Abstract
Primary tracheal adenoid cystic carcinoma (TACC) occurring in the cervical trachea and invading the thyroid is very rare. Surgical resection and airway reconstruction are the main treatment methods, and other treatments include radiotherapy and endoscopic intervention. Herein, we present the case of a 74-year-old female patient with TACC. The patient underwent surgery and postoperative pathology showed that the tumor invaded the adventitia of the trachea and bilateral thyroid, where nerve involvement was observed. The patient recovered well after the operation without adjuvant therapy. Eight months after the operation, Computed tomography showed that the trachea was unobstructed and there was no recurrence.
Introduction
Primary tracheal adenoid cystic carcinoma (TACC) is a rare, low-grade, slow-growing tumor that is prone to local recurrence and distant metastasis. 1 However, although it is low-grade, it is often combined with different degrees of large airway stenosis, which can cause breathing difficulties and be life-threatening. Surgical resection and airway reconstruction are the preferred treatments. As TACC often infiltrates the surroundings, especially the peripheral vascular and nerve bundles, the surgical boundary is difficult to determine, and the positive rate of postoperative margins can reach 45.8–63%, 2 which is the main factor for postoperative recurrence and metastasis. The incidence of TACC itself is not high, and cases that occur in the cervical trachea and invade the thyroid are even less common. 3 In this article, we describe the surgical approach for a case of TACC with bilateral thyroid invasion in which a total cervical tracheal resection and laryngectomy were performed simultaneously with a permanent ultra-low tracheostomy.
Case Description
The patient was a 74-year-old female with a history of hypertension and diabetes. Paroxysmal cough and hemoptysis occurred more than 1 month earlier. Computed tomography (CT) showed that the anterior border of the trachea had slightly low-density nodules at the lower border of the thyroid, and the trachea was compressed and narrowed (shown in Figure 1A). Magnetic resonance imaging of the neck showed that the tracheal cartilage was involved in the anterior wall of the trachea, consistent with tracheal carcinoma (shown in Figure 1B). Thyroid color Doppler ultrasound showed a solid mass at the anterior trachea–thyroid isthmus (shown in Figure 1C and D). The