Abstract
Introduction
Malignant tumors in the paranasal sinuses and nasal cavity comprise 3–5% of head and neck tumors, 1 with average annual incidence rates of 0.5–1.0 patients/10,0000 male inhabitants and 0.2–0.5 patients/10,0000 female inhabitants worldwide. 2 Squamous cell carcinoma and adenocarcinoma are the most common pathological diagnoses of head and neck tumors, and tumors without glandular and squamous features that cannot be classified in other ways are identified as sinonasal undifferentiated carcinoma (SNUC), as defined in the 2017 4th edition of the World Health Organization Classification of Head and Neck Tumors. 3 With the development of molecular biology technology, some emerging sinonasal malignancies reclassified from SUNC have been reported, such as human papillomavirus (HPV)-related multiphenotypic carcinoma, 4 nuclear protein in testis (NUT) midline carcinoma (NMC), 5 and SWItch/Sucrose Non-Fermentable (SWI/SNF)-related matrix-associated actin-dependent regulator of chromatin subfamily B member 1 (SMARCB1)-deficient sinonasal carcinoma (SDSC). 6 As a rare, highly aggressive malignant tumor, there have been few cases of SDSC in the English literature. To our knowledge, no cases of SDSC with nasal polyps have been reported. We present a case of right SDSC with left chronic sinusitis with nasal polyps (CRSwNP) that required attention from an otolaryngologist.
Case Report
A 58-year-old man who presented with a 3-month history of right intermittent epistaxis was referred to the outpatient clinic. He had a history of functional endoscopic sinus surgery (FESS) for nasal polyps 7 years previously. He underwent an endoscopic biopsy in the outpatient department of our hospital, and a biopsy of this moruloid mass was identified as SDSC.
