Abstract
Objectives
This study investigated the changes in vestibular and cochlear function in patients receiving platinum-based chemotherapy.
Methods
A longitudinal study of 32 participants (10-70 years) receiving chemotherapy participated in the study. Baseline and exit vestibular and hearing assessments that included video head impulse (VHIT) testing, cervical and ocular vestibular evoked myogenic potentials (VEMP), dynamic visual acuity (DVA) and pure-tone audiometry were performed at the patient’s treatment venue.
Results
Half (50%) of the participants showed cochleotoxicity from baseline to exit testing, with left ears significantly more affected than right ears. There was no consistent relationship between hearing loss and vestibular dysfunction. DVA yielded normal results at baseline and exit testing in all participants. VEMP responses were absent in 28.1% of participants at baseline, reflecting the challenges of using VEMP for monitoring. VEMP and VHIT results showed a statistically significant (p < 0.05) decline in results from baseline to exit testing; however, participants did not report symptoms related to vestibular dysfunction. VHIT also showed left ears significantly (p < 0.05) more affected than right ears.
Conclusion
VHIT proved to be a valuable measure of changes in vestibular function secondary to ototoxicity. Future investigations should determine vestibulotoxicity criteria and optimal protocols for vestibulotoxicity monitoring at the patient’s treatment venue.
Keywords
Introduction
Cancer is considered one of the world’s leading fatal diseases, with nearly 19.3 million new cases and 10 million deaths in 2020. It is estimated that the total number of people living within five years of a cancer diagnosis, called the five-year prevalence, is 50.6 million globally. 1 While cancer seems to be a life-altering disease, in the last two decades there has been an overall decrease (26%) in cancer deaths thanks to medical advances. 2 However, recovery may also result in long-term physical and psychological complications for survivors. 3 More focus is therefore placed on long-term effects, health-related quality of life (HRQoL), and follow-up care after cancer treatment as a result of these improved survival rates. 4
Platinum-based chemotherapy is a key antineoplastic intervention used for a variety of human cancers, including testicular, ovarian, bladder, head and neck, and non-small cell lung cancer. 4 Ototoxicity refers to medication-induced auditory and/or vestibular system dysfunction that results in hearing loss or disequilibrium. 5 Furthermore, ototoxicity is a well-known adverse effect following platinum-based chemotherapy (especially cisplatin), which causes variable-degree permanent irreversible hearing loss in 40-80% of patients. 6 As signs of ototoxicity are poorly correlated with drug dosage, peak serum levels, and other toxicities, the only way to detect ototoxicity is by assessing auditory and vestibular function directly. 4
Since the inner ear's auditory and vestibular organs share the same blood, nerve and fluid sources, ototoxicity is possible in both the hearing and vestibular systems. 5 Yet, there are some variations in physiologic function between the cochlear and vestibular end organs, which may affect the extent of ototoxicity. A major physiological difference between the cochlear and vestibular systems is the endolymphatic potential. 7 The vestibular system demands lower endolymphatic potentials for its proper function compared to the high endolymphatic potential in the cochlea. Morphological vestibular damage was not found at early stages in ototoxicity, which suggests that functional vestibular impairment may not be associated with sensory hair cell damage, but rather with other biochemical factors such as electrolyte or electro-potential disturbances. 4 Therefore, considering this shared anatomy and physiology of the inner ear structures, ototoxicity is not limited to alterations in cochlear functioning. 4
The monitoring of cochleotoxicity in platinum-based chemotherapy is well established; however, there are no universally accepted guidelines for vestibulotoxicity monitoring and it is rarely conducted in patients who are critically ill. 4,5,8 Furthermore, present ototoxicity testing recommendations include detailed test protocols. 9 The biggest challenge in monitoring vestibulotoxicity is the detection of symptoms that are only visible when patients are mobilised, which can also be falsely attributed to the patient’s weakened state. 9 By the time a patient complains of imbalance or dizziness, permanent vestibular system damage has more than likely already occurred. For patients undergoing chemotherapy, the difficulties of introducing an ototoxicity monitoring protocol include fatigue, general acute illness, travel problems and priority issues. 9 Limited research has been published about the potential effects of cisplatin on the vestibular system. Furthermore, there is a large variability (0-50%) in the rates of vestibulotoxicity reported by objective tests following treatment with platinum-based chemotherapy. 4 Other limitations of published studies are small sample sizes, various methods of vestibular evaluation and criteria to determine abnormalities in the vestibular system, and outdated studies.
