Abstract
Melanotic neuroectodermal tumor of infancy (MNTI) is a rare benign tumor. Here, we report the diagnosis and treatment of 1 case of MNTI in the maxilla and discuss its clinical and pathological features, imaging features, treatment, and prognosis.
Introduction
Melanotic neuroectodermal tumor of infancy (MNTI) is a rare tumor which occurs during infancy, often within the first year of life. MNTI was first reported in 1918 and since then there have been approximately 500 diagnoses.1,2 MNTI typically affects the face or the skull, with rapid, distended growth, and a high recurrence rate. Around 93% of MNTIs occur on the head and neck; most commonly in the maxilla (68-80%), skull (10.8%), jaw bone (6%), and head (4.3%). 4 However, the rarity and unique nature of the disease make diagnosis and treatment challenging. 3
In 1961, Borello and Gorlin found that vanillymandelic acid (VMA) levels in MNTI patients’ urine were significantly higher, but dropped back within normal limits following surgery. This is a common occurrence in many neurogenic tumors, and as such MNTI was considered a neurogenic tumor and was acknowledged by the World Health Organization (WHO).5,6 MNTIs are considered benign tumors: most MNTIs grow rapidly within 1–2 months of onset and are aggressive. Currently, the only treatment option for MNTI patients is surgical resection but the recurrence rate after surgery is not insignificant (10-20%). Tumors in approximately 6.5% of patients involve peripheral lymph nodes or have distant organ metastasis.7,8 Compounding this recurrence and metastasis rate is the fact that clinical diagnosis is difficult. If treatment is delayed, it seriously affects growth in infants. Thus, it is essential to further characterize these tumors, improve early diagnosis rates, early treatment, and implement regular postoperative follow-ups. Here, we report a case of MNTI in the maxilla, discuss the clinical features of MNTI, and summarize our diagnosis and treatment experience of this rare disease.
Materials and methods
Ethical approval and informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (name of institute/committee) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Results
Case report
The patient was an infant female, who presented at hospital at 3 months of age with “a growing rapidly painless mess in the left maxillary gingiva for one month.” The growth in the left maxillary gingiva had been noticed one month prior to the hospital visit. However, the seriousness had not been appreciated and the tumor grew rapidly. The color of gingiva turned from pink to purplish red, the face was increasingly swollen; however, the infant could eat and sleep normally, and there were no clinical or biochemical signs of infection.
Specialized examination
The lesion tissue was located in the left maxillary gingiva and alveolar region, the size was about 2.5 × 1.5 cm, it was raised on the mucosal surface, and purplish red color (Figure 1A). The mucosa was complete, the tissue was solid to touch, non-cystic, not unlike a table tennis ball. There was non-eruption of baby teeth. No other remarkable findings were observed. (A) Intraoral phase, the tumor was purplish red with complete capsule and it was solid to touch. (B) Horizontal position of spiral CT, the low-density image of anterior left maxilla, bone destruction (arrow). (C) Horizontal position of spiral CT showed multicentric growth in the lesion.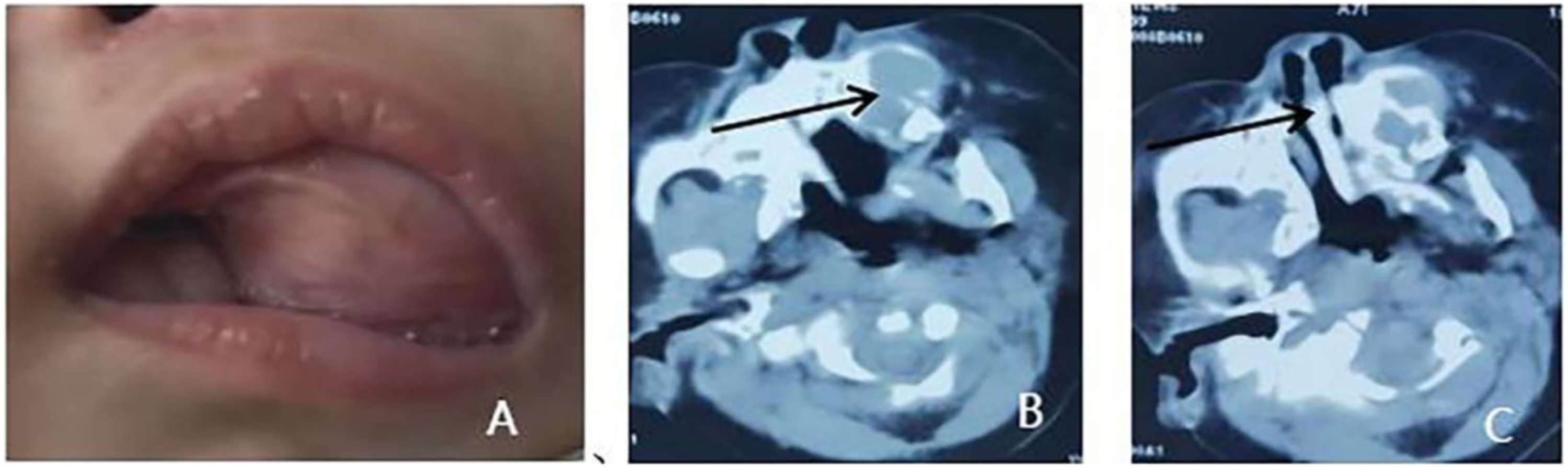
Imaging examination
Spiral CT (CT value was about 59HU) showed a compartment-like low-density region in the anterior part of the left maxilla, with buccal bulging growth, bone resorption destruction, through between bone cavities, clear boundary, irregular, lining with homogeneous low-density lesion tissues (Figures 1B and 1C).
