Abstract
Objectives: Silicone tracheostomy tubes are a popular choice for pediatric patients with chronic tracheostomies due to their pliability and increased comfort. However, the presence of wire reinforcement causes them to be labeled “MR-conditional.” The objective of this study was to understand practice patterns across radiology departments for obtaining MRI in children with MR-conditional tracheostomy tubes. Methods: A survey consisting of 7 questions aimed at determining a facility’s protocol for obtaining MRI in children with tracheostomy tubes was developed and administered via telephone to MRI technologists at US children’s hospitals. Results: 182 children’s hospitals were identified and 59 responses were obtained across 30 states (32.4%). 19 sites (32%) were excluded as they were unable to perform MRI. All 40 facilities reported that they have a standard questionnaire for medical implants, however only 20 reported that tracheostomy tubes are included on that questionnaire (50%). 6 (15%) reported all MR-conditional tubes are changed to MR-safe ones. Of the remaining 34, protocols were as follows: 1 (2.9%) scans patients with conditional tubes for a maximum of 15 minutes, 5 (14.7%) only use a 1.5T magnet, and 28 (82.3%) reported following the manufacturer conditions. In terms of artifact, 3 sites change MR-conditional tubes to MR-safe if scanning the head, neck, chest, or abdomen (8.8%), 6 (17.6%) change the tube if scanning the head or neck, 15 (44.1%) change the tube only if scanning the neck, and 10 (29.4%) were not aware of issues with artifact. Conclusion: Based on this survey of MRI technologists at US children’s hospitals, there is no unified protocol for obtaining MRI in pediatric patients with MR-conditional tracheostomy tubes. A lack of standardized protocols may be contributing to unnecessary tracheostomy changes. Future research includes clearly defining the anatomical regions affected by wire-related artifact and developing a standardized MRI protocol for these patients.
Introduction
Tracheostomy is one of the oldest surgical procedures, however, there has been an overall shift in the common indications for placement of tracheostomy tubes within the past 50 years. While tracheostomy tubes had been most commonly placed for the short term management of acute viral and bacterial infections, they are now more often placed for the long-term ventilatory management of complex congenital neurological conditions. 1 Common indications for the placement of tracheostomy tubes in the pediatric population include congenital or acquired airway stenosis, vocal fold insufficiency causing chronic aspiration which requires frequent pulmonary toilet, and neurologic conditions requiring long-term ventilatory support.1,2
Given the shift towards long-term tracheostomy placement in children with congenital neurologic abnormalities, tracheostomy tubes have adapted to meet these changing indications. Tracheostomy tubes can be made of metal, PVC, or silicone.3,4 Commonly utilized tracheostomy tubes in the pediatric population include Shiley® and Bivona® tubes. The Shiley® tracheostomy tube is made of PVC, with a more rigid shaft, and is usually changed weekly or biweekly in the pediatric population. Bivona® tracheostomy tubes are made from silicone, which increases comfort, facilitates suction, reduces blockages, and allows for monthly changing. 5 The silicone shaft is reinforced with a wire coil, which allows for the tube conform to the shape of the trachea. 6 Although the Bivona® tube is the most commonly placed pediatric tracheostomy tube, the presence of this wire coil complicates its MRI safety. In 2014, Smiths Medical launched the Bivona® FlexTend™ TTS™ tracheostomy tube. 7 This tube is embedded with a non-ferrous wire, the goal of which is to enhance MRI compatibility. 7
Given its lack of ionizing radiation and ability to allow physicians to better visualize soft tissue structures, MRI has become a commonly utilized, safe mode of imaging. 8 The main safety issue in MRI is the ability of metallic objects to become projectiles or burn due to the magnetic field.9,10 For this reason, many implanted medical devices are contraindicated and radiology departments must utilize extensive screening forms in order to minimize any risks to patients undergoing MRI. Additionally, metallic objects may cause artifact, affecting the quality of the image. 11
Many medical device materials have been altered in order to become safer for use with MRI. These devices are often labeled as “MR-conditional,” which is defined as an “item that has been demonstrated to pose no known hazards in a specific MRI environment with specified conditions of use.” 12 For example, in recent years some pacemakers have become MR-conditional. 13 These pacemakers make use of reduced ferromagnetic components and specific programming in order to prevent pacemakers from being inhibited by the MRI machine. 14 Particular conditions must be met in order for the patient to undergo MRI safely, including a maximum field strength, absorption rate, temperature rise, and gradient slew. 15
The Bivona® pediatric tracheostomy tube contains a wire coil and is considered MR-conditional. According to the Smiths Medical statement on MRI compatibility, MR-conditional devices include those which are “safe to use within an MRI environment if the user is aware of the safe conditions and takes the necessary precautions.” 16 Patients with a Bivona® wire-reinforced tracheostomy tube can undergo MRI if the following conditions are met: “static magnetic field of 3-Tesla or less, maximum spatial gradient magnetic field of 720-Gauss/cm, and maximum MR system reported whole-body-averaged specific absorption rate (SAR) of 2-W/kg for 15 minutes of scanning.” 16
The aim of this study was to assess radiology department practice patterns for obtaining MRI in pediatric patients with MR-conditional tracheostomy tubes. It was hypothesized that the labeling of these tubes as MR-conditional and the variety of conditions that must be met in order to undergo a safe MRI have led to different protocols among radiology departments at US children hospitals.