Since there is no single test that can identify vestibulotoxicity, tests for screening, such as dynamic visual acuity (DVA), dizziness handicap inventory (DHI) and head impulse tests are recommended to monitor patients. In addition, diagnostic vestibular procedures are also often impractical due to the compromised health status of the patient. 4,9,10 Vestibular symptoms reported by patients are often underappreciated due to the underlying cancer diagnosis and adverse effects of treatment, such as dehydration, nausea and vomiting, persistent weakness, anaemia and hypotension, 11 which can be due to nonspecific symptoms of imbalance. In addition, vestibulotoxicity is typically symmetrical and progressively affects both ears, resulting in insidious imbalance, postural imbalance and oscillopsia, which are less likely to undergo clinical evaluation. 10 Additionally, due to the slow progressive nature of vestibulotoxicity, vestibular dysfunction may be hidden by central compensation, obscuring peripheral vestibular pathology. 12 Lastly, platinum-based chemotherapy agents are often prescribed with other potentially ototoxic drugs and the effect of platinum-based chemotherapy can be obscured. 13
It is important to identify the presence, severity and nature of vestibular signs in patients on chemotherapy treatment so that healthcare providers can be alerted early, to mitigate debilitating vestibular symptoms affecting the patient’s HRQoL and potential earning ability after remission. 4,14 Balance concerns such as falls and impairment in mobility are more dominant in cancer survivors than in the general population. This is of importance because falling is a leading cause of injury and death in the community. 15 Early identification and monitoring of vestibulotoxicity provide audiologists with the opportunity to perform appropriate rehabilitation during and after treatment. 16,17
Vestibulotoxicity confirmed by objective vestibular assessments has been associated with cochleotoxicity (either hearing impairment or tinnitus). 5,18 There is some evidence of vestibulotoxicity associated with platinum-based chemotherapy, especially cisplatin, but this is not always validated with patient-reported symptoms. 4 Objective tests such as the video head impulse test (VHIT) provide quick and objective measurements of the vestibular-ocular reflex (VOR) and efficiently assess the dizzy patient to determine if the dizziness is related to a vestibular disorder. 19 A recent study based on patient self-reported symptoms revealed that vestibular signs after cisplatin treatment occurred in 17% (n = 65) of adult cancer survivors. 20 Patients with peripheral neuropathy were more likely to have vestibular dysfunction. No vestibular dysfunction was detected with the VHIT (testing all semicircular canals) in cancer survivors after cisplatin therapy; however, benign paroxysmal positional vertigo (BPPV) was relatively prevalent in this group of patients. 20 On the contrary, a study by Hulse et al 10 found that VHIT showed a significantly reduced median gain six weeks after chemoradiation and significantly more refixational saccades could be detected after therapy. A study in paediatric cancer patients revealed bilateral vestibular hypofunction (25%) following the VHIT. 18
Vestibular-evoked myogenic potentials (VEMP) assess otolith and vestibular nerve function with air conduction cervical VEMP (cVEMP) and ocular VEMP (oVEMP). 21 VEMP amplitudes were significantly decreased after cisplatin exposure in an animal-related study. 21 Another study revealed that no consistent trend could be found amongst VEMP responses or hearing loss in patients undergoing cisplatin-based chemoradiation. Both cVEMP and oVEMP results showed extended latencies at follow-up testing; however, these were not statistically significant. 8 Dynamic visual acuity (DVA) assesses the vestibulo-ocular reflex (VOR), which is most helpful for diagnosing ototoxicity and other bilateral vestibular pathologies. DVA as a vestibular screening test showed abnormal results (28%) in paediatric head and neck cancer patients receiving platinum-based chemotherapy. 22
Evidence of clinically significant vestibular dysfunction after platinum-based chemotherapy is still not clear. 23,24 There is also a lack of guidelines for a vestibular assessment protocol that is appropriate to detect vestibulotoxicity in a manner that is sensitive to the over-burdened treatment schedule of cancer patients. This study investigated the changes in vestibular and cochlear function in patients receiving platinum-based chemotherapy using VHIT, VEMP and DVA testing along with pure-tone audiometry.
Materials and methods
Ethical clearance was obtained from the Research Ethics Committee of the Faculty of Health Sciences and Faculty of Humanities of a University in South Africa on January 11, 2019 (665/2018).
Study design, setting and participants
A longitudinal study design was implemented. Inclusion criteria included all participants (aged >10 years) treated with platinum-based compounds (cisplatin, carboplatin and/or oxaliplatin) for the first time in private and public oncology units and hospitals. In this study, participants were in oncology units where treatment was solely chemotherapeutic ie, contained platinum based agents, and nobody in the unit underwent radiotherapy for head and neck cancers. Testing was conducted during chemotherapy treatment in oncology clinics or at the hospital bedside. Thirty-two participants above the age of 10 years participated in the study, taking into account that repeated measures (baseline and exit testing) were performed for each participant. Testing was performed in the oncology rooms during chemotherapy appointments or oncology visits, as well as in hospital wards. Participants were tested prior to initiation of treatment or within 24 h of treatment initiation (baseline testing). Post-treatment follow-up occurred at 3-6 months post treatment (exit testing).
Equipment
Hearing testing was performed with the hearTestR certified digital audiometer (IEC 60645-1, hearX Group, South Africa) for baseline and exit testing. Supra-aural Sennheiser HDA 300 headphones (Sennheiser, Wedemark, Germany) calibrated according to prescribed standards (International Organisation for Standardisation, ISO 389–1, 2017), and adhering to equivalent threshold sound pressure levels determined for this headphone were connected to the smartphone. Automated protocols were used to obtain hearing thresholds and monitor cochleotoxicity.
Vestibular assessment included VHIT, VEMP and bedside DVA. The ICS impulse VHIT device (GN-Otometrics, Denmark) and ICS impulse video goggles (GN Otometrics, Taastrup, Denmark) with a camera speed of 250 frames per second, recording motion of the right eye, was used to assess semi-circular canal function. The SOCRATES Clinical Auditory Evoked Potentials (Hedera Biomedics, Italy) was used to obtain cVEMP and oVEMP measurements. SOCRATES is a computer-based medical device that can detect auditory evoked potentials by using two independent channels. A Snellen eye chart was used for bedside DVA.
Data-collection procedures
The ototoxicity monitoring case history interview 25 was used as a guideline during case history at baseline testing. The case history included questions regarding any history of hearing loss, auditory-related symptoms, previous vestibular insults or symptoms and timing of such events. Patients were also requested to report on vestibular symptoms experienced throughout the chemotherapy treatment. Testing was performed in the oncology rooms during chemotherapy appointments or oncology visits. Participants were tested prior to initiation of treatment or within 24 h of treatment initiation (baseline testing). Post-treatment follow-up occurred at 3-6 months post treatment (exit testing). All assessments were completed at a single assessment. Participants with changes in vestibular function and hearing were advised to continue monitoring until vestibular function and hearing stabilised up to 12 months post treatment. 26 All participants, even those without a significant deterioration in vestibular and hearing function, were advised to continue annual monitoring of hearing and vestibular function.
In addition to the objective vestibular tests VHIT (to measure semicircular canal function) and VEMP (to measure otolith function), DVA was included to assess the functional VOR, which is often compromised in those with bilateral vestibular loss. 27 Pure-tone audiometry was performed using an mHealth supported device.
