Abstract
Objective
The aim of this study was to investigate the incidence of stoma recurrence and the therapeutic strategy outcomes in relation to survival that have been adopted over the past few decades using a monoclonal antibody, specifically nivolumab.
Methods
This study included a total of 487 patients diagnosed with laryngeal carcinoma undergoing either a laryngectomy or salvage surgery after conservative interventions at the ENT Unit of Federico II University in Naples, Italy, between 2011 and 2021. Following a minimum 2.5-year follow-up and a maximum 21-year follow-up, the results revealed that only 38 patients suffered a stomal recurrence.
Results
Despite various adopted treatment strategies, the literature reports lower patient survival rates. Following a total laryngectomy, stomal recurrence represents a therapeutic management challenge due to a poor prognosis for nearly every treated case. According to the literature, in fact, despite a low incidence (ie, 0.8–31.3%), the overall mortality rate increases from 77% to 100% after three years. Nevertheless, introducing immunotherapy into cancer treatment has resulted in an observable revolution in the treatment of different types of cancers over the years.
Conclusion
In light of recorded data on survival following the use of the nivolumab, the case presented in this study allows a new perspective of successfully treating recurrences of squamous carcinoma of the head and neck.
Introduction
While a stomal recurrence (SR) following a total laryngectomy (TL) is rare—only a 0.8–3.1% incidence rate, according to the literature—serious complications due to a poor prognosis for nearly all treated cases challenge the effectiveness of therapeutic management. In fact, the 77% overall mortality of patients undergoing a total laryngectomy without peristomal recurrence increases to 100% in patients undergoing total laryngectomy with a peristomal recurrence. 1
Distant metastasis was the cause of mortality in 24.3% of these cases. 2 An SR is characterized by a diffuse infiltrate of neoplastic tissue at the junction of the amputated trachea and skin and is commonly diagnosed within the first year following a TL. Notably, some cases occurring after three-and-a-half years have been described in the literature. 1
Sisson divided stomal recurrences according to stages and localization from the stoma and structures involved (ie, trachea, esophagus, and mediastinum), each of requires a different surgical procedure. 3 While the first treatment stage only involves an exeresis of the recurrence, a sternotomy with an extension to the sternoclavicular joint and the esophagus is practiced in some Stage II cases, and in all Stage III and Stage IV cases. Reconstructive procedures involve the use of pectoral myocutaneous flaps; despite extensive surgical excisions in the 75 cases studied, however, Sisson reported that only 45% of Stage I, 29% of Stage II, 33% of Stage III, and 16% of Stage IV patients survived for 3 years.
Bolla and Scolari also reported unsatisfactory results for seven patients undergoing radiation therapy. 4 A plesiotherapy patient expired 2 months after treatment and another patient expired 10 months afterward; a cobalt therapy patient expired after two months; and a betatron therapy patient expired after two months, while another expired after four months.
Balm reported modest results for eight patients with stomal recurrence between 1978 and 1985 and proposed a radio–polychemotherapy protocol that combined vincristine, bleomycin, and methotrexate with radiotherapy. 5 While two patients expired from lung metastases and one reported ongoing stomal recurrence, the survival expectancy of the remaining five patients was seven, three, and two-and-a-half years, and 14 and eight months, respectively.
In recent decades, studies have recommended chemotherapy—specifically a combinational use of platinum, 5-fluorouracil (5-FU), and cetuximab (platinum, 5-FU, cetuximab [PFE])—as a treatment protocol for squamous cell carcinoma of the head and neck (R/M SCCHN). It should be noted, however, that this treatment has led to an average survival of stomal recurrence patients that does not exceed 6 months. 1
Over the last few years, the introduction of immunotherapy in oncology has brought about a real revolution in the treatment of different cancers. Many in the area of head-and-neck-cancer research have advocated for the use of nivolumab, a human immunoglobulin-G4 (IgG4) monoclonal antibody (HuMAb) that binds to the programmed death-1 (PD-1) receptor—which is a negative regulator of T cell activity that controls cellular T immune responses and cytokine secretion—which enables this treatment to activate the immune system against cancer cells and hinder interactions between the PD-1 receptor and the programmed death ligand-1 (PD-L1) and programmed death ligand-2 (PD-L2). Nivolumab, which represents an important therapeutic novelty, was the first anti-PD-1 drug approved in Italy and significantly improved patients’ overall survival (OS) and quality of life when it was used in a Phase III study to treat R/M SCCHN relapses (Checkmate 141).
The aim of this study is to compare and analyze the incidence of stomal recurrence and the survival results of various treatments that have been adopted over the course of 20 years, with new experimental protocols using monoclonal antibodies.
Materials and methods
This study included a total of 487 patients in the Federico II University ENT Unit in Naples, Italy from 2001 to 2021 who were diagnosed with laryngeal carcinoma and underwent either a TL or a salvage surgery after conservative interventions.
Ethical approval and informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional Review Board Committee of Federico II University in Naples, Italy (2020/20457) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. This article does not contain any studies with human participants performed by any of the authors.
Total laryngectomy: Number of cases.

