Abstract
Mucosal melanoma of the oral cavity is rare and highly aggressive, thought to represent less than 1% of melanomas. Within this subgroup, melanoma in situ has been rarely described. We describe the case of a 54-year-old male with history of tobacco use presented with extensive pigmented changes to the hard and soft palate. Biopsy demonstrated melanoma in situ. Mucosal surgical resection was performed with all peripheral epithelial margins involved and negative deep margins. After extensive multidisciplinary discussion, remaining mucosal margins were re-resected to the teeth and posteriorly onto the soft palate. Deep margins remained negative with melanoma in situ still present peripherally. The patient is routinely surveilled without evidence of recurrence. Oral cavity melanoma in situ has been rarely described. The treatment of choice is surgical excision, ranging from wide local excision to composite resections, with consideration given to medical adjuncts. This unique entity should be considered in pigmented oral abnormalities.
Introduction
Mucosal melanoma of the oral cavity is exceedingly rare, thought to represent less than 1% of all melanomas. Similarly, mucosal melanoma represents less than 1% of oral cavity malignancies. 1 Within the oral cavity, the palatal mucosa is the most common site of occurrence, followed by the maxillary gingiva.1,2 Oral mucosal melanoma is known to be highly aggressive with a 15% 5-year survival. 1 The primary management of malignant mucosal melanoma is typically surgical resection, though radiation therapy and chemotherapy can be used as adjuncts.1-3
When describing oral mucosal melanoma, histopathologic patterns include in situ, invasive, and combined in situ and invasive. 1 Specifically, in situ melanoma demonstrates atypical melanocytes proliferating in the epithelium without stromal invasion. Oral mucosal melanoma in situ has been rarely described in the literature, with described cases most commonly localizing to the hard palate and maxillary gingiva.4-10 Given lack of histopathologic invasion and a paucity of reported cases, recommendations for management are limited and vary greatly.