Pure-tone audiometry
Prior to baseline testing, participants were provided with simple instructions and a demonstration of the testing procedure. An automated protocol was employed for baseline and exit audiometry (hearTestR) to determine participant thresholds. Participants were expected to indicate when they heard the tone by pressing a button on the smartphone. The Shortened Threshold Ascending method was used in the automated protocol to obtain thresholds. 26
The pure-tone average (PTA) was calculated as the better ear average for four frequencies of 500, 1000, 2000, and 4000 Hz. The WHO grades of hearing impairment were used to determine severity of hearing loss. A PTA of <25 dB indicates normal hearing, 26-40 dB HL slight hearing loss, 41-60 dB HL moderate hearing loss, 61-80 dB HL severe hearing loss and >81 dB profound hearing loss. 27
Ototoxicity monitoring criteria were regarded as significant when there were threshold shifts of 20 dB decrease or greater at one frequency, a 10 dB decrease or greater at two adjacent frequencies, and loss of response at three consecutive frequencies where there was a previously recorded response. 28
Video head impulse test
Participants were tested in a well-lit room with an eye-level target at a distance of 1 m in front of them while seated in a chair. Spectacles were removed for this assessment. VHIT goggles were tightened on the head until movement of the goggles at the bridge of the nose was minimal to avoid goggle slippage. 19
Calibration of the eye position signal was performed with the subject successively fixating on two projected laser dots separated by a known horizontal angle. For each of the canal planes, the researcher aimed to deliver a range of velocities in random order and direction so as to achieve at least 10 artefact-free impulses in each of the following ranges: horizontal: 10 <120°/s, 10 in the range 120–180°/s, and 10 over 180°/s in each direction. For vertical impulses, the ranges were: 10 <110°/s; 10 between 110° and 140°/s; 10 >140°/s. 19
For the horizontal VHIT stimulus, the researcher delivered small, passive, abrupt horizontal head rotations, with an unpredictable direction and magnitude. All tests were performed by the same right-handed researcher. Horizontal tests were performed with both hands on the top of the head, well away from the goggles strap and forehead skin.
Vertical VHIT included left anterior, right posterior (LARP) and right anterior left posterior (RALP) semi-circular canals. For LARP, the participant’s head was rotated 30-40° to the right of the fixation point. The participant was instructed to keep fixating on the target on the wall. Thereafter, a diagonal head pitch forward (toward the fixation target) activated the left anterior canal and caused an upward eye movement, and a head pitch back (away from the fixation target) activated the right posterior canal and caused a downward eye movement. Similarly, the RALP was performed with the participant’s head turned 30-40° to the left of the target, while still fixating on the target. A head pitch forward activated the right anterior canal, and a head pitch back activated the left posterior canal. 19 The entire VHIT took 10-15 min to complete.
Test results were interpreted as abnormal if (i) the VOR gain value <0.8 for lateral canals and <0.7 for vertical canals or (ii) if overt (saccades after the head movement) or covert (saccades during the head movement) catch-up saccades were present. 19
Vestibular evoked myogenic potentials
Participants were seated on a standard chair for both cVEMP and oVEMP testing. Ipsilateral electromyography recordings were performed for cVEMP testing. The participants had to obtain sufficient tonicity of the sternocleidomastoid (SCM) muscle with minimum discomfort in order for the cVEMP to be recorded. 29 The participants turned their head contralateral to the side of stimulation and neck flexion of the SCM muscle was achieved while being instructed to gaze at a target point in order to generate cVEMP with the most robust amplitudes and without premature fatigability. 29 An electromyography (EMG) monitor was used to ensure consistent and sufficient muscle contraction. Disposable wet-gel electrodes were used for recording after mild scrubbing of the electrode sites. The active (inverting) electrode was positioned on the ipsilateral mid-portion of the SCM muscle of the test ear, the reference (non-inverting) electrode was placed on the sternum, and the ground electrode was positioned on the forehead. 29 Impendences were kept below 5 kΩ. The stimulus was presented using insert earphones and an air-conduction tone burst stimulus of 500 Hz was presented at an intensity of 97 dB nHL using alternating polarity. A 2-ms rise/fall time and plateau time was used with band pass filters ranging from 10 to 1000 Hz at a repetition rate of 5.1 per second. One hundred sweeps were averaged for each cVEMP test. For the cVEMP waveform interpretation, the first positive peak on the waveform was marked as P1 and the first negative deflection was marked as N1. Normal P1 latency was ≤19 msec and for N1, ≤28 msec was considered normal. 29 The inter-peak (peak-to-peak) amplitude was the sum of the amplitudes of the repeated cVEMP responses.
