Abstract
Spontaneous regression of a neoplasm is a rare oncologic phenomenon. Certain neoplasms, such as melanomas and neuroblastomas, display this phenomenon. To date, spontaneous regression of oral cavity squamous cell carcinomas has been documented in only a handful of case reports. We present a novel case of spontaneous regression of an oral tongue squamous cell carcinoma following biopsy. We discuss the tumor’s unique genetic profile, immune response to cancer, and review the literature on possible mechanisms of spontaneous regression. Small-volume persistent cancer in our patient reinforces that tissue confirmation remains crucial to avoid missing remaining tumor. Further investigation is required to understand mechanisms of spontaneous regression and how these may be exploited to improve head and neck squamous cell carcinoma treatment.
Keywords
Introduction
Spontaneous regression is a rare but well-documented phenomenon in which malignant tumors disappear partially or completely without treatment, with no apparent cause. 1 It is not necessarily a complete absence of tumor, but rather a notable reduction in tumor burden. The frequency of this event is difficult to determine due to unspecified criteria for spontaneous regression and likelihood of many unreported cases. Spontaneous tumor regression has been observed in a variety of tumors, most commonly malignant melanoma, hypernephroma, lymphoma, leukemia, and neuroblastoma. 1
Spontaneous regression of head and neck squamous cell carcinoma (HNSCC) is exceedingly rare, with a rate of 3:504 in one review. 1 To our knowledge, there have been 4 prior case reports on spontaneous regression of oral cavity SCC, including regression of recurrent tumors and lymph node metastases.2-5 As such, there has only been one report on spontaneous regression of primary oral cavity SCC. 2 Here, we present the novel case of a patient with primary SCC of the right tongue whose tumor spontaneously regressed in one month without any treatment. Our report is the first to include tumor genomic profiling. We also review the literature on spontaneous regression of HNSCC and possible mechanisms to explain this rare but significant event.
Case Report
A 77-year-old woman was seen by the Albany Medical Center Otolaryngology department in August 2020. She complained of a 70-pound weight loss and worsening right tongue pain with increased dysphagia over the last two weeks. She was bothered by oral tongue irritation for several months, although it had not previously affected her diet. She had been a lifelong non-smoker, rarely drank alcohol, and her past medical history included active Meniere’s disease and trigeminal neuralgia. Oral examination revealed a 2x3 cm endophytic tumor on her right tongue. We performed a punch biopsy at several locations in the right anterolateral tongue, which revealed invasive, poorly differentiated squamous cell carcinoma with predominantly lymphocytic inflammatory infiltrate. Further evaluation including an enhanced computed tomography (CT) of the neck with contrast demonstrated extensive streak artifact from dental amalgam and no definite cervical lymphadenopathy by size criteria.
The patient was diagnosed with T3N0 SCC of the right oral tongue. She had a positron emission tomography (PET) scan that showed focal asymmetric uptake at the right lateral tongue, consistent with carcinoma. In the right neck, a small lymph node in level III was present and likely consistent with metastatic disease given a maximum standardized uptake value(SUVmax) of 5.1. In the left neck, there was a small level II lymph node and a small level III lymph node with SUVmax of 3.8 and 3.1, respectively. Each of these lymph nodes could not be differentiated as reactive or representative of metastatic disease. The surgical treatment plan consisted of a right partial glossectomy with primary closure or free flap reconstruction, and bilateral neck dissection. The likelihood of adjuvant therapy was discussed.
Approximately two weeks after the PET scan, the patient arrived for surgery. Shockingly, the tongue mass had almost completely regressed. Further discussion with the patient revealed that she was feeling well and could tolerate a regular diet. She only noticed a very small area of tenderness in the right lateral tongue, consistent with a small 5x5 mm nodularity on palpation. Surgery was canceled to allow for further evaluation. 6 weeks following her initial PET scan, an ultrasound of the neck was ordered for interval evaluation, which revealed that multiple small (< 1 cm) bilateral lymph nodes were present with normal appearing tissue; the largest lymph node was 1.2x0.6x0.9 cm in level I of the right neck. FoundationOne genomic testing was ordered, which demonstrated that the tumor was microsatellite stable with 0 mutational burden as well as TERT 124 (C > T) and TP53 818 (G > A) mutations. The TERT and TP53 variant allele frequencies were 21.6% and 16.9%, respectively. In part, the testing was ordered to verify that the regressed tumor had mutations consistent with malignancy.
