Abstract
Salivary megaduct with stricture is characterized by recurrent pain, swelling, and infection, leading to reduced quality of life. Surgical management includes sialodochoplasty, however, recurrence is common and repeated surgery can lead to further scarring. In the sinuses, drug-eluting stents (DES) are used to reduce scarring after surgery, yet no such technology exists for salivary megaduct with stricture.We trialed DES for this condition with promising results which may pave the way for future development.
Significance Statement
A 49-year-old male with a remote history of facial trauma developed chronic left parotid ductal stenosis with proximal ductal dilation, ultimately becoming a salivary megaduct. Initial evaluation with CT demonstrated a widely dilated parotid megaduct with distal stenosis (Figure 1). The patient underwent a pull-through sialodochoplasty as previously described by Kandl et al
1
and initially did well, but redeveloped stenosis over the following months. Five months later, the patient underwent revision surgery with repeat marsupialization of the distal duct and placement of a 2 mm silicone salivary stent (Schaitkin Salivary Duct Cannula™; Hood Laboratories, Pembroke MA). The silicone stent was removed 14 days after surgery, the patient did well initially, but two weeks later, he again developed stenosis with megaduct. Unfortunately, this clinical scenario is common among patients with this condition. A non-contrast CT scan demonstrating stricture and associated megaduct of the patient’s left parotid duct, indicated by the yellow arrows.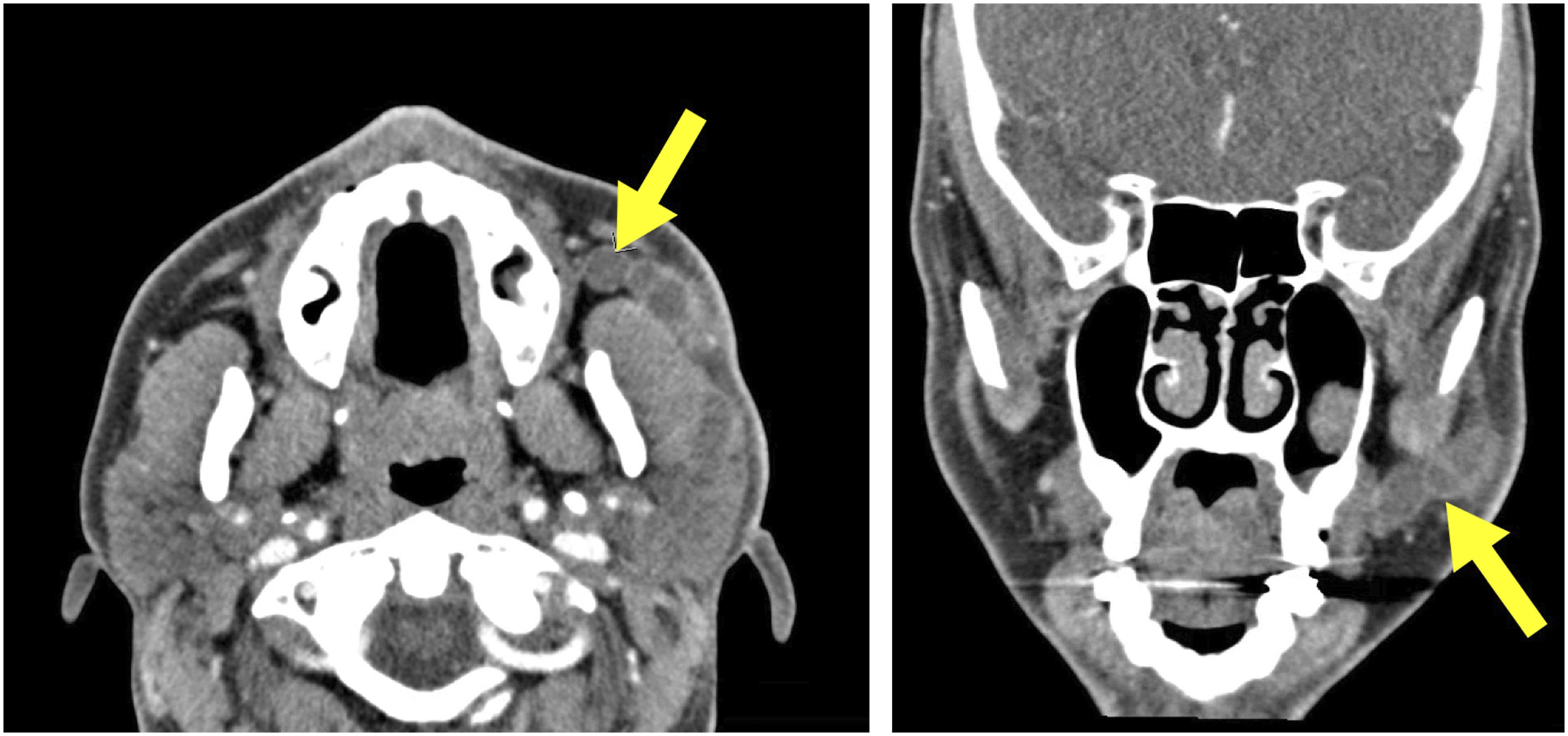
At eight months from initial surgery, the patient was offered revision surgery with a novel, off-label use of a steroid-eluting stent to attempt to achieve permanent duct patency. The patient elected to proceed and signed informed consent.