Regarding oVEMP testing, electromyography recordings from the extra-ocular muscles in the infra-orbital region are recorded while the stimulus is presented in the contralateral test ear. An upward gaze during the stimulation and recording of oVEMP is required. Participants were asked to maintain their gaze on a stationary target on the ceiling. The active (inverting) electrode was positioned under the opposite eye on the inferior oblique muscle from the test ear. The reference (non-inverting) electrode was placed on the nose bridge, and the ground electrode was positioned on the forehead. 30 A 1-ms rise/fall time and 2-ms plateau time with band pass filters ranging from 2 to 500 Hz. One-hundred and fifty sweeps were averaged for each oVEMP test. For the oVEMP waveform interpretation, the first negative deflection was marked as N1 and the first positive peak was marked as P1. 30 Normal latencies for N1 were ≤11.1, and a latency of ≤17.6 msec was considered normal for P1. The interpeak amplitude was the sum of the amplitudes of the repeated oVEMP responses. 30
The VEMP asymmetry ratio (AR) was calculated using the following formula: (AR): [(AL-AS)/(AL + AS)] x 100, where “AL” represents the larger P1-N1 amplitude and “AS” the smaller P1-N1 amplitude. In order to confirm the presence of VEMP responses, the responses and the peaks had to be repeated within the correct latencies to test for wave reproducibility and to disregard potential artefacts. The VEMP responses were interpreted according to the following parameters: (i) classified as normal in the presence of identifiable P1 and N1 waveforms; (ii) the presence of identifiable P1 and N1 waveforms and latencies above the upper limits of the waveform were considered present yet delayed, and recorded as abnormal; (iii) the presence of an AR of ≥40% was considered abnormal, as it confirms amplitude differences between the ears and (iv) absent VEMPS could not be interpreted and were not useful for ototoxicity monitoring. 31
Bedside dynamic visual acuity
The participant was seated approximately 3 m from a Snellen eye chart, which was placed at eye level. Eyeglasses were permitted during this test. To determine static visual acuity, the participant was asked to read the smallest line, while reading all of the letters correctly. After verifying and recording the line of static visual acuity, the examiner stood behind the participant and rotated his/her head side to side at a speed of 2 Hz to effectively elicit a VOR response. A metronome was used to ensure that the appropriate speed was maintained throughout. To determine the DVA, the participant was again asked to read the smallest line possible in which all of the letters were read correctly, while his/her head was moving. A decline of more than two lines from static head recordings was considered abnormal. 32
Data analysis
SPSS was used for all data analysis (IBM SPSS Statistics 27) except for the achieved power where G*Power version 3.1.9.4 33 was used. Descriptive statistics (averages and standard deviation) were used to determine the decline in vestibular function from baseline to exit testing. The Shapiro-Wilk test 33 was used to test for normality, and since the P-values were less than 0.05, the data differed significantly from normality, and non-parametric tests were used. A within-subject statistical test (Wilcoxon signed-rank (WSR)) was used to determine whether there were statistically significant differences in the vestibular function from baseline to exit testing. If the P-value is < 0.05, then there is a statistically significant difference between baseline and exit. Non-parametric Spearman correlations were used to report on statistically significant (P-value < 0.05) correlations. The achieved power for a level of significance of 0.05, and sample size of 32 with an effect size of 0.573 (calculated from the data), equaled 0.973. In order to show an association between cochleotoxicity and vestibulotoxicity, correlations were used between VEMP and VHIT responses and average hearing thresholds. If the P > 0.05, then there was no significant correlation. On the other hand, if the P < 0.05, the correlation was significant and could be be interpreted. A positive correlation was used to conclude that as cochleotoxicity increases, so does vestibulotoxicity.
Results
Characteristics of participants (n = 32).
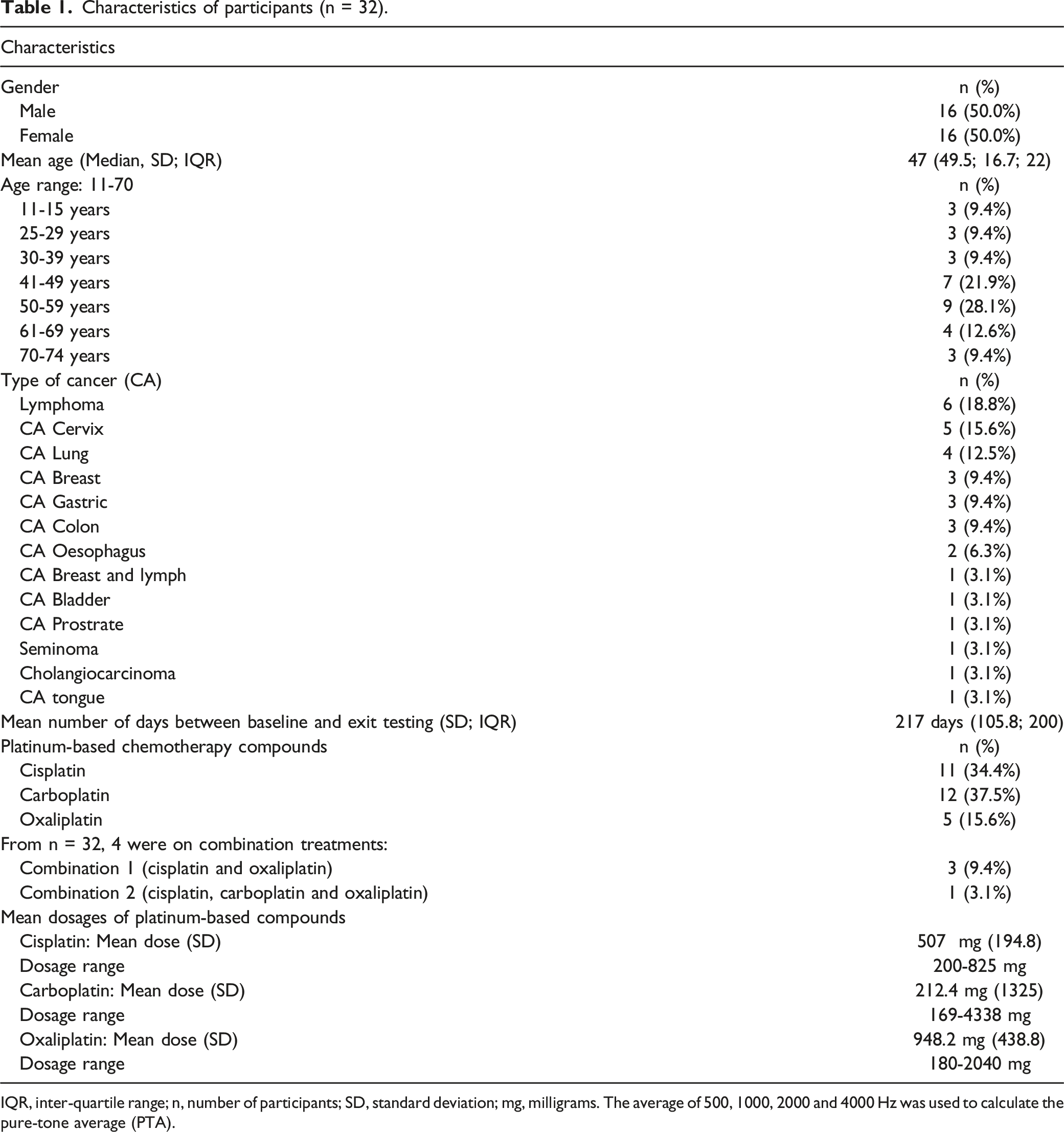
IQR, inter-quartile range; n, number of participants; SD, standard deviation; mg, milligrams. The average of 500, 1000, 2000 and 4000 Hz was used to calculate the pure-tone average (PTA).
