Abstract
Surgical repair of esophageal perforation is a challenging procedure with a high risk of secondary complications, such as early esophageal leakage and late esophageal stricture, which can significantly reduce the patient's quality of life. A 34-year-old man underwent anterior cervical corpectomy decompression and fusion. On the ninth day post-operation, the patient developed fever and neck swelling. A computed tomography scan of the neck showed multiple subcutaneous pneumatosis. An esophageal perforation of approximately 1.5 cm in diameter was identified by esophagoscopy. During the operation, the fistula was first located using an esophagoscope. The distal end of the esophagoscope was then placed into the stomach to support the damaged segment of the esophagus. The esophageal mucosa was sutured under the microscope, and the perforation was successfully repaired. Postoperatively, the patient's body temperature decreased, and the infection indexes gradually returned to normal. Three months after the operation, the esophagoscopic review showed complete healing of the perforation. Esophagoscopy plays an important role in diagnosing and repairing esophageal perforations. The esophagoscope provides direct visualization of the perforation during diagnosis and detects smaller and not yet fully penetrated esophageal injuries. During the repair process, the esophagoscope immobilizes the esophagus, prevents its movement and facilitates suturing, maintains proper dilatation of the esophagus, provides space for suturing, and prevents esophageal stricture.
Introduction
Anterior vertebral resection and fusion have become effective methods for treating various diseases, including cervical degenerative disease, posterior longitudinal ligament ossification, trauma, and tumors. Esophageal perforation is a rare complication of anterior cervical spine surgery. 1 According to reports, the incidence of esophageal perforation is low, ranging from 0.04 to 0.25%,2-4 but it has a mortality rate as high as 20%.5,6
Surgical repair of esophageal perforation is extremely challenging due to several reasons. The first is the difficulty in locating the fistula; the second is the movement of the esophagus during the suturing process, which is not conducive to suturing; and the third is severe edema of the esophageal muscular layer and erosion by gastric acid, which requires finding healthy tissue for suturing.
Primary repair also carries a high risk of secondary complications such as esophageal leakage and stricture, leading to reduced quality of life and high mortality rates. 7 Gerald et al. 8 reported a 54% incidence of esophageal strictures after esophageal perforation repair, requiring multiple (3–9) postoperative endoscopic surgical dilations. Tanaka 9 reported that anastomotic stricture occurs in 