Abstract
Objective:
Lymphoepithelial-like carcinoma of the salivary glands (LELCSG) is a rare tumour of unknown aetiology. No studies have reported the imaging features of primary LELCSG.
Methods:
The clinical information and imaging features of eight patients with LELCSG were reviewed. Computed tomography (n = 4 patients) and magnetic resonance imaging (n = 4 patients) features were analysed by two radiologists to identify the location, number, size, shape, boundary, signal intensity and enhancement of LELCSG.
Results:
The study included four women and four men, and the mean size of the tumours was 32.88 ± 3.41 mm (range, 27–38 mm). The tumours affected the parotid gland in six cases and the submandibular gland in two cases. The eight cases were evaluated by
Conclusions:
LELCSG is a lobulated, multi-nodular tumour, with some fused nodules. LELCSG lesions showed isointensity or slight hyperintensity on T1WI MRI, slight hyperintensity on T2WI MRI and
Introduction
Lymphoepithelioma-like carcinoma (LELC) is a rare type of cancer that typically occurs in pharyngeal and foregut-derivative organs. 1 However, LELC has also been observed in other anatomical locations,2-8 including the salivary gland, bladder, lacrimal glands, ovaries, cervix and skin. Pulmonary LELC typically affects young, non-smoking individuals and is associated with Epstein–Barr virus infection. 9 When LELC occurs in the salivary glands (i.e. lymphoepithelial-like carcinoma of the salivary glands [LELCSG]), the most common site is the parotid gland. 10 LELC shows specific regional and ethnic distributions; the incidence of LELCSG is highest among Eskimo, Greenlandic Inuit, southern Chinese and Japanese populations. 11
Lymphoepithelioma consists of poorly differentiated cells with large nuclei and nucleoli within the lymphoid stroma; it occurs mainly in the nasopharynx. 12 The World Health Organization considers LELC, large-cell undifferentiated carcinoma and small-cell undifferentiated carcinoma to be three subtypes of undifferentiated carcinoma of the salivary gland. 13 Histologically, LELC and large-cell undifferentiated carcinoma have similar morphologies in terms of malignant epithelial cells, although large-cell undifferentiated carcinoma lacks lymphocytic stroma around tumour cells. 14 Patients with large-cell undifferentiated carcinoma of the salivary gland have poor outcomes; the 5-year overall survival ranges from 0% to 35%. The prognosis of patients with LELCSG is comparatively better. 14
Qi et al. 15 reviewed the relevant LELC literature over the past 50 years in both PubMed and Web of Science and found only a few articles regarding LELCSG.14,16-21 Notably, the imaging features of LELC have not been summarized. In the present study, we analysed the imaging features of eight patients with primary LELCSG to identify the imaging features of this rare tumour, which may aid in its diagnosis.
Materials and Methods
Patients