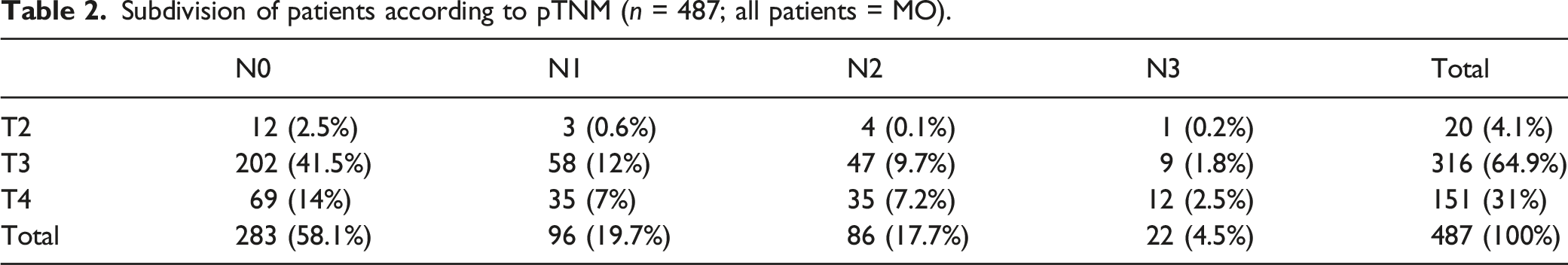
Subdivision of patients according to pTNM (n = 487; all patients = MO).
Subdivision of patient according to stage and grading.

After a histopathological examination of the surgically removed specimens, we concluded that the pan most prevalent histotype was the squamocellular oncotype (98.2%), followed by adenoid cystic carcinoma (0.4%) and warty carcinoma (1.4%). Further examination of the squamocellular oncotypes revealed 39 (8%) cases with basaloid characteristics, 15 (3%) with acantholytic characteristics, and three (0.6%) with spindle-cell characteristics (ie, sarcomatoid carcinoma); of these cases, 101 (20.7%) were G1-grade, 106 (21.8%) were G2, and 280 (57.5%) were G3. These data are also presented in Table 3. Regarding the risk of recurrence when an emergency tracheotomy was performed at least 24 hours prior to the TL, only 53 (10.9%) of the patients underwent this procedure, while the remaining patients were tracheotomized during the TL. 6
Therapeutic strategies for SR and survival in months.
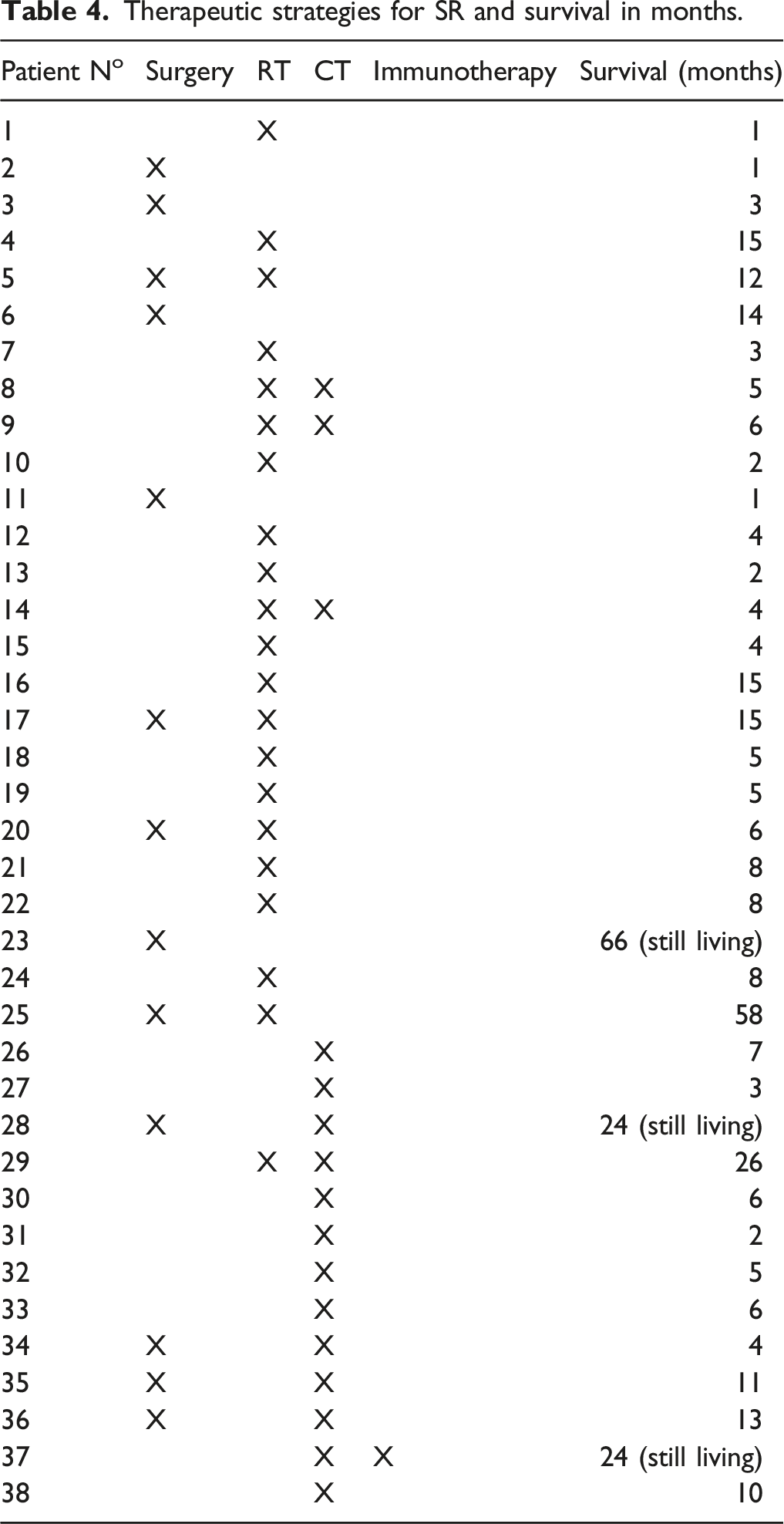
The obtained data were statistically analyzed by applying the chi-squared test using IBM® SPSS® Statistics 28.0 software.