pathological findings of bronchoscopy biopsy: (new organisms in the trachea): salivary gland tumors and dysplasia of epithelial-myoepithelial components. Immunohistochemistry (IHC) results were as follows: Syn (–), CgA (–), CD56 (–), Ki67 (20%), TTF-1 (–), NapsinA (–), CK (epithelial +), CK5/6 (myoepithelial +), P40 (myoepithelial +), and pan-CK (+). Based on the morphology and IHC, a diagnosis of adenoid cystic carcinoma was considered. A. Sagittal computed tomographic images showing irregular thickening of the tracheal wall at the level of the lower border of the thyroid, forming soft tissue shadows with unclear boundaries, uneven enhancement, and patchy non-enhancing areas of liquefaction necrosis inside. B. T1-weighted magnetic resonance imaging showed irregular soft tissue nodules in the anterior wall of the trachea and bulging into the lumen, showing low signal on T1WI, and narrowing of the tracheal lumen at the corresponding level. The anterior and thyroid borders were not clear. C–D. Ultrasound showed a hypoechoic area in the trachea with unclear boundaries, uneven internal echoes, and unclear demarcation between the anterior trachea and the thyroid.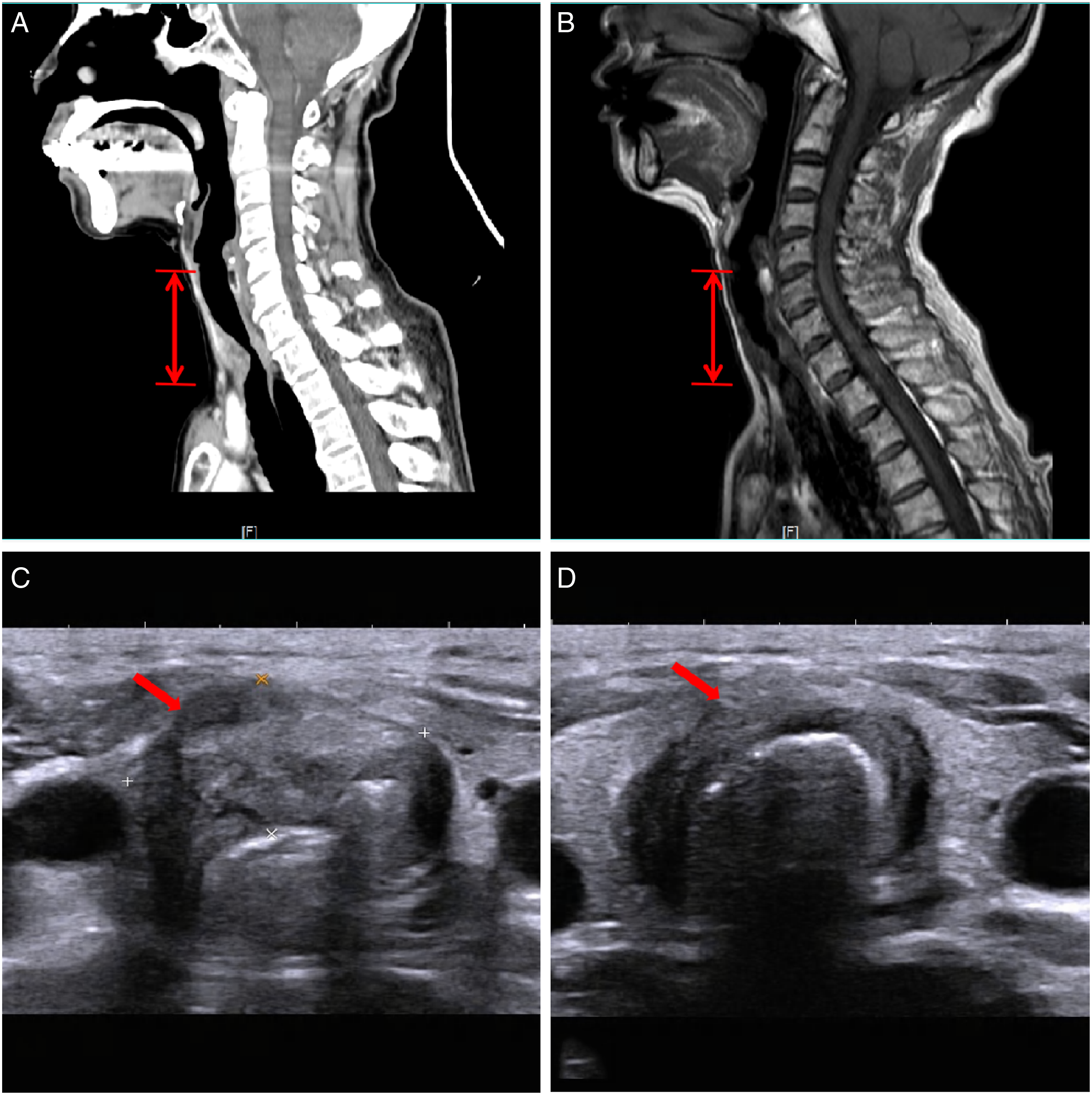
After a multidisciplinary discussion, surgical treatment was decided upon. On March 20, 2020, cervical tracheal resection + bilateral thyroidectomy + total laryngectomy + hypopharyngoplasty + tracheostomy + left lower parathyroid autograft was performed under general anesthesia. During the operation, the 1st to 6th tracheal rings were found to have cancer. The intratracheal mucosa of this segment was white, with small granules and micro-bulges. The lower edge of the 7th cartilage ring was horizontally excised. No cancer was found in the 8th tracheal ring. The incisal margin of the 8th tracheal ring and cricoid cartilage were taken and sent for frozen pathology. No cancer was found in the left or right margin of the 8th tracheal ring or the lower border of the right cricoid cartilage. A few epithelial nests were severely squeezed at the lower edge of the left cricoid cartilage, but the diagnosis needed to be confirmed by paraffin sections. Total laryngectomy was performed due to a positive left upper margin (shown in Figure 2A and B). A. Surgical situation. Tracheal tumor specimen and invaded thyroid. B. Intraoperative situation: ① hypopharynx, ② recurrent laryngeal nerve, and ③ stump of trachea. C–D. Postoperative pathology. (C) Tumor cells lined up in irregular epithelial masses with many round or oval cyst-like cavities in a cribriform pattern and invasion of the thyroid. (D) Tumor invasion of the nerve.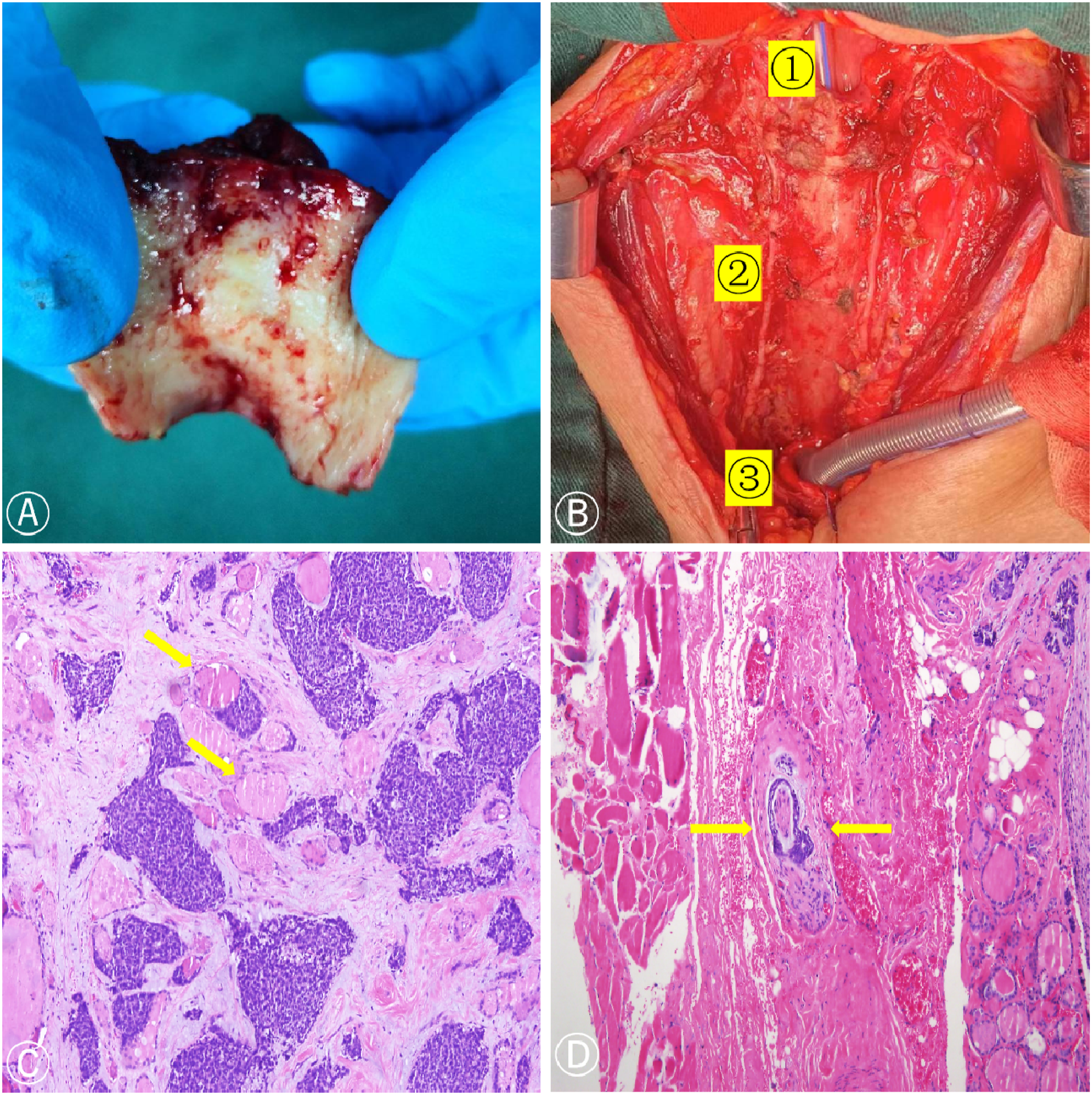
