Abstract
Facial paralysis secondary to acute otitis media (AOM) is rare in the post-antibiotic era.1,2 In immunosuppressed patients, atypical bacteria are more commonly encountered as the cause. Mycoplasma hominis, normally found in the genitourinary tract, uncommonly causes extragenital infection.3 We report a case of AOM secondary to M. hominis, complicated by facial paralysis in an immunosuppressed patient. A 24-year-old male with multiple sclerosis, on rituximab, presented to the emergency department with otalgia and facial paralysis. He was diagnosed with Bell’s palsy and subsequently referred to the otolaryngology service. Examination revealed right facial paralysis and purulent otorrhea. Computed tomography (CT) of the temporal bone showed right tympanic and mastoid opacification. The patient was admitted and started on IV ampicillin/sulbactam, IV dexamethasone, and ciprofloxacin/dexamethasone otic drops. Debridement and examination under anesthesia revealed a right tympanic membrane perforation and granulation tissue. Biopsy and cultures were obtained. Final cultures were positive for Mycoplasma hominis. Facial paralysis is an uncommon complication of AOM but typically resolves once appropriate antibiotic therapy is regimented. Because Mycoplasma can be missed on routine culture, clinicians should consider workup for atypical organisms in settings of immunosuppression so appropriate antibiotic therapy can be initiated.
Introduction
Facial paralysis secondary to acute otitis media (AOM) is a rare complication in the post-antibiotic era.1,2 In immunosuppressed patients, atypical bacteria are more commonly encountered as the cause. Mycoplasma hominis, normally found in the genitourinary tract, uncommonly causes extragenital infection. 3 We report a case of AOM secondary to M. hominis, complicated by facial paralysis in an immunosuppressed patient.
Case Report
A 24-year-old male with past medical history of multiple sclerosis on rituximab presented to the emergency department with right ear pain. He was seen at an urgent care and prescribed ofloxacin ear drops. He subsequently followed up with his primary care provider for increased otalgia and was switched to polymyxin/neomycin/hydrocortisone ear drops with no improvement. Three days following the onset of otalgia, he presented to the emergency department with right-sided facial paralysis and was discharged home with the diagnosis of Bell’s palsy. Based on this diagnosis, his neurologist had ordered an outpatient MRI, and subsequently referred him to the emergency department for further evaluation based on the imaging findings suggestive of mastoiditis.
Laboratory findings included an elevated white blood cell count of 15.1, absolute neutrophil count of 13.3, erythrocyte sedimentation rate of 2 mm/hr. Temporal bone CT described opacification of the right mastoid air cells and right tympanic cavity without bony erosion (Figure 1). Based on these findings and clinical presentation, the otolaryngology service was consulted. Examination revealed right facial paralysis with House-Brackmann score (HB) of 5 out of 6 and purulent otorrhea in the right external auditory canal. The patient was admitted to the otolaryngology service, started on IV ampicillin/sulbactam, IV dexamethasone, and ciprofloxacin/dexamethasone ear drops. On hospital day 2, the patient underwent examination under anesthesia with debridement and biopsy. The patient was then discharged home on oral amoxicillin/clavulanate and ciprofloxacin/dexamethasone ear drops with follow-up with the otology service. Noncontrast axial CT of right mastoid opacification without bony erosion.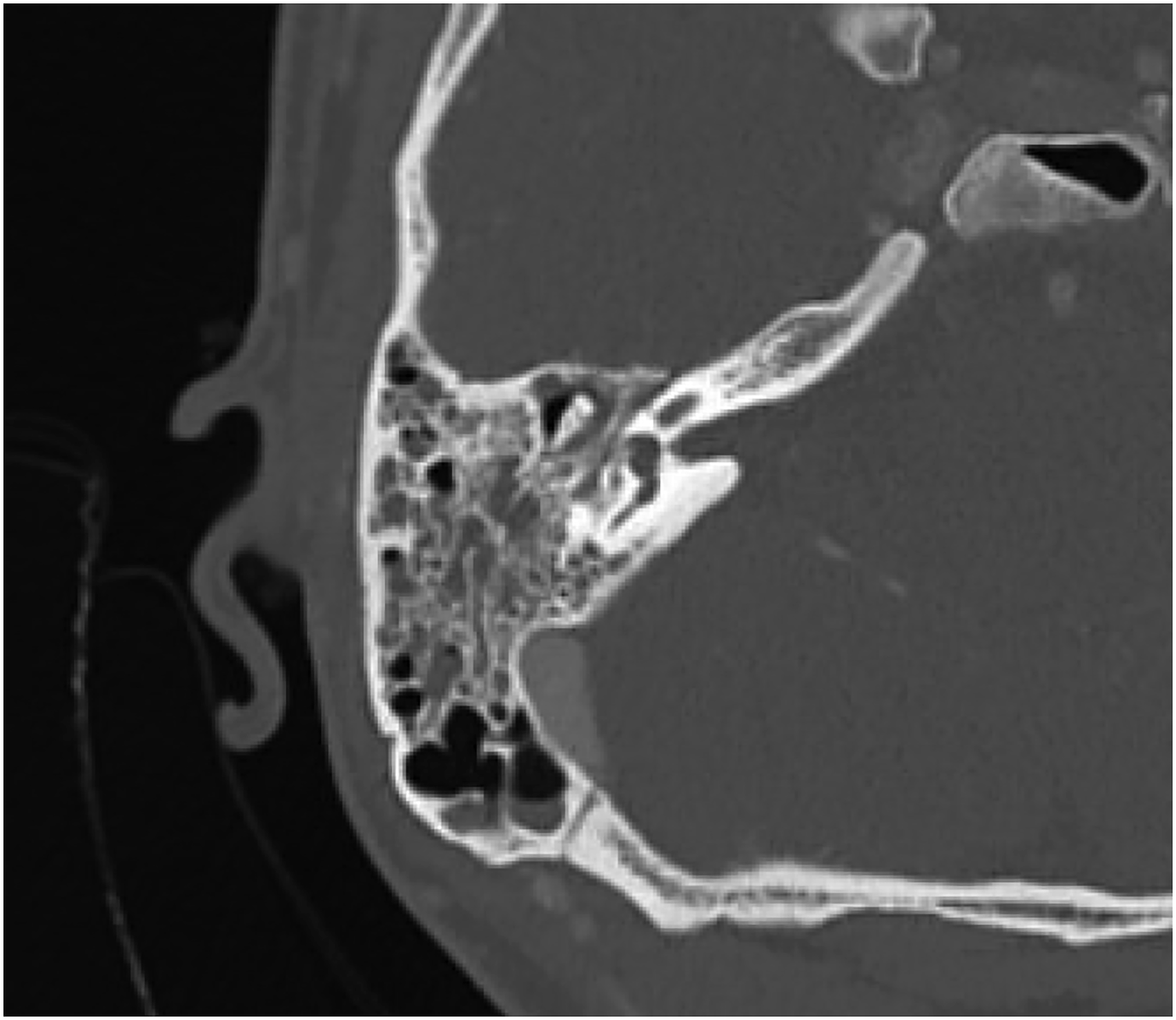