The patient subsequently underwent further examination. Computed tomography (CT) of the sinuses showed soft-tissue shadows in the right frontal, left sphenoid, and bilateral ethmoid sinuses, and partially missing bone in the right anterior ethmoid sinus (Figure 1A–C). Contrast-enhanced magnetic resonance imaging (MRI) revealed a nodular shadow in the right frontal and anterior ethmoid sinuses measuring 0.7 cm × 1 cm × 1.9 cm with moderately intense enhancement (Figure 1D–F). (A–C): CT scan showed soft-tissue signal (arrow) in the right frontal sinus, left sphenoid sinus, and bilateral ethmoid sinuses, and displayed partially missing bone of the anterior edge of the right ethmoid sinus (axial, coronal, and sagittal CT scans); (D and E): Contrast-enhanced T1-weighted MRI scan demonstrated a neoplasm with moderately intense enhancement (arrow) and the degree of enhancement was lower than the sinus mucosa (axial and coronal contrast-enhanced T1-weighted MRI scans); (F): T1-weighted MRI scan of the paranasal sinuses revealed nodular hypointense signal shadows (arrow) in the right frontal sinus and anterior ethmoid sinus (sagittal T1-weighted MRI scans). CT, computed tomography; MRI, magnetic resonance imaging.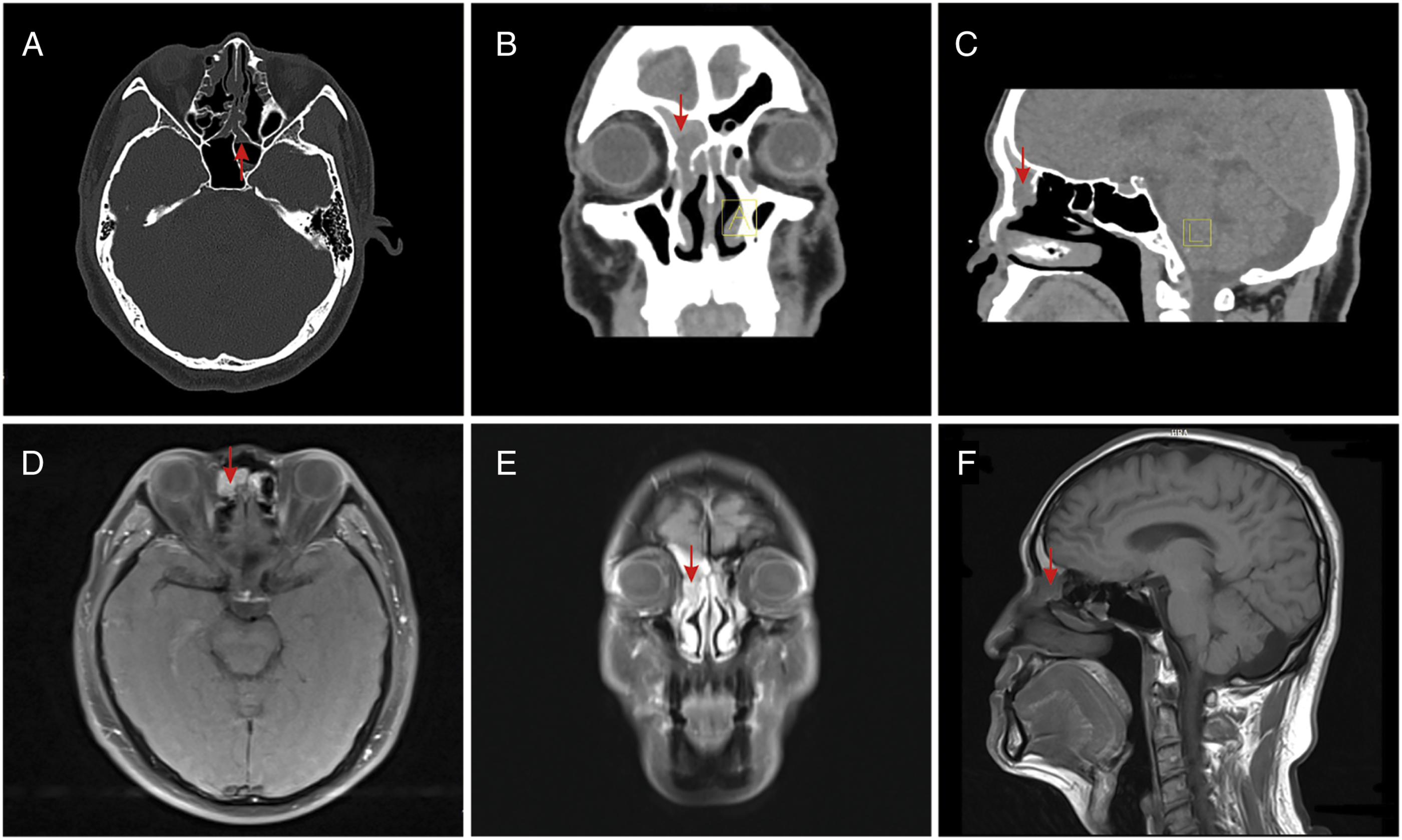
The patient underwent a planned FESS for complete resection of the tumor under general anesthesia in the operating room. Intraoperatively, a left mucosal polypoid hyperplasia originating from the nasal septum in the olfactory fissure area was observed. In the right nasal cavity, tumors originating from the nasal spine of the frontal bone area blocked the opening of the frontal sinus, the frontal sinus mucosa was smooth without neoplasm and dark red discharge in the right frontal sinus, and the bone of the nasolacrimal duct and lacrimal sac was partially destroyed.
