Abstract
This is a brief communication stressing some critical points associated with cystic nodal metastasis (CNM) in human papillomavirus (HPV) positive oropharyngeal squamous cell carcinoma (OPSCC) and its implication on treatment protocols and outcomes.
Keywords
To the Editor
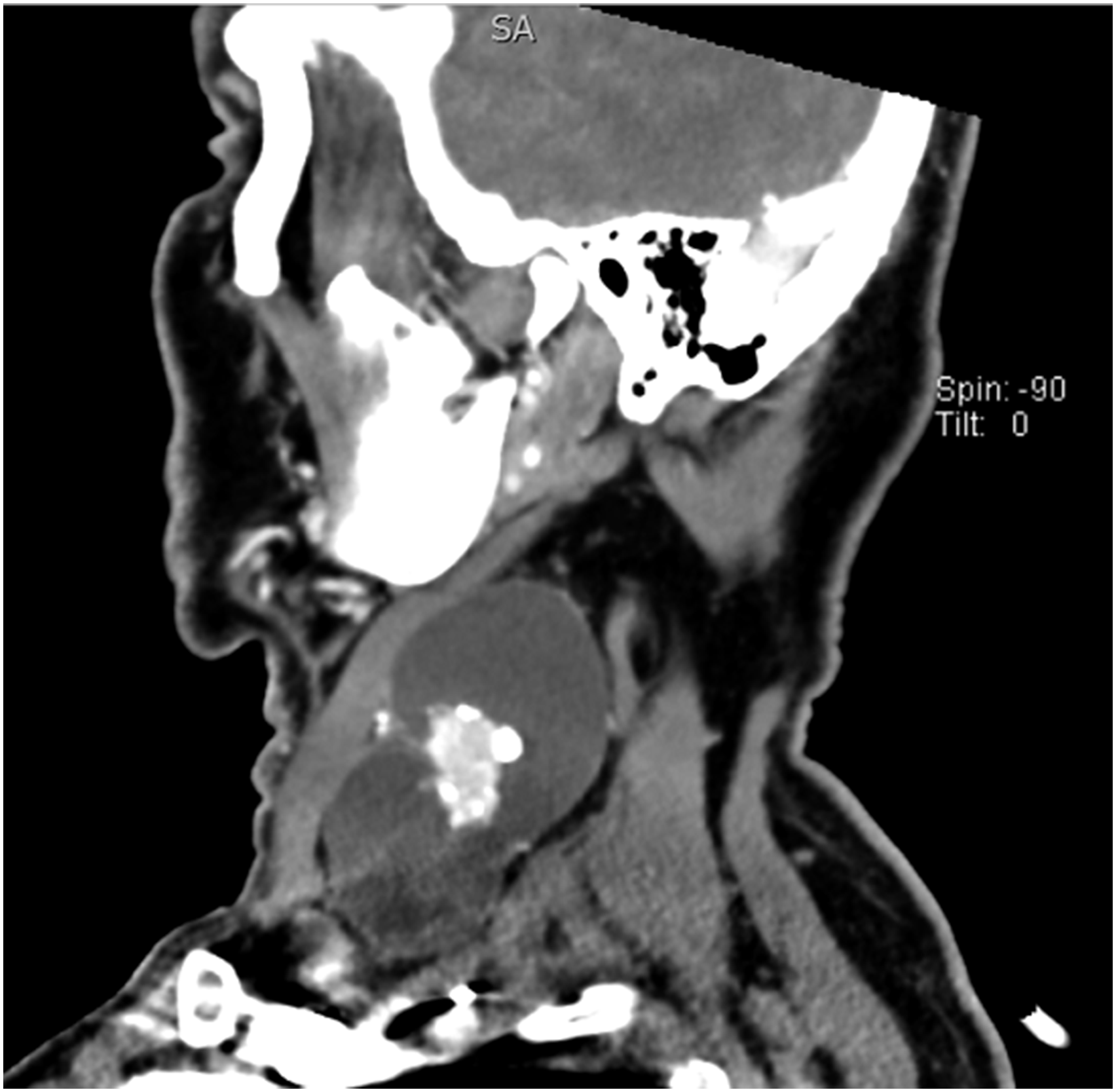
Cystic nodal metastasis (CNM) is a well-recognized clinical entity defined as a cystic mass with homogenous fluid content and an enhancing capsule <2 mm in thickness but without internal complex, irregular, or solid areas (Figure 1). 1 It is observed in various head and neck malignancies, such as primary squamous cell carcinoma (SCC) originating in the epithelium within the Waldeyer’s ring, papillary carcinoma in the thyroid gland, and adenoid cystic carcinoma in the salivary glands. 2 Goldenberg et al. 3 reported that in 85% of the patients with CNM, primary head and neck SCC (HNSCC) was found in the oropharynx and was associated with human papillomavirus (HPV) in 87% of the cases.