Hearing status of participants at baseline and exit testing (n = 32).

IQR, inter-quartile range; n, number of participants; SD, standard deviation.
The average of 500, 1000, 2000 and 4000 Hz was used to calculate the pure-tone average (PTA).
A P-value < 0.05 was used to indicate if there is a statistically significant difference between baseline and exit testing.
*Statistically significant difference from baseline to exit testing (p = 0.001).
Cochleotoxicity according to ototoxicity monitoring criteria was present in 50% (n = 16) of participants. The most affected frequencies according to ototoxicity monitoring criteria were in the high frequencies from 4000 to 16 000 Hz. From the 16 participants who showed cochleotoxicity, 50.0% (n = 8) were treated with carboplatin, 31.3% (n = 5) were treated with cisplatin, 6.3% (n = 1) was treated with oxaliplatin and 12.5% (n = 2) were treated with a combination of cisplatin and oxaliplatin. No vestibulotoxicity criteria exist in order to confirm the presence of early signs of vestibular damage. A significant association between cochleotoxicity and vestibulotoxicity was present in 3.1% (n = 1) for left ear PTA values and oVEMP P1 results (P < 0.05). No further significant associations between cochleotoxicity and vestibulotoxicity were identified. Therefore, no consistent relationship between cochleotoxicity and vestibulotoxicity could be identified.
DVA yielded normal results at both baseline and exit testing (100%, n = 32). cVEMP could be elicited from 65.6% (n = 21) participants. cVEMP were absent in the remainder of the participants (28.1%, n = 9) and cVEMP could not be performed in 6.2% (n = 2) due to large lymphoma neck masses. From the cVEMP elicited at baseline and exit testing, 28.6% (n = 6) were on carboplatin, 42.9% (n = 9) were on cisplatin, 14.3% (n = 3) were on oxaliplatin and 14.3% (n = 3) were on a combination of platinum based compounds.
VEMP testing at baseline and exit assessments (cVEMP n = 21; oVEMP n = 22). VEMP were absent in 9/32 participants.
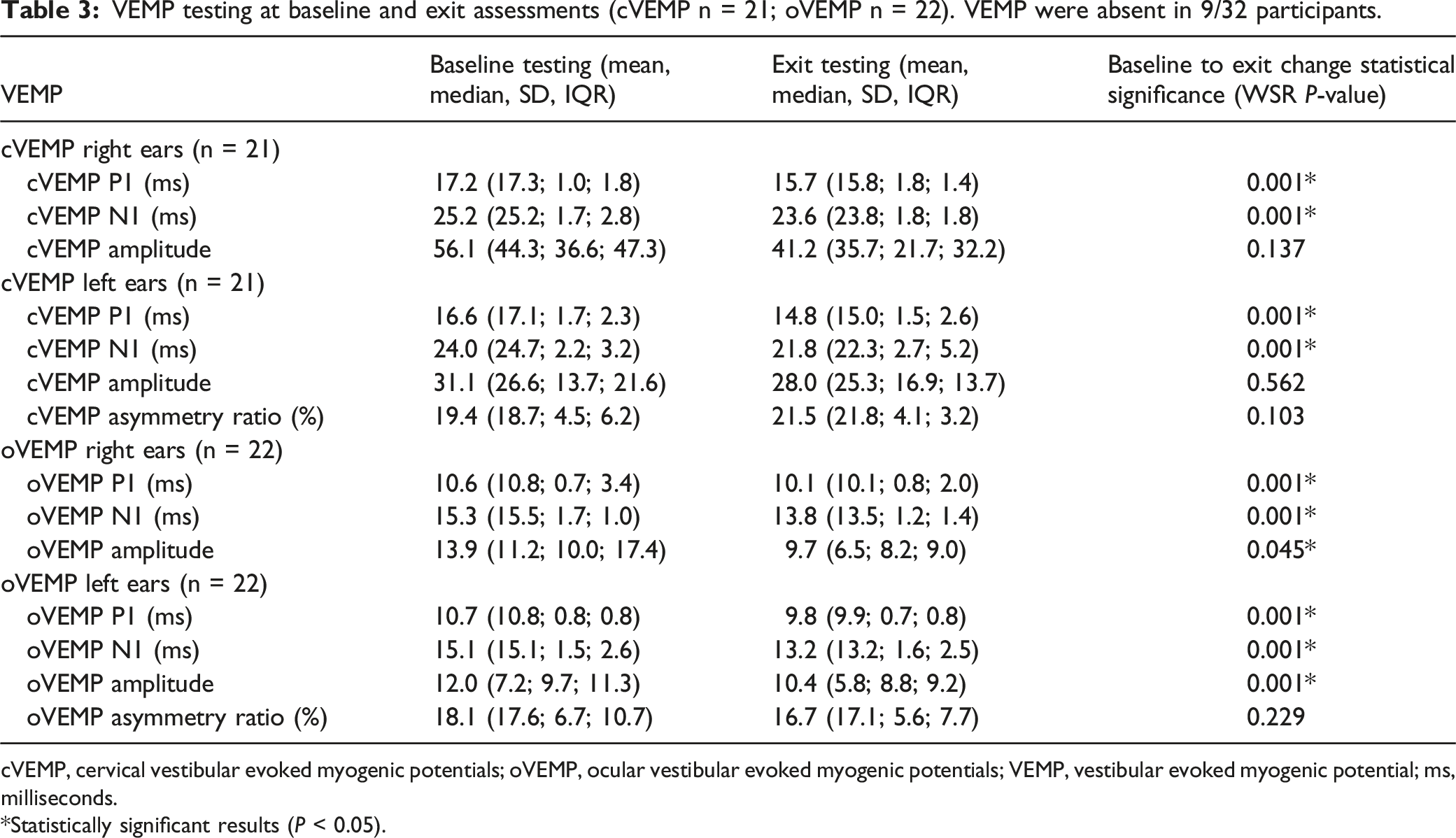
cVEMP, cervical vestibular evoked myogenic potentials; oVEMP, ocular vestibular evoked myogenic potentials; VEMP, vestibular evoked myogenic potential; ms, milliseconds.
