Abstract
Objective
Perioperative risk stratification of pediatric patients undergoing airway intervention remains crucial in identifying those at a higher risk of requiring postoperative intensive care unit (ICU) care. Here we determined the likelihood of and possible risk factors for developing perioperative adverse respiratory events (PAREs) requiring ICU care after various pediatric endoscopic airway surgeries (EASs).
Methods
We conducted a retrospective chart review of pediatric patients who were aged <18 years and underwent EAS between 2015 and 2021. Early postoperative adverse events within 24 h of surgery were recorded and analyzed.
Results
Overall, 99 patients who underwent EAS were included. The age at the time of the intervention ranged from 8 months to 18 years. Fifty-eight patients, median age was 4.83 years, underwent papilloma debulking with no high likelihood of PARE in this patient subgroup (OR = 0.48; 0.16–1.44). Twenty-five patients, median age was 9.72 years, underwent balloon dilation of laryngotracheal stenosis with no increase in the likelihood of PARE in this patient population (OR = 2.02; 0.65–6.28). Early postoperative respiratory events occurred in 16 patients (16.2%). Most of these events (75%) manifested within 4 h after surgery. In a univariate analysis, intervention at the level of the subglottis or 2 or more laryngeal subsites increased the risk of PARE (OR = 6.57; 1.11–12.52 and OR = 3.73; 1.93–22.34, respectively). In a multivariate analysis, only intervention in the subglottic area maintained its effect (OR = 6.84; 1.82–25.65).
Conclusion
Respiratory adverse events following pediatric EAS are not uncommon, and the majority are encountered shortly after surgery. Intervention in the subglottic area was an independent predictor of PARE.
Keywords
Introduction
Endoscopic airway surgeries (EASs) are indicated for a variety of laryngotracheal pathologies either as primary or consequent procedures following open surgery. Over the years, the efficacy of EAS has increased due to several surgical innovations, including laser, powered microdebrider, and balloon dilator, along with the use of adjuvant pharmaceuticals, such as mitomycin-C.1-3
Pediatric airway endoscopy with intervention is considered an intermediate to high-risk surgery as it carries a significant risk for postoperative airway edema with a possible need for urgent airway management. 4 Several factors have been shown to increase the incidence of postoperative adverse events and the likelihood of unplanned intensive care unit (ICU) admission, including emergency or airway interventions, a higher American Society of Anesthesiologists physical status classification (ASA) score, prolonged anesthesia time, and age <1 year.5-7 Therefore, many pediatric otolaryngologists prefer routine overnight observation of patients in the ICU after EAS. 8 This practice often results in the overutilization of ICU resources, especially in centers with a high number of cases per year.
Previous reports found that the majority of adverse respiratory events, if encountered following certain types of pediatric EAS, occurred shortly after surgery.9,10 Therefore, Lavin et al 11 developed the Gray Zone model of care in which pediatric patients undergoing intermediate-risk surgery on the aerodigestive tract were safely observed for 2–5 h in the post-anesthesia care unit (PACU); the need for ICU care was determined based on the patient’s status at the end of the observation period. Despite the reported beneficial effect on ICU resource utilization, adopting such a model is not feasible for all hospitals as it requires a PACU that is not overwhelmed and the availability of respiratory therapists and airway skilled nursing staff. Hence, perioperative risk stratification of pediatric patients undergoing airway intervention remains crucial in identifying those at a higher risk of requiring postoperative ICU care.
In this study, we aimed to determine the likelihood of developing early adverse respiratory events after various EASs and explore the possible factors that might contribute to the development of these events.
Patients and Methods
King Saud University Institutional review board approval (E-21-5792) was obtained for this retrospective chart review. Need for informed consent was waived because of the retrospective nature of the study. We included a consecutive series of all patients who were aged < 18 years and underwent endoscopic airway interventions at King Saud University Medical City between June 29, 2015 and July 5, 2021.
Patients were identified from a search through the surgical logbooks of the three available pediatric otolaryngologists during the study period. Patients with tracheostomy, aerodigestive foreign bodies, and those who had undergone only diagnostic airway endoscopy with or without injection laryngoplasty were excluded. Patients who were referred to another facility for postoperative management, those who were planned to be shifted to the ICU intubated at the end of surgery, and those with incomplete postoperative records were also excluded.