Surgical treatment
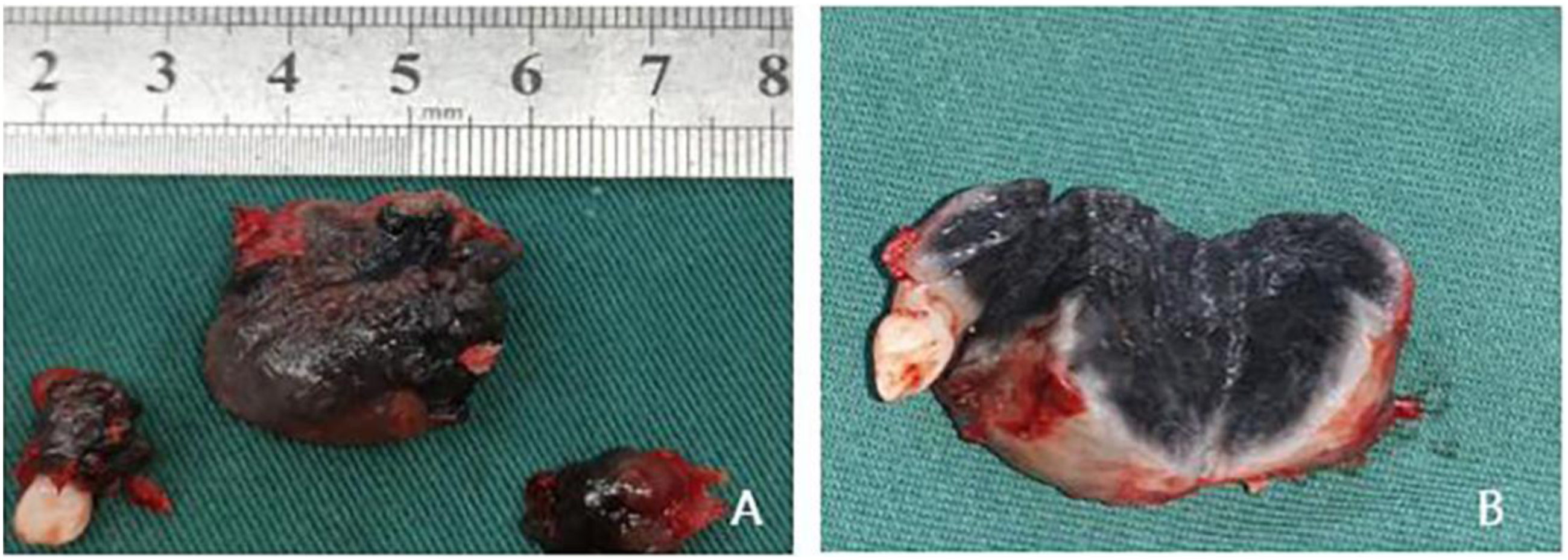
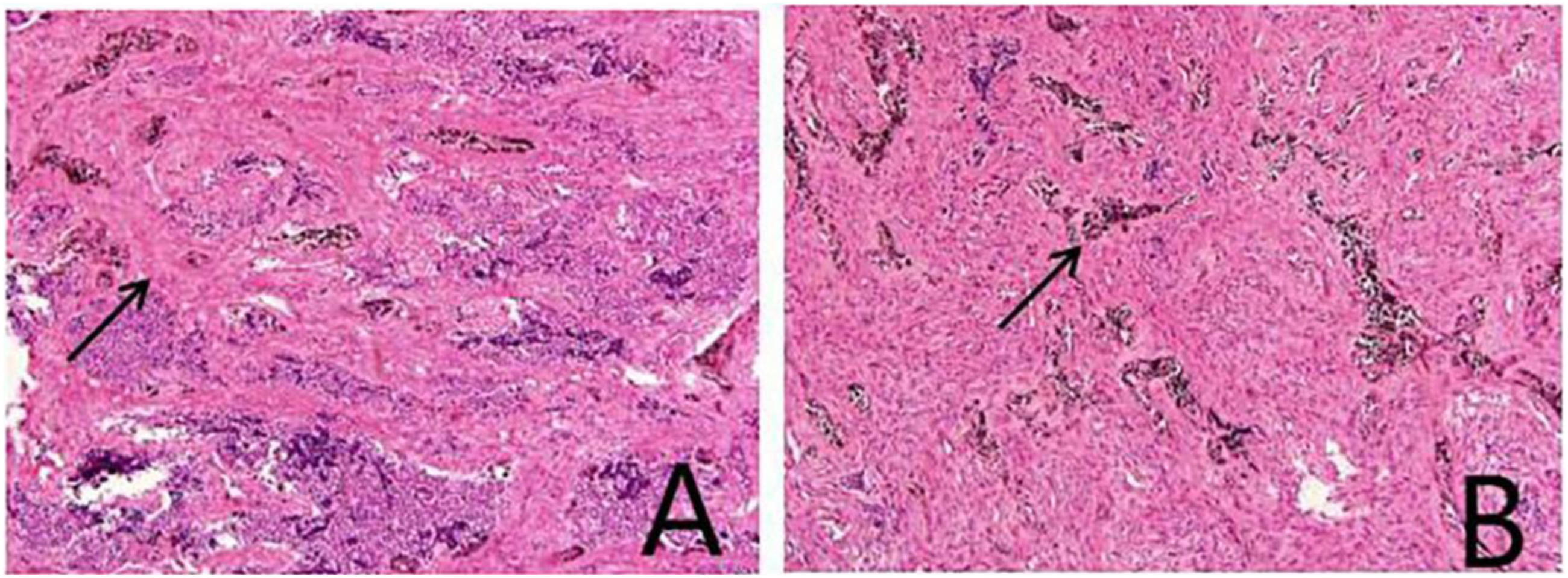
Surgical contraindications were excluded (Figure 2). After general anesthesia with intraoral intubation, a left maxillary alveolar ridge incision was made, and the mucosa and submucosal tissues were incised to expose the tumor. The tumor was solid with a clear boundary, incomplete capsule and purplish red color, surrounding the 61 tooth germ. There were three lobes growing along the bone cavity, which were located in the side of the nose, the alveolar ridge of the anterior part of the left maxilla, and the maxilla in the low position of the maxillary sinus, which were independent of one another. Surgical curettage was performed, the lesion tissues were completely curetted along the bone wall and the surrounding tooth germ was removed. A secondary resection was performed on the visibly pigmented tissue to preserve the bone as much as possible. The gross appearance of the intraoperative tumor showed 3 lesions surrounding the tooth germ, while an incomplete capsule could be seen during dissection of the tumor and the inside was homogeneous solid lesional tissue: the center was black, and the boundary was white (Figure 3). Intraoperative freezing indicated a benign tumor. Rapid paraffin pathology revealed a mixture of epithelioid cells and lymphocyte-like cells which formed a clumped cord-like structure, and some epithelial cells were pigmented (Figure 4). After the pathological consultation performed by Xijing Hospital, the diagnosis of MNTI was confirmed. Immunohistochemical examination revealed Vimentin/NSE/HMB45/Syn(+), CK/EMA (large cell+), GFAP/S-100(+), Desmin (scattered+), Ki-67 (3%+). The patient had recovered well one year after surgery. Close follow-up observation was recommended. (A) Incomplete capsule and solid lesion tissue could be seen by dissecting the tumor. (B) Remaining 2 tumors located in the nasal floor and the left maxillary sinus floor, between bone cavities, no obvious adhesion of lesion (arrow). (C) The resection phase, complete curettage, clean bone cavity. (A) Three lesions, independent, surrounding the tooth germ. (B) Dissection phase, incomplete capsule of the tumor, black center, homogenous. (A) Epithelioid cells and lymphocyte-like cells mixed (arrow) to form a clumped cord-like structure. (B) Epithelial cells contained pigments (arrow).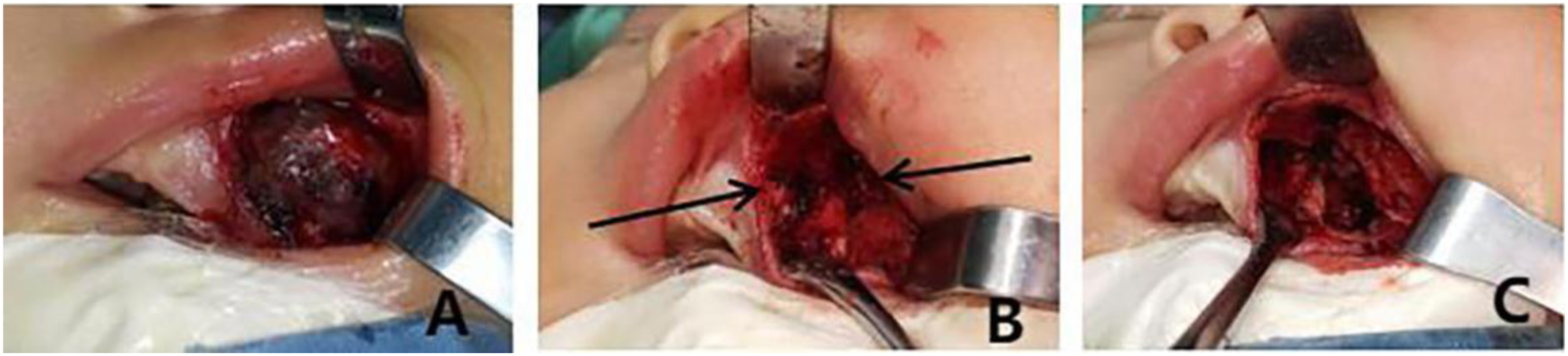
Discussion