Results
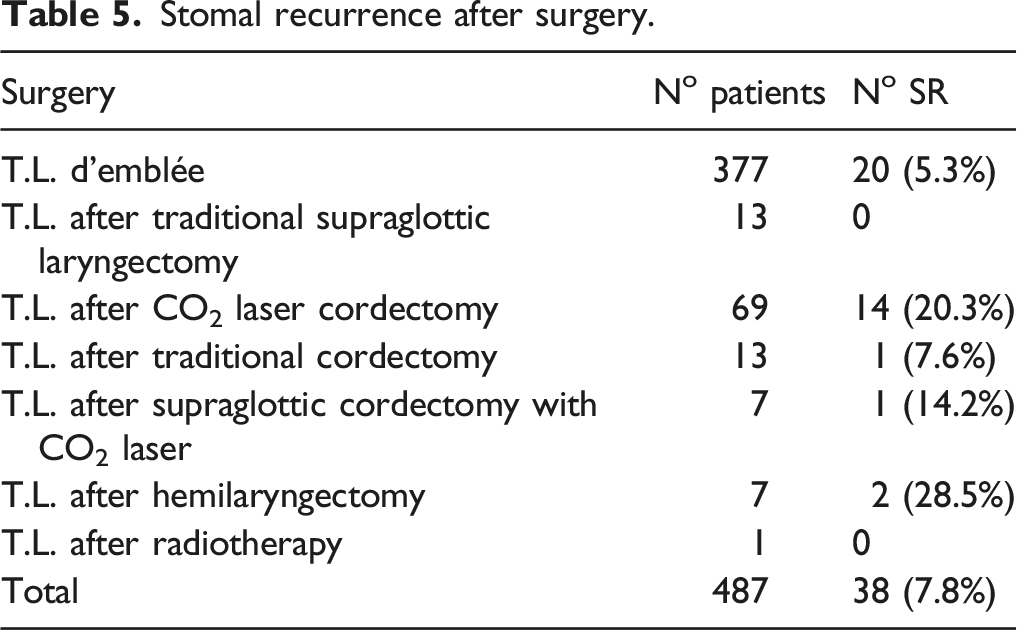
Stomal recurrence after surgery.
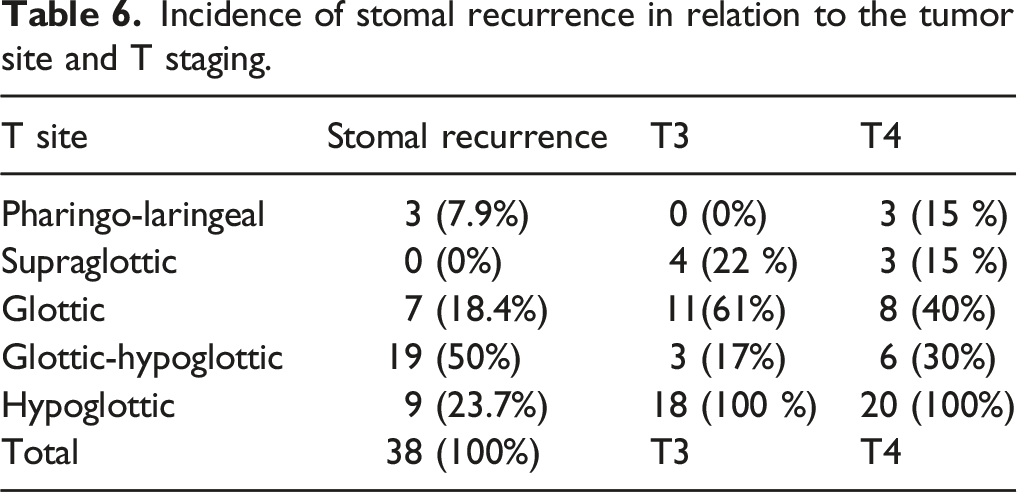
Incidence of stomal recurrence in relation to the tumor site and T staging.
As was previously stated, five (13.15%) cases underwent an exclusive surgical treatment consisting of relapse excision, four (10.5%) chose to combine surgery with radiotherapy, another four (10.5%) opted for surgery combined with chemotherapy, five (13.15%) were treated with a chemoradiotherapy protocol, 12 (31.6%) were only treated with radiation therapy, six (15.8%) were treated solely with chemotherapy, and two patients (5.3%) were lost during follow-up.
The case of patient n. 37 (Tab.4), who was treated with nivolumab and presented an optimal therapeutic response and improved life prospects.
A 64-year-old smoker patient (n.37, tab.4) reported a history of dysphonia for which he had undergone micro-laryngoscopy surgery for squamous carcinoma of the left vocal fold without evident signs of infiltration (pT1N0M0) at a different hospital in May 2018. The patient was admitted to our hospital in January 2019 due to a relapse involving the entire right vocal cord, the anterior commissure, the anterior third of the left cord with fixity, the ventricle of Morgagni, the aryepiglottic region, and the ipsilateral pyriform sinus with inhomogeneous impregnation; small reactive lymph nodes were also bilaterally present in the submandibular and latero-cervical regions.
