Abstract
Only 0.3–1% of laryngeal cancer are non-squamous cell neoplasms. Of these, a rare entity is inflammatory myofibroblastic tumour (IMT), in which anaplastic lymphoma kinase-1 (ALK-1) is frequently expressed. Just 50 cases of IMT have been reported. Therefore, many otolaryngologists may be unfamiliar with this type of tumour and be prone to its over- or undertreatment.
We report a case of ALK-1–negative IMT treated with transoral endoscopic excision and disease-free 6 months after surgery.
Inflammatory myofibroblastic tumour (IMT) is an uncommon neoplasm composed of spindle-shaped cells that have characteristics halfway between fibroblasts and smooth muscle cells and that are known to play an essential role in tissue and organ matrix remodelling in normal and pathologic conditions. 1
Previously, IMT was considered an abnormal inflammatory response and not a neoplastic lesion. However, some features indicate its neoplastic nature, such as its potential for local recurrence, local infiltrative growth, the development of non-contiguous multifocal tumours, vascular invasion and even distant metastasis.2,3 According to the 2020 ‘WHO Classification of Soft Tissue Tumours’, it is included within the category of ‘Fibroblastic/Myofibroblastic tumors’ of the ‘Intermediate (Rarely metastasising) type’. 4 This soft-tissue tumour has intermediate biologic potential; it is characterized histologically by myofibroblastic spindle cells on a background of inflammatory cells. The true causative factors leading to IMT are unknown. Potential links with smoking, trauma and viral/bacterial exposure have been suggested. 5 It is rare in the larynx where the most common involved site is the glottis.5-10 The most common symptoms are voice change (74%), stridor (29%) and dyspnoea, shortness of breath (22.5%) and globus sensation (16%). 6
