintensity on T1-weighted imaging and hyperintensity or iso-hyperintensity on T2-weighted imaging, with heterogeneous enhancement after gadolinium. 12 The similar radiographic manifestation of the 2 tumors makes it difficult to differentiate them using MRI examination before surgery. Third, histopathology examination is the gold standard for identifying the origin and malignancy of tumors. PA typically presents as round or oval masses. The tumor body is an encapsulated submucosal lobular mass with a gray section. Microscopically, it consists of a mixture of glandular epithelial cells, myoepithelial cells, myxoid, or chondroid stroma. Tumor cells are organized in sheets, irregular cords, or glandular tubes, and are cuboid, polygonal or fusiform in shape. The nuclei are uniform in size, with no mitoses or degenerative development.16,17 Acoustic neuroma is spherical, oval or dumbbell-shaped, with a smooth gray, light yellow, or white surface and a complete envelope.18,19 Microscopically, acoustic neuromas appear as compact spindle cells with palisading nuclei and verocay bodies, or as fewer cells with loose reticular arrangements and microcystic changes.
The accuracy of fine needle aspiration for PAs is poor.18,20 There is also a tendency for PA to be aggressive and malignant, after a biopsy of fine needle aspiration. In addition, due to the special location of an IAC, a biopsy of PA cannot be performed before surgery. Compared to tumor identification, total intracapsular resection of PA in the IAC is more important. Similarly, a malignant transformation of the PA can also occur after radiotherapy. Meanwhile, PA is also not sensitive to radiation. While gamma knife radiosurgery is a popular option for treating patients with IAC tumors, surgeons should be aware that an ineffective treatment or malignancy may result from radiotherapy of PA. Therefore, biopsy, preoperative radiotherapy, and gamma knife radiosurgery are not recommended for PA of IAC. Previous study showed that the prognosis is excellent (95%) after complete excision. 21 Based on our experience, the key points in properly treating PA include total removal of the tumor, reduction of the recurrence, and preservation of the neural function. The recurrence rate of PA is related to the surgeon’s skill, the integrity of the tumor capsule, the location of the tumor, and the surrounding of tissues and nerves. In our patient, preoperative manifestations included severe profound sensorineural hearing loss in the right ear, obvious irritation of the facial nerve, and a tumor mass occupying the IAC. Although the histopathology of the tumor was not identified before the surgery, the translabyrinthine approach was a suitable method for exercising the PA in the IAC. Our surgical procedure and postoperative treatment were consistent with the diagnosis and therapeutic principle for PA in the IAC. The good prognosis of this patient demonstrates that surgery is a good treatment option for PA in the IAC for patients with a definitive clinical diagnosis and no contraindications to surgery. Furthermore, a regular follow-up is necessary to monitor for recurrence of PA in the IAC.
In conclusion, PA of the IAC is an extremely rare disease, originating from the ectopic salivary glands or the same tissue of the germ layer. The development of the tumor compresses the surrounding tissues and nerves, leading to various clinical manifestations, including hearing loss and facial paralysis. The translabyrinthine approach under a microscope is a good option for total resection of the tumor. Follow-up is imperative to observe the recurrence of PA in the IAC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Yaowen Wang is currently receiving a grant (2019KY584) from Zhejiang Province Medical Science and Technology Project (China), and a grant (2023ZL6448) from Zhejiang Province Traditional Chinese Medicine Science and Technology Project (China).
Ethical Approval
This study was approved by Ethics Committee of Ningbo First Hospital (Approval number: 2022RS024).
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article.