A TL and a bilateral functional neck dissection were consequently performed. Histological examination revealed “high-grade infiltrating squamous carcinoma with numerous tumor necrosis areas. The mass infiltrated the cartilage, extended to the fibro-adipose and muscular tissues, and reached—but did not infiltrate—the thyroid parenchyma.” Endovascular neoplastic emboli were present. An immunohistochemical study of the tumor revealed positivity for cytokeratin pan (ie, clone AE1/AE3/PCK26; Ventana-Roche); moderate positivity for cytokeratin 5/6 (ie, clone D5/16B4; Ventana-Roche) and cytokeratin 7 (ie, clone SP52; Ventana-Roche); and intense, uniform positivity for protein P63 (ie, clone 4A4; Ventana-Roche). Detection of the synaptophysin, chromogranin, calcitonin, cytokeratin, and mucin neuroendocrine markers were negative. No neck metastasis was observed (pT4N0 SCC3).
The postoperative course was regular. The patient underwent 60 Gy, 30-fraction adjuvant radiotherapy cycles from March 2019 to May 2019. A suspected peristomal recurrence was identified at the January 2020 when we observed a lesion affecting the upper portion of the stoma without the apparent involvement of the esophagus (Figure 1A). (A-D): Stomal recurrence before and during immunotherapy.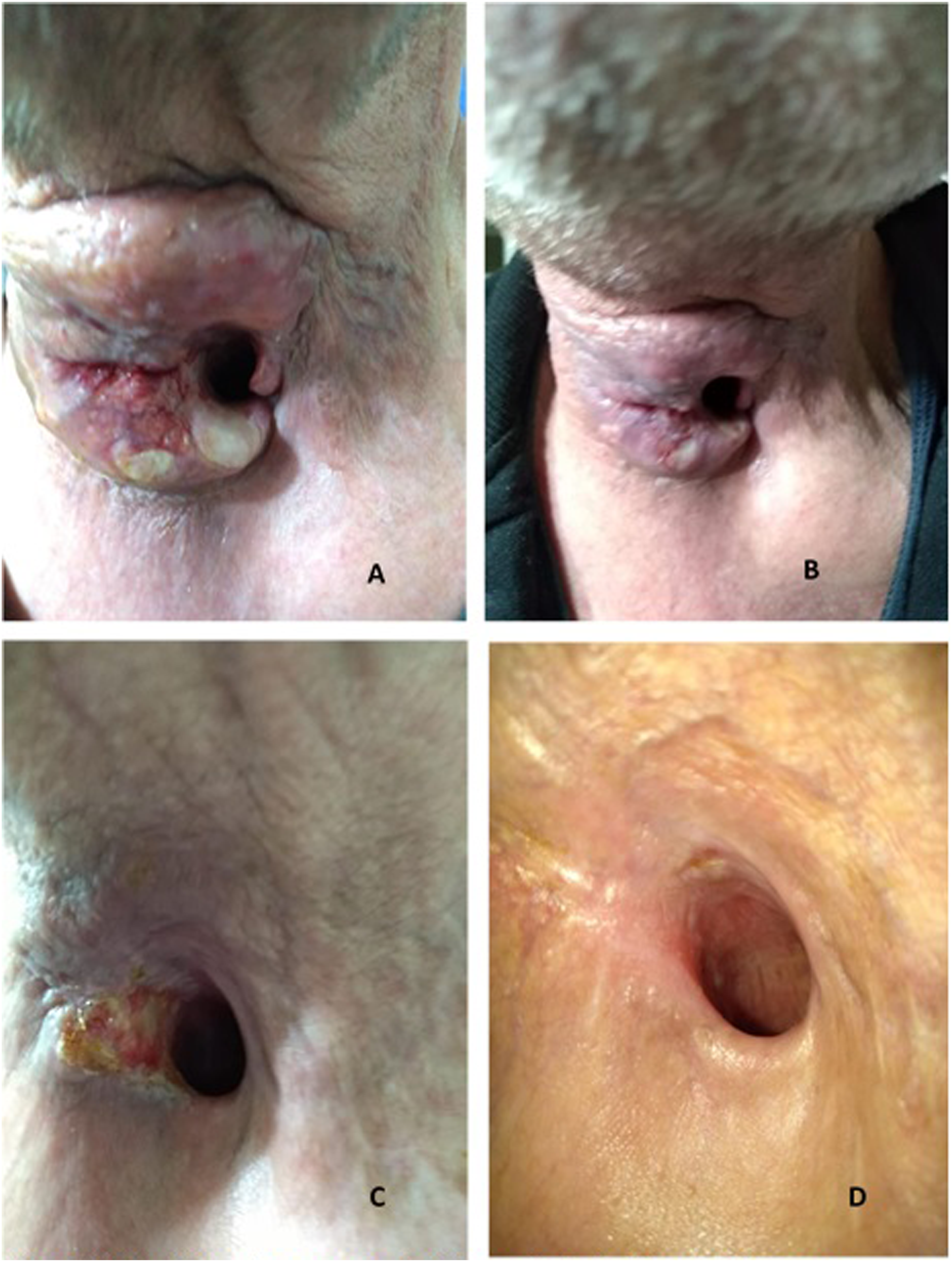
At fine-needle aspiration cytology (FNAC) was observed a moderately differentiated squamous carcinoma. CT revealed a hypodense solid swelling in the right paramedian position close to the stoma with an approximate overall diameter of 45 mm involving the musculocutaneous plane and the right thyroid lobe (Figure 2). Accordingly, MRI revealed a mass extended