A pull-through sialodochoplasty was performed. In brief, a circumferential mucosal incision was created around the site of prior scar. This was gently retracted, and the underlying parotid duct was identified and carefully skeletonized. The duct was opened using a T-shaped incision to fillet the duct and maximize the diameter of the dochoplasty. The redundant ductal tissue and distal scar were excised and the dochotomy site was widely marsupialized to the buccal mucosa, resulting in a widely patent dochoplasty (Figure 2). A widely patent sialodochoplasty is performed. After dissecting the papilla and duct away from the oral mucosa, scarred and fibrotic ductal tissue is excised. The remaining duct is marsupialized and secured to the oral mucosa with 4-0 PDS suture.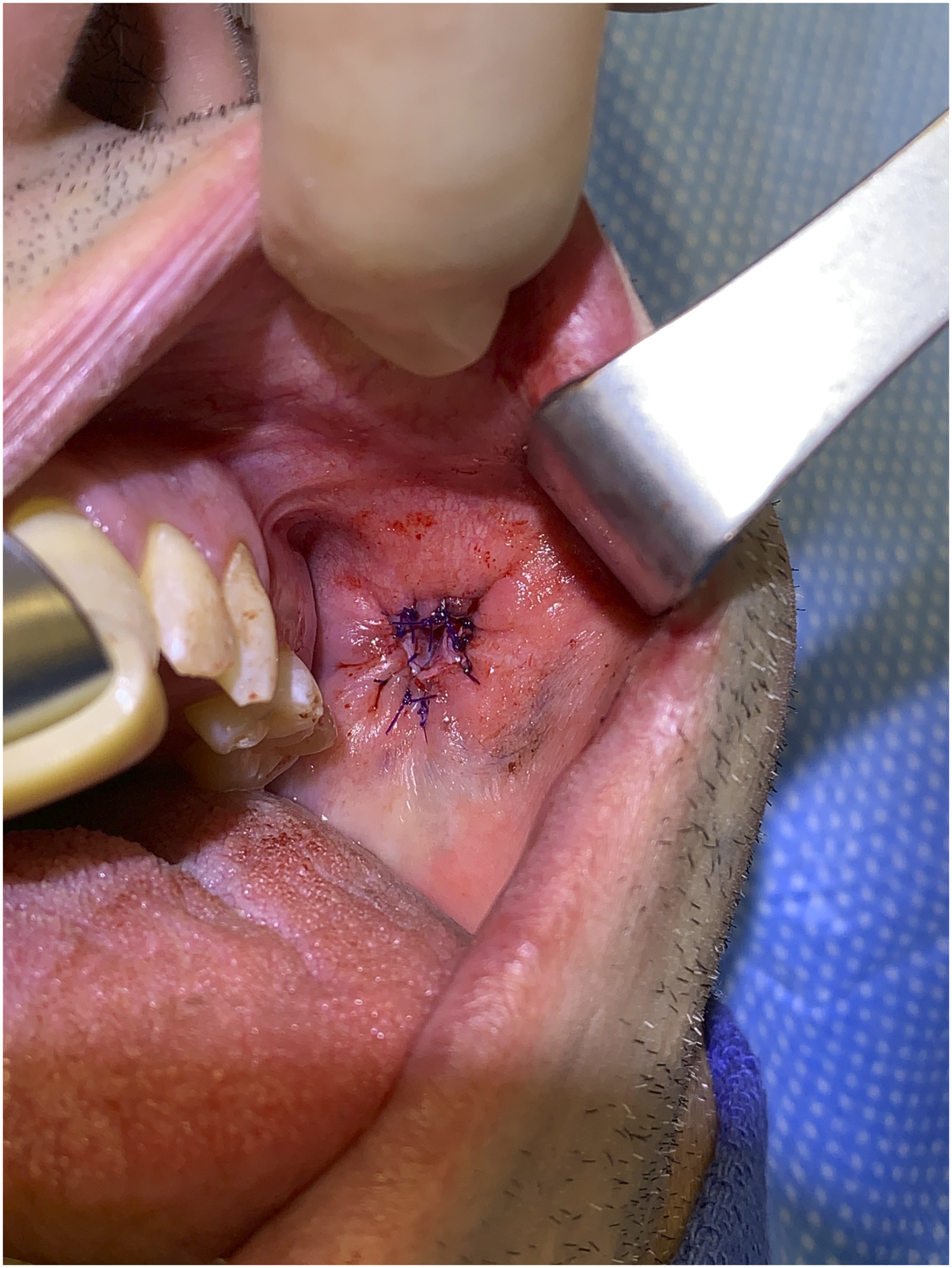
A Propel Contour sinus stent (mometasone furoate, Intersect ENT, Menlo Park, CA) was loaded onto the insertion device and carefully deployed in the distal parotid duct under direct visualization (Figure 3). This resulted in good placement within the distal duct extending to the level