13.6% of patients with esophageal repair after surgery. Iannettoni 10 reported long-term outcomes in 42 patients with esophageal perforation. Forty percent of patients with first-stage repair required postoperative dilatation due to stenotic dysphagia. Esophageal anastomotic leak is a potentially life-threatening complication of esophageal repair. In a significant number of cases, postoperative esophageal anastomotic leakage has proven fatal and the risk should not be underestimated. 11 Wang et al. reported leakage rates from 25 to 50% following the initial repair of esophageal perforations.12,13 The question of how to minimize the incidence of leakage and stricture after esophageal repair through intraoperative techniques is an urgent issue that needs to be addressed.
The combined use of an esophagoscope and microscope may reduce the procedure's difficulty and reduce the likelihood of secondary esophageal strictures and leaks after surgery. Microscope-assisted surgery with a clear surgical field facilitates the distinction between healthy and unhealthy tissues, facilitates fine and tight suturing, can maintain uniform stitch spacing, flatten the anastomosis, avoid poor blood flow, and prevent leakage at the repair site. The esophagoscope fixes the esophagus and prevents movement during suturing, which facilitates suturing and maintains the proper dilatation of the damaged segment of the esophagus, providing room for manipulation, and avoiding late stricture of the esophagus.
This article reviews a case of esophageal perforation following cervical spine surgery, which was successfully repaired surgically with microscopy and esophagoscopy and without postoperative esophageal stricture or leakage.
Case Presentation
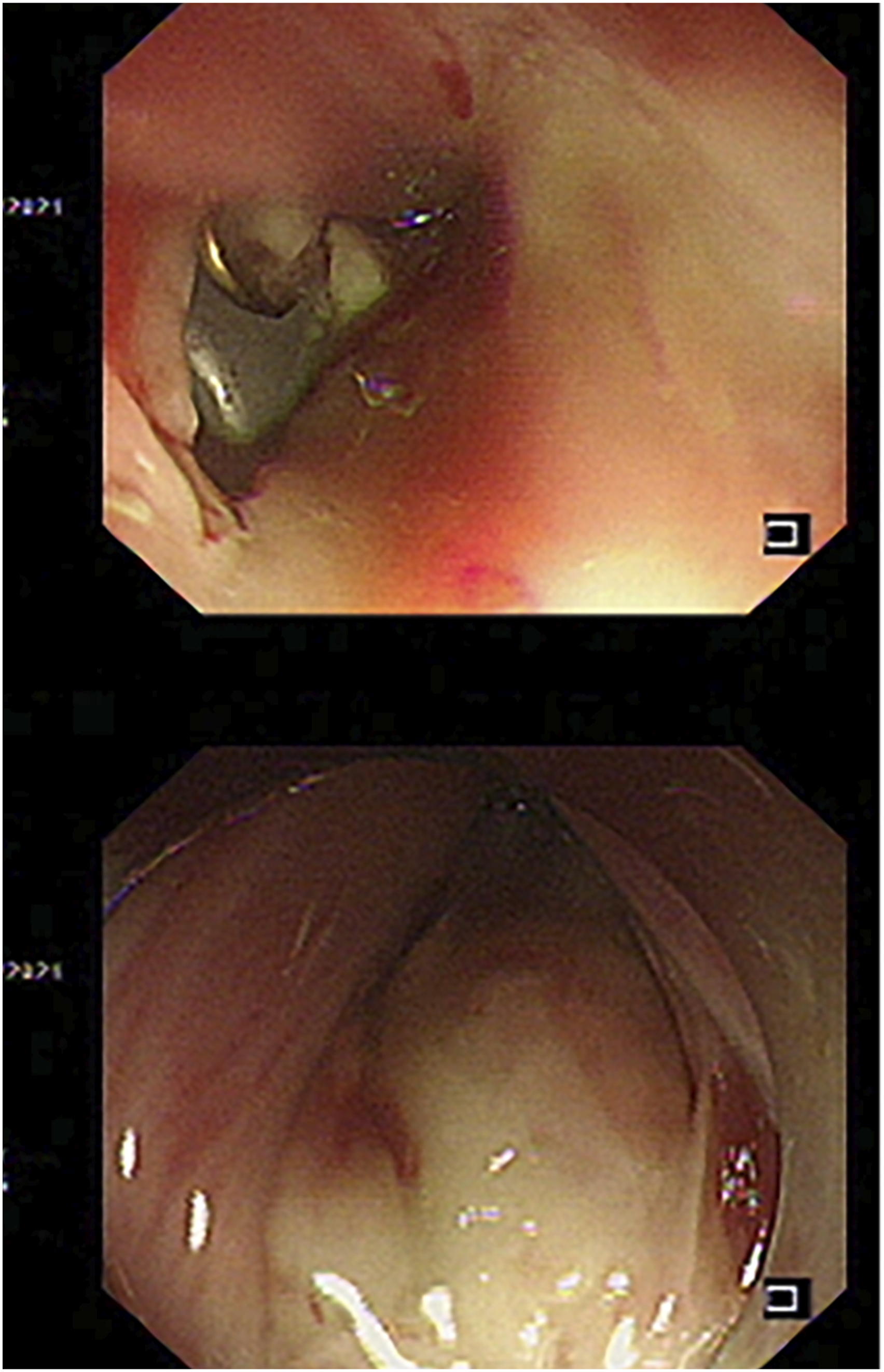
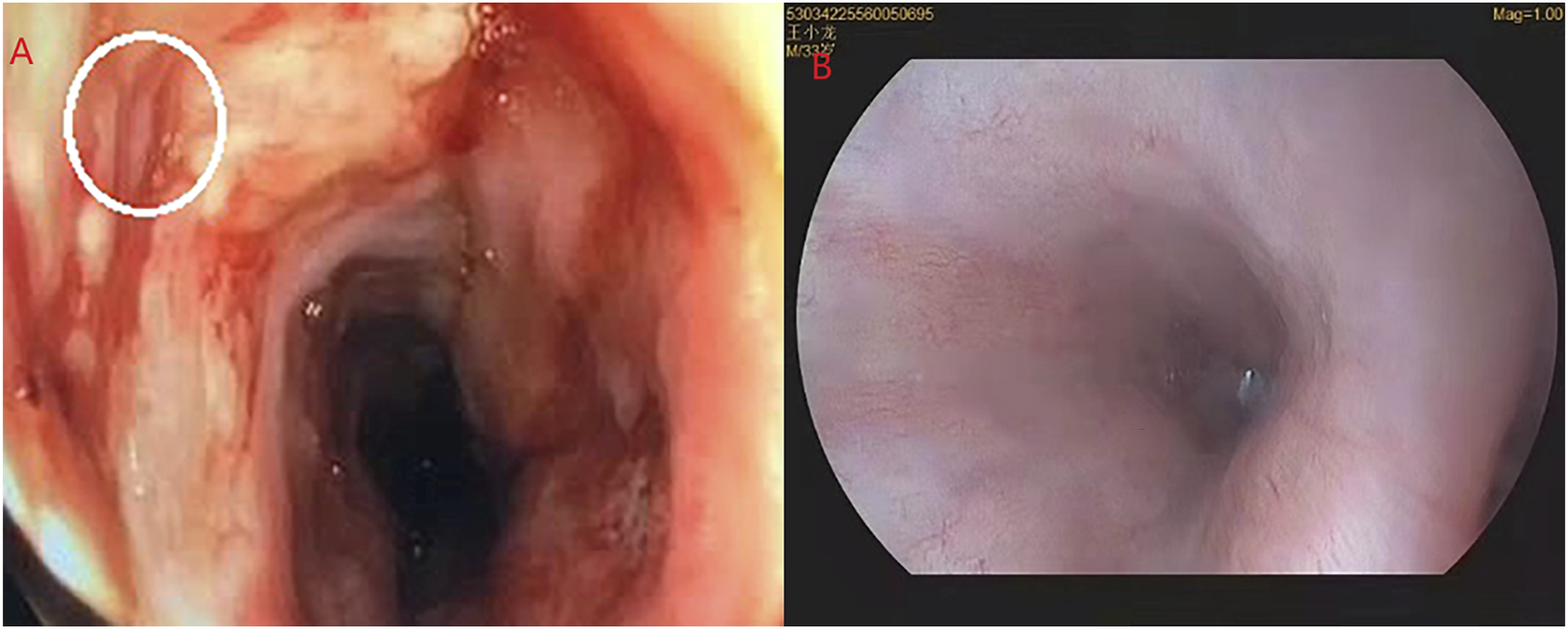
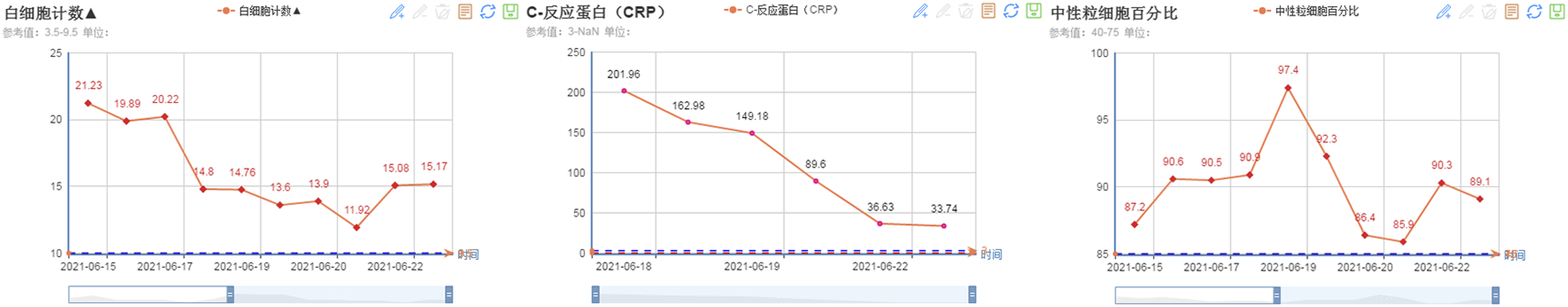
A 34-year-old male underwent anterior cervical corpectomy decompression and fusion for cervical 7 vertebral compression burst fracture with spinal cord injury and paraplegia. The patient developed local swelling and fever on the right side of the neck incision nine days postoperative. Computed tomography scan data of the neck suggested fluid accumulation in the subcutaneous soft tissue at the cervical 6-thoracic 1 vertebral body (Figure 1), suspected esophageal perforation. A “cervical spine postoperative wound exploration + indwelling irrigation and drainage” was carried out urgently. Further esophagoscopy was performed that afternoon and revealed an esophageal perforation with a diameter of approximately 1.5 cm at the entrance of the esophagus, 15 cm from the incisors (Figure 2). A gastrostomy was performed with the assistance of gastroscopy, and a jejunal nutrition tube was placed. Later that day, the patient underwent esophageal repair surgery. Cervical 6-thoracic 1 vertebral body level anterior subcutaneous soft tissue accumulation of fluid and gas. The esophagoscopy showed a small perforation with a diameter of 1.5 cm.