of 43 mm × 27 mm. While this lesion deviated from the homologated thyroid lobe, there was no evidence of bilateral latero-cervical lymphadenopathy, so the patient was administered a PFE scheme of chemotherapy per the first-line metastatic treatment guideline (Table 7). CT scan of stomal recurrence. Chemotherapy treatment (PFE scheme).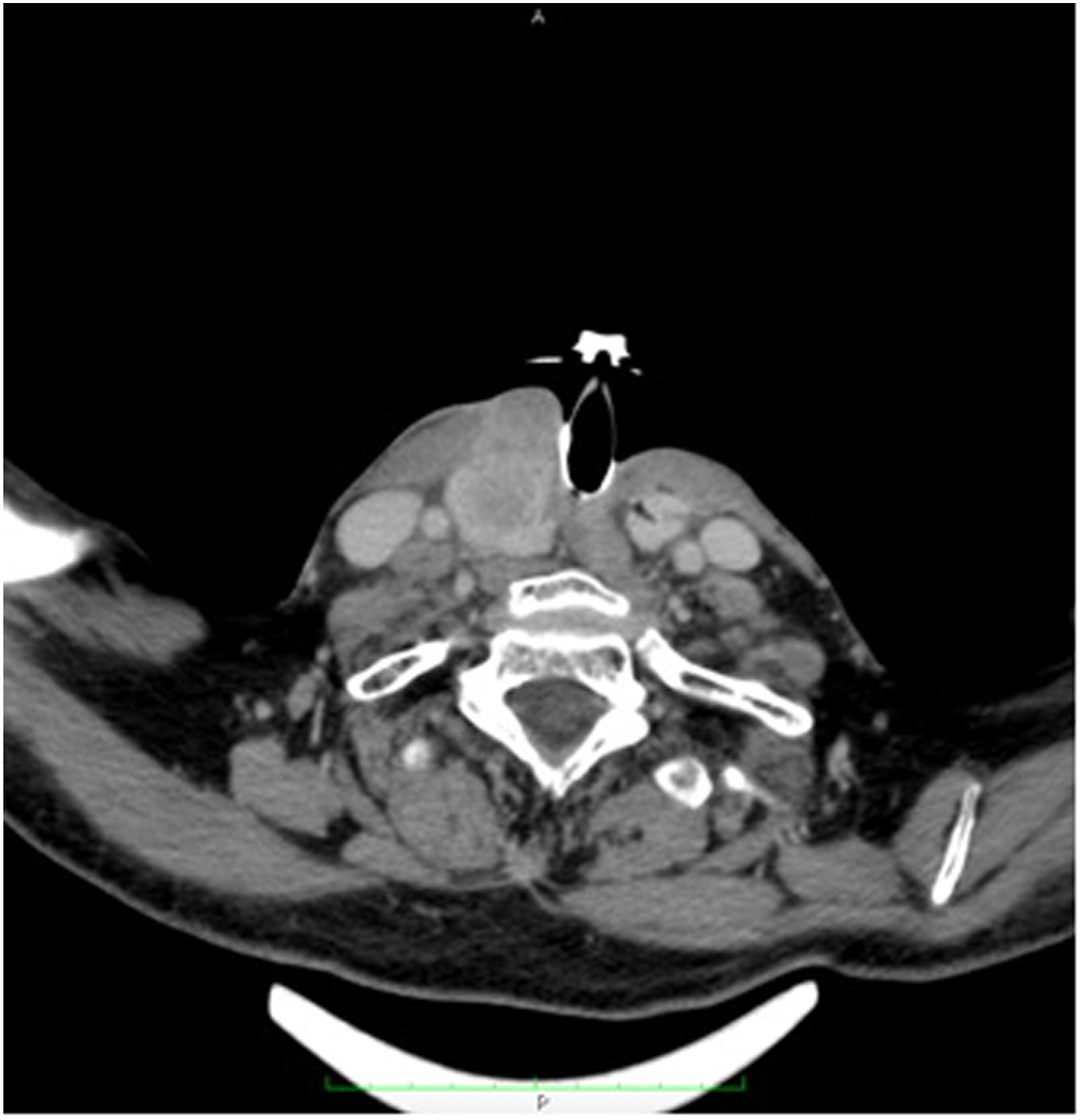
The chemotherapy to which the patient was subjected, however, did not lead to an improvement in his clinical condition. In fact, the CT revealed a dimensional increase of a known solid formation des At this time, this mass was found to have extended approximately 46 mm along the caudal cranial plane, 47 mm along the anteroposterior plane, and 40 mm along the lateral plane, and it presented an inhomogeneous post-contrast impregnation due to the presence of a necrotic hypovascular component infiltrating the musculocutaneous plane, which confirmed the disease progression.
The MRI performed at this same time further confirmed the disease progression. Concomitant hyperintense thickening in the T2 images and hypointensity in the T1 images corresponding to the cutaneous and subcutaneous planes close to the stoma, and infiltrative phenomena that partially extended to the contralateral site was also evident. The pathological tissue appeared near the right common carotid artery; lymphnodal metastasis with approximate axial dimensions of 15 mm in the right submandibular region was observed. We also observed diffuse edematous inhibition of the subcutaneous planes in the right-side latero-cervical area on the right side with a para-esophageal-area extension to the superior mediastinum.
