Abstract
Objective
The objective is to describe the clinical features, treatments, and outcomes of a case series of patients with Onodi cell mucocele, with or without cholesterol granuloma (CG).
Material and methods
We retrospectively reviewed the medical records of eight patients diagnosed with Onodi cell mucocele at a single tertiary care university hospital in Beijing, China, between January 2017 and September 2020. Data regarding nasal symptoms, ocular symptoms, sinus computed tomography findings, treatments, histopathological results, and clinical outcomes were extracted.
Results
We identified eight patients (six men and two women) of an average age of 48.1 (range, 26–70) years. Four patients presented nasal symptoms. Three patients presented ocular symptoms. Among them, one patient experienced concurrent nasal and ocular symptoms. Two patients were diagnosed based on a physical examination in the absence of nasal or ocular symptoms. All patients underwent endoscopic sinus surgery. The pathological specimens showed mucocele in four cases and mucocele with CG in the other four cases. Among the four cases with CG, three cases presented with decreased vision. After endoscopic sinus surgery, one patient recovered completely, and two patients showed significant improvement.
Conclusion
If Onodi cell opacity is observed, especially with optic neuropathy, mucocele and CG are important differential diagnoses. The combination of mucocele and CG is more likely to promote bone destruction and cause serious optic neuropathy than simple mucocele. Endoscopic sinus surgery is appropriate. Diagnoses, treatments, and follow-up should be performed by a multidisciplinary team.
Introduction
An Onodi cell is a posterior ethmoid cell lateral and/or superior to the sphenoid sinus. The optic nerve may lie within this sphenoethmoidal cell. 1 According to radiological findings, its incidence is 8–24%, with a rate as high as 60% in cadaveric studies. 2 Given the proximity of this cell to the optic nerve, pathological processes (e.g., sinusitis, mucocele, cholesterol granuloma [CG], and tumors) can cause ophthalmologic complications.
Sinonasal mucoceles are benign, locally expansile masses in the paranasal sinuses. 3 CG is a histological entity defined by a chronic granulomatous reaction to the presence of cholesterol crystals in an enclosed space. 4 The two lesions occur rarely in Onodi cells.3,5 We report diagnoses and treatment of eight patients with Onodi cell mucocele, four concomitant to CG.
Methods
We retrospectively reviewed a series of patients diagnosed with Onodi cell mucocele at our hospital between January 2017 and September 2020. Data regarding nasal symptoms, ocular symptoms, sinus computed tomography (CT) findings, treatments, histopathological results, and clinical outcomes were obtained from the patients’ medical records. This study was approved by the Clinical Research Ethics Committee of Peking University People’s Hospital (2021PHB064-001) and was conducted following the Helsinki Declaration.
All surgeries were performed under general anesthesia using transnasal endoscopy. The nasal mucosa was locally anesthetised using neuropatties infiltrated by 20 mL 1% tetracaine and 4 mL 0.1% epinephrine. The basal lamella of the middle and superior nasal concha was removed to reveal the posterior ethmoidal sinus. The sphenoid sinus was widely opened from the natural ostium or anterior wall. The Onodi cell was opened, and the cyst contents removed without damaging the optic nerve. The mucocele was marsupialized to expand drainage to the nasal cavity. Postoperative nasal packing was performed without compressing the optic nerve canal and was removed during follow-up.
Pathological reports were issued according to the conventional hospital protocol, and a senior pathologist subsequently re-analyzed the sections and reviewed the pathological results.
Results
We identified eight eligible patients (six men and two women) with a mean age of 48.1 (range: 26–70) years. A total of six of these eight patients presented with nasal and/or ocular symptoms: four patients presented with nasal symptoms (e.g., nasal obstruction and olfactory disorder); three patients had ocular symptoms (e.g., decreased vision, visual field defect, and exophthalmos); and one patient experienced concurrent nasal and ocular symptoms. The other two patients included in this study were diagnosed based on a physical examination in the absence of nasal or ocular symptoms. All patients denied having experienced nasal surgery or trauma.