Herein, we present a 62-year-old Caucasian male with 6 months of worsening dysphonia and hoarseness. He was healthy, denied smoking history and previous laryngeal surgery. We performed a flexible trans-nasal pharyngolaryngoscopy with narrow band imaging (NBI) which revealed a white tumour involving the right true vocal cord and the anterior commissure with minimal extension into the homolateral subglottic region (Figure 1A). Subsequently, we performed a transoral excision with cold steel instruments under microlaryngoscopy. Multiple firm, fleshy, loose tissue fragments were removed, the dimensions of which were approximately 23x18x6 millimetres (Figure 1B). Pre-operative picture of the IMT lesion in narrow band imaging nasoendoscopy (A). Macroscopic lesion after the excision (B).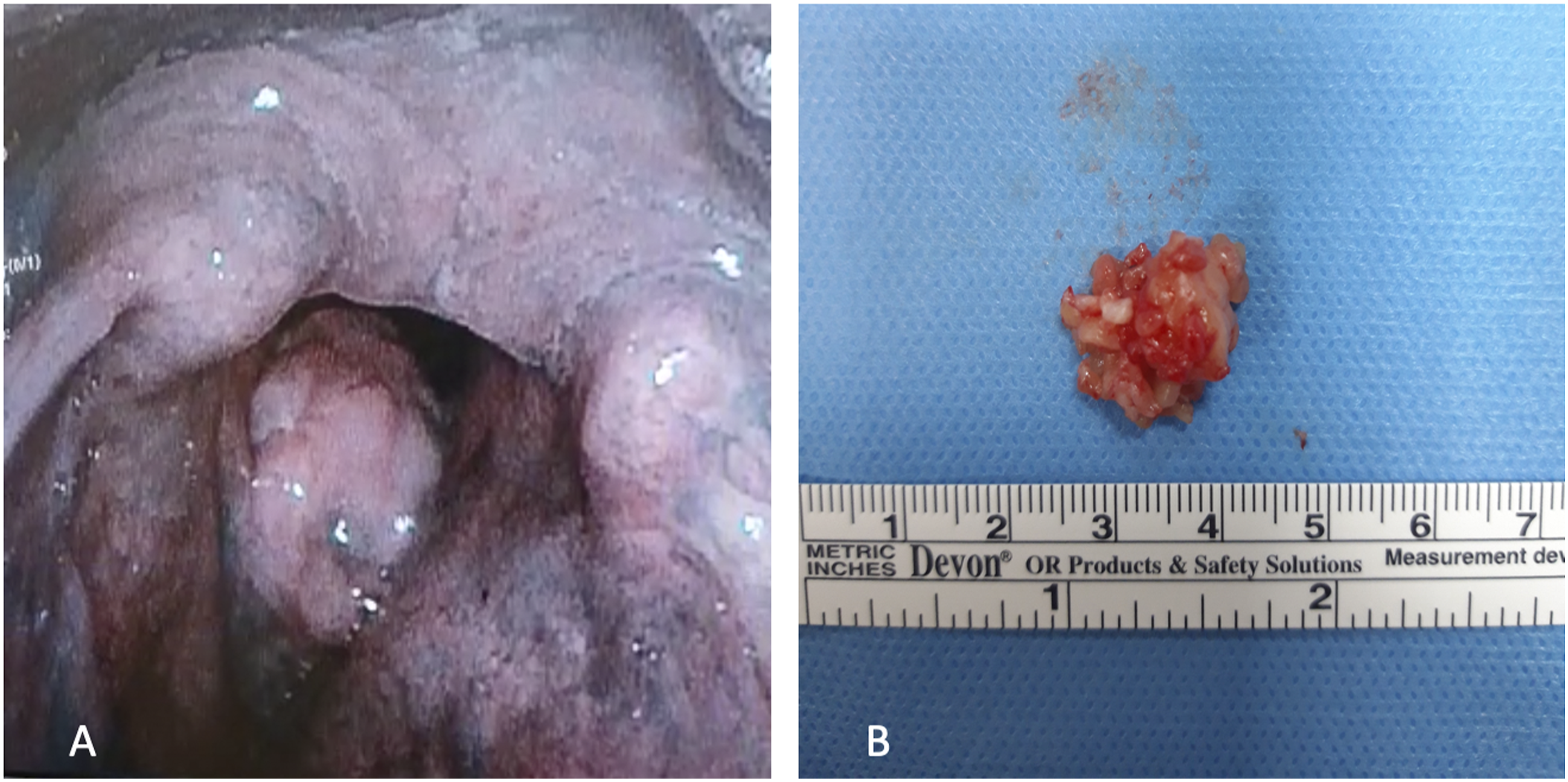
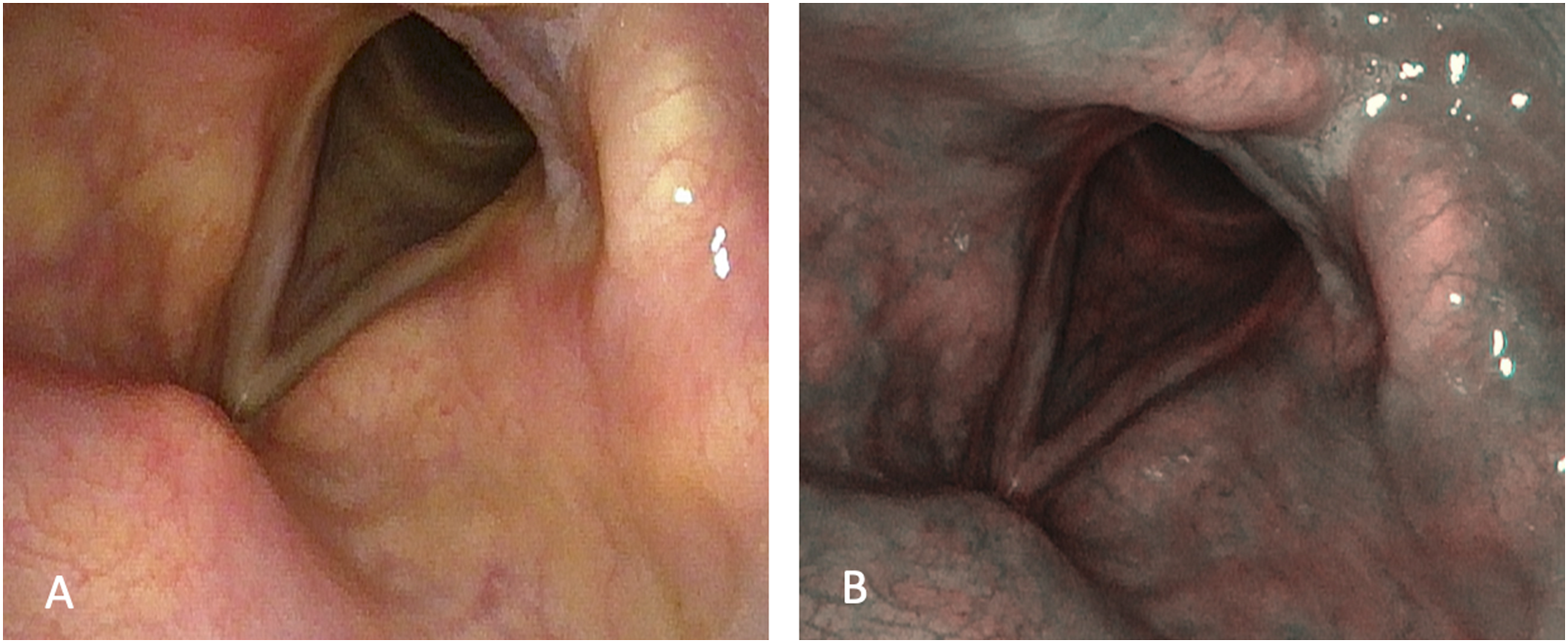
Histological examination revealed an ulcerated tumour consisting of spindle cells associated with a prominent admixture of inflammatory cells (Figure 2A and B). The spindle cells were arranged in variably oriented fascicles and showed elongated vesicular nuclei and ill-defined cytoplasmic borders (Figure 2B and C). Inflammatory cells included including neutrophils, small lymphocytes, histiocytes and plasma cells. Necrosis was absent. Immunohistochemical analysis, performed as described previously,7,11-16 showed