This situation led to the decision to enroll the patient. in an experimental study wherein 240 mg of nivolumab would be intravenously administered for 60 minutes every two weeks; this was due to support in the literature for the lack of therapeutic options for chemotherapy-resistant peristomal relapses and the patient’s encouraging immunohistochemical results, which indicated positive PDL1-protein expression in the patient’s tumor tissue. After six treatment cycles, a total-body CT with a contrast medium conducted in September 2020 confirmed the absence of solid pathological tissue in the right paramedian region close to the stoma; neck mass were not observed. A January 2021, MRI and Total-body CT did not detect relapsing pathological tissue and revealed the locoregional and right submandibular lymphopathies had decreased in volume and were several centimeters smaller than before (Figure 3). MRI scan after immunotherapy.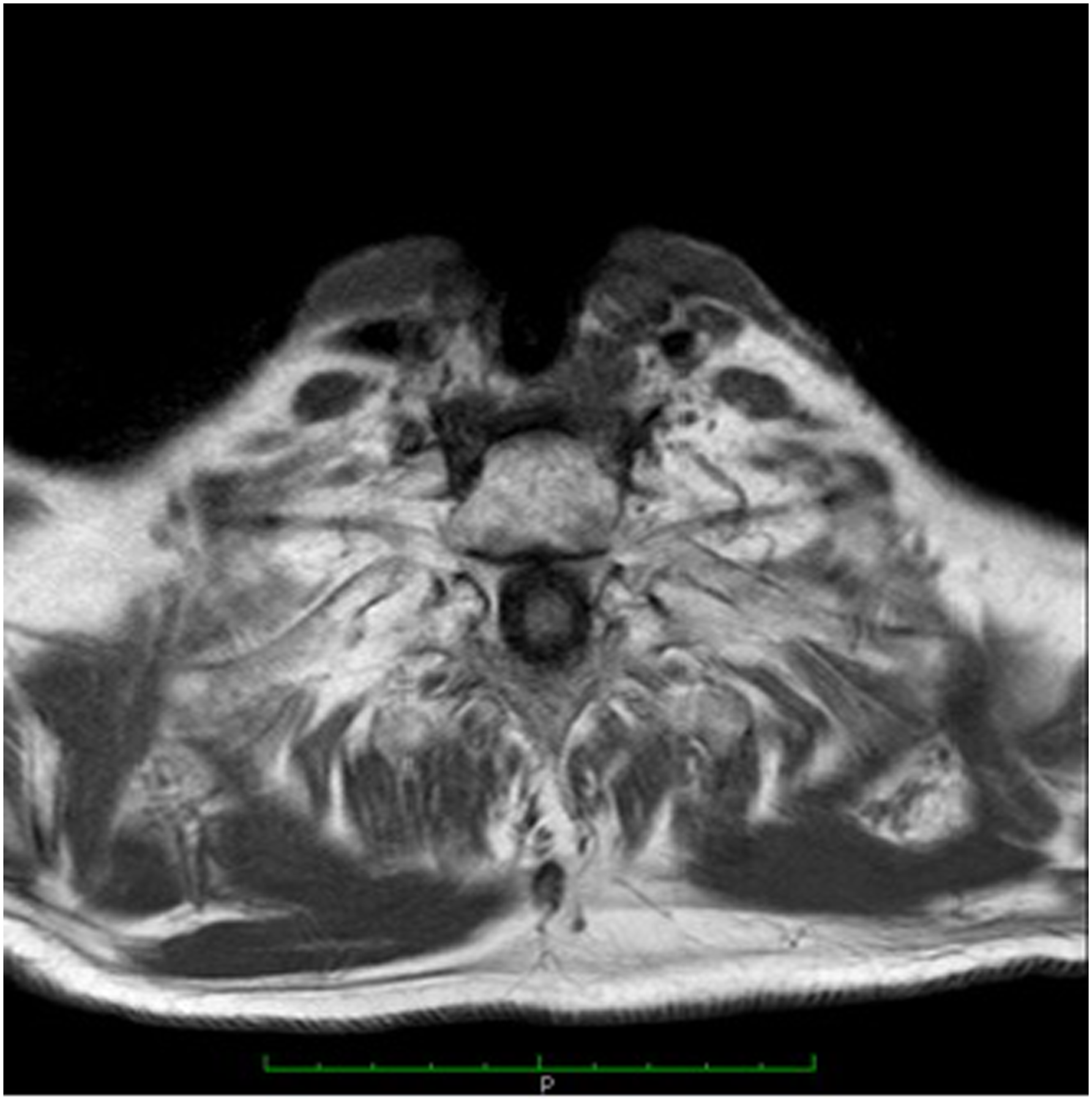
Actually, the patient was undergoing the 40th cycle of nivolumab immunotherapy and planned to continue treatment until no further disease progression or relapse was detected (Figure 1D).
Discussion
Peristomal recurrence is one of the most dreaded TL side effects. According to the literature, 1.7–25% of patients treated for TL experience tracheostoma recurrences; in the case studies we examined, 38 (7.8%) patients experienced peristomal recurrence. We evaluated the incidence of several risk factors for peristomal recurrence several authors detailed in the literature, specifically any conservative interventions prior to the TL; the extent, location, stage, and degree of differentiation of the primary tumor; the presence of metastatic lymphoglandular adenopathies; and the type of tracheotomy performed (ie, emergency or election) in relation to the time of the surgery.
Poor prognoses and limited treatment options with poor outcomes, peristomal recurrence has been challenging for head and neck surgeons. Rather than proposing therapeutic strategies other than those described above—which did not yield satisfactory survival and prognosis outcomes—many authors in the literature review focused on preventing risk factors for peristomal recurrence. 6 In a retrospective study on treatment options in which peristomal relapses were classified, for example, Ampil et al. analyzed patient treatment choices and outcomes and noted that even though surgery was only the primary option in carefully selected cases, chemoradiotherapy was an effective alternative when this option was not viable. 7 Here, the absence of distant metastases and good performance status were fundamental prerequisites, and two patients experienced complete resolution with local tumor control; there was a 10% overall two-year survival rate, and the median survival time with and without repeated treatment was 11 months and seven months, respectively.