The institutional review committee of our hospital approved this retrospective study and waived the requirement for informed consent. A total of 11 LELCSG cases were retrospectively analyzed, including 8 cases treated in the First Affiliated Hospital of Sun Yat sen University from 2018 to 2021, and 3 cases treated in Zhuhai Hospital of Guangdong Provincial Hospital of traditional Chinese medicine from 2015 to 2021. Among the 11 cases, 2 cases were excluded because they were limited to postoperative assessment and 1 case with only CT scan was excluded. A final total of eight cases were included in the analysis. Surgically resected specimens were subjected to conventional fixing, dehydration, paraffin embedding, sectioning and haematoxylin-eosin staining, followed by analysis by light microscopy. The specimens from the eight patients were subjected to immunohistochemical staining with Envision two-step technology (Figures 1A and 1B). H-E staining, immunohistochemistry and MRI T2WI image of LELCSG lesion found in a 33-year-old male. (A) Haematoxylin-eosin staining (×100). Many epithelioid tumour cells are present in a nested or scattered distribution; the tumour cell nucleus is large and darkly stained, while some nuclei are vacuolated with obvious nucleoli. Large numbers of lymphocytes are evident around the tumour tissue. (B) Immunohistochemistry analysis shows positive CK5/6 staining. (C) Left submandibular gland with multi-nodular shape, (red arrow), blurred edge and incomplete capsule (blue arrow). The signal intensity are similar in metastatic lymph nodes and LELCSG (orange arrow).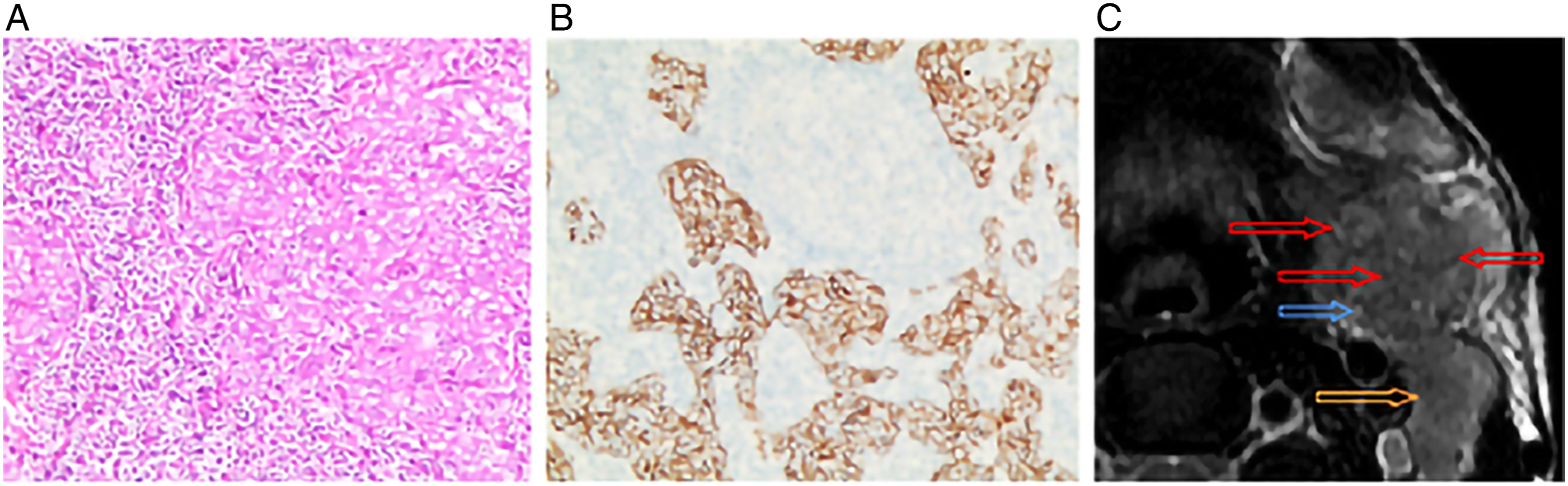
Magnetic Resonance Imaging (MRI) Protocols and Computed Tomography (CT)
Four patients were examined with magnetic resonance imaging at 3.0 T (two patients: Magnetom Verio; two patients: Signa Excite 3.0 T); spin echo or fast spin echo T1-weighted imaging (T1WI) (337–840/8–13 ms repetition time [TR]/echo time [TE]) and T2-weighted imaging (T2WI) (2000–4350/87–130 ms TR/TE) were used to obtain images in the axial, coronal and/or sagittal planes. MRI contrasted enhancement was performed after intravenous injection of gadopentetate dimeglumine (0.1 mmol/kg; Magnevist, Schering, Germany); the image thickness was 3–3.5 mm. Enhanced fast spin echo fat-suppressed T1WI (337–840/8–13 ms TR/TE) was also performed. Four patients received CT scan with contrast enhancement; the image thicknesses were 1–3 mm (two patients: Aquilion ONE Genesis, tube voltage, 120 kV; tube current (mA), smart milliampere; two patients: Somatom Definition Flash, tube voltage, 120 kV; tube current (mA): smart milliampere).
Image Analysis
The images were retrospectively interpreted by two experienced radiologists using the hospital image archiving and film reading systems; the radiologists were blinded to the clinical and pathological data of the patients, although they were aware that the patients had been diagnosed with LELCSG. Any disagreements between radiologists were resolved by discussion until a consensus was reached. The following imaging features were evaluated for each tumour: location, number, size (maximum diameter), shape, edge, T1WI signal intensity, T2WI signal intensity, T1WI contrasted enhancement signal intensity, plain and contrasted enhancement intensity of CT, necrosis, haemorrhage, degree of enhancement and multi-nodular changes.