MNTI is a rare benign congenital tumor. Until relatively recently the tissue source of the tumor had not been determined, therefore several different names were used to describe the same disease (congenital melanoma, melanotic adamantinoma, primordial retinal tumor). 6 However, it is currently believed to be of neural ridge origin. Despite being considered a benign tumor, MNTI grows rapidly, is aggressive and has reasonably high rates of recurrence. There are several reported cases with distant metastasis and even reports of malignancy; 9 therefore, early diagnosis and treatment of the tumor is critical. Based on review of the literature review and case analysis, this paper focuses on the clinical manifestations, diagnosis, differential diagnosis, treatment and prognosis of the tumor.
Most MNTI patients are under 1-year-old: 77% cases are identified in infants less than 6 months old, and few occur in patients over 1-year-old. Further, there are also reports of adult patients. 10 There is a slight imbalance in gender of patients where the tumor is more common in males (the male to female ratio is 1.27:1). More than 70% of MNTIs occur in the anterior part of the maxilla; however, other predilection sites include the skull, mandible, and brain. There are also reports of cases in the epididymis, mediastinum, uterus, and femur.11,12 The anterior part of the maxilla is the most common site, which may be caused by a reversion of the sensory neuroectoderm, or related to the sinking of the retinal primordium cells at the fusion line of the maxillary embryo. 13 It presents as a painless growth which progresses rapidly. Unless there is a secondary infection or the tumor is advanced, the surface of the mass gradually changes color toward visible pigmentation; purplish red or blue-black with homogeneous texture. On imaging, there is extensive jaw destruction and tooth displacement, and additional manifestations such as cystoid bone destruction, bone hyperplasia, and bone sclerosis. Typical CT images reveal low-density masses with irregular margins, occasional reports of high-density, clear boundary with surrounding tissue, and lobulated, multicentric growth. 14 Both T1 and T2 weighted MRI scans show low signal masses. 15
According to the unique clinical manifestations and imaging characteristics, the possibility of MNTI should be considered in patients who present with similar growths. Preoperative urine examination of patients can be performed, where VMA abundance in the urine of patients with MNTI is significantly increased. However, abnormal levels of VMA can also be diagnostic of retinoblastoma, ganglioneuroblastoma or pheochromocytoma. 16 In a recent study, there were 69 patients where urine VMA was quantified, but of these only 24 MNTI cases sdfd identified. 