While standard first-line therapy for recurrent and/or metastatic R/M SCCHN includes combinational use of platinum, 5-fluorouracil (5FU), and cetuximab, this regimen is associated with severe adverse effects and poor outcomes. 8 Other studies have concluded that if a patient’s disease progresses within six months of the platinum-based chemotherapy protocol, their average OS is 6 months or less. 1
In this study, we evaluated the incidence and associated risk factors of peristomal relapses and the survival rates of patients receiving several different types of treatment: (1) Of the patients in our case studies, 38 (7.8%) experienced peristomal recurrences. Notably, the incidence rate of patients who were primarily treated with conservative interventions was significantly higher than that of patients who underwent a total laryngectomy; the difference was statistically significant (ie, 16.4% vs. 5.3%; p < 0.01). (2) Our data confirmed that a tracheotomy performed a minimum of 24 hours prior to the TL was a risk factor for the onset of peristomal recurrence; we concluded that peristomal relapses occur in 20% of patients who undergo an emergency tracheotomy and 6.2% of patients who undergo a tracheotomy during TL (p < 0.01). It is essential, however, to consider other factors that frequently coexist with an emergency tracheotomy and independent risk factors for peristomal recurrence, specifically the frequent involvement of the subglottic region and the extent of the primary tumor. 3) We discovered that 359 (73.7%) of the peristomal relapses in our case studies occurred in patients with lesions that were primitively or secondarily located in the subglottic region; a comparison with other locations demonstrated a statistically significant difference (ie, 73.3% vs. 26.3%; p < 0.001). 4) As it relates to the primary tumor (pT) extension, peristomal recurrences were only observed in patients with large tumors—18 T3 patients and 20 T4 patients—and no recurrences were observed in patients with T2 neoplasms. 5) Concerning treatment, our data confirmed the difficult prognoses associated with various therapeutic options. Even though multiple treatments were administered, most of these patients were given a poor prognosis: 32 (89%) patients died within 1–15 months with an average of 6 months, four (11%) survived for 24–66 months with a mean of 43.5 months, one died 26 months after their peristomal recurrence diagnosis; and another patient died after 58 months due to a neuroendocrine tumor of the pleura. Three of the four patients underwent surgical treatments, some of which included radiotherapy and/or chemotherapy (see Figure 4). (6) In terms of survival, we were unable to identify any predictive factors or responses to therapy based on our examination of the general characteristics. Case survival curves.
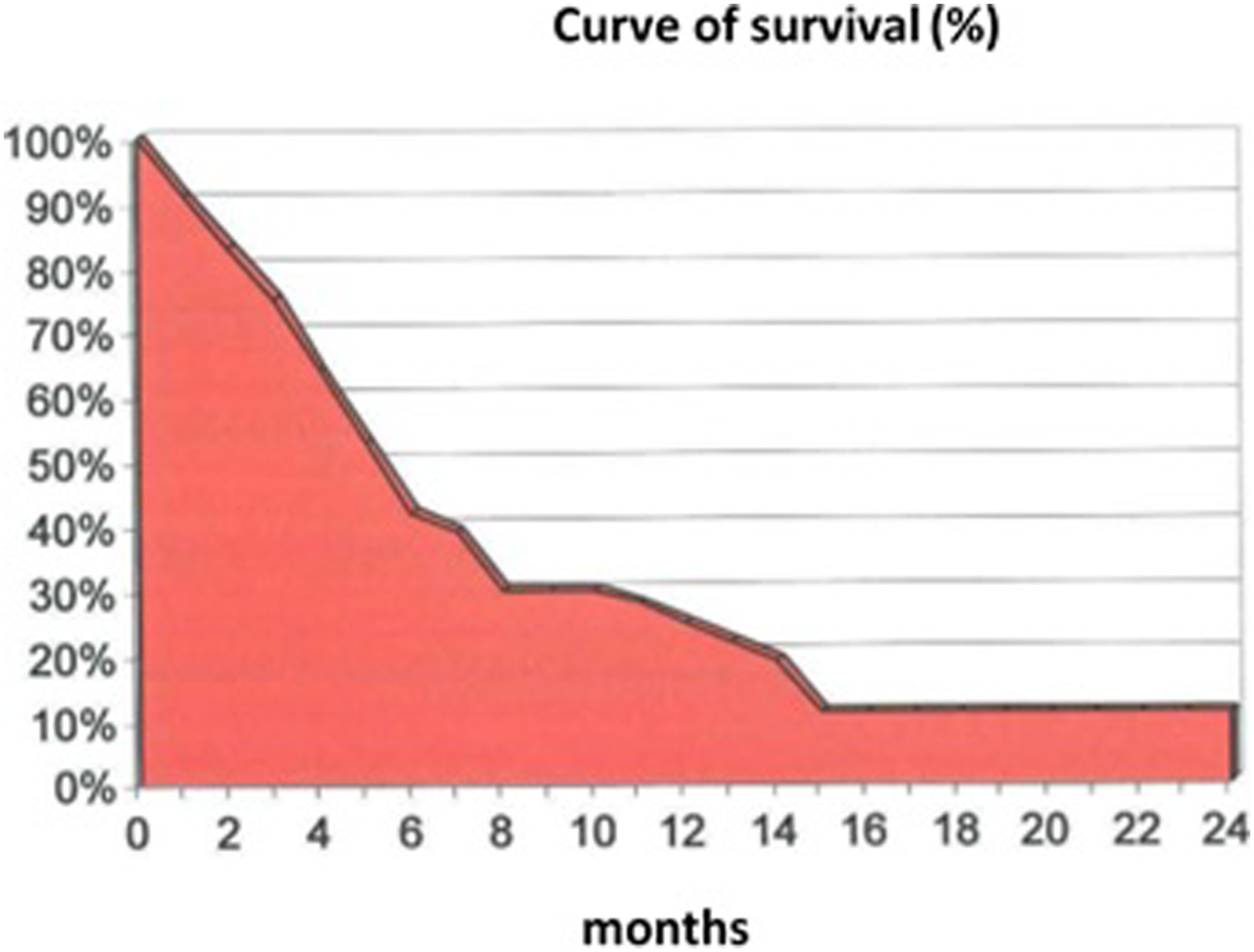
The severity of an SR prognosis was evidenced by the results reported in the literature and our inability to identify specific factors that either contribute to therapeutic success or provide prognostic indicators. As a result, the significance of adopting preventive measures was primarily represented during the initial intervention by precise oncological radicality criteria, and molecular antibodies were utilized to represent the importance of new therapeutic strategies in delayed cases.