Histological examination showed that approximately 60% of the tumor tissues were separated into solid nests or cords (Figure 2A). The tumor cells were basaloid and showed focal necrosis in the center of the nest (Figure 2B). Approximately 40% of the tumor tissues were papillary and flaky around blood vessels (Figure 2C). Under high-power magnification, the tumor cells were rhabdoid and plasmacytoid (Figure 2D). No findings of squamous or glandular differentiation were observed (Figure 2A-D). Immunohistochemical analysis of the right sinonasal neoplasm revealed a complete loss of SMARCB1 expression in all tumor nuclei (Figure 2E). The tumor cells were partially or focally positive for pancytokeratin (PCK) (Figure 2F), epithelial membrane antigen (EMA), P40 (Figure 2G), P63, cytokeratin 5/6 (CK5/6), synaptophysin (Syn), and CD56. No loss of SMARCA4 was observed. The expression of other antibodies was uniformly negative, including chromogranin A (CgA), S-100, Sry-related HMg-Box gene 10 (SOX10), CD34, desmin, P16, and nuclear protein in the testis (NUT). The Ki67 proliferation index was approximately 80% (Figure 2H). No cancer cells had infiltrated the excised right lacrimal sac. A mass in the left olfactory cleft area was pathologically diagnosed as a chronic inflammation with focal polypoid changes. Based on the morphological characteristics and findings of the immunohistochemical examination, combined with the clinical and imaging findings, the patient was diagnosed with right SDSC T2N0M0 and left CRSwNP. (A): 60% of the tumor tissue was divided into solid nests or