In our center, most pediatric endoscopic airway interventions were performed under general anesthesia with spontaneous tubeless field ventilation. Intermittent intubation with a small-for-age cuffless tube was sometimes required during surgery if the patient desaturated. Postoperatively, a routine single stat dose of racemic epinephrine was prescribed for most patients along with three doses of dexamethasone (1 mg/kg/day), which were administered during the surgery initially. Unfortunately, we did not have a step-down or high-dependency unit; therefore, all patients were either shifted from the PACU to the surgical ward or were electively admitted to the ICU for airway observation. The airway surgeon and anesthesiologist collectively decided on the need of elective ICU admission based on the patient’s clinical picture, intraoperative findings, and type of intervention without fixed criteria.
Eligible participants’ electronic health records were probed for all perioperative adverse respiratory events (PAREs) that occurred within 24 h of surgery. PAREs were previously defined as events necessitating intubation or other supportive measures in the form of multiple doses of racemic epinephrine, high-flow supplemental oxygen > 4 L/min, or positive pressure ventilation. 9
Statistical Analysis
All de-identified data were analyzed using SPSS version 23.0.0.0 (IBM, New York, NY, USA) for statistical analysis. Categorical variables were presented as absolute values and percentages. Normally distributed continuous variables were presented as mean and standard deviation (SD), while non-normally distributed continuous variables were presented as median with interquartile range (IQR). A univariate binary logistic regression analysis was set for all included variables, and the occurrence of PARE was considered as a dependent variable. A modified approach by Sahoo et al 12 was applied by creating a multivariate stepwise backward binary regression model, in which only the statistically significant clinical variables from the univariate analysis were included. As suggested by Heinze and Dunkler, 13 the α value was set to .157 in the stepwise backward model. P-values ≤ .05 were considered significant.