Results
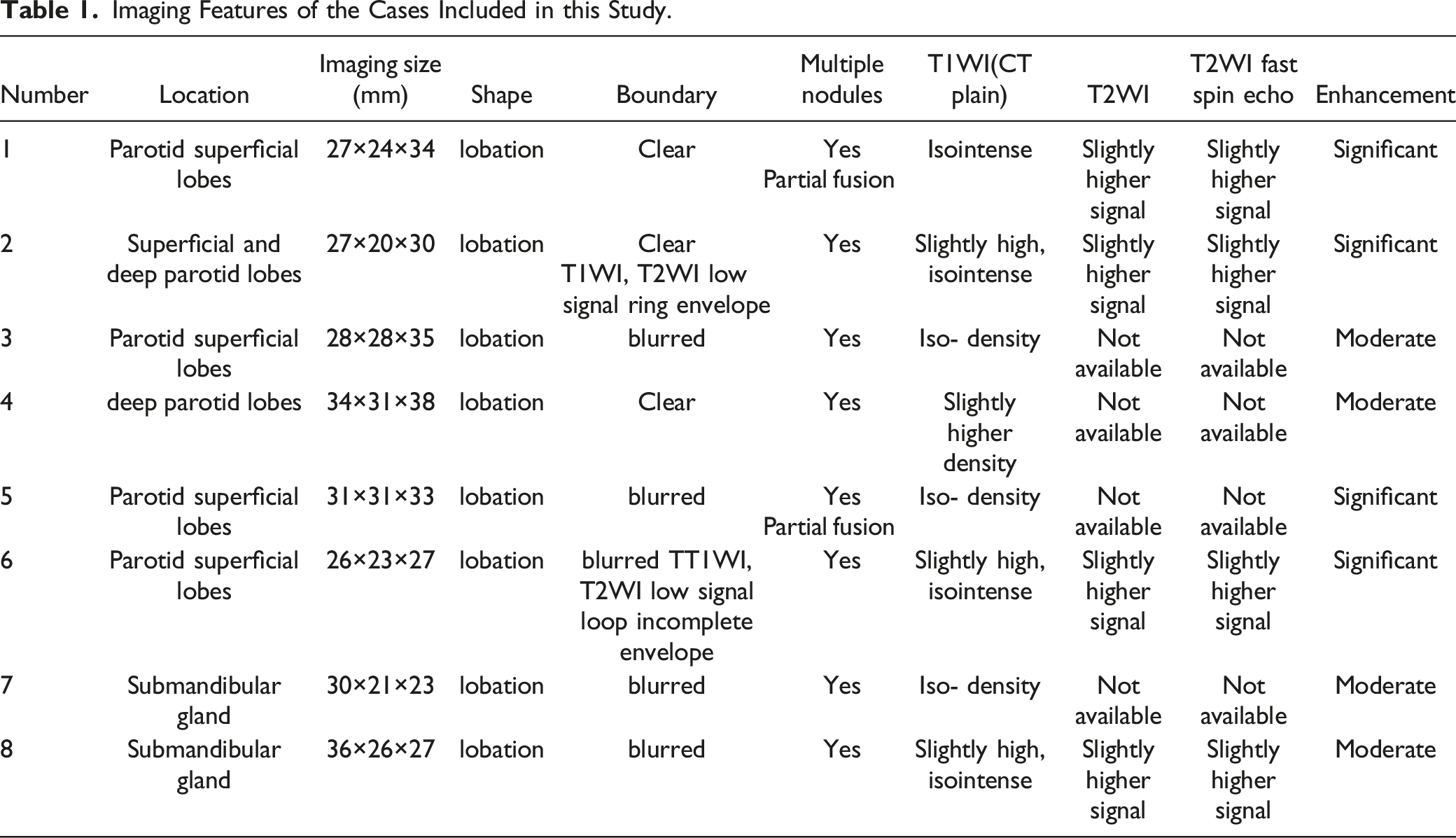
Imaging Features of the Cases Included in this Study.