One month later, the tongue had a stable 4-mm deep pocket with some thickened tissue, but no tumor was visible. Multidisciplinary tumor board conversation recommended excising the remnant oral scar to rule out small persistent disease. The patient had a right partial glossectomy that was completely excised to negative margins (T1N0 SCC). Pathology demonstrated marked tumor regression with only a minute focus of residual invasive SCC with surrounding chronic inflammatory cells and tumor apoptotic figures (Figure 1). Partial glossectomy demonstrating marked tumor regression without prior treatment. (A) The right-sided partial glossectomy specimen shows a largely regressed tumor with only a minute focus of residual invasive squamous carcinoma (irregular scattered nests of atypical squamous cells in the lamina propria) spanning 2 mm in greatest dimension with a depth of invasion of 0.8 mm (H&E, 40x). (B) There are lymphocyte predominant chronic inflammatory cells throughout the adjacent lamina propria, tumor cell apoptotic figures, and intraepithelial lymphocytes (H&E, 100x).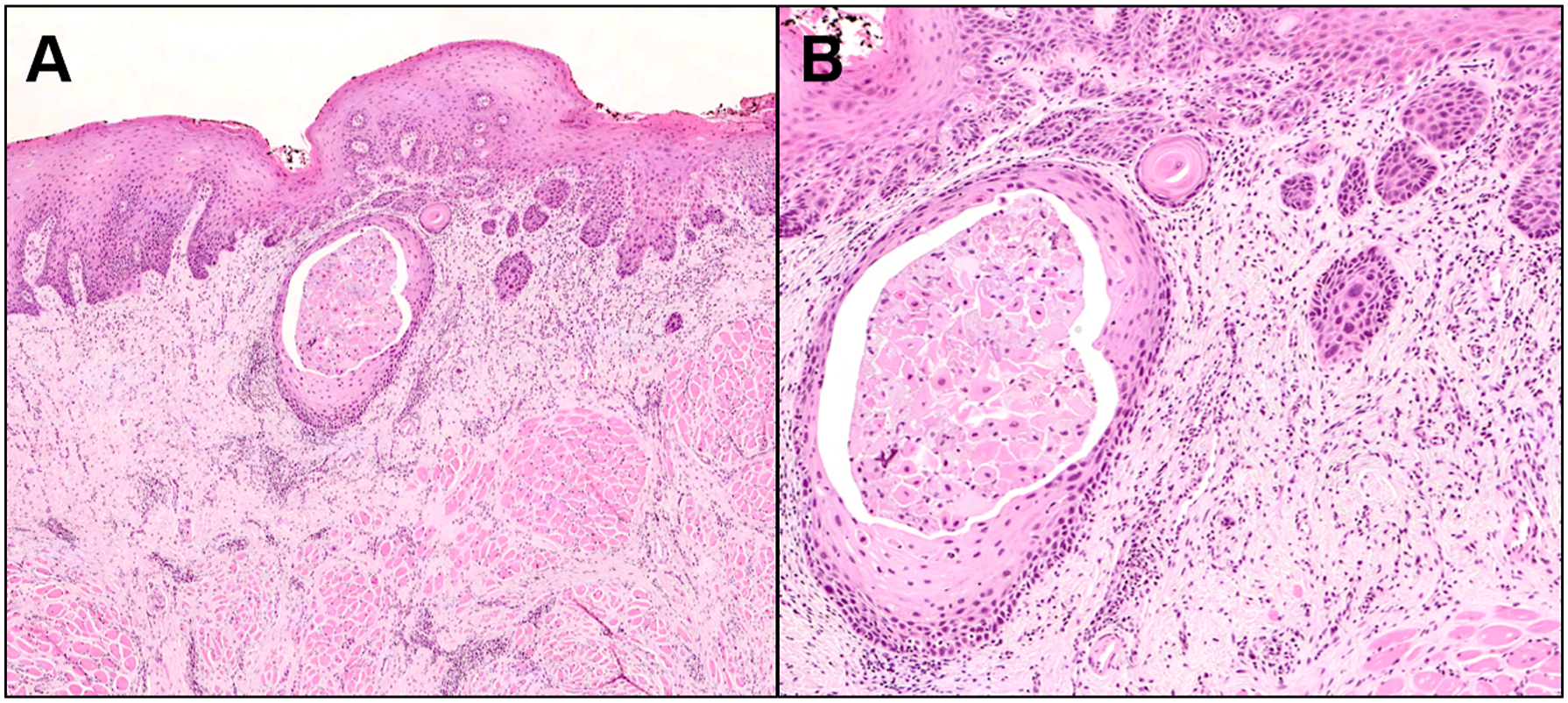
The patient healed well after right partial glossectomy. Due to only a partial regression in the oral cavity, the initially suspicious cervical lymphadenopathy was reinvestigated. Repeat CT scan was correlated with the previous PET scan and confirmed that the metabolically active right lymph node was now measuring 5x4 mm. Out of an abundance of caution, a right neck dissection (levels I, II, and III) was performed. A 1.4-cm level II lymph node with an internal 0.6-cm focus of moderately differentiated non-keratinizing SCC with intratumoral inflammatory cells but without extranodal extension was removed (Figure 2). Our patient’s final diagnosis was T1N1 right oral cavity SCC. About 1 year after her partial glossectomy and 9 months after her neck dissection, the patient is doing very well, with no persistent disease clinically or on 6-month post neck dissection CT imaging. She received no adjuvant therapy. She is eating a regular diet and denies any unintentional weight loss. Lymph node metastasis with intratumoral inflammatory cells. (A) Lateral neck dissection specimen at levels 2 and 3 shows the presence of a metastatic moderately differentiated non-keratinizing squamous carcinoma (black arrows) that is 0.6 cm in greatest dimension and without extranodal extension (H&E, 20x). (B) There are frequent intratumoral inflammatory cells comprising mostly lymphocytes and a few eosinophils (H&E, 100x).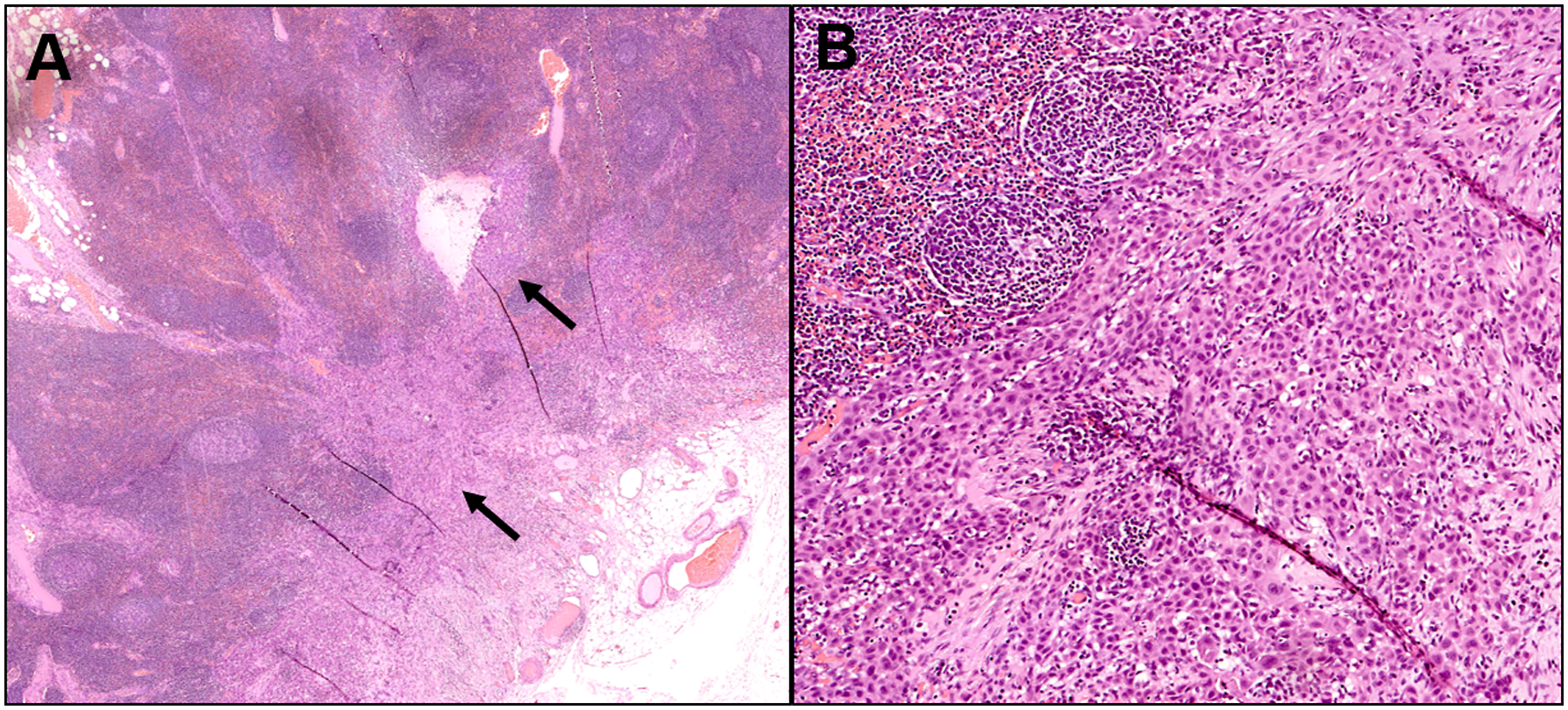
Discussion