17 Therefore, urine VMA quantification should be used as an auxiliary examination. Pathological biopsy is required to diagnose and there is also a report that surgical resection could be performed without biopsy to avoid stimulation of the lesion. 18 Visual cross-section findings are solid, tough or hard, gray to blue-black, and under microscopic analysis the tumor is composed of epithelioid cells and small neuroblast-like cells. 19 The lining is an irregular acinar, tubular, slit-shaped structure, and separated by dense fibrous tissue. Immunohistochemistry of MNTI shows strong heterogeneity as HMB-45 positive and occasionally S-100 protein positive. Expression of Ki-67/CD99 is seen in some cases, suggestive of faster tumor growth. 20 Clinical manifestations need to be differentiated from developmental cyst, ameloblastoma, and eosinophilic granuloma. Some MNTI cases display characteristics of malignant tumors, therefore, it should be differentiated from neuroblastoma. When neurogenic cells are most abundant, as determined by microscopy, MNTI should be differentiated from melanoma where melanocytes are the most abundant cell type. Immunohistochemical examination is often necessary. If immunohistochemical staining is performed on melanoma, melanoma are characterized by cytokeratin(CK) (−), melanoma-associated antigen HMB45 (+), S100 (+), but MNTI cases are generally CK (+), HMB45 (+), S100 (−). Neuroblastoma markers are NB84a (+), however MNTI is NB84a (−). Rhabdomyosarcoma is Desmin (+) , MyoD1 (+), neuronspecificendolase (NSE) (−), however MNTI is NSE (+), MyoD1 (−), while Desmin can be either positive or negative. Other small round cell malignant tumors of children are rare before 5 years old (such as Ewing's sarcoma, peripheral primitive neuroectodermal tumor and desmoplastic small round cell tumor).
Surgery is currently the main treatment option for MNTI patients. Chemotherapy and radiotherapy are not suitable for surgical patients or to be used as adjuvant therapies before or after surgery. 21 There has been one report of treatment of MNTI using only chemotherapy; 22 however, the optimal treatment regime is not yet known. Surgical treatment (total resection) of MNTI remains the best method to prevent recurrence. At present, surgical approaches tend to be conservative. However, where possible, it is recommended to extend the resection by 5 mm to do a complete resection. Complete resection should be considered only where function is not greatly affected; local resection and curettage of the underlying bone are usually sufficient for treatment. 21 For recurrent lesions, radical surgery is recommended with postoperative adjuvant chemoradiotherapy. 