According to the latest tumor–node–metastasis (TNM) classification system for head and neck cancers by the American Joint Committee on Cancer (AJCC 8th edition), oropharyngeal SCC (OPSCC) was divided into HPV-positive and HPV-negative OPSCC. It is known that HPV-related OPSCC has a gene expression profile and response to chemoradiation that is distinct from that of OPSCC related to tobacco and alcohol. However, prognostic significance of CNM in the setting of metastatic HPV-positive OPSCC is poorly established, although it is found in approximately 40% of these patients. 4
The pathogenesis of CNM is not completely understood. However, the following pathogenic mechanisms in the substrate have been proposed: (a) Pseudocystic transformation resulting from spontaneous degradation of keratin and cellular debris within carcinomatous lymph node deposits; (b) True cystic change originating from malignant salivary type cells or transformed keratinocytes, which have intrinsic properties for cyst formation; and (c) Metastasis into branchial cleft cyst.2,5 As for differential diagnosis, CNM should be differentiated from necrotic nodal metastasis (Figure 2), which is a completely different pathological entity. The latter is characterized by thicker walls (>30% of the circumference) and irregular, complex, central, low attenuation. 3 It is significantly more prevalent in the HPV-negative OPSCC cases than in the HPV-positive ones. Moreover, it has low radiosensitivity and is an independent predictor of regional failure. 4 Another important malignancy in the differential diagnosis of CNM is primary branchiogenic carcinoma. It is known that CNM frequently metastasizes from an unknown primary site, with the occult primary tumor located in the Waldeyer’s ring. Therefore, diagnostic work-up should follow clinical practice guidelines for cancers of unknown primary.1,5
Unfortunately, the prognostic significance of CNM in HPV-positive OPSCC cases has been poorly investigated. A recent study by Rath et al.
6
on 183 patients with HPV-positive OPSCC showed that CNM at imaging analysis was associated with significantly lower risk for treatment failure (locoregional failure and occult distant micrometastasis following primary chemoradiotherapy) with better disease-free survival than solid nodal metastasis. In contrast, Huang et al.
4
reported that CNM did not correlate with the relative risk of local and regional failure, but it was a negative independent predictor of distant failure in patients with metastatic OPSCC receiving chemoradiotherapy. However, the vast majority of patients (65/98) from this study were HPV-negative. Previous studies by Munck et al.
7
and Janot et al.
8
showed that isodense metastatic lymph nodes had significantly better response rates to chemotherapy in HNSCC as compared to hypodense lymph nodes (68% vs. 8% and 63% vs. 15%, respectively). Similarly, in patients with N2 stage of nasopharyngeal carcinoma undergoing chemoradiotherapy, the disease-specific survival and overall survival rates were poorer and the risk of distant failure was higher with CNM than solid nodal metastasis.