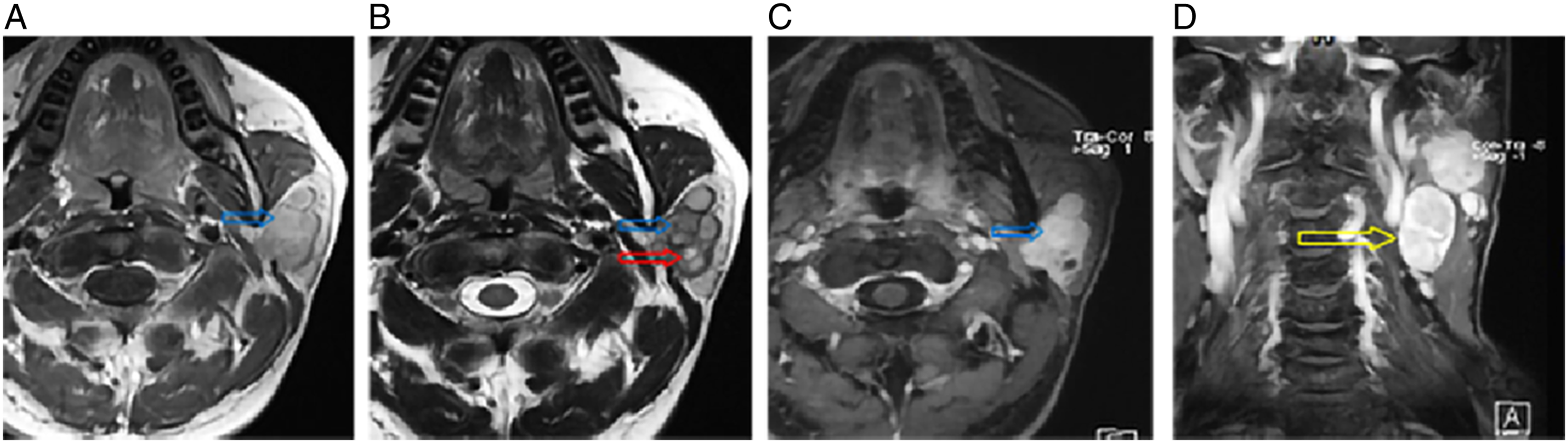
MRI images of LELCSG in a 43-year-old male. (A–C) Axial T1WI, T2WI and enhanced T1WI. (D) Coronal T1WI enhancement shows multiple nodules, incomplete medial capsule (blue arrow), and a few necrotic foci (red arrow). The degrees of enhancement are similar in metastatic lymph nodes and LELCSG (yellow arrow).
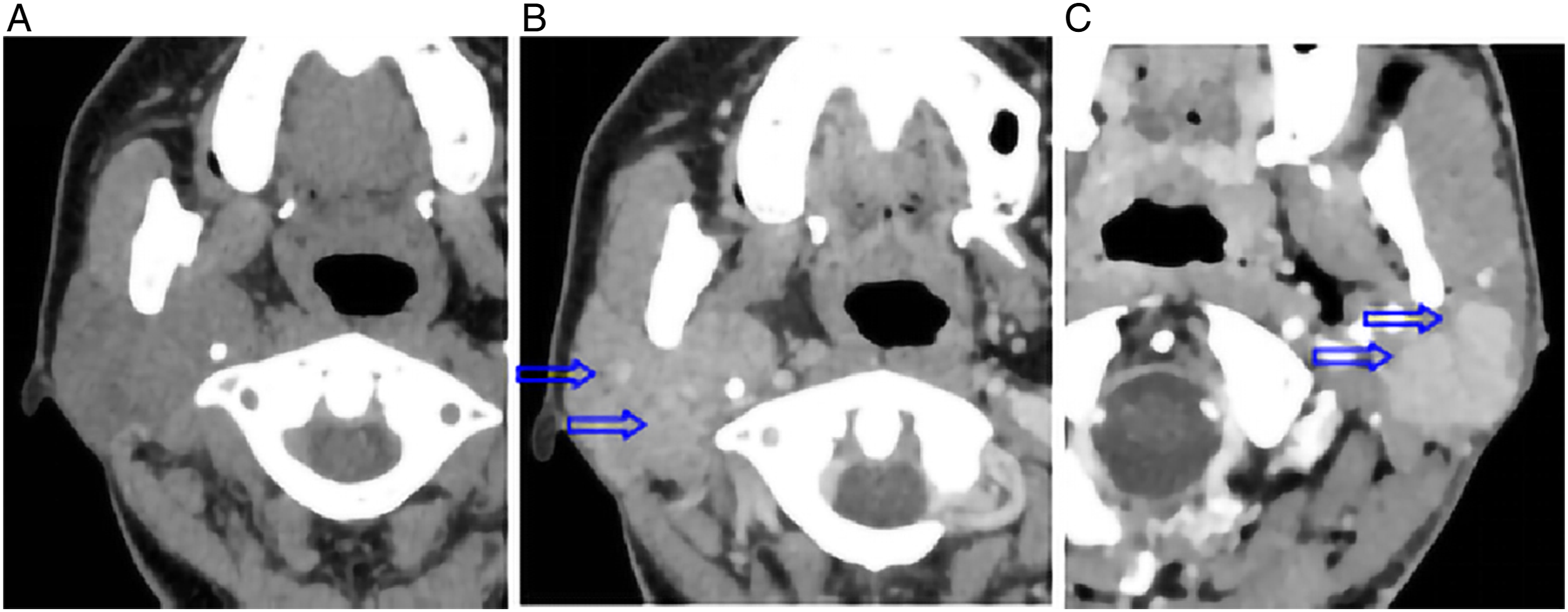
CT images of a 37-year-old female (A, B) and enhanced CT image of a 57-year-old female (C). (A, B) LELCSG tumour in CT image and enhanced arterial phase CT image. The tumour exhibits moderate enhancement with an unclear lesion boundary and multiple internal nodules (arrows). (C) Enhance Computed tomography shows obvious enhancement and multiple nodules with clear boundary.