*Statistically significant results (P < 0.05).
VHIT results at baseline and exit testing (lateral SCC n = 31; anterior SCC n = 30; posterior SCC n = 31).
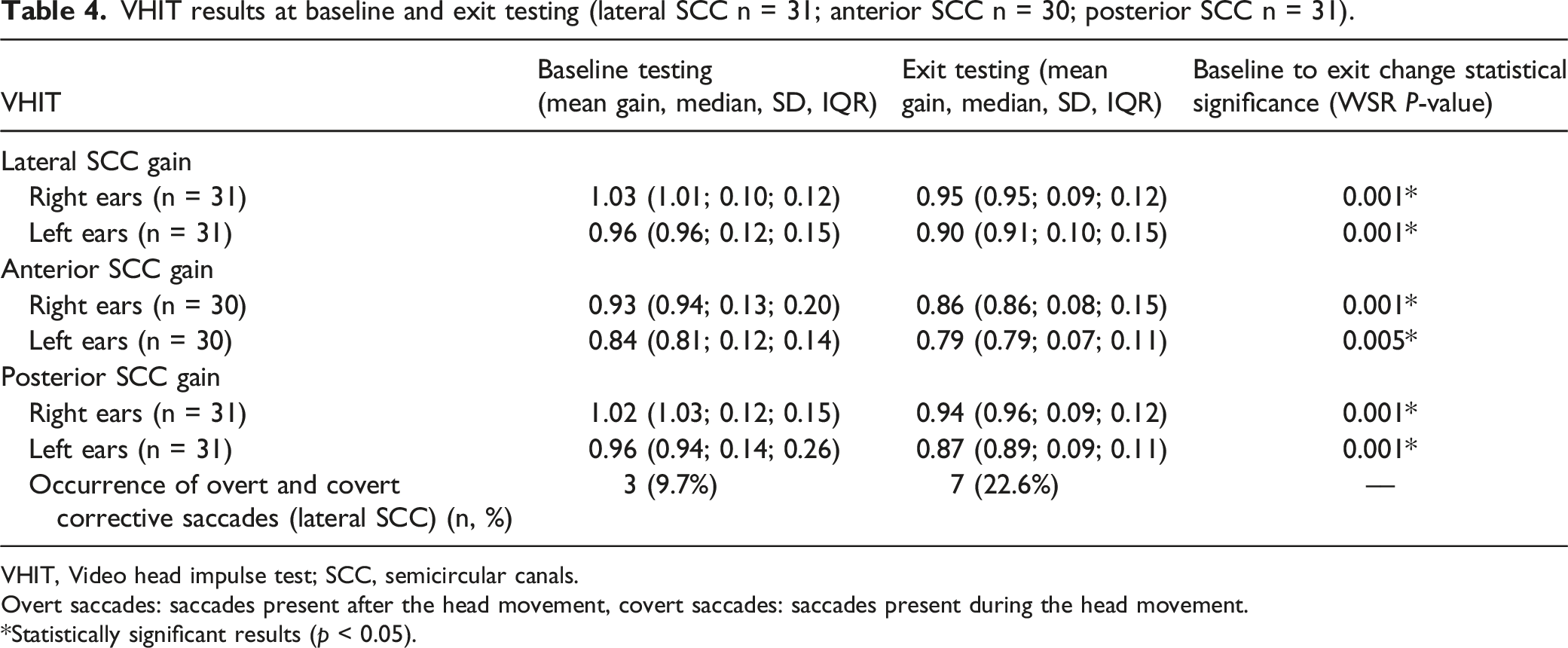
VHIT, Video head impulse test; SCC, semicircular canals.
Overt saccades: saccades present after the head movement, covert saccades: saccades present during the head movement.
*Statistically significant results (p < 0.05).
Although gain values remained within normal limits, a significant decline in gain was noted at exit testing (Table 4). The Supplementary Table in the appendix shows the comparison of baseline and exit testing VHIT gain values per participant for lateral, posterior and anterior semi-circular canals. A decrease in gain values is noted in the majority of participants in all six semi-circular canals. A typical set of RAW VHiT data from baseline to exit testing for one participant is included as Supplementary Data.
Comparison of VEMP and VHIT results in left and right ears: cVEMP n = 21; oVEMP n = 22; lateral SCC n = 31, anterior SCC n = 30; posterior SCC n = 31).