We describe the clinical and histopathologic features of an in situ melanoma of the hard palate and discuss options for management.
Case Report
A 54 -year-old male presented 5 months after incidental discovery of extensive pigmented changes to the hard palate during a routine dental visit. A biopsy performed by the referring dentist revealed in situ melanoma. His past medical history was unremarkable, with a remote history of tobacco use, but no history of alcohol use or occupational exposures. The patient was Burmese, and had moved to the United States 15 years prior. Physical exam revealed extensive pigmented changes of the anterior hard palate spreading posterior toward the soft palate (Figure 1A). Oral cavity exam demonstrating: (A) exam on initial presentation with extensive pigmented changes of the hard palate extending posterior to the soft palate, (B) palate 2 months s/p initial wide local excision, (C) palate 1 month s/p revision wide local excision, and (D) palate 5 months s/p revision wide local excision.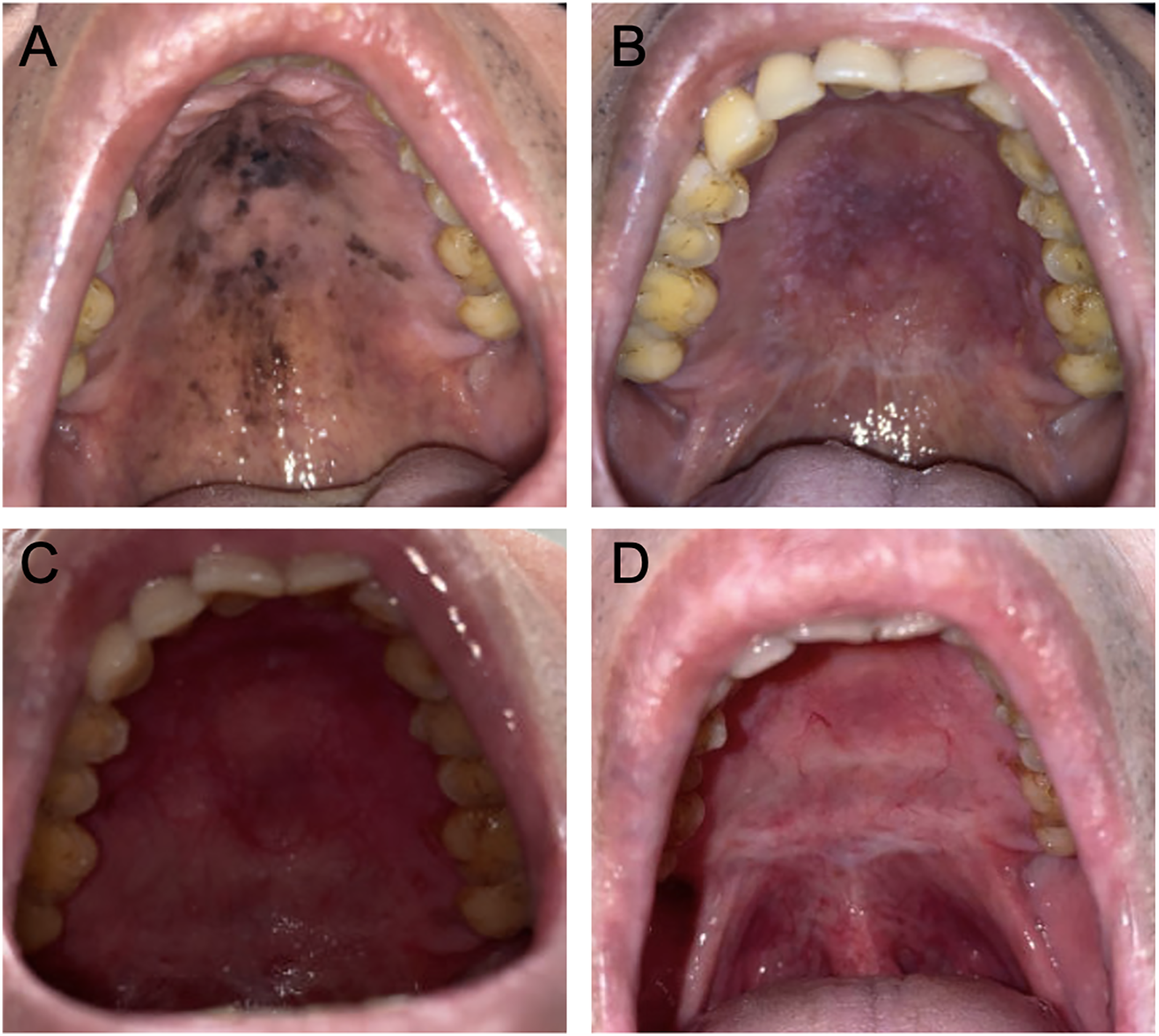
The patient subsequently underwent wide local excision of the lesion with near-total resection of hard palate and anterior soft palate mucosa including all visible lesion with a 5 mm margin. A Biodesign® graft was secured and bolstered with a temporary silastic splint. Surgical pathology demonstrated mucosal melanoma in situ of the hard and soft palate, Clark level I, extending to all peripheral epithelial margins without involvement of the deep margin (Figure 2A). Cells were diffusely positive for p16 and negative for BRAF V600E mutation within the lesion. The neoplastic melanocytes were reactive to a PRAME (Preferentially expressed Antigen in Melanoma) immunostain that assisted in assessment of surgical margins. (Figure 2B). Surgical pathology demonstrating: (A) palatal mucosa with enlarged rounded cells that are present singly and in nests in the basal layer and scattered in the spinous layer, representing melanoma cells that sometimes contain melanin pigment and that have large hyperchromatic nuclei with prominent nucleoli (H&E 20X); (B) a PRAME immunostain reacts with the malignant melanoma cell nuclei and helps to distinguish them from normal melanocytes and epithelial cells (PRAME alkaline phosphatase 20X).