The recurrence and metastasis of the squamous cell carcinoma was facilitated by immune evasion that was partially mediated by the expression of PD-L1 and PD-L2, which bind the suppressive immune checkpoint receptor (ie, the PD-1) of T cells; specifically, the PD-L1 on tumor cells combines with the PD-1 on cytotoxic T cells to transmit a second inhibitory signal to T cells, resulting in the exhaustion, dysfunction, and progression of effector T cells. PD-L1 also binds to CD80 (ie, B7–H1), which provides inhibitory signals to T cells, and PD-1 inhibitors function as immunomodulators by blocking PD-1 receptors, stimulating an immune response, and mediating tumor regression. 9
Theodoraki et al. demonstrated the presence of soluble PD-L1 (sPD-L1) in the plasma of patients with SCCHN. 10 The mechanism underlying PD-L1 expression has been studied extensively over the past decade; like PD-L1, PD-L2 inhibits T cell activation, reduces cytokine production, and stimulates T cell cytolysis. 10 Notably, neither Yearley et al. nor Schoenfeld et al. were able to detect PD-L2 expression was not detected in the SCCHN tumor parenchyma in most cases.11,12
The U.S. Food and Drug Administration (FDA) has approved two PD-1 inhibitors—nivolumab and pembrolizumab—for the treatment of advanced SCCHN. Furthermore, several clinical trials involving the PD-L1 inhibitors durvalumab and atezolizumab have either concluded or are ongoing; to date, PD-1/PD-L1 blockades have been shown to reduce tumor growth and achieve recurrent tumor regression in SCCHN. 9
Nivolumab is a high-affinity IgG4 HuMAb that inhibits PD-L1 binding to PD-1 and CD80. The FDA approved nivolumab in 2016 to treat recurrent/metastatic SCCHN with or without PD-L1 expression based on the Phase III clinical trial results of CheckMate141, which demonstrated lower OS, fewer benefits, and less toxicity than the standard treatment. In this study, nivolumab prolonged patients’ median overall survival (mOS) compared to standard therapies (ie, methotrexate, docetaxel, or cetuximab)—7.5 months vs. 5.1 months—even though the patients’ median progression-free survival (mPFS) was not affected. The overall response rate (ORR) was 13.3% in the nivolumab group and 5.8% in the standard therapy group, and the incidence of grade 3-4 adverse events (AEs) was 13.1% and 35.1%, respectively. According to preliminary findings, PD-L1-positive (ie, with a cut-off that is ≥ 1%) and p16-positive tumors are more likely to benefit from nivolumab monotherapy.13-17
The use of nivolumab in this study enabled us to achieve highly satisfactory clinical response and survival rates. As described above, a patient suffering from squamous carcinoma of the larynx (ie, pT4N0 SCC3) presented a peristomal recurrence one year after TL; the lesion measured 43 mm × 27 mm and involved the musculocutaneous plane of the upper MPs portion of the stoma. Even though the patient received chemotherapy per PFE protocol, the disease progressed approximately 6 months later. In light of the literature supporting nivolumab's efficient treatment of SCCHN recurrences and considering the presence of PD-L1 in the immunohistochemical analysis of the patient’s tumor, the patient opted for nivolumab; in addition to extending the patient’s survival, nivolumab also improved their quality of life.
While it should be noted that a more extensive series of cases is needed to support this therapeutic strategy, this study can be viewed as a pilot case for a new therapeutic approach that led to highly satisfactory results, especially when compared to other chemoradiotherapy or surgical options. Immunotherapy yielded a result that suggested a potential new treatment for this once-fatal complication; even though there are AEs associated with nivolumab therapy, these were significantly more tolerable than those caused by other treatment protocols.
Implication for practice
In light of our findings, it is evident that conservative surgery, subglottic involvement by primary neoplasm, an advanced T stage, and an emergency tracheotomy were all statistically associated with peristomal relapses, the prognosis for which is decidedly dismal, even when the most precise therapeutic measures currently available; as such, it is essential that preventive measures are implemented at the time of the primary tumor intervention. In the event of inevitable risk factors in this context, immunotherapy leads to new therapeutic possibilities, especially when surgery and chemotherapy fail, and a standardized protocol with stringent eligibility requirements is needed. The survival data obtained with nivolumab in the present case provides a new perspective on the possibility of successfully treating SCCHN recurrences, even in the rare-but-ominous event of a peristomal recurrence.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest concerning this article’s research, authorship, and/or publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