of the dochoplasty. The stent was secured to the adjacent mucosa using three 4-0 polydioxanone (PDS) sutures (Figure 4). The patient was discharged home with instructions to use Chlorhexidine mouth wash four times daily for 10 days and to limit diet to soft foods for 5 days. The Propel Contour stent (mometasone furoate, Intersect ENT, Menlo Park, CA) is placed into the parotid duct. The drug-eluting stent is seen emerging from the parotid duct. The stent is secured to surrounding oral mucosa with three 4-0 PDS sutures.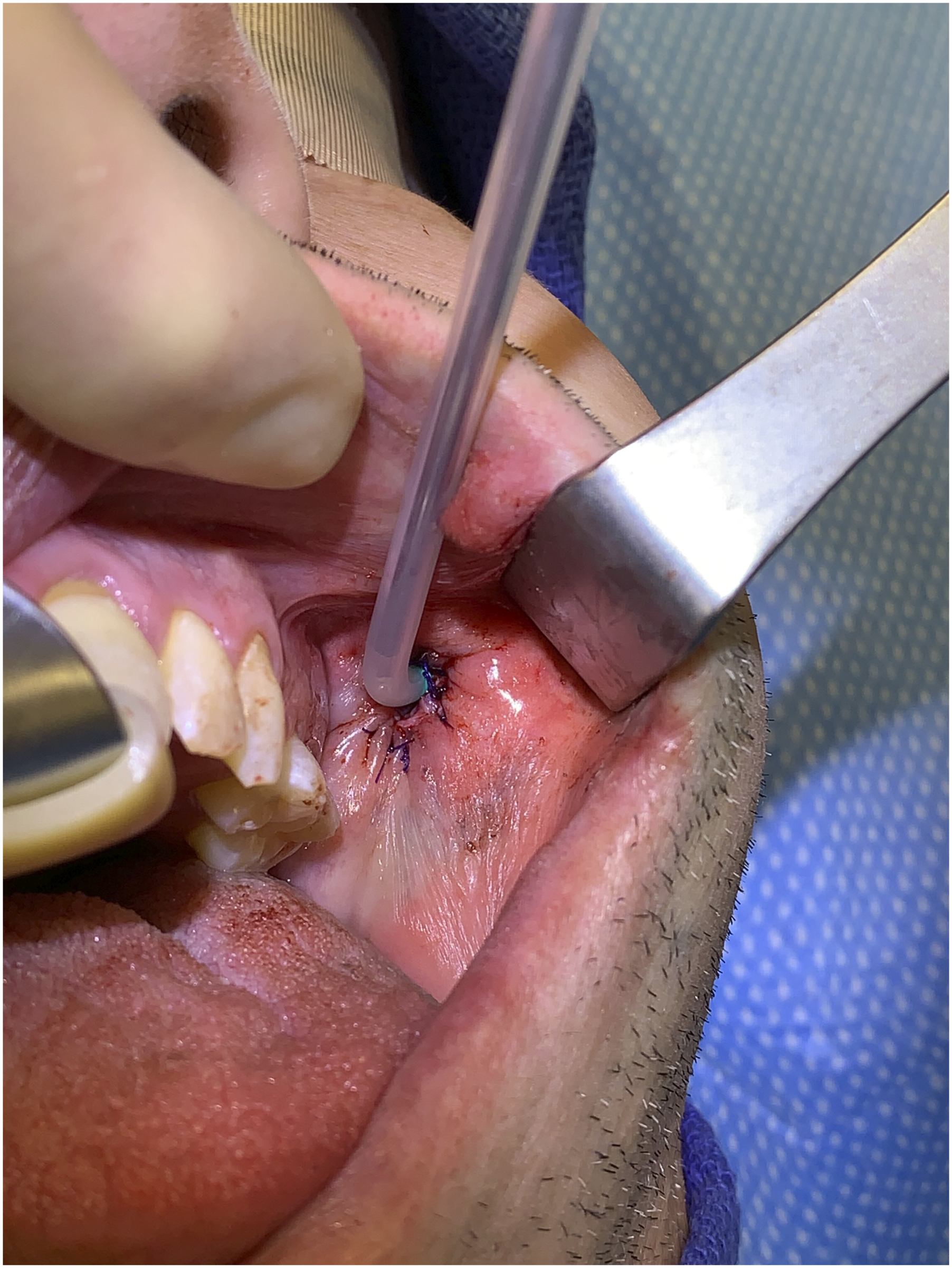
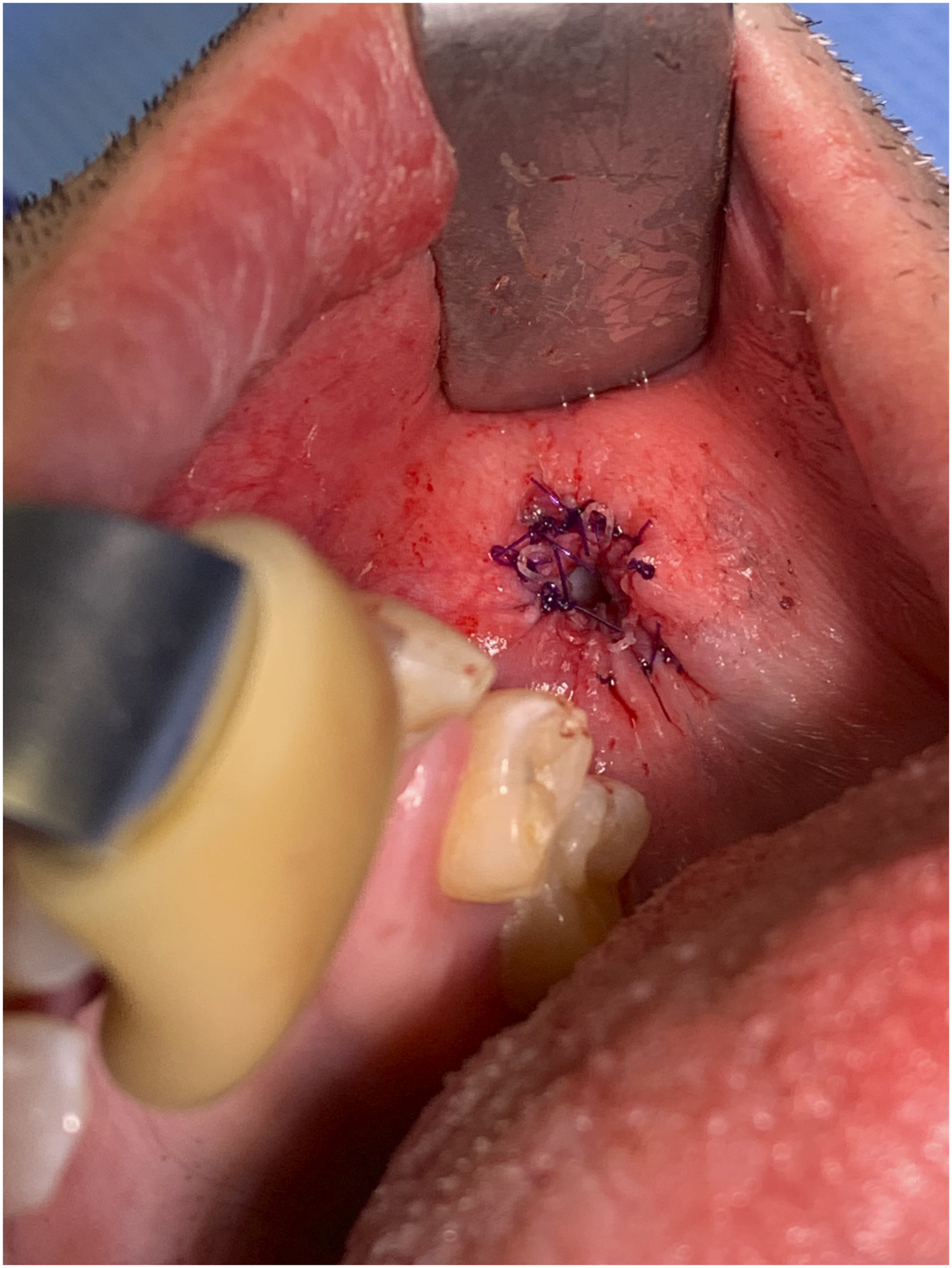
At 10 days post-operative follow-up, the parotid duct remained widely patent with the stent intact. The patient denied complications. At 5 weeks, the parotid duct remained widely patent. Residual filaments of the stent were removed in office. The patient denied complications or further episodes of swelling or pain since surgery.
Drug-eluting stents (DES) are currently approved by the United States Food and Drug Administration for use in the sinuses; intraoral use is off-label. In the sinuses, the