Specialist Examination of the Cases Included in this Study.
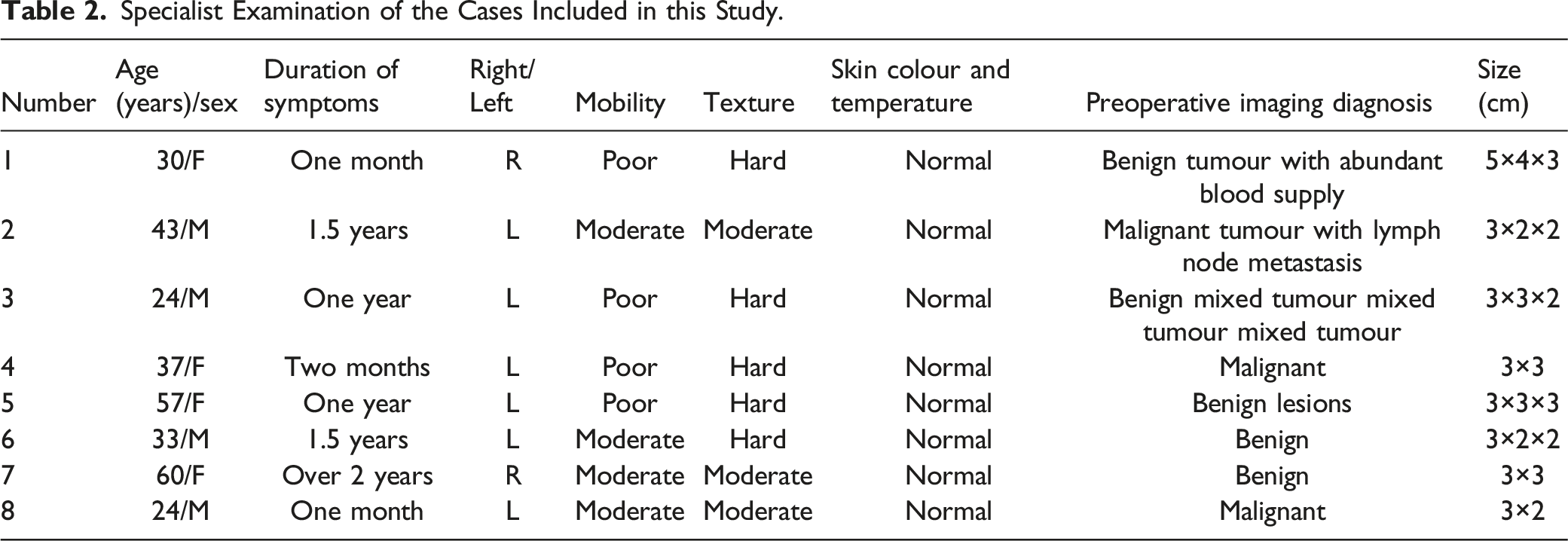
Concerning location, all tumours were unilateral and mainly located on the left side (seven tumours). Five lesions occurred in the superficial lobe of the parotid gland or concurrently across the deep and superficial lobes (Figures 2 and 3C); only one lesion occurred in the deep lobe (Figures 2A and 2B). All patients had a single lesion (size, 27–38 mm) with multiple aggregated nodules. Larger lesions with partial fusion demonstrated incomplete separation, considering that extracapsular invasion involved multiple nodules with more incomplete capsules and mass integration (Figure 2). In terms of morphology, all tumours were lobulated (Figures 1, 2, and 3).