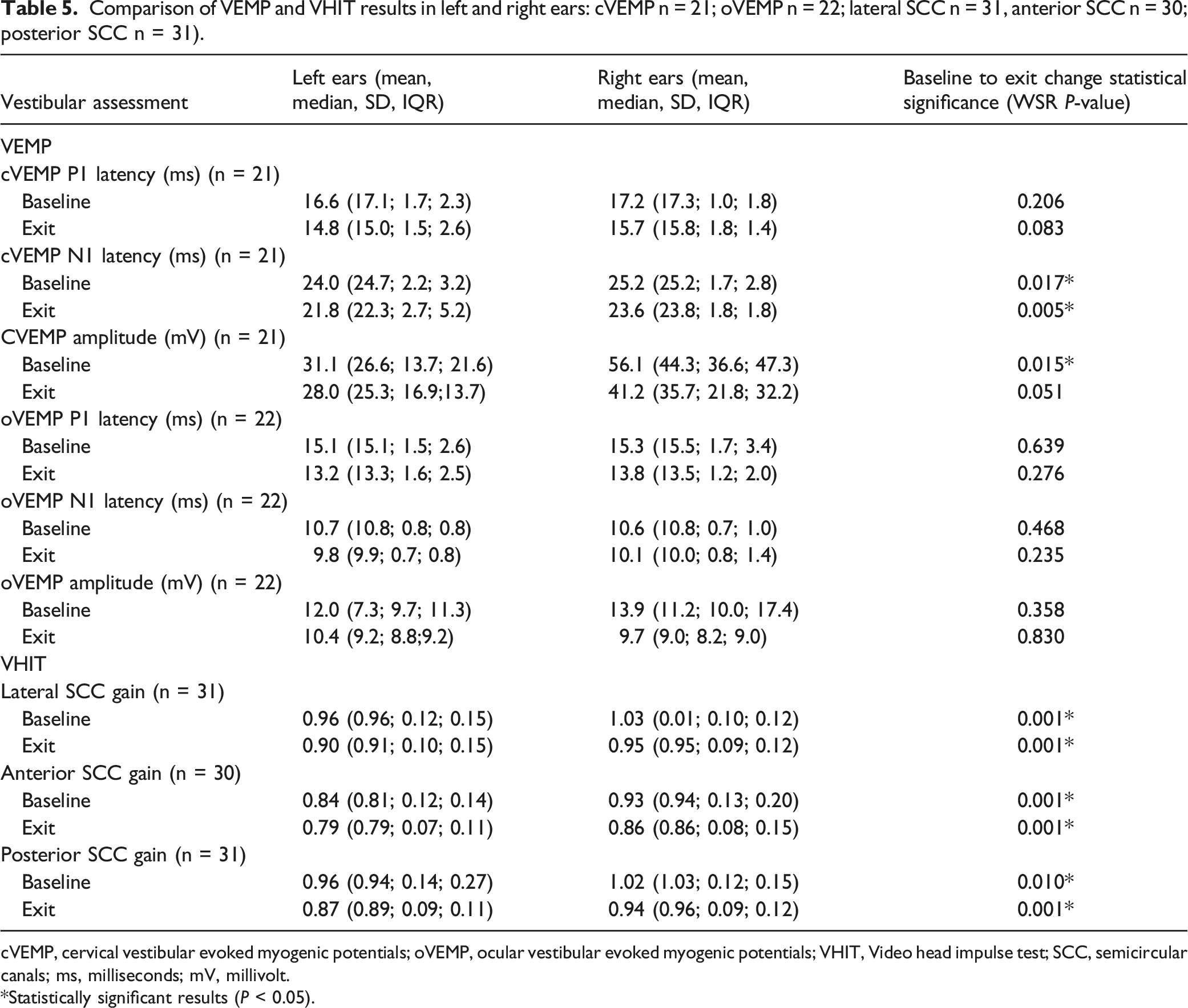
cVEMP, cervical vestibular evoked myogenic potentials; oVEMP, ocular vestibular evoked myogenic potentials; VHIT, Video head impulse test; SCC, semicircular canals; ms, milliseconds; mV, millivolt.
*Statistically significant results (P < 0.05).
Discussion
This study investigated the vestibular and cochlear function in patients receiving chemotherapy. When testing at the patient’s chemotherapy treatment venue, VHIT proved to be a suitable measure for changes in vestibular function following ototoxicity treatment, however VEMP was challenging to perform. VEMP was not possible for patients that present with cervical masses; those who were too weak to ensure sufficient SCM muscle contraction, and the setup was time consuming and challenging due to space limitations in the oncology units. Self-report did not reveal any vestibular symptoms at baseline or exit testing in the current study. Self-reported impact of the cochlear and vestibular handicap should be included in a monitoring and surveillance programme for appropriate investigation and management. 23 Hulse et al 10 reported that none of their patients had subjective dizziness or balance problems during vestibular monitoring and after being treated with chemoradiation. Another study based on patient self-reported symptoms indicated that dizziness was prevalent in 17% of the participants, and patients with peripheral neuropathy were more likely to have balance symptoms, as confirmed by objective vestibular tests. 24 Dizziness and lightheadedness (not related to vestibular function) were reported by some participants in the current study who also suffered from hypotension and neuropathy following chemotherapy. Although balance problems can also be associated with vestibular toxicities, the participants in the current study felt that other comorbidities, such as weakness and neuropathy, were responsible for the dizziness and lightheadedness. 34
Vestibular dysfunction has been reported with cochleotoxicity (either hearing impairment or tinnitus), 18 whereas no hearing loss was found in some patients with abnormal objective vestibular assessments. 4 The current study demonstrated no consistent relationship between cochleotoxicity and vestibular dysfunction. Vestibular damage may remain undetected, as patients and healthcare professionals assign imbalance symptoms to other causes and no vestubulotoxicity criteria exist to identify early damage caused by chemotherapy. Patients in the current study did not report vestibular related symptoms. Moreover, normal auditory function does not imply that vestibular function is also unimpaired. 8 Further research with larger sample sizes is required to confirm if hearing dysfunction can serve as a proxy for vestibular dysfunction during ototoxicity monitoring. Both cochleotoxicity and vestibulotoxicity (including at minimum patient self-report of symptoms) should be included when testing patients being treated with platinum-based agents at the treatment venue or hospital ward. Participants in the current study who showed significant shifts from baseline to exit testing were mostly on carboplatin, cisplatin or a combination of platinum-based treatments. Cisplatin and carboplatin related otooxicity is more common, while oxalipltin related ototoxicity is rare. 5
Although VEMP in the current study stayed within the normal range for both cVEMP and oVEMP at baseline and exit testing, statistically significant (P < 0.05) changes were evident. There was a definite decrease in N1 and P1 latency values, as well as amplitude from baseline to exit testing in both cVEMP and oVEMP, with oVEMP more affected. VEMP in patients receiving chemoradiation have demonstrated statistically significant changes following treatment. 10 An important limitation of using VEMP is the fact that VEMP were absent in all participants over 60 years in the current study. 34 When VEMP are absent bilaterally in an older adult, it can be challenging to interpret. The individual may have a bilateral otolith impairment, or an impairment occurring anywhere along the VEMP reflex pathway, inability to hold the muscle contraction, possibly be due to recording and/or stimulus parameters used to elicit the responses, or just be absent due to age above 60 years. 34 Furthermore, cancer patients on platinum-based compounds are often in older age groups 22 and the validity and reliability of measurements involved in vestibular evaluations must be considered in effective monitoring programmes.