stent delivers 370 µg of mometasone furoate over a period of approximately 30 days until dissolution. The dosing, along with the risks and benefits of DES in the sinus environment have been well-studied. 2 Importantly, there exist limited data on the viability or safety of DES in the oral environment. While DES in sinus surgery has an established half-life in vivo, continuous exposure to saliva, food, and liquids of varying acidity in the oral cavity may reduce the half-life of these implants. Mometasone furoate is hydrophobic, stable under aqueous and acidic conditions, and nearly insoluble in water, suggesting it would be suitable for local release in the oral cavity. The contraindications to the use of Propel stents include patients with a known intolerance of mometasone furoate or hypersensitivity to lactide, glycolide, or caprolactone copolymers. Safety has not been studied in pregnancy. Potential complications include displacing or swallowing parts of the implant, hypersensitivity reaction, or localized infection. Further investigation into the safety and pharmacologic parameters of DES in the oral environment is warranted.
The greatest limiting factor upon widespread adaptation of this technique is the current lack of DES designed for salivary ducts. Most normal salivary ducts are on the order of 2-3 mm in diameter, and the most commonly used sialoscopes are 0.9 to 1.6 mm in diameter. In their current configuration, these stents do not fit in normal or dilated salivary ducts and were only able to be used here due to the existing megaduct. There may be additional applications of DES for more narrow strictures and smaller ductal lumens. Based upon the initial success described, research and development are now needed. Innovation in this domain will primarily involve scaling down the stents to fit the relatively smaller ductal diameters along with development of a new stent deployment method specific to salivary applications.
This case represents the first reported use of a steroid DES for treatment of refractory salivary megaduct, establishing a new approach for treatment of this challenging condition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