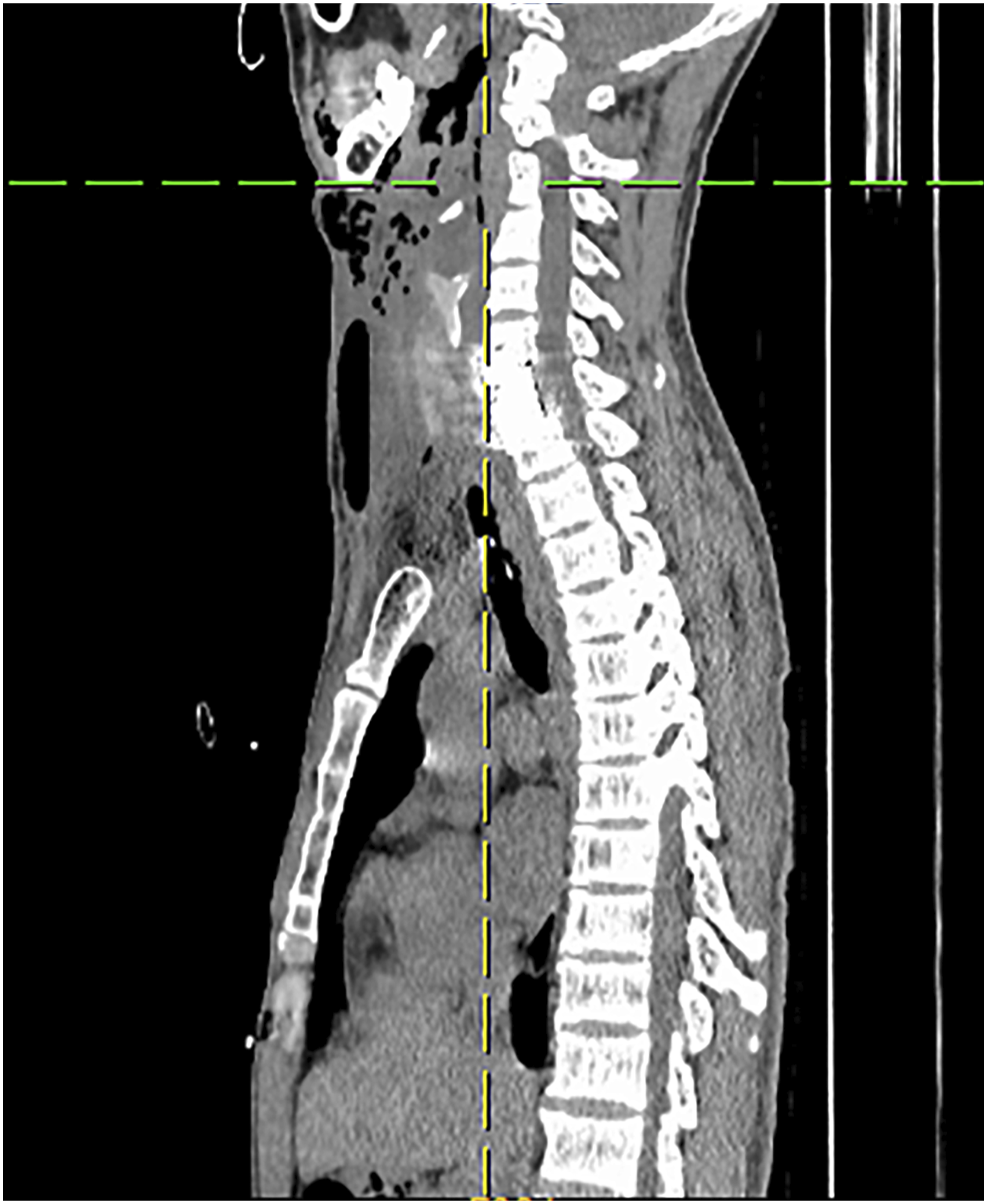
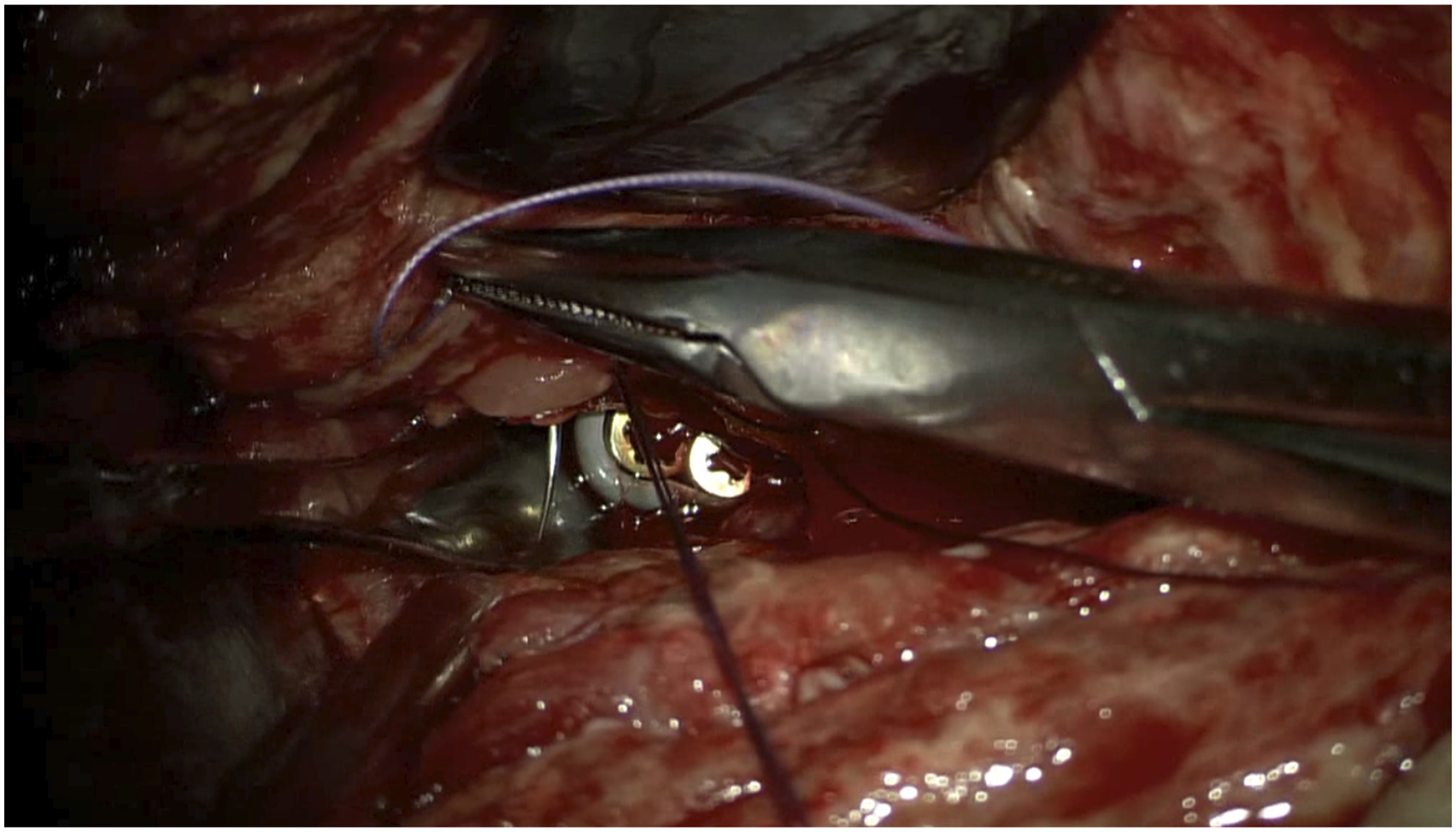
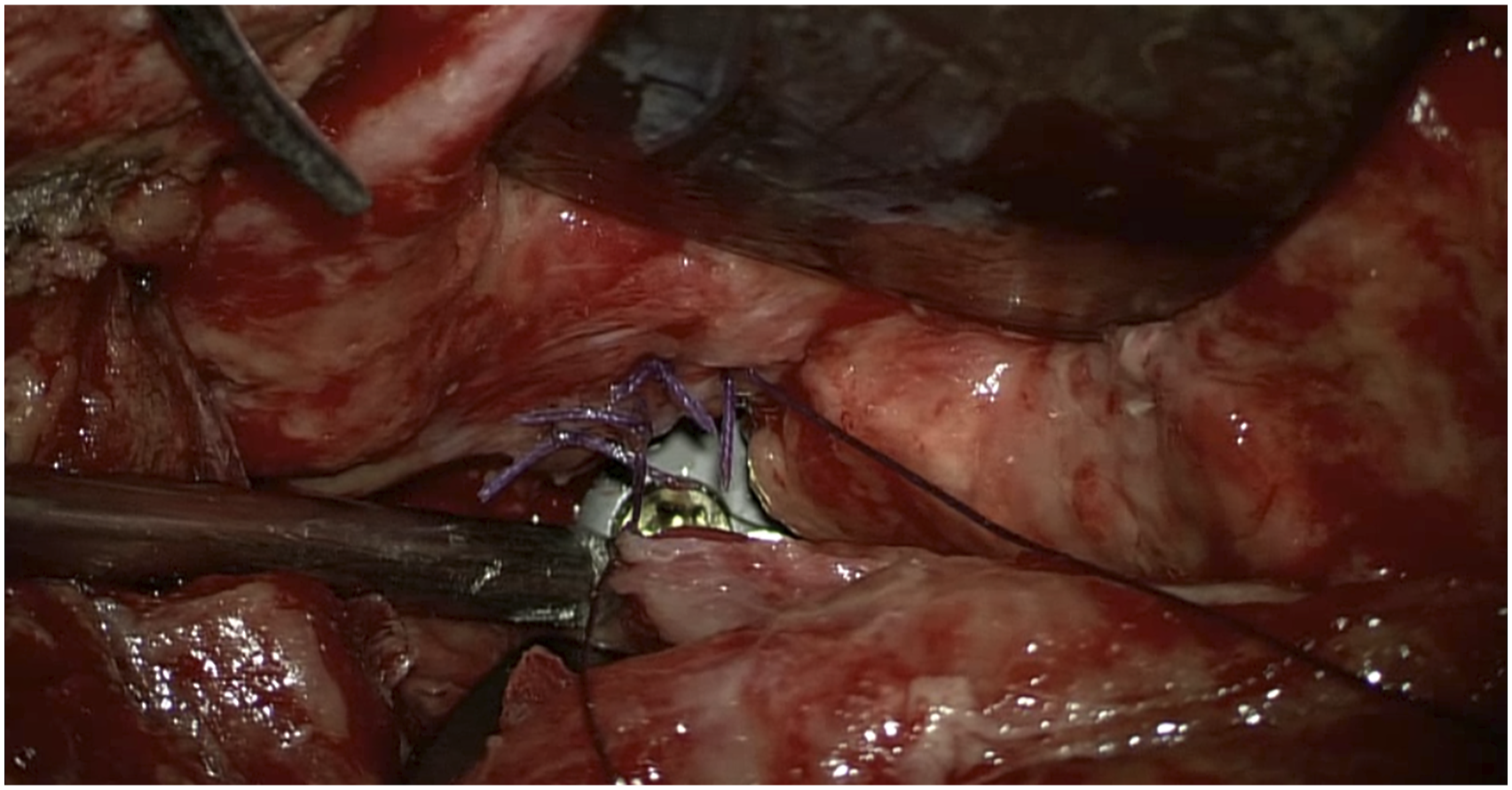
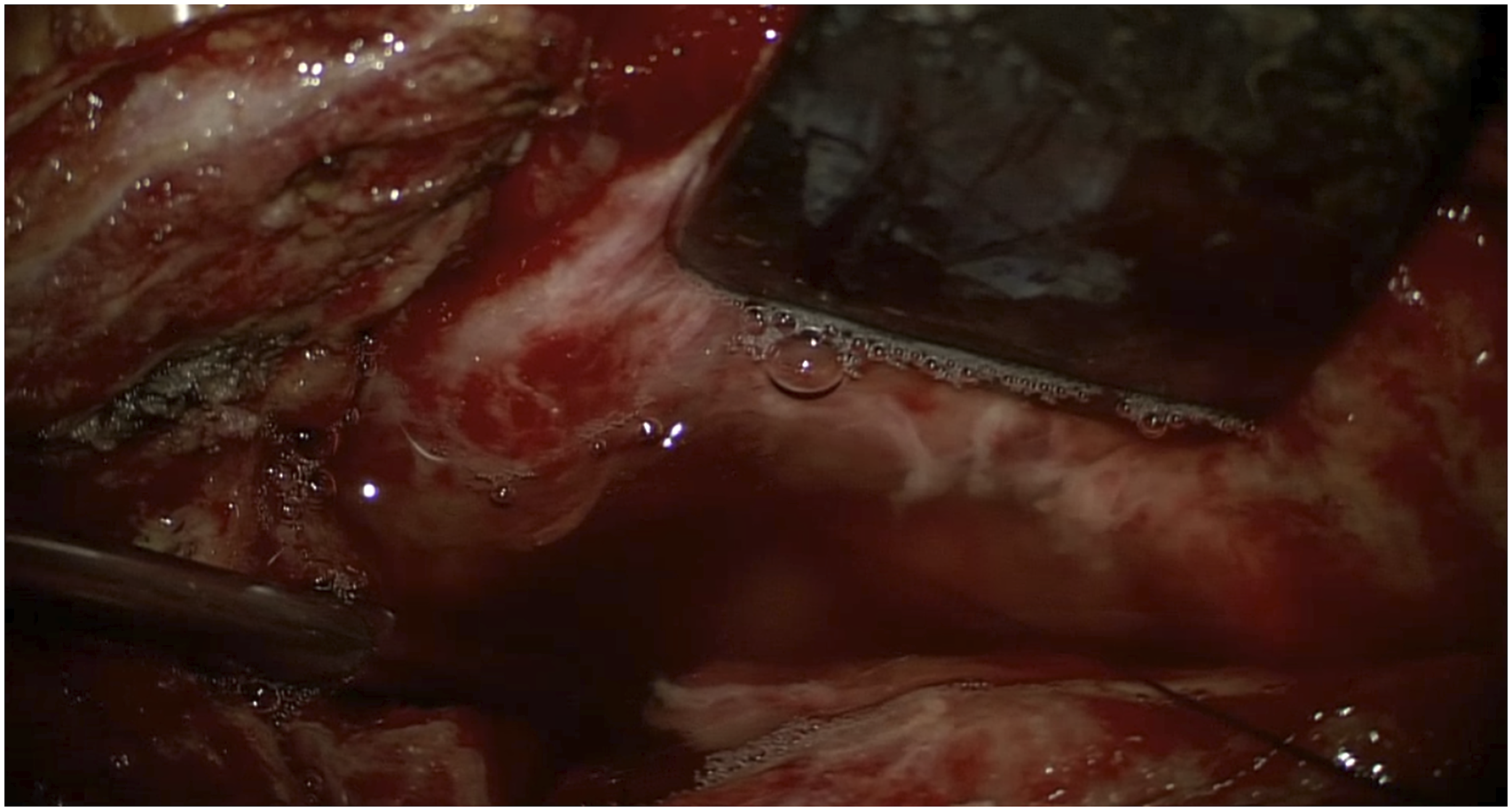
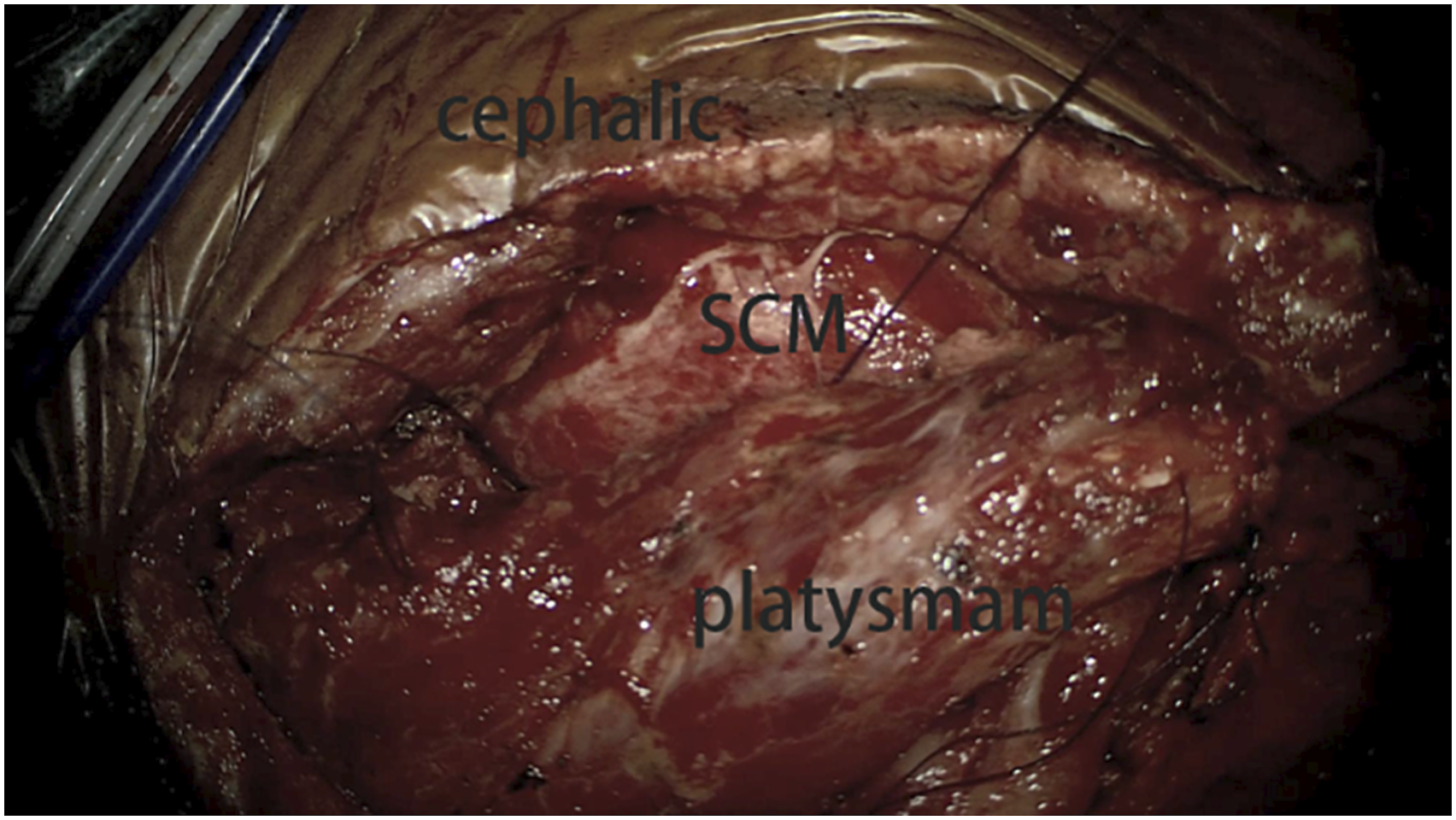
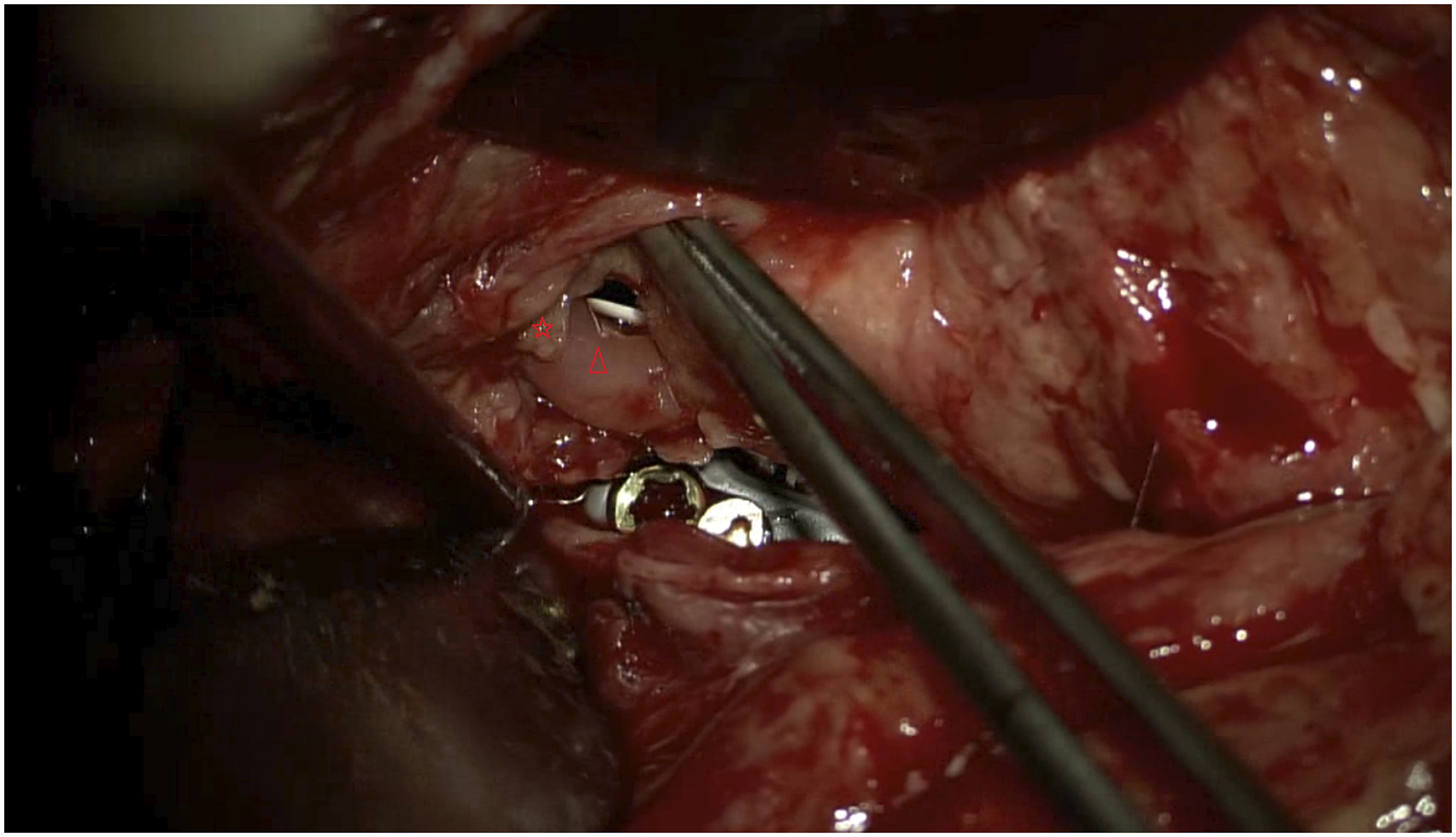
During the operation, the patient was placed in the supine position. The surgical repair began with a skin incision along the lesion site of the previous surgical scar, a broad under-neck incision along the front edge of the sternocleidomastoid muscle (SCM), and blunt dissection using a standard spinal approach. The perforation was explored, and a drain was inserted through the esophageal perforation to the surgical area with the assistance of an esophagoscope (Figure 3). With the help of this drain, the esophageal lesion could be easily localized. The distal end of the esophagoscope was then placed in the stomach to fix the esophagus and facilitate subsequent repair. Later, with the aid of a microscope, the posterior wall of the esophagus was peeled off correctly so that the left- and right-wound edges could be closed and sutured. The esophagoscope maintained proper dilatation of the damaged esophagus segment. The mucosa of the esophageal perforation was sutured with approximately