Nasal endoscopy revealed small amounts of purulent secretions in the olfactory fissure in two patients with decreased vision; the other patients had no positive endoscopic findings. All patients completed sinus CT examination, and three patients had additional magnetic resonance imaging (MRI) examination. Sinus CT findings revealed expansion of the lesions, compression and thinning of the surrounding bone of Onodi cell, and bone destruction occurred in seven patients. MRI showed that the cystic masses exhibited T1-W shortening and T2-W prolongation or mixed signals without evidence of enhancement.
All patients’ white blood cell level, percentage of lymphocytes and neutrophils, hemoglobin value and platelet counts were within the normal range, and there were no abnormalities in their coagulation function analysis. In the blood biochemical analysis, four older patients had excessive triglycerides, but their cholesterol and low-density lipoprotein were in the normal range. Only one patient had a slightly lower high-density lipoprotein (HDL) level, and the others’ HDL results were normal.
The preoperative preparation time ranged from one to six days depending on patients’ symptoms. The preoperative preparation time of patients with decreased vision was significantly shorter (i.e., one, two, and four days, respectively), while the preoperative preparation time of the other five patients varied more (i.e., one, two, five, five, and six days, respectively).
Summary of cases.
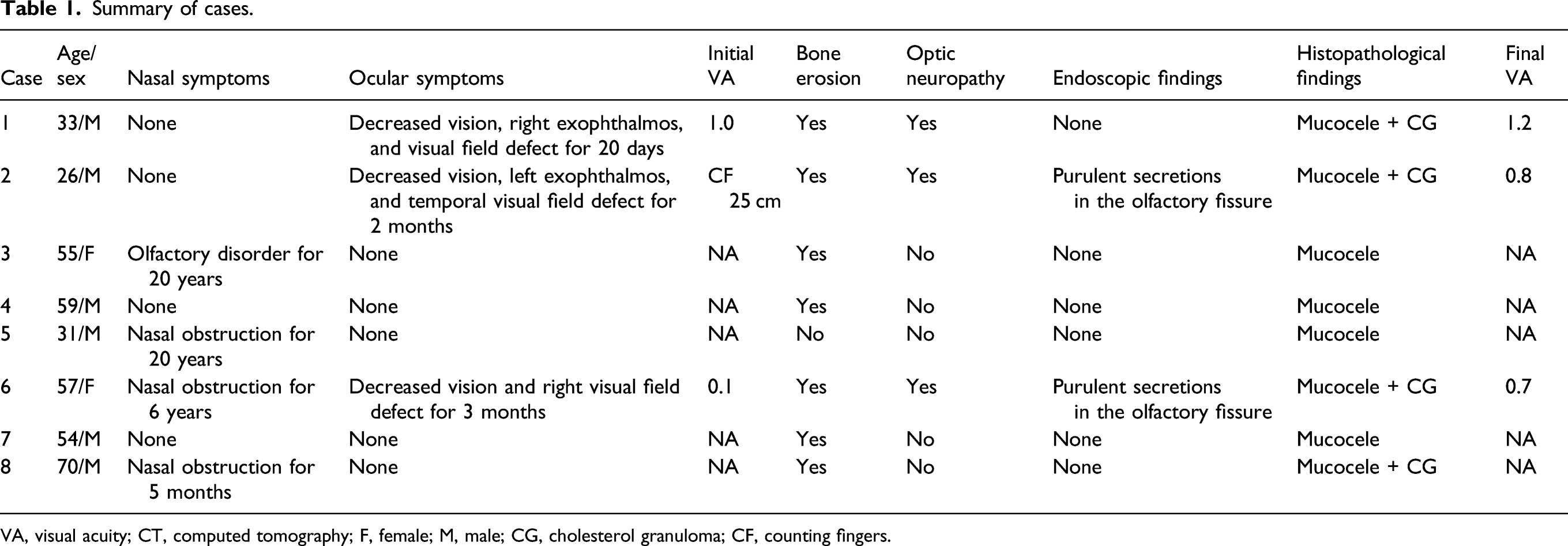
VA, visual acuity; CT, computed tomography; F, female; M, male; CG, cholesterol granuloma; CF, counting fingers.