Results
Baseline Characteristics (n = 99).
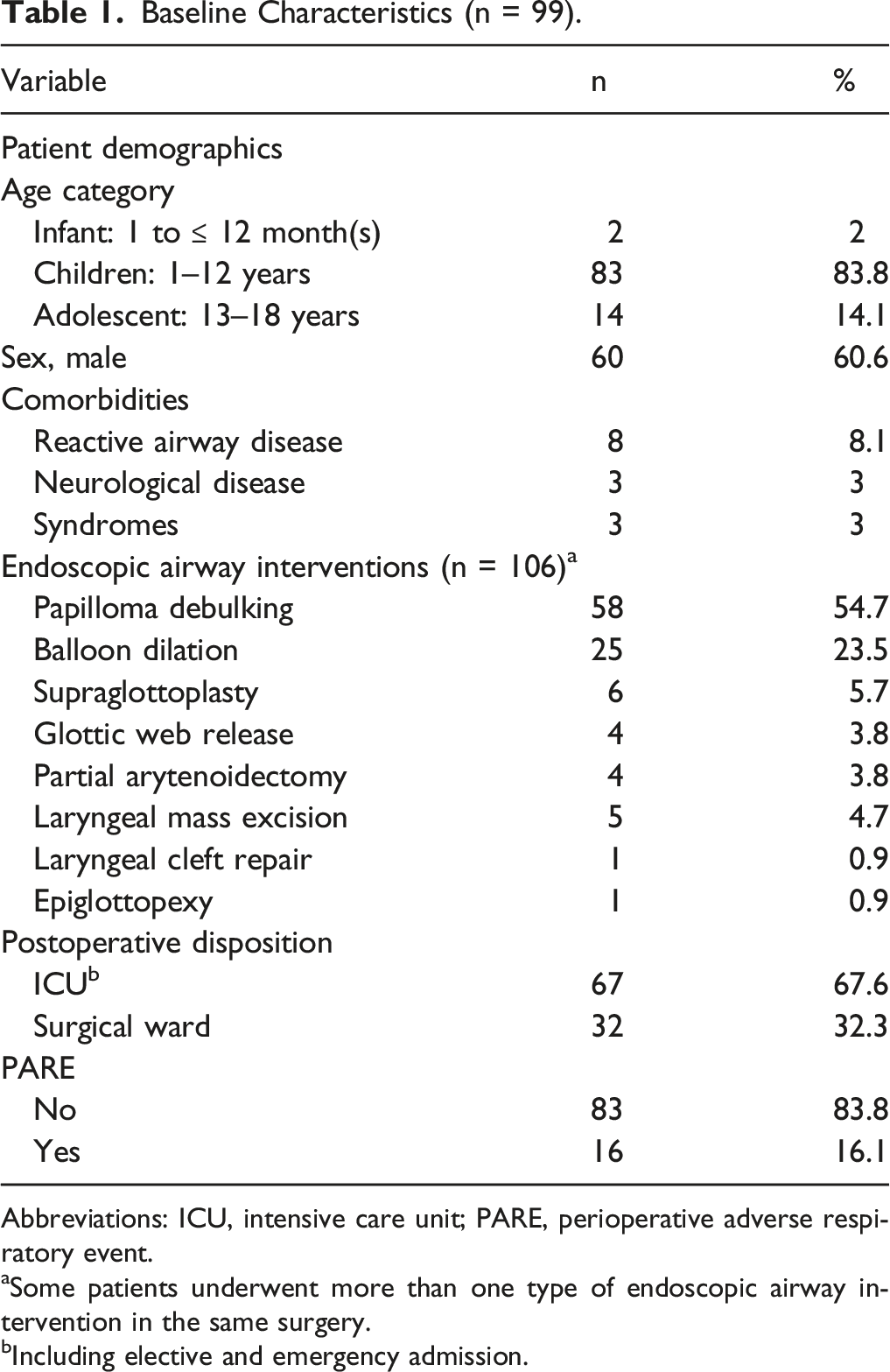
Abbreviations: ICU, intensive care unit; PARE, perioperative adverse respiratory event.
aSome patients underwent more than one type of endoscopic airway intervention in the same surgery.
bIncluding elective and emergency admission.
Recurrent respiratory papillomatosis was the most prevalent diagnosis (51.7%), followed by subglottic/tracheal stenosis (22.4%), bilateral vocal fold paralysis (6%), posterior glottic stenosis (5.2%), laryngomalacia (4.3%), glottic web (3.4%), laryngeal cyst (3.4%), prolapsed arytenoid (1.7%), laryngeal cleft (0.9%), and vocal fold mass (0.9%). A variety of endoscopic airway interventions were performed, of which papilloma debulking was the most common, followed by laryngotracheal stenosis release and balloon dilation. Other less performed procedures included supraglottoplasty, partial arytenoidectomy, glottis web release, and laryngeal cyst/mass excision.
Fifty-eight patients, median age was 4.83 years (IQR, 2.62–8.10 years), underwent papilloma debulking and all of those patients did not have an associated comorbidity. There was no high likelihood of PARE in this patient subgroup (OR = .48; .16–1.44).
Twenty-five patients, median age was 9.72 years (IQR, 4.56–15.49 years), underwent balloon dilation of laryngotracheal stenosis. The majority (88%) had no associated comorbidities. There was no increase in the likelihood of PARE in this patient population (OR = 2.02; .65–6.28).
The median duration of the surgery was 40 minutes (IQR, 39–55 minutes), and laser was used in 12.1% of surgeries. Most patients (64.4%) were electively admitted to the ICU for airway observation after surgery, while only three patients experienced immediate postoperative respiratory distress necessitating immediate intubation and emergency ICU admission. The mean duration of the ICU stay was 16.2 h (SD, 7.62 h) for patients electively monitored in the ICU. Almost one-third (32.3%) of the cohort was shifted to the surgical ward after a short period of observation in the PACU; none suffered from postoperative adverse events.