9
Negative prognostic value of CNM in these papers was attributed to increased radio- and chemo-resistance secondary to impaired angioperfusion and hypoxia and increased extracapsular spread.7-9 Multi-slice computed tomography scan of the neck in sagittal projection showing cystic nodal metastasis. Multi-slice computed tomography scan of the neck in sagittal projection showing necrotic nodal metastasis. 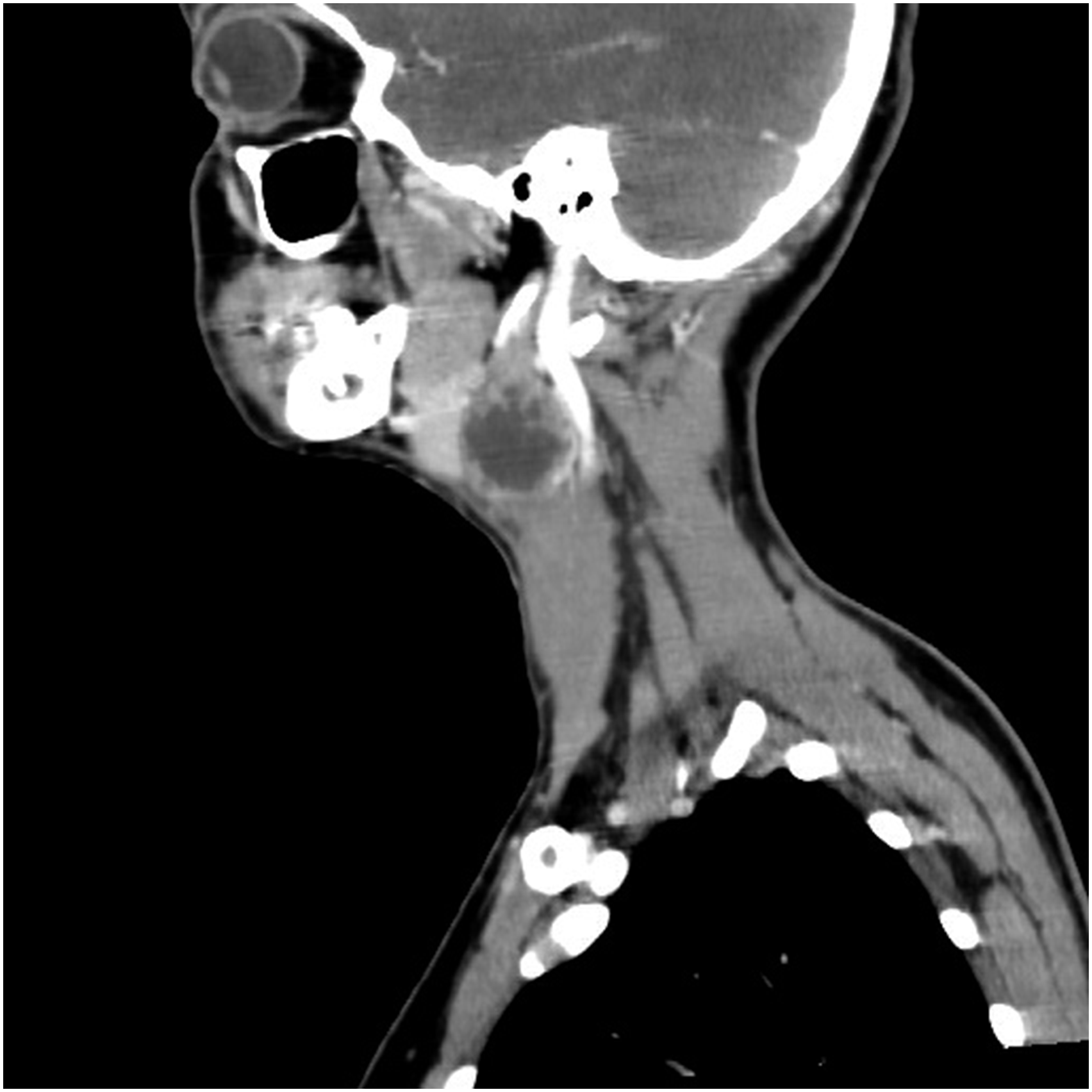
In the last several decades, the reported prevalence of HPV in OPSCC in the United States has been over 70%, and it continues to increase rapidly. 3 Although the AJCC 8th edition of the TNM classification system incorporated the specific staging for HPV-positive OPSCC, further identification of prognostic factors is warranted. Novel clinical trials should provide a definitive answer regarding the prognostic significance of CNM in HPV-positive OPSCC and its potential use as a predictive marker in future editions of the AJCC TNM classification system. In our institutional experience, the sole presence of CNM in HPV-positive OPSCC might be associated with more favorable clinical outcome than solid nodal metastasis, posing a question about treatment de-escalation in these patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The authors assert that all procedures contributing to this work complied with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1964, as revised in 2013. No ethical approval was required as this is a Letter to the Editor with no original research data.