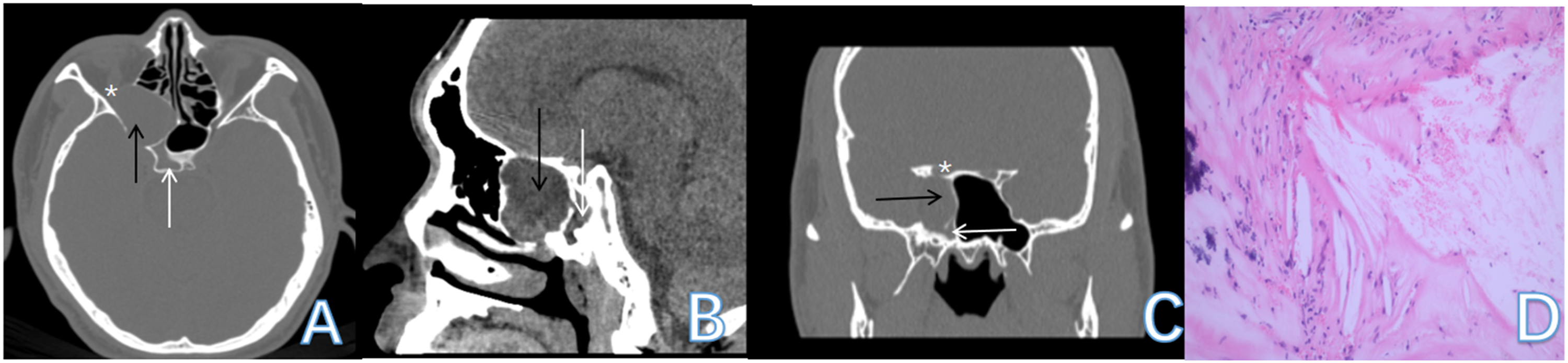
Sinus computed tomography and pathological findings from Case 1. The Onodi cell mucocele (black arrow) lies lateral and superior to the sphenoid sinus (white arrow), compressing the right optic nerve (white asterisk). Pathological evaluation reveals mucocele with cholesterol granuloma, by acicular cholesterol crystal clefts, foamy histiocytic reaction, giant cells, and lymphocytes (D).
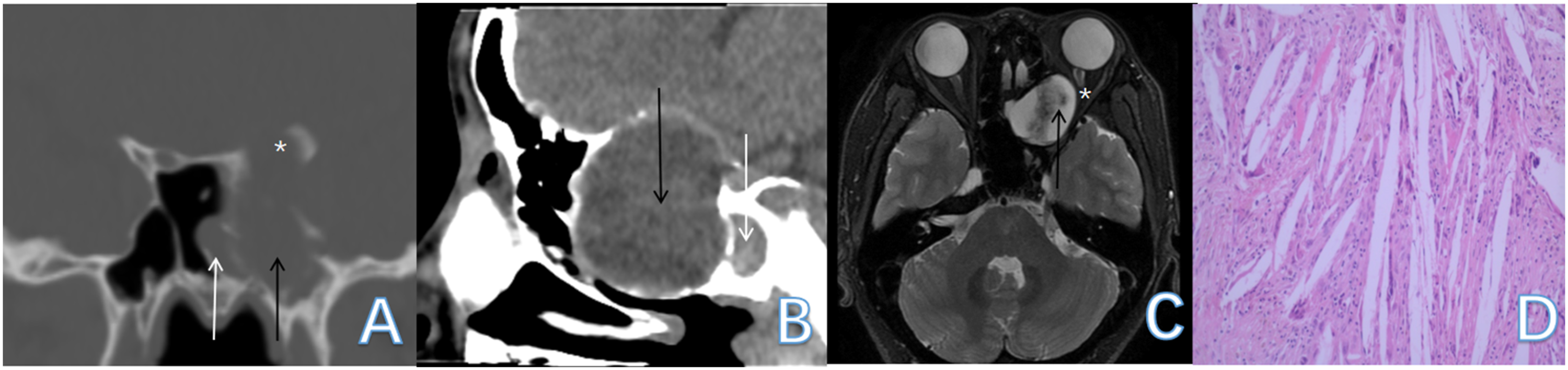
Sinus computed tomography, magnetic resonance imaging, and pathological findings from Case 2. The Onodi cell mucocele (black arrow) lies lateral and superior to the sphenoid sinus (white arrow), compressing the left optic nerve (white asterisk). Pathological evaluation reveals mucocele with cholesterol granuloma, by acicular cholesterol crystal clefts, giant cells, and lymphocytes (D).