During the first 24 postoperative hours, 16 patients developed respiratory adverse events in a mean time of 2.54 h (range: 0–13 h); 75% of these events occurred within the first 4 h. Most of these events (68.8%) were managed medically with multiple doses of racemic epinephrine, salbutamol and budesonide nebulization, and/or high-flow supplemental oxygen, while 6 patients required intubation; three of them were immediately reintubated in the operating room and the remaining were intubated in the ICU after failed supportive management. Delayed adverse events (> 8 h) were observed in only 2 patients who underwent glottic-subglottic web release with keel placement; both were eventually intubated, one of them during the first 24h after surgery (Figure 1). One patient developed sinus tachycardia as an adverse effect of racemic epinephrine with spontaneous resolution. Time and management of initial adverse respiratory events after various pediatric endoscopic airway surgeries.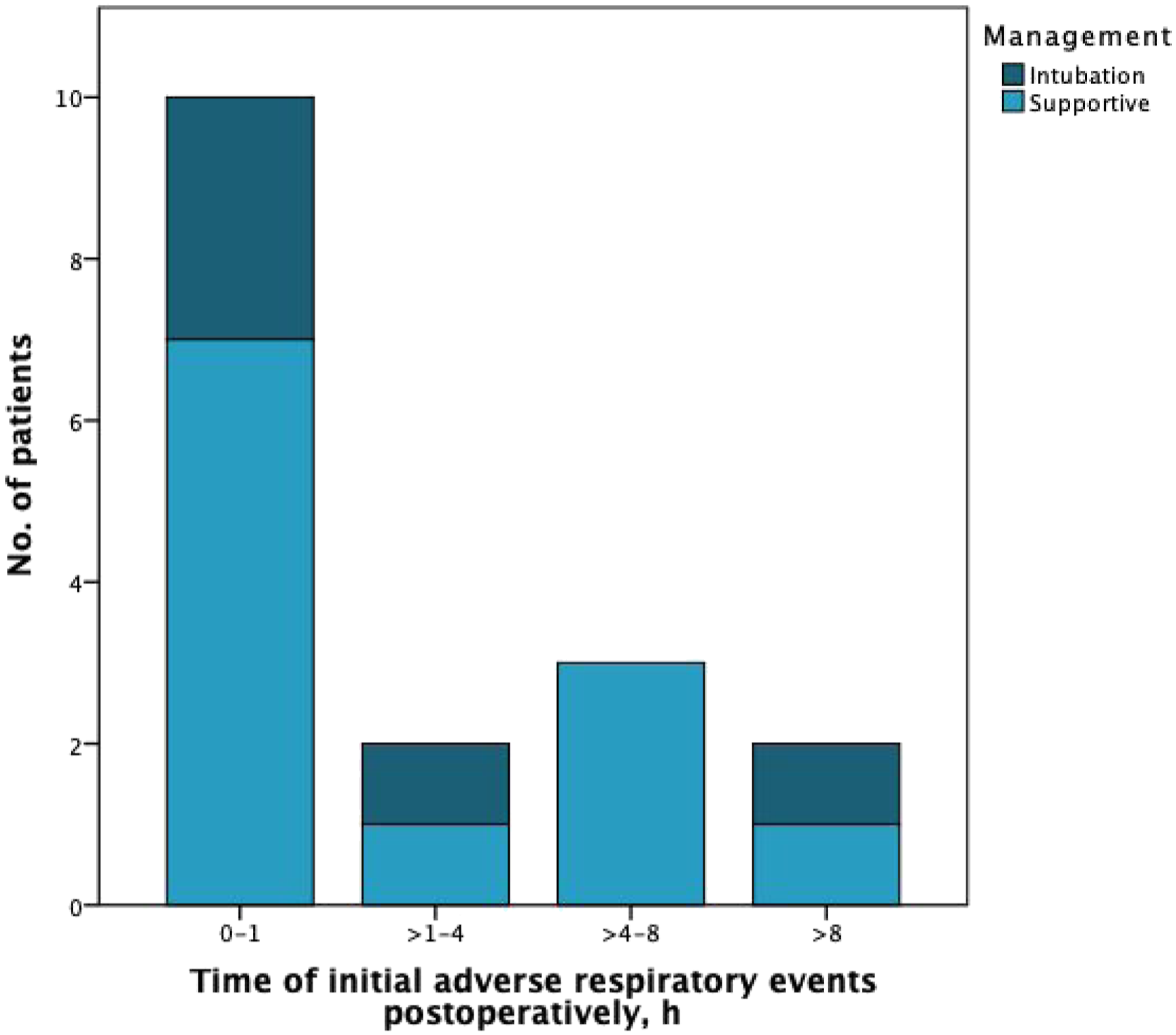
Factors Associated with PARE in Univariate and Multivariate Logistic Binary Regression Modeling.