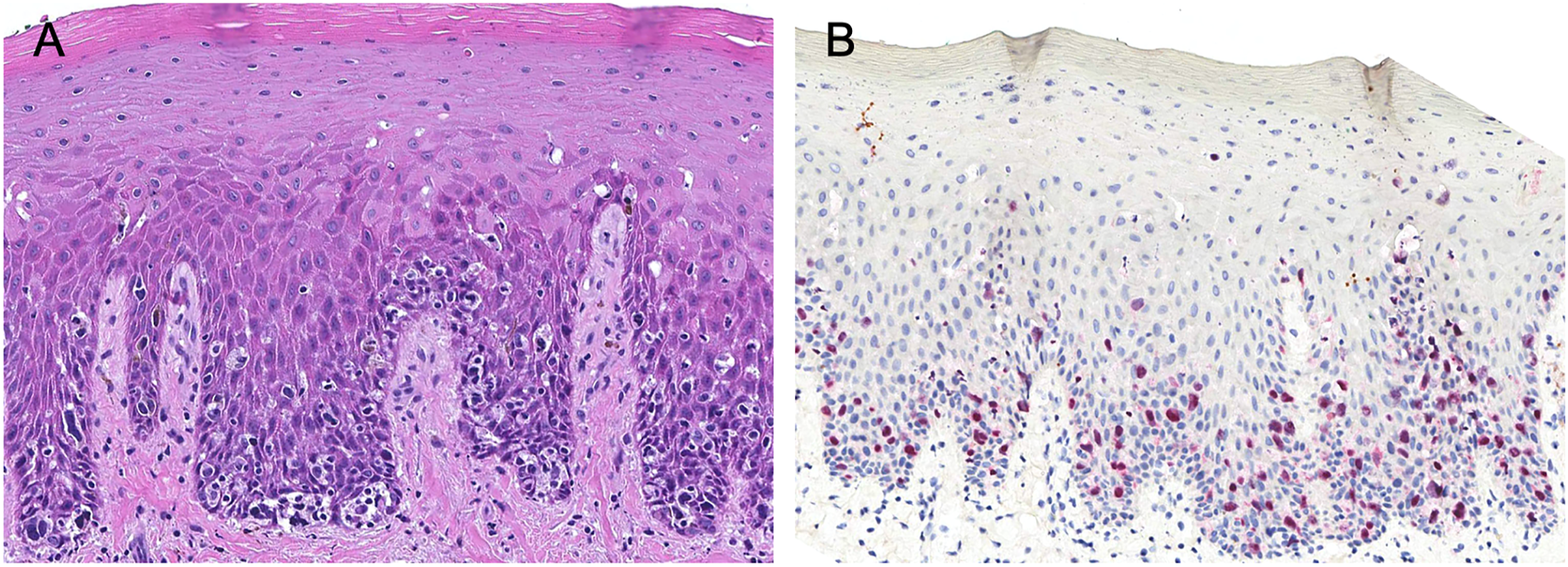
The patient was subsequently seen by Medical Oncology and Dermatology with recommendations for positron emission topography scan (PET), with consideration of proceeding with adjuvant medical therapy versus repeat surgical excision. Medical management considerations included topical chemotherapeutic agents imiquimod or 5-fluorouracil. PET was performed 2 months after surgical excision and demonstrated focal uptake in the oral cavity, thought to be post-surgical, without FDG-avid lymph nodes or distant metastases. At his 2 month follow-up visit, his palate was noted to be well healed (Figure 1B). The case was discussed at several multidisciplinary tumor board conferences (head and neck, melanoma, and complex cutaneous), with recommendations to proceed with revision surgical excision.
Revision wide local excision of the palate lesion was subsequently performed 4 months after the initial surgery, with re-excision of mucosal margins to the level of periosteum along the hard palate to the alveolus (interdental papilla), and re-excision of the mucosal margin of the soft palate. In similar fashion to the initial surgery, a Biodesign® graft was secured and bolstered with a silastic splint. Pathology demonstrated positivity in multiple regions of the hard palate specimen, as well as positivity of all soft palate epithelial margins, again with negative deep margins and remaining in situ melanoma deep to the mucosal margin. After extensive multidisciplinary discussions, the decision was made in conjunction with the patient to proceed with active surveillance. He was healing appropriately at 2 week follow-up (Figure 1C) and was seen in clinic for 5 month post-surgical follow-up without melanotic patches or other evidence of disease recurrence (Figure 1D).