Further workup included obtaining cultures for aerobic, anaerobic, mycobacterial, and fungal elements. Joint decision was made between the otology service and the ear, nose and throat (ENT) service to re-admit the patient for infectious disease evaluation. Final cultures were positive for Mycoplasma hominis. Patient was started on intravenous clindamycin and oral doxycycline.
The patient’s facial paralysis had significantly improved to a HB 1/6 on subsequent follow-up.
Discussion
Facial paralysis is an uncommon complication of AOM but typically resolves once appropriate antibiotic therapy is regimented. However, in cases of immunosuppression and atypical bacteria, AOM-induced facial paralysis can remain unresolved with routine antibiotic therapy and warrants further investigation and a different antibiotic regimen. Here, we present a case of AOM-induced facial paralysis secondary to M. hominis.
The pathophysiology of facial paralysis secondary to otitis media is still open to question. Most cases are believed to be related to a congenital dehiscence of the fallopian canal as this puts the nerve at risk from intrafallopian inflammation and edema mediated by bacterial toxins. 4 This dehiscence is seen in 55% of temporal bones 4 and is, thus, a strong predisposing factor. Other risk factors include immunosuppression and latent neurotropic viral infections, which can be reactivated by middle ear suppuration. 2 Facial paralysis associated with AOM is predominately acute; onset of paralysis is on average 6 days after onset of AOM 4 but can appear as early as one day after. 2 Paralysis typically completely resolves through conservative treatment involving antibiotics and myringotomy (with or without ventilation tube insertion); however, more severe paralysis have a worse prognosis and might require a mastoidectomy if conservative treatment is ineffective. 4 The grades of paralysis and time from symptom onset to recovery are variable. 2 A retrospective chart review found that after treatment, initial paralysis improvements occurred as early as 3 days and as late as 74 days, and complete recover occurred as early as 19 days and as late as 312 days. The later improvement and recovery were seen in the same patient who had a grade IV paralysis and required a mastoidectomy. 2 Another series conducted on 22 children found that complete recovery took on average 133 days. 4
Acute otitis media most commonly occurs secondary to bacterial and viral infections. The most common bacterial pathogens include Haemophilus influenzae, Streptococcus species, Moraxella catarrhalis, and Staphylococcus aureus, while the most common viruses include respiratory syncytial virus, influenza A virus, parainfluenza viruses, adenoviruses and rhinovirus. 4 Acute otitis media induced by the common bacterial pathogens typically resolves with empiric antibiotic therapy; however, if empiric therapy fails, atypical infections should be suspected.