Spontaneous regression is an extremely rare phenomenon in HNSCC. Both comprehensive otolaryngologists and head and neck oncologic surgeons may face questions from patients regarding non-treatment of cancer. Knowledge about the natural history of tumor progression is difficult to find in the literature. One retrospective review of 808 patients with untreated HNSCC found no cases of spontaneous regression, with about 50% of patients dying within 4 months of diagnosis. 6 A larger review of 6477 patients with untreated HNSCC also reported no cases of spontaneous regression. 7 Oya et al. reported spontaneous regression of recurrent tongue carcinoma, 3 and cases of regression of lymph node metastases have also been described.4,5 In 2014, spontaneous regression of primary SCC of the floor of the mouth was reported, with regression within 3 months and no signs of recurrence at 70 months. 2 While the mechanism(s) of spontaneous tumor regression remain unknown, several theories have been proposed including operative trauma, infection, immunomodulation, hormonal changes, induction of apoptosis, tumor inhibition by growth factors and cytokines, inhibition of angiogenesis, and tumor necrosis.1,8
One primary hypothesis for the mechanism of tumor regression is immune modulation, which may be triggered by local trauma or infection. Operative trauma from biopsies and surgery on the primary tumor or metastases may induce host immune response to the remaining tumor burden, with several reports of such cases. 1 In a review of 176 cases of spontaneous regression, 40% were associated with operative trauma. 9 The immune system’s role in fighting cancer has led to efforts to develop cancer vaccines and other immunotherapies that induce host response, primarily via cytotoxic CD8+ T cells that are specific to tumor antigens. 10 Butterfield theorizes that traditional treatments, including surgery, tumor ablation, chemotherapy, and radiation, may induce antitumor immunity by increasing antigen expression, acting as generalized “cancer vaccines.” 10
Acute infections may also trigger an immune response to cancer given that infection can induce fever and a local inflammatory response. There are several reported cases of spontaneous tumor regression in the setting of infections including influenza, diphtheria, measles, malaria, syphilis, and tuberculosis.11,12 Fever has been reported in 25–80% of documented spontaneous tumor regression cases, which can be triggered by bacterial, viral, fungal, and protozoal pathogens. 12 Thus, acquired or induced infection could cause a pyrogenic immune response leading to tumor regression.
Aside from immunomodulation, other less common mechanisms of spontaneous regression have been hypothesized. Necrosis and vascular insufficiency have been reported as causes of tumor regression. 1 Operative trauma may disrupt tumor vasculature in addition to causing an immune response. 1 Spontaneous regression likely involves a variety of mechanisms, with apoptosis (programmed cell death) playing a central role as the endpoint of most pathways.5,13
