Abstract
Significance Statement Posterior reversible encephalopathy syndrome (PRES) is characterized by variable neurological findings such as headache, deterioration in mental condition, loss of consciousness and visual loss. The disease results with impairment of the cerebral blood flow autoregulation. Ondansetron, serotonin (5HT-3) receptor antagonist, may cause the haemorrhagic PRES.
Keywords
Introduction
Posterior reversible encephalopathy syndrome (PRES) has typical computed tomography (CT) and magnetic resonance (MR) imaging findings. The disease is characterized by variable neurological symptoms such as headache, deterioration in mental condition, loss of consciousness and visual loss.1,2 PRES has multiple heterogeneous etiologies. The disease is thought to be associated with impaired cerebral blood flow autoregulation and damaged endothelial function. Ondansetron is a serotonin (5HT-3) receptor antagonist antiemetic agent. It is used in the treatment of nausea and vomiting due to chemotherapy. Our patient was diagnosed larynx cancer and he was using chemotherapeutic agents and ondansetron. We aimed to present MR imaging features of the haemorrhagic PRES that we thought to be associated with ondansetron.
Case Presentation
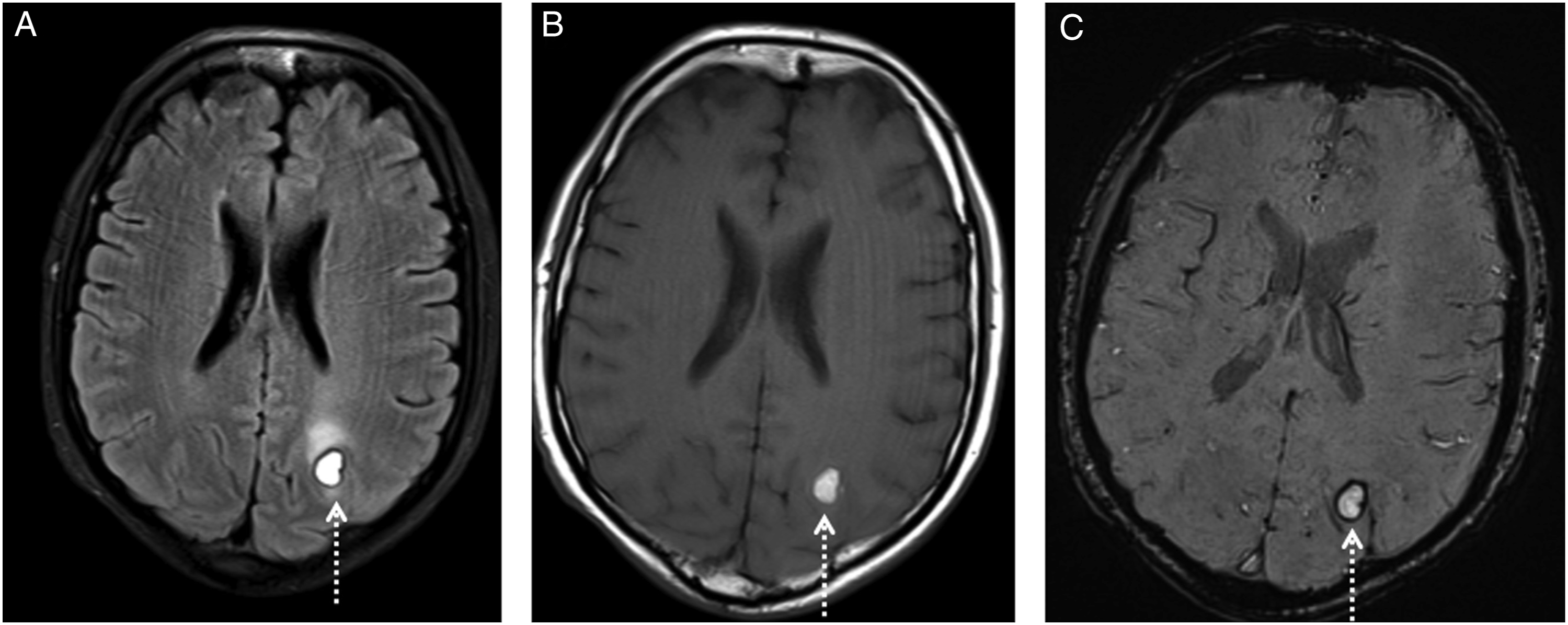
A 49-year-old male diagnosed with larynx cancer presented with headache. He was using a set of chemotherapy drugs and ondansetron for nausea and vomiting prophylaxis. MR imaging was performed for severe headache aetiology. At the time of presentation, T2 and FLAIR (fluid attenuated inversion recovery) weighted images showed symmetrically increased pathological signals in the white matter of the parietal and occipital lobes (Figure 1A and C). These areas were isointense according to the grey matter (Figure 1B). MR image findings were compatible with PRES. MR image one week after presentation revealed subacute stage haemorrhage in the subcortical white matter of the left occipital lobe (Figure 2A and B). Susceptibility weighted imaging (SWI) also showed magnetic susceptibility or blooming artifact