Materials and Methods
In order to determine current protocols for obtaining MRI in pediatric patients with MR-conditional tracheostomy tubes at United States children’s hospitals, a survey study was designed. The study was approved by the Institutional Review Board (IRB) at our institution. (Pro202001387).
An anonymous survey consisting of 7 questions focused on determining a facility’s protocol for obtaining MRI in children with tracheostomy tubes was developed by the authors.
The survey was administered via telephone to MRI technologists at US Children’s Hospitals. 182 children’s hospitals were identified via the Children’s Hospital Association Directory. Verbal informed consent was obtained prior to survey administration after respondents confirmed their job title to be “MRI technologist.” Respondents remained anonymous and were assured confidentiality of their answers. No incentives were distributed as part of survey completion. Each survey took approximately 5 minutes to administer via telephone.
Descriptive statistics based on the survey responses were obtained for protocols for obtaining MRI in pediatric patients with MR-conditional tracheostomy tubes. Data was analyzed using Microsoft Excel version 16 (Redmond, WA).
Results
Respondent Characteristics
Geographical Distribution of Survey Responses.

All 40 facilities reported that they have a standard questionnaire for medical implants; however, only 20 sites reported that tracheostomy tubes are included on that questionnaire (50%). MRI technologists/technicians at 39 sites reported they were aware of MRI compatibility coding and were able to correctly state that pediatric tracheostomy tubes are labeled as MR-safe, MR-conditional, or MR-unsafe (97.5%).
Protocols for Obtaining MRI in Pediatric Patients with Tracheostomy Tubes
With regard to specific protocols for pediatric patients with tracheostomy tubes, MRI technologists/technicians at 38 sites reported that they determine the make and model of the tracheostomy tube to determine if safe for MRI (95%). Technologists/technicians at the remaining 2 sites were unaware of any particular protocol for these patients. 1 site reported that they have a dedicated staff member who is responsible for determining the type of tracheostomy tube a patient has prior to the MRI and this person is specifically aware of tube compatibility with MRI (2.5%). Three sites reported that they have a physical manual that describes MRI compatibility for different tracheostomy tube models (7.5%). The remaining sites (36) reported using the manufacturer’s website or calling the manufacturer to determine MRI safety (90%).