Discussion
This case demonstrates a rare occurrence of in situ melanoma of the hard palate. As in the described case, in situ lesions most commonly present as melanotic patches. Mucosal melanomas typically present late due to lack of symptoms such as pain or bleeding until the lesion is of an advanced stage, which can make distinguishing between benign pigmented lesions, melanoma in situ, and malignant melanoma difficult.1,11 Subsequently, histopathologic analysis with absence of stromal invasion is paramount in diagnosis of in situ mucosal melanoma. 1 However, it is important to note that distinction between invasive tumors with junctional activity and in situ tumors can be difficult.1,4,5 Melanocytic immunohistochemical stains for reactivity to HMB-45 and Melan A can be utilized to identify invasive tumor.6,11 Recently, reactivity to the cancer testis antigen PRAME has been used to aid in identification of oral malignant and in situ melanomas, with described use for differentiating reactive hyperplasia from in situ melanoma along specimen margins. 12
Differentiating between in situ and invasive mucosal melanoma has significant implications on course of disease and management. Malignant mucosal melanoma is highly aggressive and purports a poor prognosis, with reported 15% 5-year survival. 1 Subsequently, the mainstay of management is complete surgical resection with minimum margins of 1.5 cm, with larger margins recommended for tumors larger than 3 cm. 3 Particularly in more advanced lesions or with distant metastases, radiation, and chemotherapeutic agents such as targeted immunotherapy may be used as adjuncts.1,2
In contrast, the course of in situ mucosal melanoma has been rarely described in the literature, subsequently limiting knowledge of prognosis and standardized course of treatment. Surgical intervention for oral mucosal melanoma in situ ranges in the literature from local mucosal excision alone to composite resection.4-10 Local mucosal excision was performed in multiple cases of palatal or gingival in situ melanoma,7,8,11 with recurrence requiring composite resection described in one case. 7 Initial composite resection with partial maxillectomy or mucosal excision with adjacent bone or dentition has also been described for palatal or gingival lesions.4,6,9 Two reported in situ melanomas of the lip underwent complete surgical resection with reconstruction.13,14 While several cases demonstrated no evidence of disease on subsequent follow up after initial surgical resection, recurrence was described in multiple cases.5,6,13 One such case demonstrated recurrence after initial partial maxillectomy, and after a second surgical excision and adjuvant radiation demonstrated no evidence of disease. Other reported cases of recurrence utilized the “watch and wait” strategy of close clinical observation after either multiple surgical excisions with persistent disease or recurrence after a long period with no evidence of disease after initial excision. 5 A case of in situ melanoma of the lip developed invasive metastatic disease. 13
Adjuvant therapy is less commonly utilized for in situ oral melanoma than for invasive oral melanoma; however, use of radiation 6 and topical chemotherapeutic agents such as imiquimod have been reported. 10 In general, melanoma is not very radiosensitive, but radiation can be used in some cases of early melanoma with good response. 2 Spieth et al. describe a case of topical imiquimod use for a recurrence limited to the epithelium of an invasive oral mucosal melanoma which had previously undergone surgical resection and systemic chemotherapy, with no evidence of disease after 13 months. 10 The use of topical imiquimod has been used as an adjunct for recurrent oral melanoma, with complete pathologic response in conjunction with systemic immunotherapy. 15 Importantly, in situ oral mucosal melanomas have also been likened to cutaneous acral lentiginous and lentigo maligna melanomas, 1 in which topical chemotherapeutic agents have been studied more extensively in the literature. For lentigo maligna melanoma, both topical imiquimod or topical 5-fluouracil alone have been used as primary treatment.16-19 Topical imiquimod has also been used as adjuvant therapy for in situ cutaneous melanomas with positive margins, with described cases reporting complete response and no recurrence at interval follow up.20,21 However, as was discussed in our case, the method of keeping the imiquimod on the surface of interest in the mouth is challenging and contributed to our management decisions.
In conclusion, oral cavity melanoma in situ is rarely described in the literature. Diagnosis of invasion on histopathology has significant implication on management; thus, it is vital to consider in situ melanoma in the differential for pigmented oral cavity lesions and perform appropriate biopsy and histopathologic analysis. More investigation is needed to completely understand the prognosis of this rare entity and the preferred management, necessitating the need for reporting in the literature.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