In treatment of AOM, empiric antibiotic therapy is typically instituted without obtaining bacterial cultures. However, cultures should be obtained to identify any resistant or atypical bacterial strains and to aid in antibiotic selection; this is particularly important in immunosuppressed patients or in cases where AOM is unresponsive to therapy.
Since Mycoplasma hominis can cause extragenital infections in immunosuppressed patients, proper testing for this pathogen should be conducted in cases where atypical bacterial infection is suspected. Reliable detection of M. hominis requires specialized growth media, thus extragenital infections are often underdiagnosed. 5 M. hominis samples are typically first incubated in an arginine broth. After an arginine hydrolysis-induced color change, the sample is subcultured on A7 agar, where identification can be confirmed via characteristic “fried-egg-type” colonies. 5 A more complex and expensive method to identify M. hominis is a PCR assay. This method is considered more sensitive than culture for M. hominis detection, accounts for bacterial heterogeneity, helps eliminate human subjectivity involved with colony identification, and is time saving as bacterial incubation can take up to 5 days. 6
Mycoplasma species do not have a rigid cell wall and are therefore resistant to most beta lactamase antibiotics, which are commonly prescribed in AOM. These pathogens are also difficult to eradicate in immunosuppressed patients due to their intracellular location. Unlike other Mycoplasma species, M. hominis is uniquely erythromycin-resistant. 7 Historically, Mycoplasma hominis displayed sensitivity to doxycycline. However, resistance to select tetracyclines due to mutations in the tetM gene has been observed; for these species, clindamycin, lincomycin, or fluoroquinolones, such as ofloxacin, are recommended. 7
Since M. hominis can be recovered from sites not usually subject to colonization, such as the conjunctiva, blood, anal canal, and oropharynx, it displays characteristics of an opportunistic pathogen. 8 M. hominis typically arises from the urogenital tract and spreads to other tissues via hematogenous dissemination. 9 A study reported 36 cases in which most of the patients had an injury or surgery at the site of M. hominis infection; 9 were immunosuppressed and 7 of had undergone kidney transplantation. In this cohort of patients, M. hominis was associated with extragenital infection including septicemia, septic arthritis, prosthetic valve endocarditis, wound infection, and brain abscesses. 5
While extragenital infection with M. hominis is often associated with immunosuppression, it has also been identified in immunocompetent individuals. Huminer et al. found that M. hominis was cultured from about 15% of tonsils and 5% of adenoids in children who underwent an adenoidectomy and/or tonsillectomy. Mycoplasma was isolated at significantly higher rates in children with recurrent adenotonsillitis (34.5%) than in children with chiefly obstructive symptoms (3.7%). Most of the children who underwent tonsillectomy also mainly had symptoms of recurrent infections (67%). 10
In a study of 67 patients with M. hominis infection, 32 patients were categorized as immunocompromised, while the rest had predisposing factors such as surgical trauma or prosthetic joints. 11 Of these cases, they found that the use of immunosuppressive agents in transplant recipients or patients with collagen vascular disease were the main predisposing factors to extragenital Mycoplasma hominis infection. 11
In summary, in cases of persistent AOM despite empiric antibiotic therapy, atypical bacteria should be considered and adequate cultures obtained, particularly in immunosuppressed patients. Because Mycoplasma can be missed on routine culture, clinicians should consider workup for atypical organisms in settings of immunosuppression so appropriate antibiotic therapy can be initiated. As seen in our patient, adequate treatment after identification of M. hominis infection resulted in resolution of facial paralysis secondary to AOM.
There are no financial disclosures and there are no sources of funding or grants. This submission has not been presented or published elsewhere. The Authors declare that there is no conflict of interest. Verbal consent from the patient was obtained.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