four absorbable sutures (Figure 4), and the perforation was sutured in position (Figure 5). The esophagoscopy showed that the esophagus was sutured entirely, with no stenosis (Figure 6A). No air bubbles emerged or were observed following water injection into the operation area and inflation of the esophagus (Figure 7). The absence of perforation was then reconfirmed. The SCM was separated, keeping the pedicle at the bottom. The upper end of the SCM was disconnected and then trimmed to protect the muscle membrane. The prepared SCM flap was then placed between the esophageal perforation and the cervical spine and fixed. The muscle flap entirely covered the injured posterior wall of the esophagus (Figure 8). A vacuum sealing drainage tube was placed on the neck, and the neck incision was sutured in layers. Jejunal nutrition tube feeding was continued postoperatively, as was symptomatic anti-inflammatory treatment. Postoperatively, the patient's temperature decreased, and the infection indicators gradually returned to normal (Figure 9). The vacuum sealing drainage tube was removed on the eighth postoperative day. One month after surgery, the patient started to eat via the mouth. Postoperative esophagoscopic review at three months showed complete healing of the perforation and no strictures (Figure 6B). Intraoperative esophagoscope positioning. After inserting the catheter through the esophagus, the operator can quickly locate the esophageal lesions by touching the catheter Intraoperative suturing of the mucosal layer. Absorbable sutures were used to close the esophageal mucosa at the perforation site with about four stitches, in basic alignment. Postoperative esophagoscopy: (A) Immediately. (B) Two months after surgery. No air bubbles were seen after the perforation was sutured and inflated into the esophagus. The SCM flap is located between the anterior vertebral plane and the esophagus to protect the esophageal wall. The front edge of the SCM flap is sutured and fixed with the platysma muscle. Changes in postoperative infection indicators.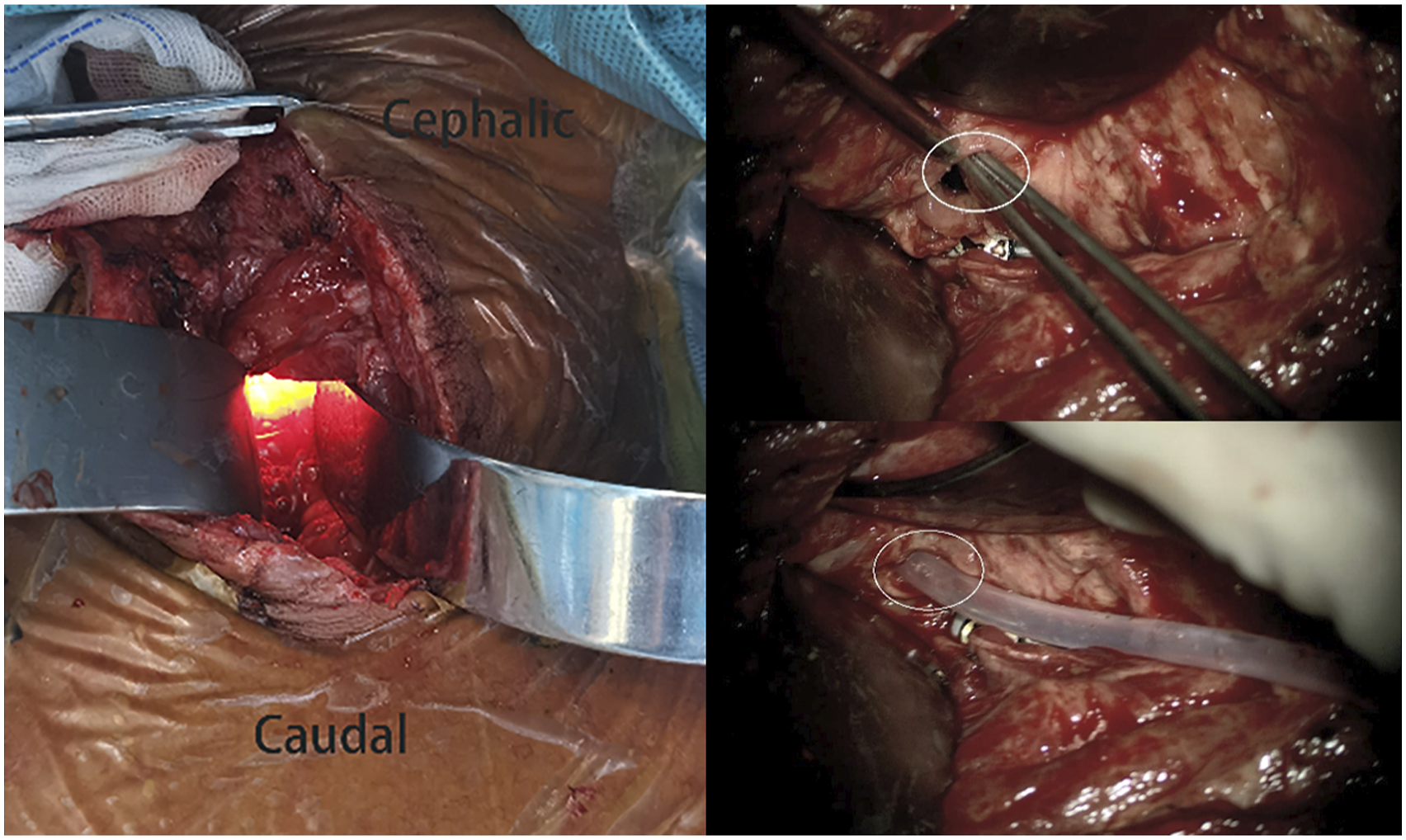
Discussion
The fine suturing of the mucosal layer played a key role in the successful repair of the esophageal perforation in this case. First, the mucosal and submucosal layers have a rich capillary network with good blood flow and relatively active tissue production,14-16 and the fast-growing mucosa can cover the wound surface and facilitate perforation healing. In addition, the submucosa contains tough connective tissue and elastic fibers,
16
which are adequately docked to strengthen the firmness and tensile properties of the anastomosis and are not easily torn off. Second, the mucosa contains a variety of structural and functional components and is the first line of defense against gastric acid corrosion.