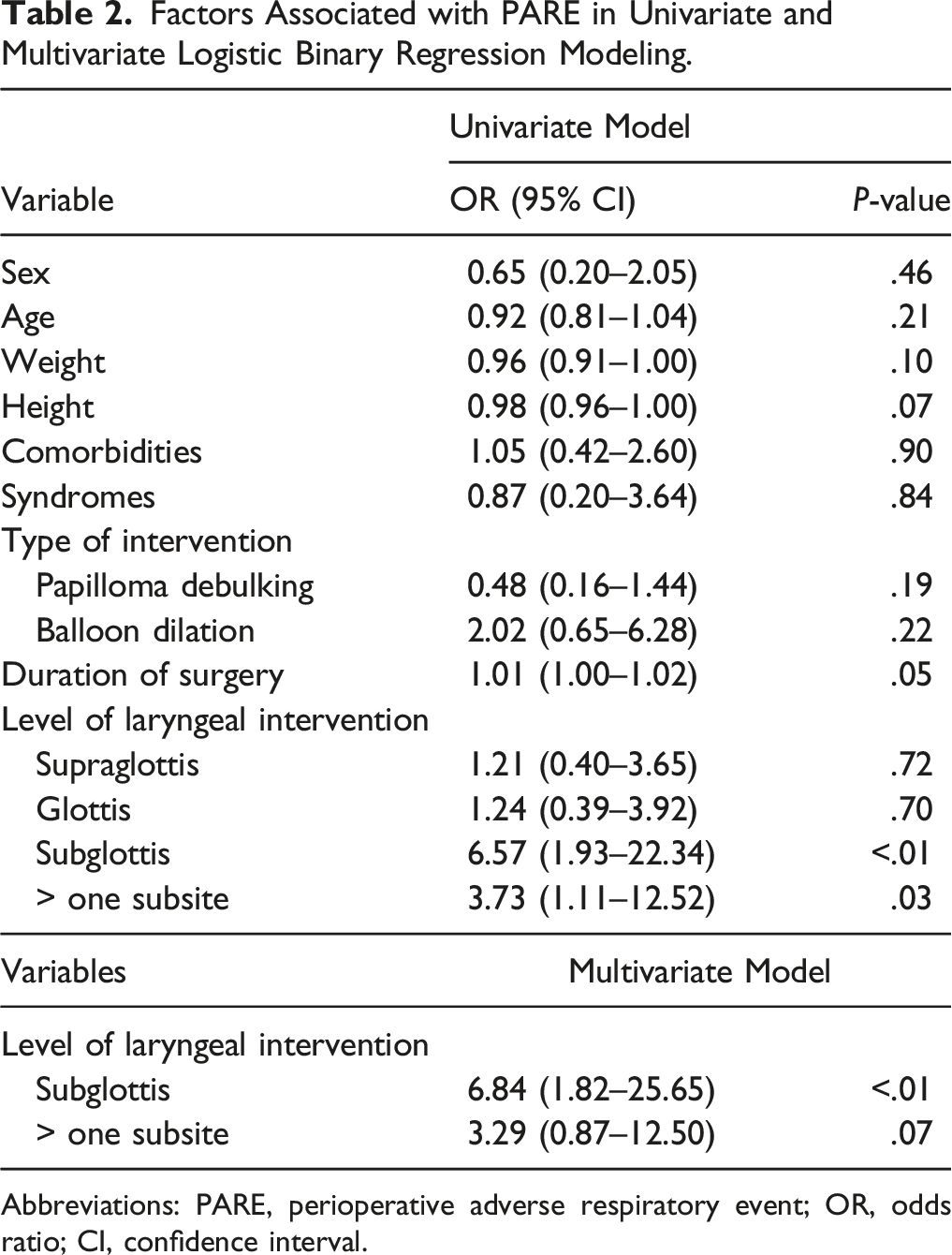
Abbreviations: PARE, perioperative adverse respiratory event; OR, odds ratio; CI, confidence interval.
Discussion
Endoscopic airway intervention in adults has a low complication rate, and many patients can be managed safely as surgical outpatients. 14 By contrast, pediatric EAS carries a significant risk of postoperative adverse events and has been associated with a high rate of readmission. 4 Due to the lack of step-down or high-dependency unit in our center, routine ICU admission after pediatric airway intervention became the preferred clinical practice of some surgeons. As airway intervention in patients increases the risk of postoperative airway compromise, an assessment of other risk factors related to a specific type of intervention is needed to stratify these patients and to identify those at a higher risk of requiring ICU care.5,6 Furthermore, determining the critical postoperative period when these adverse events mostly develop will be beneficial in building a center-specific clinical pathway for endoscopic airway interventions.