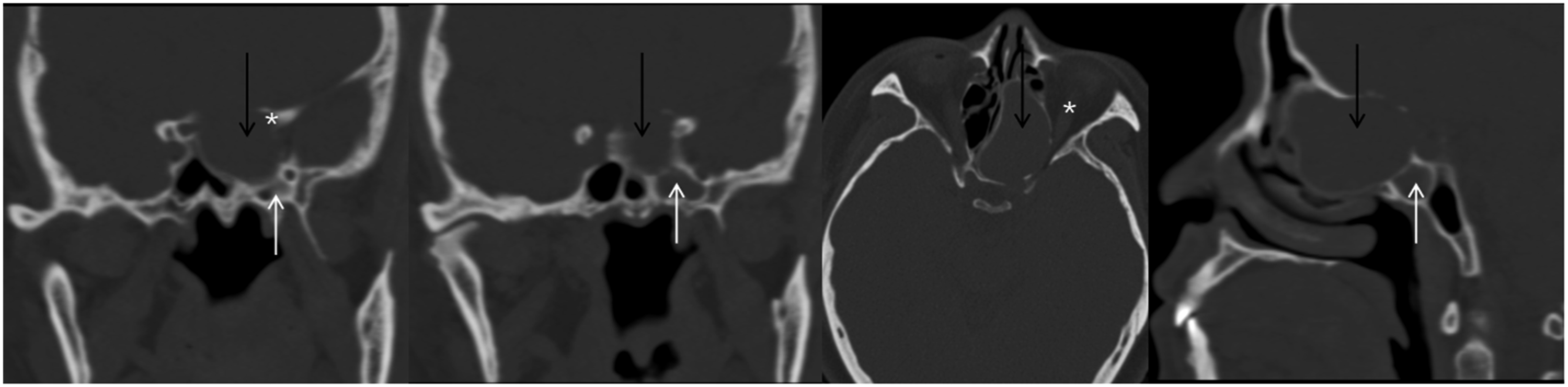
Sinus computed tomography from Case 3. The Onodi cell mucocele (black arrow) lies superior to the sphenoid sinus (white arrow), compressing the left optic nerve (white asterisk).
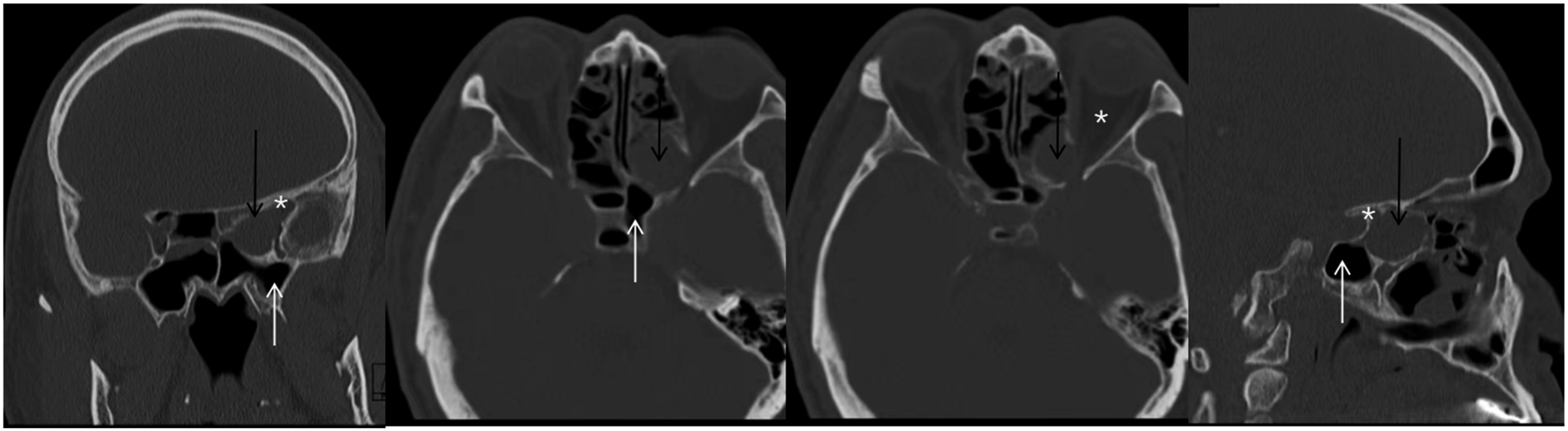
Sinus computed tomography from Case 4. The Onodi cell mucocele (black arrow) lies superior to the sphenoid sinus (white arrow), compressing the left optic nerve (white asterisk).