immunoreactivity of the neoplastic cells for vimentin, smooth muscle actin (Figure 2D) and muscle-specific actin, but not for pan-cytokeratin, CD34, S100, desmin and anaplastic lymphoma kinase-1 (ALK-1, Figure 2E). The overall expression of Ki-67 was about 10%. The pathological findings were consistent with IMT. The patient did not receive adjuvant therapy. At follow-up, the patient was completely disease-free 6 months after surgery (Figure 3A-B). Low-power magnification of the lesion is illustrated in (A). It involves the sub-epithelial connective tissue (ep epithelium) and is composed of a mixture of spindle and inflammatory cells (B and C). At high-power magnification (C), the spindle cells show elongated vesicular nuclei and ill-defined cytoplasmic borders. Spindle cells are uniformly positive for smooth muscle actin (D) and negative for ALK-1 (D). A–C: Bars: 200 µm in A, 100 µm in B, D and E and 50 µm in C. A–C: hematoxylin and eosin. Fiberoptic trans-nasal flexible endoscopy of the larynx after 6 months from surgical excision (A) and narrow band imaging (B).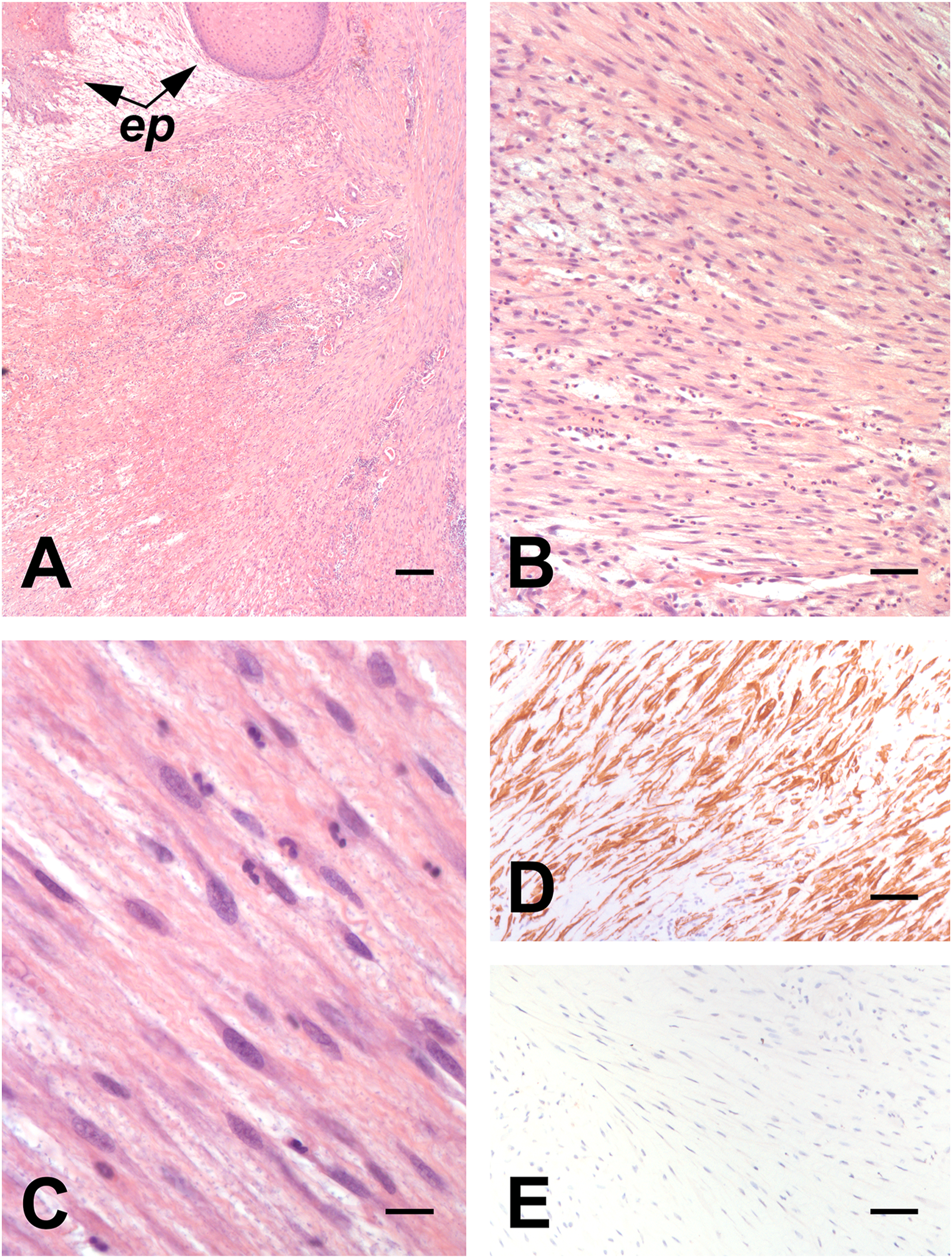
Recent data demonstrate ALK‐1 gene translocations, resulting in the overexpression of ALK-1 protein that can be detected by immunohistochemistry, in a very large part of IMTs.5,9,10 ALK‐1 immunoreactivity is relatively specific for IMT, and is frequent in laryngeal IMT.5,10 However, in our case, immunoreactivity for ALK-1 was not detected. In spite of this, the diagnosis of IMT was proposed based on the histological and immunohistochemical findings.