Evidence of early respiratory events after pediatric EAS is limited and focused mainly on supraglottoplasty, which was reported to be high (up to 41%). 15 Despite adopting conservative criteria when defining PARE, our results showed that only 16.2% of patients required ICU care after various EASs. The hours immediately after surgery were found to be the most critical period, with 75% of PARE observed within 4 h post-surgery. Delayed events after 8 h only occurred in patients who underwent glottic-subglottic web release with additional placement of a silastic keel. This may be explained by the repeated airway trauma caused by the silastic keel, which worsens edema over time.
In the series of supraglottoplasty cases in a study by Albergotti et al, 9 11.2% of patients developed respiratory events requiring postoperative ICU care, and 80% of these events developed within 4 h after surgery (mean: 2.66 h, range: 0–22 h). Harris et al 10 studied patients who underwent endoscopic type 1 laryngeal cleft repair and showed that 3 out of 34 (8.8%) patients had a significant oxygen drop within 3 h of surgery; supportive management with supplemental oxygen, mask ventilation, or oral airway was found to be sufficient. Although we included patients who underwent various endoscopic interventions at different levels of the airway not limited to the supraglottic area, we observed similar results regarding the incidence rate of PARE and the time during which most adverse respiratory events occurred after surgery.
In the univariate logistic regression analysis, intervention at the subglottic level or intervention at more than one laryngeal subsite was a significant factor in predicting PARE. However, intervention in the subglottic area was the only independent factor that could predict PARE in the multivariate model. This was mostly related to the small diameter of the subglottis, which was subjected to a significant narrowing with minimal edema.16,17 A longer duration of surgery, which may indicate a more complex disease or an aggressive intervention, was close to being statistically significant in the univariate model as a predictor of PARE. However, it has been previously reported that a longer surgery duration (> 30 minutes) along with other factors such as non-white race, preoperative ICU admission, and perioperative desaturation were associated with increased odds of adverse respiratory events and the need for postoperative ICU observation.9,15,18 In our study, papilloma debulking and balloon dilation were not associated with an increased likelihood of PARE (OR = 0.48; 0.16–1.44 and OR = 2.02; 0.65–6.28, respectively). The risk associated with other types of interventions remains unknown as a proper analysis was limited by the small sample size.
Anticipating the need for ICU care after airway intervention is challenging, especially in patients without other risk factors. 11 Several studies have proposed that preserving ICU admission for selective cases while observing others in a step-down or observation unit after certain types of EAS increases ICU utilization efficiency without compromising patient safety.19,20
Approximately two-thirds of our cohort were electively monitored in the ICU with a mean ICU stay duration of 16.2 h (SD, 7.62 h). Although the available resources for airway monitoring in the surgical unit are inferior to the ICU, none of the patients who were shifted to the surgical ward postoperatively were observed to have adverse respiratory events. This might indicate that our threshold for ICU admission was low, resulting in ICU overutilization. Beside the financial savings, decreasing the postoperative ICU stay helps to lessen parents’ traumatic stress and anxiety and allow better management of hospital resources.21,22
Our study had several limitations as it was a single-center, retrospective study on patients who underwent heterogenous airway interventions with a small sample size. Only 2 of our included cohort were infants, thus limiting our findings in that age group. The criteria for elective ICU admission were not standardized which reflects practice variability; patients who were considered high risk were more likely to be observed in the ICU, thus reflecting an inherent bias. Additionally, several factors, such as the number of laryngeal suspension attempts, intraoperative hypoxia events, and intermittent intubations, which might be associated with PARE, were not assessed due to limited documentation. Future prospective studies with a larger sample size that permit comprehensive comparative analysis between different types of EAS should be performed to draw a solid conclusion.
Conclusion
Adverse respiratory events following pediatric EAS are not uncommon, and the majority were encountered shortly after surgery. Therefore, a short period of airway observation in a step-down or high-dependency unit following such interventions may improve hospital resource utilization. Intervention in the subglottic area was considered an independent predictor of PARE.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