Concerning signal strength and degree of enhancement, plain T1WI showed isointensity or slight hyperintensity and moderately obvious homogeneous enhancement on contrasted enhancement T1WI scan, while T2WI showed slight hyperintensity.
The overall texture was generally solid, and small areas of necrosis were evident in larger tumours. Furthermore, adjacent fat and skin infiltration was observed in combination with regional lymph node metastasis. Although normal neck tissue often has multiple lymph nodes with different sizes, the metastatic lymph nodes of the patients had signal and enhancement characteristics similar to the parotid tumour characteristics; thus, they could be differentiated from normal neck lymph nodes. Examination by specialists revealed that the metastatic lymph nodes showed medium texture, hard and clear boundaries, normal or poor mobility, normal skin colour and temperature and occasional swollen lymph nodes (Figures 1 and 2).
Discussion
In this study, we explored the MRI and CT imaging features of LELCSG in eight patients and summarized the characteristics. These findings may help provide some guidance for the diagnosis of LELCSG in clinical practice.
Our analysis revealed the following characteristics in patients with parotid gland lymphoepithelioma-like carcinoma
Preoperative imaging examination plays an important role in the diagnosis and treatment of LELCSG. Misdiagnosis from imaging negatively impacts the treatment decision, with potential consequences such as delayed operation, which influences the survival and life quality of patients. Therefore, ensuring accurate imaging diagnosis is critical. The other published studies did not address the imaging features of LELCSG.14,16-21 Thus, our findings may provide a deeper understanding of the MRI or CT imaging features for LELCSG, which might help guide the diagnosis and treatment of LELCSG.
Nasopharyngeal lymphoepithelioma is a radiosensitive tumour, and radiotherapy is the standard treatment. 22 Non-nasopharyngeal lymphoepithelioma of the head and neck is also reportedly radiosensitive with high rates of locoregional tumour control. 23 At present, treatment of LELCSG mainly consists of surgical treatment, sometimes with postoperative local radiotherapy when necessary. The recurrence rate is low in patients who undergo postoperative local radiotherapy.12,23,24 In the present study, five patients were misdiagnosed with benign tumours, resulting in a misdiagnosis rate of 62.5%. Misdiagnosed benign tumours of the parotid gland should be reviewed regularly because of the potential for harm from delayed treatment.
Currently, surgeons and radiologists have insufficient knowledge of LELCSG; our patients were misdiagnosed with mixed parotid tumours or benign lesions based on imaging findings. Instances of lymph node metastasis were diagnosed as malignant tumours. As stated above, misdiagnosis may lead to delayed treatment and negative consequences. Characteristics of mixed parotid tumours25,26 include good mobility, location in the superficial lobe of the parotid gland, round shape, inhomogeneous signal and enhancement, visible cystic degeneration, necrosis, calcification and moderate enhancement. Compared with the characteristics of other types of salivary gland tumour, such as adenolymphoma, the characteristics of adenolymphoma include greater frequency in older men (in contrast to LELCSG), frequent location at the posterior and inferior poles of the parotid gland, bilateral occurrence, multiple nodules, cystic degeneration without regional lymph node metastasis and no invasion of adjacent subcutaneous fat and skin.26,27 Physical examination by specialists revealed good activity and medium texture. Characteristics of lymphoma 28 include mild intensity and rare occurrence solely in the salivary glands. The characteristics of malignant parotid gland tumours 26 include differentiation from common mucoepidermoid carcinoma, adenoid cystic carcinoma and mixed tumour malignant transformation, primarily because of obvious necrosis and cystic transformation. For a more accurate diagnosis of LELCSG, pathological examination is also needed, which is the standard diagnosis strategy for many types of tumours.
Conclusion
Lymphoepithelial-like carcinoma of the salivary glands tumours are rare and most commonly occur in patients with Epstein–Barr virus infection. These tumours are lobulated, multi-nodular and partially fused. LELCSG also exhibits isointensity or slight hyperintensity on T1WI, as well as slight hyperintensity on T2WI and
Footnotes
Author’s contributions
All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
Radiology Department, Zhuhai hospital Guangdong Hospital of Traditional Chinese Medicine.
Ethics Statement
This study was approved by the institutional review board of ----Ethics Committee of Guangdong Provincial Hospital of Traditional Chinese Medicine, and all patients provided written informed consent.
Consent for Publication
Written informed consent for publication was obtained from all participants.
Availability of Data and Material
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