17
In this case, gastric acid could be seen pouring out of the perforation during the repair, and gastric acid erosion may have contributed to the perforation formation (Figure 10). Mucosa repair can further reduce gastric acid damage to the sutured segment and aid healing. In addition, it was found intraoperatively that the esophageal muscular layer was severely edematous and eroded due to gastric acid corrosion and infection. There was often insufficient tissue at the sutured end to close the fistula, while the mucosal layer remained relatively fresh and healthy (Figure 11). In this case, the end of the perforation was difficult to close with full sutures, so the mucosa was closed tightly. In summary, the mucosa was an effective and available repair material that facilitated the rapid healing of perforations and created a barrier to prevent leakage of digestive tract contents. However, the esophageal mucosa is formed by loose connective tissue and has a fragile texture,18,19 resulting in a lack of tensile force to withstand sutures during esophageal suturing, which requires fine suturing and makes the procedure more difficult. Gastric acid and gas were seen pouring out of the perforation during the repair process. The muscular layer was severely edematous and vesiculated, while the mucosal layer remained relatively fresh and healthy.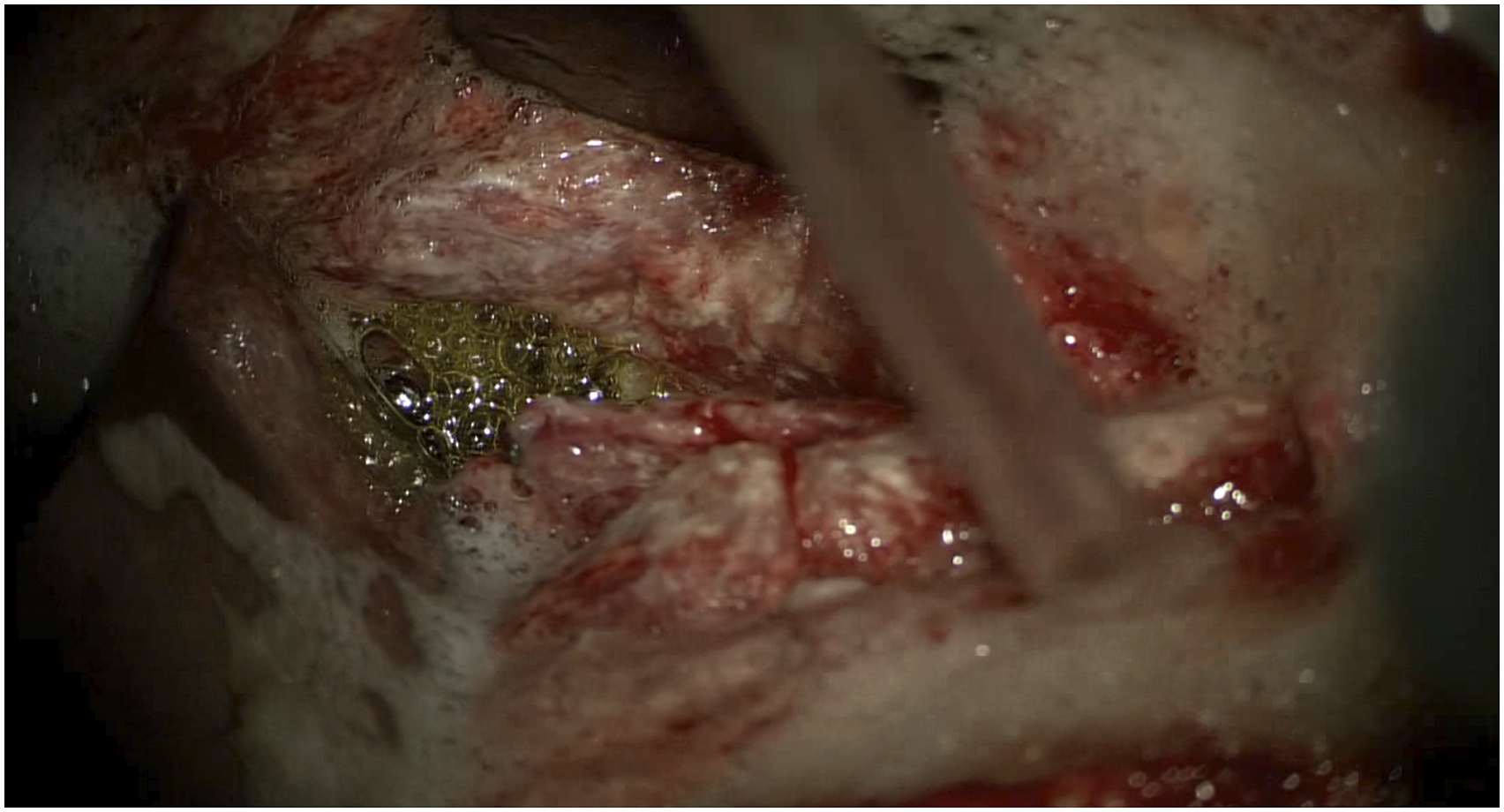
Repair complications, including leakage and stricture, are a significant cause of postoperative mortality and morbidity. The outcome of esophageal perforation surgical repair depends on whether the procedure is complicated by early postoperative leakage and late anastomotic stricture. In this case, the perforation was successfully repaired by the combined use of esophagoscopic and microscopic biomicroscopy, with no postoperative strictures or leaks.
The microscope provides favorable illumination conditions and magnification for deep and narrow surgical views, resulting in better tissue recognition, more precise hemostasis, less tissue damage, uniform operating stitch spacing, flat anastomosis, smooth blood flow, and a lower likelihood of stenosis or embolism. 20 In this case, microscope-assisted surgery also facilitated meticulous exposure and fine repair of the mucosa.
The perforation was repaired intraoperatively with the assistance of esophagoscope. Ahn et al. used the intraoperative nasogastric tube to locate the perforation position, 21 but it is difficult to find a small perforation in this way. Using an esophagoscope had the following advantages: (1) It assisted in placing a drainage tube from the esophageal perforation to the surgical area, which facilitated the localization of the perforation. It also detected smaller and not yet fully penetrated esophageal injuries, and made it possible to ensure the integrity of the mucosal repair, thus avoiding omissions and incompleteness in the repair process. (2) Intraoperative esophagoscopy maintained the properly dilated state of the esophagus, which not only facilitated suturing the perforation but also prevented the perforation repair from causing secondary esophageal stricture. (3) The esophagoscope was placed in the esophagus to hold it in place, preventing movement during suturing and facilitating suturing. (4) It inflated the esophagus, injected water into the surgical area, and reconfirmed the perforation position and the tightness of the suture.
The combined use of the esophagoscope and microscope greatly reduced the difficulty of the procedure and effectively avoided early leakage and late stricture of the esophagus after surgery. This case study shows that microscope and esophagoscope-assisted esophageal mucosal suturing is an effective surgical procedure for repairing esophageal perforation.
Footnotes
Authors Contributions
Y C: Data curation, writing—original draft preparation, writing—reviewing and editing. Y J: Formal analysis, data curation, writing—original draft preparation, writing—reviewing and editing. Z P: Resources, supervision, writing—reviewing and editing. Y Q: Writing—reviewing and editing. Z L: Conceptualization, methodology, project administration. G W: Conceptualization, methodology, project administration. All authors have read and approved the manuscript.
Ethics Approval
This study was approved by the local ethics committee. Informed consent was obtained from patient.
Informed Consent
We have obtained the patient’s consent and signed the patient consent. A copy of the written consent is available for review from the Editor-in-Chief of this journal.
Data Availability
Data was already included in the manuscript.