Postoperative pathology showed that the tumor invaded the adventitia of the trachea and bilateral thyroid and parathyroid tissues, where nerve involvement was observed. Carcinoma involvement was found on the lower margin of the left cricoid cartilage, and there was no invasion at the rest of the margins. Immunohistochemistry showed the following results: CK (+), CK5/6, P63 myoepithelial (+), S-100 (–), CD117 ductal epithelium (+), Ki-67 (+, about 35%), Syn (–), and carcinoembryonic antigen (–) (shown in Figure 2C and D).
The patient recovered well after the operation without adjuvant therapy. Eight months after the operation, CT showed that the trachea was unobstructed and there was no recurrence.
The authors confirm that written informed consent was provided for the patient for publication of case details and any accompanying images.
Discussion
Primary malignancies of the cervical trachea are rare, with only 2.6 new cases per million people per year. 4 Tracheal adenoid cystic carcinoma accounts for about 30% of primary tracheal malignancies, which is the second most common tumor after squamous cell carcinoma. 5 Tracheal adenoid cystic carcinoma is a salivary gland tumor that originates from the glands in the trachea and bronchial wall. About two-thirds of TACC cases present in the lower trachea and the starting level of the left and right main bronchi, and the other one-third occurs at the starting point of the large bronchi. There is no significant sex predilection, 6 the peak age of onset is 40 to 50 years old, 7 and is not related to smoking. 8 The initial symptoms of TACC are non-specific, mainly manifesting as symptoms of airway irritation and obstruction, such as cough, expectoration, hemoptysis, shortness of breath, dyspnea, and fever. In our case, the patient presented with cough and hemoptysis. Due to the non-specific symptoms and the tumor growing in the lumen or along the wall, diagnosis is often missed. 9 The diagnosis of TACC depends on CT findings of airway masses and is mainly confirmed by bronchoscopy and biopsy histology. In our case, the diagnosis was also confirmed by bronchoscopy. Therefore, timely fiberoptic bronchoscopy and biopsy are particularly important for the diagnosis of TACC.