compatible with haemorrhage in left parieto-occipital lesion (Figure 2C). At the time of presentation, T2 and FLAIR (A and C) weighted images showed symmetrically increased pathological signals (arrows) in the white matter of the parieto-occipital lobes compatible with PRES. (B) These areas are isointense according to the grey matter on T1 weighted images. On control MR images, FLAIR and T1 (A and C) weighted images reveal subacute stage haemorrhage (dashed arrows) in the subcortical white matter of the left occipital lobe. (C) SWI sequence also shows peripheral blooming artifact compatible with haemorrhage in the left parieto-occipital lesion.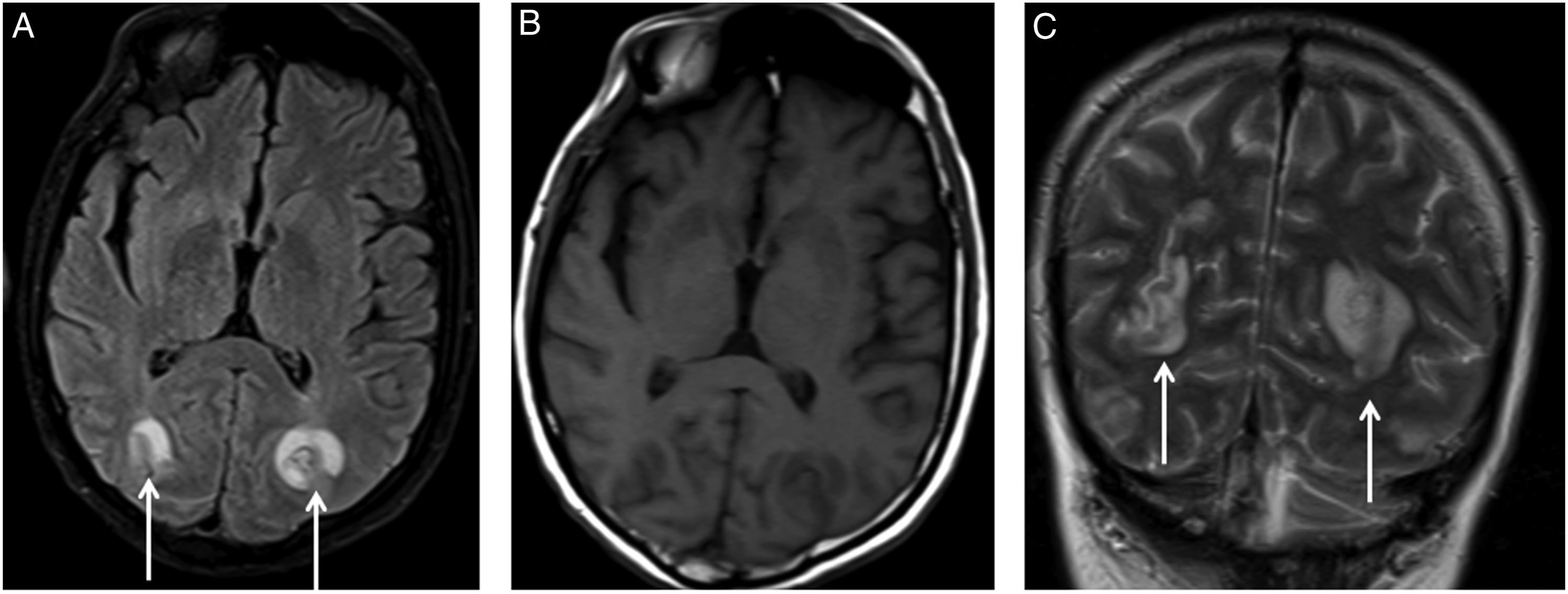
Conclusion
Pathogenesis of PRES is uncertain, but the disease appears to be related to impaired cerebral autoregulation and damaged endothelial function. The disease is characterized commonly with vasogenic oedema in the white matter of the bilateral parietal and occipital lobes. It affects frequently both cortex and subcortical white matter. 3
Haemorrhage is an un-expected condition in this disease. Ondansetron is a selective serotonin 5-HT3 receptor antagonist agent with low affinity for dopamine receptors. 4 Because 5-HT3 receptors are broadly expressed in the intracranial pial vasculature, ondansetron can effect cerebral blood vessels.4,5 By this mechanism, it may contribute to the development of haemorrhagic PRES.
Radiologic features in PRES are a symmetric pattern of parieto-occipital vasogenic oedema. This appears as a signal increase in T2 and FLAIR weighted MR images. PRES can be found in a non-posterior distribution such as the inferior temporal, superior frontal, brainstem and cerebellum. 3 Parenchymal haemorrhage is associated with PRES in 15% of cases. 5 These lesions have variable contrast enhancement. Haemorrhagic PRES lesions are demonstrated easily using SWI sequences as in our case.
Our case showed a rare potential neurological complication of ondansetron. In this case, PRES had complicated by intracerebral haemorrhage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