The role of tumor genomics in spontaneous regression has yet to be characterized. Microsatellite instability is caused by defective DNA mismatch repair and predisposes tumor cells to mutation. Microsatellite stable tumors like our patient’s are significantly less likely to respond to immunotherapy (PD-1 checkpoint receptor targeting agents) compared to tumors with microsatellite instability. 14 Similarly, tumors with low mutational burden are less sensitive to immunotherapies across cancer types, 15 while high tumor mutational burden has been associated with improved outcomes in HNSCC. 16 Only one case series has explored microsatellite instability as a potential cause of spontaneous regression, reporting 3 cases of spontaneous regression of microsatellite unstable colorectal cancers. 17 There has been one reported case of spontaneous regression of hepatocellular carcinoma with confirmed high tumor mutational burden. 18 Our patient’s tumor mutational burden of 0 is unusual given that HNSCC have an average tumor mutational burden of 5, with 10% of cases demonstrating greater than 20 mutations per megabase. 19 Microsatellite instability and high mutational burden may induce an antitumor immune response; thus, it is perhaps counterintuitive that our patient’s microsatellite stable tumor with no mutational burden would spontaneously regress.
Further profiling of our patient’s tumor yielded TERT and TP53 mutations, which have been associated with antitumor activity. TERT promotor mutations have been observed in 17% of HNSCC, and most frequently in oral tongue SCC. 20 hTERT-positive tumor cells have been observed to induce cytotoxic T lymphocyte and T helper response, 21 with evidence of tumor regression in some studies. 22 TP53 mutations are more frequently reported in HNSCC than TERT mutations, with rates of 38–58%.23,24 While TP53 mutations have been associated with lower recurrence-free survival, 23 some studies suggest that they may confer antitumor activity. The relationship between TP53 and antitumor immunity is complex, with variations by cancer type. 25 Of note, this patient’s tumor genomic profiling yielded mutations in three additional genes [NBN (L421S), NOTCH2 (L2408H), and SPEN (V3191M)]. While mutations in the family of NOTCH genes have been identified in HNSCC, 26 the relevance of these genetic variants for this case is not immediately apparent.
Given the rarity of spontaneous tumor regression, we hope this report gives general practitioners a framework for approaching such cases. Specifically, some consideration for the genetic heterogeneity of HNSCC as well as the high index of suspicion that a “disappearing tumor” may simply represent a regression and that cancer that requires treatment is still present. Ultimately, it seems likely that this patient’s spontaneous regression of tongue SCC is better explained by antitumor immunogenicity due to biopsy rather than her tumor genomic profile.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