The VHIT gain results remained within normal limits from baseline to exit testing. However, there was a significant (P < 0.05) decrease in gain at exit testing, suggesting signs of early vestibulotoxicity. From baseline (9.7%), there was also an increase of overt and covert corrective saccades (22.6%) at exit testing in the lateral canals. Research by Hulse et al 10 reported saccades present in 39% of participants following chemoradiation treatment, and significantly reduced median gain six weeks after treatment. Increased corrective saccades and horizontal VOR abnormalities have been reported and can be an indicator of vestibular loss. 4,10 Other studies have found posterior labyrinth damage following treatment with cytostatics such as cisplatin. 35 A study by Prayuenyong et al 24 found that no vestibular dysfunction was detected by VHIT; however, benign paroxysmal positional vertigo (BPPV) was relatively prevalent in this group of cancer patients.
Average hearing thresholds showed a statistically significant (P < 0.05) decline from baseline to exit testing, with left ears more affected than right ears. A study examining the role of EHF in ototoxicity monitoring demonstrated that among the 45 patients affected by ototoxicity, hearing loss was unilateral in 31.1% (n = 14) before bilateral hearing loss was reported. 36 Similar to hearing threshold changes, the VHIT results in the current study showed that left ears were significantly more affected than right ears. Vestibulotoxicity may well follow a similar trajectory because of the shared blood, nerve and fluid sources. 8 Hypothetical explanations for unilateral involvement in ototoxicity include the fact that asymmetry and the genetic difference of bilateral organs are well-known; therefore, a correlation of a genotype with unilateral ototoxicity is possible. It is assumed that two molecular mechanisms with different speeds may cause ototoxicity. Due to the asymmetry of organs and expression of enzymes, the slow toxicity becomes unilateral first and then bilateral. Another theory may be related to the unilateral noise–induced effect during treatment, as the ears are more susceptible to extreme noise during treatment. This sensitivity may also be related to some gene variants. 37 This potentially provides the opportunity to adjust the patient’s drug regimen to prevent progression to bilaterality.
DVA was not sensitive in identifying vestibulotoxicity in the current study, as normal results were present at baseline and exit testing for all participants. The results of vestibular objective testing in this study (with significant changes from baseline to exit testing) did not always correspond to patient symptoms, as participants did not report symptoms related to vestibular dysfunction. Studies have shown that the prevelance of vestibular dysfunction after chemotherapy administration varied from 0 to 50%. 4 Patients with vestibular dysfunction at baseline are at greater risk for vestibulotoxicity following treatment with cisplatin. 4
The limitations of the current study include a limited sample size and follow-up conducted only up to 3-6 months after chemotherapy. Longer follow-up is potentially needed as platinum-based compounds remain in the bloodstream for an extended period, and the effects of possible central compensation could therefore be monitored. Long-term follow up could identify the possible development of other vestibular disorders such as BPPV, and ensure early management of those disorders.
Conclusion
This study suggests that both VHIT and VEMP testing showed significant changes from baseline to exit testing and may prove to be effective measures of changes in vestibular function secondary to ototoxicity. VHIT can easily be performed at the patient’s bedside or treatment venue during an ototoxicity monitoring programme. However, VEMP at the patient’s treatment venue has proven to be logistically challenging and time-consuming when performed as part of a cochleotoxicity and vestibulotoxicity monitoring programme. Furthermore, considering that VEMP responses are absent in patients >60 years and the prevalence of cancer is higher in this age group, VEMP may not be practical as part of a vestibulotoxicity monitoring tool for older cancer patients. Criteria for vestibulotoxicity and optimal protocols for monitoring vestibular function during chemotherapy treatment, and preferably in the patient’s treatment venue or hospital ward, should be explored. By the time a patient complains of imbalance or dizziness, permanent vestibular system damage has more than likely already occurred. The opportunity for early identification and possible prevention of further damage may be missed if only self-report symptoms or handicap scales are used. The practicality and ease of providing vestibular assessment protocols must, however, be considered, especially for those patients receiving medical care, who may already be weak or debilitated in hospital, or may merely not have access to more comprehensive testing facilities. Where objective testing is possible, the VHIT proved to be fast to perform in a patient treatment venue and sensitive in identifying vestibulotoxicity. This study showed statistically significant (P < 0.05) changes in vestibular function from baseline to exit testing, however, patients did not report vestibular symptoms that had a functional impact on daily life. These significant changes may be due to central compensation that occurs, damage that is bilateral, as well as possibly an early indicator of vestibular dysfunction, before subjective symptoms are reported, motivating the need for vestibular monitoring during ototoxicity surveillance. Patients did not report symptoms related to vestibular dysfunction but symptoms like dizziness may be masked by disease processes, general lethargy, and side effects of medications. 4 Patient self-report of symptoms such as headaches, a feeling of ear fullness, imbalance and oscillopsia may be valuable to monitor vestibulotoxicity in the treatment venue for patients who are ill and incapacitated, and referrals can be made for further in-depth vestibular assessments when symptoms are reported. However, this approach may overlook the opportunity for early identification of vestibulotoxicity, before symptoms are reported.
Supplemental Material
Supplemental Material - Changes in vestibular and cochlear function following platinum-based chemotherapy: A preliminary report
Supplemental Material for Changes in vestibular and cochlear function following platinum-based chemotherapy: A preliminary report by Katerina Ehlert, Barbara Heinze, Marien A Graham and De Wet Swanepoe in Ear, Nose & Throat Journal
Footnotes
Author contributions
KE conceived of the presented idea, developed the theory, and performed the computations. MG conducted the statistical analysis. BH and DS verified the analytical methods. BH and DS supported KE to write the findings of this work. All authors discussed the results and contributed to the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DS has equity interest, consultancy and potential royalties in the hearX Group.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sefako Makgatho Health Sciences University Research Development Grant (D200).
Ethical approval
This study was approved by a University in South Africa Faculty of Health Sciences and Faculty of Humanities (ethics reference number: 665/2018). All study procedures adhered to the tenets of the Declaration of Helsinki. Written informed consent was obtained from the participants after all study procedures had been explained in detail.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