The first choice of treatment for a laryngeal IMT is complete surgical excision with laser or cold steel instruments. Microlaryngoscopy or endoscopy is preferred over an open procedure, which is reserved for cases of recurrence, poor visualization or when malignancy cannot be ruled out. If the tumour is completely removed, the risk of recurrence and distant metastasis is low.2-10,17 In partially excised ALK-1–positive IMT, tyrosine kinase inhibitor can be used.10,18 Indeed, remission (partial or complete) has been reported with crizotinib after partial tumour resection. 18 Interestingly, crizotinib therapy resulted in remission of the tumour also in the absence of previous surgery. 18 However, as already noted,5,18 further studies are needed because these data ‘are limited by publication bias, complex treatment regimens, and variable follow‐up time’.
The main problem facing IMT in the larynx is the risk of overtreatment with the first-line use of open procedure (such as total laryngectomy), which should be reserved only for disease recurrence after endoscopic resection, and regarding the need for close follow-up due to the risk of local recurrence.
In conclusion, we reported here the clinico-pathologic findings of an uncommon tumour, the IMT, in an unusual site, the glottic larynx.
Footnotes
Authors’ Note
The data sets used and/or analysed during the current study are available from the corresponding author on reasonable request. All the clinic-pathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards. Written informed consent for publication of data and images was obtained from the patient.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