Scanning MR-Conditional Tracheostomy Tubes
Six sites reported that MR-conditional tracheostomy tubes are always changed to MR-safe Shiley tubes for MRI (15%). These sites indicated the tracheostomy tube may be changed by the patient’s parent, respiratory staff, nursing staff, or anesthesia depending on the circumstances. The remaining 34 sites were questioned regarding any adjustments made for scanning MR-conditional tubes. 1 site reported that they are able to scan MR-conditional tracheostomy tubes, but only for a maximum of 15 minutes (2.9%). Five sites reported that they only scan patients with MR-conditional tracheostomy tubes using the 1.5T machine (14.7%). The remaining 28 sites reported that they obtain and follow the conditions listed by the manufacturer of the particular MR-conditional tracheostomy tube (82.3%). These protocols are summarized in Figure 1. Protocols for the handling of pediatric patients with MR-conditional tracheostomy tubes.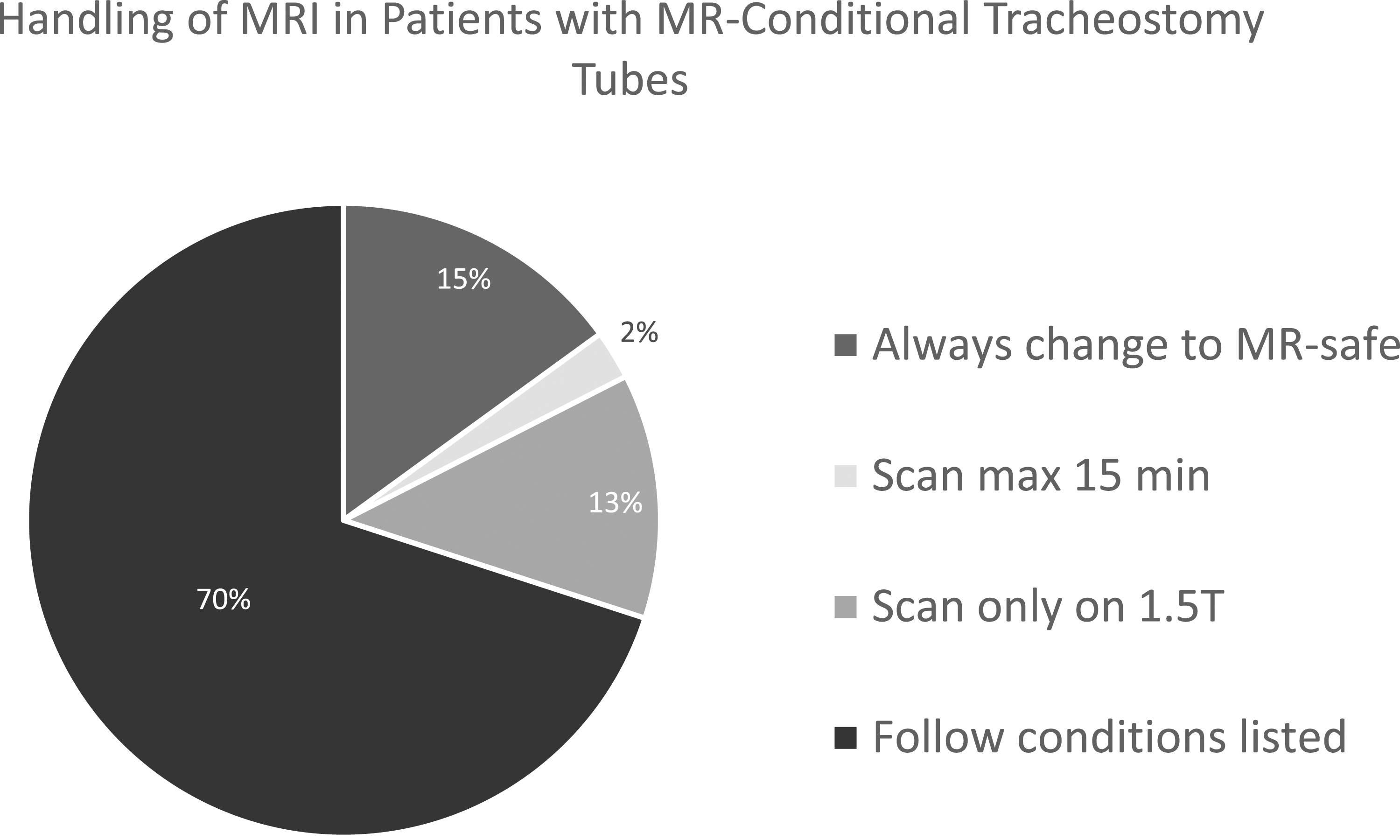
The remaining 34 sites were also questioned regarding tracheostomy changes due to artifact. 