Tracheal adenoid cystic carcinoma often spreads by direct extension, submucosal or peripheral nerve infiltration, and hematogenous metastases. 10 More than 50% of TACC patients have hematogenous metastases, the most common site of metastases being the lung, but can also be found in the brain, bone, liver, kidney, skin, abdomen, and heart.11,12 Tracheal adenoid cystic carcinoma that invades the thyroid is very rare, leading to a misdiagnosis of thyroid cancer.9,13 Tracheal adenoid cystic carcinoma mainly involves the thyroid through direct invasion, with few cases of hematogenous metastasis. In this case, TACC involved the bilateral thyroid glands through direct invasion; therefore, the bilateral thyroid tissue was completely resected during the operation.
Surgical resection is the preferred treatment method for TACC. The 5-year survival rate after surgery is 85%, while that for patients without surgery is only 63.7%. 14 Since TACC often infiltrates to the periphery and especially tends to infiltrate the peripheral vascular and nerve bundles, the surgical boundary is difficult to determine, and positive postoperative margins are common. Positive postoperative margins been reported in up to 15% of intramural cases and 85% of extramural cases, 15 which is a major risk factor for postoperative recurrence and metastasis. The postoperative pathology of this case also confirmed that the nerve was involved with the tumor. Therefore, we performed extended resection to minimize the risk of recurrence. The surgical approach to such tumors depends on the surgeon’s experience and the tumor’s size, location, and degree of local invasion. 16 The extent of surgical resection is limited by tracheal length, and it is generally accepted that the resected length cannot exceed 6 cm or 50% of the total length. Xie et al 17 reported a case of TACC with a tumor length of 6.5 cm. They removed approximately 7.5 cm of the tracheal membranous wall (about 64.1% of the total length of the trachea). To reduce the anastomotic tension, they used a pedicled pectoralis major myocutaneous flap to repair the gap in the tracheal wall. Tracheal adenoid cystic carcinoma is usually operated on by direct surgical resection of the trachea and airway reconstruction, while Jiao et al 16 introduced a new technique of thoracoscopic tracheal resection with end-to-end anastomosis, which is less invasive. However, it is limited to lesions of the lower trachea or bronchus, and difficult airway reconstruction cannot be completed, so it is not suitable for the patient described in this report. The main surgical complications include tracheoesophageal fistula, hypopharyngeal fistula, poor anastomotic healing, and vocal cord paralysis, resulting in difficulty in expectoration and pulmonary infection often requiring tracheotomy.
Tracheal adenoid cystic carcinoma is more sensitive to radiotherapy. 18 Most studies have shown that for patients with positive surgical margins or inoperable patients, postoperative adjuvant radiotherapy or radiotherapy alone is an effective treatment with a local control rate of 20%–70% without affecting survival. 19 It is now generally accepted that patients with positive surgical margins should receive radiation therapy. However, for severe life-threatening airway stenosis, radiation therapy does not work immediately, and there are many complications. In the case we have reported, surgical margins were negative, and thus no further radiation therapy was performed. Most literature reports that chemotherapy alone is not effective for TACC. For patients who are not suitable for surgery or radiotherapy, treatment methods such as bronchoscopy ablation and stent placement can also be used. 5 Bronchoscopic interventional techniques, such as transbronchial cryotherapy, high-frequency electrocautery, laser, and argon plasma coagulation can quickly open the airway, relieve clinical symptoms, and prolong survival time. 20 However, such methods have a long treatment period and are associated with many complications, which can lead to recurrence.
Conclusion
Tracheal adenoid cystic carcinoma is a rare primary malignant tumor of the trachea. Complete surgical resection is the best treatment, and airway reconstruction is key. Radiation therapy is an important treatment for patients with positive surgical margins or those not indicated for surgery. The local control rate with radiation therapy is acceptable, but it has no effect on overall survival and there are many complications. Endoscopic interventional therapy has recently become a popular treatment method for patients with locally advanced disease who cannot undergo surgery as it can quickly relieve symptoms and prolong survival for further treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Natural Science Foundation of Fujian Province (grant number: 2021J01446) and Fujian Provincial Health Technology Project (grant number: 2019-CXB-7).