cords by proliferating collagen fibrous connective tissue (arrow), showing a basal cell-like morphology (HE 100×); (B): Basaloid tumors are characterized by less cytoplasm, increased nucleo-plasma ratio, vacuolar nucleus, obvious nucleolus, and necrosis in the center of the tumor nest (arrow) (HE 400×). (C): 40% of tumor tissues grew in papillary and flaky shapes around blood vessels, with reduced adhesion (arrow) (HE 100×). (D): The tumor cells were rhabdoid and plasmacytoid, with abundant cytoplasm, red staining, and nuclear deviation (arrow) (HE 200×). (E): The expression of SMARCB1 (INI-1) in tumor cells was completely absent, while the nucleus of interstitial cells was positive (IHC 200×); (F): PCK was diffusely and strongly positively expressed in the cytoplasm of rhabdoid and plasmacytoid tumors (IHC 200×). (G): P40 was diffusely and strongly positively expressed in the nucleus of rhabdoid and plasmacytoid tumors (IHC 200×). (H): Ki67 showed high proliferation of tumor cells (IHC 200×). HE, hematoxylin and eosin staining; IHC, immunohistochemistry; SMARCB1, SWItch/Sucrose Non-Fermentable (SWI/SNF)-related matrix-associated actin-dependent regulator of chromatin subfamily B member 1; INI-1, integrase interactor 1; PCK, pancytokeratin.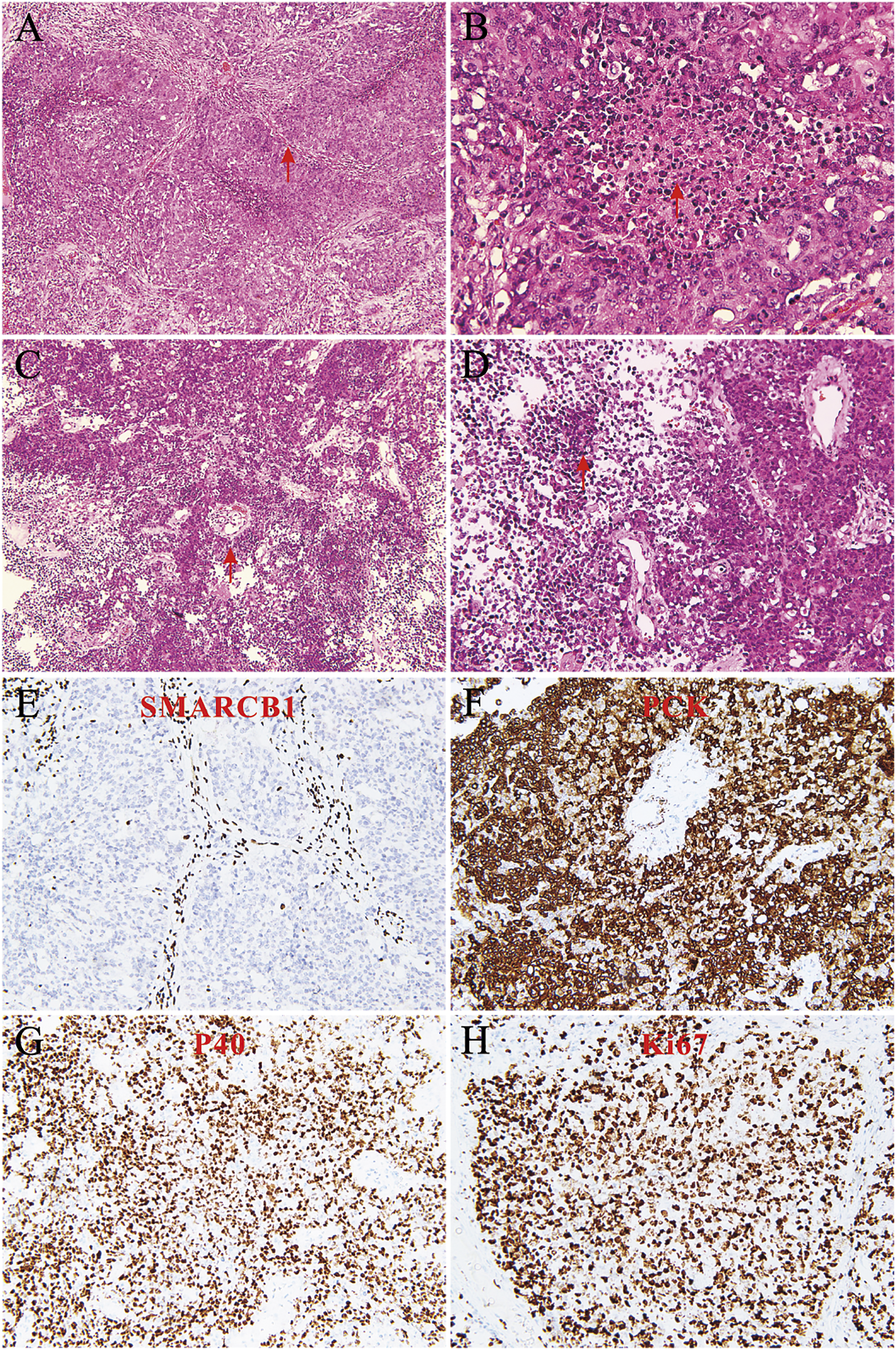
The patient was administered etoposide and platinum (EP) combined with durvalumab (a programmed cell death protein 1/programmed death-ligand 1 [PD-1/PD-L1] inhibitor) therapy at the Cancer Center, Wuhan Union Hospital, with no evidence of recurrence or metastasis at the 3-month follow-up.