3 sites reported that they change MR-conditional tubes to MR-safe if scanning the head, neck, chest, or abdomen due to artifact from the tube (8.8%). Six sites reported that they change MR-conditional tubes to MR-safe if scanning the head and neck, including the brain, due to artifact (17.6%). Fifteen sites report that they change MR-conditional tubes to MR-safe only if scanning the neck due to artifact (44.1%). Ten sites reported they were not aware of any issue with artifact when scanning with an MR-conditional tube (29.4%). These results are summarized in Figure 2. MR-conditional tracheostomy changes due to artifact.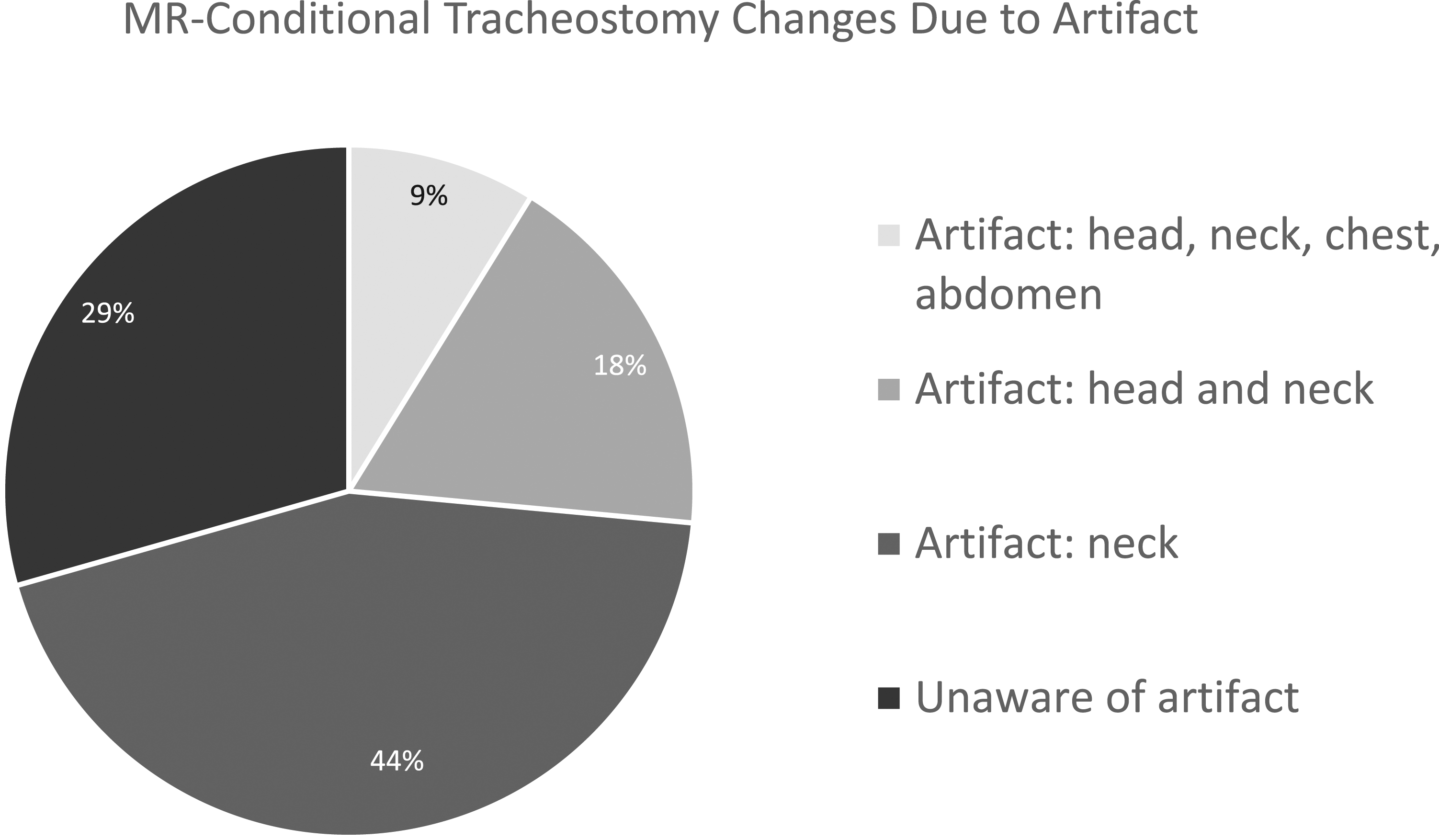
Discussion
The Bivona® silicone tracheostomy tube is a popular choice for many children with chronic tracheostomies. This tube contains a flexible wire, which causes it to be labeled as “MR-conditional,” meaning certain conditions must be met in order for patients to safely undergo an MRI.3,4 Patients specifically with a Bivona® wire-reinforced tracheostomy tube can undergo MRI if the following conditions are met: “static magnetic field of 3-Tesla or less, maximum spatial gradient magnetic field of 720-Gauss/cm, and maximum MR system reported whole-body-averaged SAR of 2-W/kg for 15 minutes of scanning.” 16 The goal of this study was to understand protocols for obtaining MRI in pediatric patients with MR-conditional tracheostomy tubes.