23 Metformin has been used to treat patients with recurrent MNTI, which controlled the tumor effectively. 24 Additionally, neoadjuvant chemotherapy performed before surgery resulted in adequate tumor shrinkage for enhanced resectability and easier surgical access, therefore minimizing the need for extended margins. 25 In summary, the main focus of MNTI treatment is to minimize surgical intervention and its sequelae. Conservative surgical resection is recommended to preserve organs and functions as the first choice of treatment. After successful surgery, adjuvant treatment is no longer required, but close postoperative review is needed. 26
MNTI is considered a benign tumor, with local recurrence rates around 10–15% after conservative resection. However, recurrence rates of 40–60% have been reported. 6.5% of cases have distant metastasis, 27 and as a result there is some evidence to suggest that MNTI should be considered a low grade malignant tumor and postoperative chemoradiotherapy should be needed. In some cases, recurrence occurred a few weeks after surgery. 28 One report suggested that a surgical microscope should be utilized to remove invisible pigmented lesions during the surgical resection to avoid recurrence. 29 Recurrence may be related to the infiltration of the tumor margin into the bone, and where the tumor has no, or an incomplete, capsule then it can be challenging to completely resect by surgery. Recurrence may also be related to intraoperative implantation or multicentric growth of the tumor. 30 There is also a case of non-recurrence with incomplete resection of the tumor. The reason may be that the de-bulking effect induced an effective anti-tumor immune response. Some studies hypothesized that the cells surrounding the tumor are maintained by a group of central “stimulating” cells, and when the central stimulating cells were resected then the surrounding tumor cells died through lack of stimuli.30 -33 Ultimately, death of MNTI patients may be caused by extensive accumulation of unresectable tumors, but there is evidence of a recurrence-free follow-up period of 19. At present, close follow-up within half a year post-surgery, especially within 1 month, remains effective for the early detection of postoperative recurrence signs and allows early intervention measures.
Conclusion
The maxilla is the most common site of MNTI, while patients are primarily under 1 year old. Tumors grow rapidly and are accompanied by color change of the mucosal surface. Conservative surgery is currently the first choice for treatment, and close follow-up is needed for 1 year after surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