Discussion
SMARCB1-deficient sinonasal carcinoma, also known as integrase interactor 1 (INI-1) deficient carcinoma and characterized by the loss of immunohistochemical staining of SMARCB1, is an emerging rare malignant cancer of the nasal cavity and paranasal sinuses that accounts for approximately 3–6% of cancer cases of the nasal cavity and paranasal sinus.6,7 In 2014, Bishop et al. and Agaimy et al. independently described SDSC for the first time.6,8 In 2017, the World Health Organization proposed SDSC as a differential diagnosis and classified it as a SNUC. 3
SMARCB1 is a tumor suppressor gene located on chromosome 22q11 that is universally expressed in the nucleus of all normal tissue cells. SMARCB1 regulates cell proliferation and gene transcription. 9 The encoded protein is the core subunit of the SWI/SNF chromatin remodeling complex and is involved in activating polycomb proteins (PRC2) to activate epigenetic pathways and chromatin remodeling, inhibiting the cell cycle by suppressing cyclin D1, activating retinoblastoma tumor suppressor genes, inducing tumor suppressor protein p16, and inhibiting the Wnt/β-catenin oncogenic signaling pathway. 10 Loss of SMARCB1 expression is associated with various malignancies, including epithelioid sarcoma, 11 epithelioid malignant peripheral nerve sheath tumor, myoepithelial carcinoma of soft tissue, 12 and extraskeletal myxoid chondrosarcoma. 13 The genetic basis for the loss of protein expression is mainly considered to be due to homozygous (biallelic) deletion of SMARCB1 identified in fluorescence in situ hybridization (FISH) analysis.6,14
The histological spectrum combined with uniform SMARCB1 deficiency suggests that SDSC is a unique tumor in poorly differentiated sinonasal neoplasms.6,8,14,15 The 2 main morphological forms of SDSC are basaloid and plasmacytoid/rhabdoid, with the basaloid pattern the most common. 16 Basaloid tumors are characterized by undifferentiated or “blue cell” tumors with high nuclear: cytoplasmic ratios; prominent nucleoli; scant cytoplasm; and sheet-like, nest-like, or palisading arrangements.6,8,14,17 Plasmacytoid/rhabdoid tumors are characterized by “pink cell tumors” with abundant eosinophilic cytoplasm and eccentric nuclei.6,14,17 All SDSC tumors contain rhabdoid cells, which vary greatly in number and distribution.6,8,14 High mitotic rates and tumor necrosis are frequent findings. SDSC manifests aggressive histological features but there is no obvious squamous or adenoid differentiation, surface epithelial dysplasia, or carcinoma in situ.6,8,14,15 The morphology in the present case was consistent with the typical basaloid (Figure 2A–B) and rhabdoid (Figure 2C–D) architectures of SDSCs.
As a subtype of SNUC, SDSC has diverse morphological manifestations and must be differentiated from poorly differentiated sinonasal carcinomas, other subtypes of SNUC, and other rhabdoid malignant tumors. Immunohistochemical examination of SDSC often shows diffuse positivity for PCK; variable positivity for P63, P40, and P16; focal positivity for neuroendocrine markers; complete loss of SMARCB1; and no loss of SMARCA4 or ARID1A. 15 Although differentiated tumors have some histomorphological similarities to SDSCs, all of them retain SMARCB1 expression. Based on morphological basaloid and rhabdoid tumor cells, the complete loss of SMARCB1 expression (Figure 2E), relevant immunohistochemical examination observations, and with clinical findings, the tumor in the present case was diagnosed as SDSC. The patient refused the FISH test that would have further supported the diagnosis.
According to previous reports of SDSC, the age at presentation ranged from 11 to 89 years,14,17 with slightly more males than females. SDSC is a highly invasive malignant carcinoma that often invades adjacent sinuses (mainly the ethmoid sinus), orbits, and the anterior skull base and can grow into the intracranial epidural or subdural regions. The frequent clinical manifestations include nasal congestion, headaches, epistaxis, and impaired vision. The prognosis is poor, with a median overall survival time of approximately 2 years. 16
Due to insufficient relevant cases and studies, knowledge of the exact biological characteristics and pathogenesis of SDSC has been limited. Moreover, the best treatment options remain unclear.
Conclusion
The patient in this case presented with right intermittent epistaxis, and endoscopic biopsy showed SDSC. After further examination, FESS was performed in the operating room. Surgical histopathological examination confirmed a diagnosis of right SDSC and left CRSwNP. To our knowledge, no related research has been reported.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Bethune Charitable Foundation (grant nos. BQE-TY-SSPC(8)-E−01) and Health Commission of Hubei Province Scientific research project (grant nos. WJ2021M250).
Authors’ Note
HW Yang, LQ Zhou, G Zhong, and XX Li contributed equally to this work. All authors contributed to data collection, drafting or revising the article, and all authors have read and approved the final manuscript.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