Our results found that protocols for obtaining MRI in patients with MR-conditional tracheostomy tubes varied across children’s hospitals. 15% of our sample reported changing all MR-conditional tubes to MR-safe ones, 1 hospital reported that they only scan patients with MR-conditional devices for 15 minutes, and others reported that they only use the 1.5T machine. As indicated above, tracheostomy changes are not required for MR-conditional tracheostomy tubes if the manufacturer conditions are met. Therefore, these may be unnecessary tracheostomy changes for patients. A recent review addressed common misconceptions for MR-conditional devices. 17 The authors emphasized that no generalizations of MR safety can be made from encountering a device labeled MR-conditional, as the conditions that must be met in order for a patient to safely undergo MRI depend on the specific device. 17 The make and model of each device must be obtained so that the instructions for use can be accessed by radiology staff. Given the highly specific conditions that must be met for MR-conditional devices, this information should be readily accessible for radiology staff.
This study also revealed that there is no consensus on the anatomical area affected by artifact due to MR-conditional tracheostomy tubes. Various regions were listed by the children’s hospitals as affected by artifact, including just the neck, the head and neck, and the head, neck, chest, and abdomen. Other sites did not report any issues with artifact from the wire-reinforced tube. It is clear that there is no consensus on the area affected by artifact and sites may be changing MR-conditional tracheostomy tubes to MR-safe ones due to concerns of artifact without evidence of this. There have been several studies analyzing the anatomical region affected by artifact for cochlear implants as well as research on ways to reduce this artifact.18,19 In recent years, metal artifact reduction sequences (MARS) have been utilized to reduce artifact from metal. 11 These techniques use particular sequences which optimize imaging in the vicinity of metal, which may have a role in MRI in patients with MR-conditional tracheostomies.
Lastly, in our survey, all 40 facilities reported that they have a standard questionnaire for medical implants, however only 20 sites reported that tracheostomy tubes are included on that questionnaire (50%). Perhaps including tracheostomy tubes on this questionnaire would help trigger a protocol that radiology staff can follow to determine the safety of MRI for these patients.
There are several limitations of the current study. First, this was a cross-sectional study and the sample size was relatively small at 59 responses out of the 182 children’s hospitals called. This low response rate increases the risk of bias and limits the ability of our results to be generalized. Additionally, 19 of these facilities did not have an MRI machine and had to be excluded from the analysis. Furthermore, the exact MRI capabilities of each institution were not recorded (e.g., presence of both a 1.5T and 3T machine), and thus some manufacturer recommendations may not be able to be followed at those sites. Additionally, our survey was not validated prior to being administered. We also did not obtain or confirm specific credential information from each MRI technologist surveyed. Lastly, at sites that reported changing the tracheostomy tubes, it was noted that there were several different possibilities for staff who changed the tube (e.g., anesthesia and nursing). At other sites, it is possible that a lack of comfortable personnel might affect whether or not a tracheostomy tube change was performed prior to the MRI.
Future research should focus on clearly defining the anatomical regions affected by wire-related artifact. Additional research should include the development and implementation of a standardized MRI protocol for these patients.
Conclusions
Given the lack of radiation and clarity of images generated, the use of MRI, especially in children, has steadily increased since the year 2000. 8 Children with complex neurological conditions often require silicone, wire-reinforced tracheostomy tubes for long-term ventilatory support and may require frequent imaging to monitor their conditions.1,2 It is important for radiology departments to have a protocol for obtaining MRI in children with MR-conditional tracheostomy tubes. Overall, this study showed that there is no unified protocol for obtaining MRI in pediatric patients with MR-conditional tracheostomy tubes. A lack of standardized protocols may be contributing to unnecessary tracheostomy changes. Future research includes clearly defining the anatomical regions affected by wire-related artifact and developing a standardized MRI protocol for these patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics
The study was approved by the Institutional Review Board (IRB) at our institution. (Pro202001387). All participants provided verbal consent.
Data Availability
The data that support the findings of this study are available from the corresponding author (EC), upon reasonable request.