Discussion
A literature review on Onodi cell mucocele shows only case reports describing one or two cases.6,7 We treated eight patients, which is the largest case series reported thus far. Notably, in our series, histological examination revealed mucocele in all eight cases, as well as CG in four cases (including the three cases with decreased vision). However, none of the four patients without CG had vision loss, despite bone destruction in most of these cases. It is reasonable to suppose that mucocele combined with CG is more likely to cause optic neuropathy than simple mucocele.
The pathological mechanism of mucocele formation is considered to involve paranasal sinus or air cell obstruction, which leads to retention of secretions. In 1981, Evans et al. reported that mucocele was related to increased protein content in secretions of the paranasal sinuses, leading to increased osmotic pressure and filling of the sinus cavity, and ultimately increasing the pressure in the sinus. Destruction of the surrounding bone wall by the mucocele is related to osteoclasts in the bone wall being activated by parathyroid hormone, prostaglandin, vitamin D, and other substances, while lymphocytes produce osteoclast activators to destroy the sinus wall, and prostaglandins may also contribute to bone resorption. 8
CG is a nonspecific disease commonly found in the aerated portions of the temporal bone, and it has also been documented in the thyroid, liver, kidney, testis, breast, brain, and atheromatous plaques. 9 Various factors can lead to obstructed drainage and impaired ventilation, which can cause middle ear hemorrhage, plasma exudation, tissue edema, and tissue necrosis; these problems may ultimately lead to red blood cell rupture, fat degeneration, and cholesterol release. The increasing cholesterol concentration eventually leads to saturation and formation of cholesterol crystals, which stimulate granulation and CG formation in the surrounding tissues. CG formation in the paranasal sinuses generally follows this physiological process, and the increasing sinus pressure and osteoclast activation by inflammatory factors can ultimately lead to bone destruction.
There are many similarities in the characteristics of mucocele and CG, including the involvement of poor drainage, blocked ventilation, inflammatory reaction, and an osteoclast reaction. Therefore, these two pathological processes may share the same etiology and interact with each other. Moreover, their coexistence may exacerbate bone destruction and its effects on adjacent nerves or other structures, which agrees with our findings.
Previous research has suggested that the mechanism of vision loss caused by Onodi cell mucocele includes direct mechanical compression of the optic nerve, local ischemia by vascular nerve reflex, and inflammation. 10 Different pathological mechanisms represent various prognoses. Mechanical compression is mainly characterized by a gradual appearance of clinical symptoms and a better prognosis. 11 Fukuda et al. suggested that ischemia and inflammation induce an acute onset of ophthalmologic signs and symptoms, which may indicate a poor prognosis. 12 Kim et al. reported that patients with infectious optic neuropathy secondary to a mucopyocele showed significantly less visual improvement after surgery than those with mucocele and no superinfection. 13 Foreign body giant cell reaction caused by Onodi cell mucocele capsular hemorrhage or tissue catabolic cholesterol crystallization may be a new secondary mechanism of vision loss.
In our case series, all three patients with vision loss experienced complete or significant recovery after endoscopic sinus surgery, and bacterial culture results were negative; together, these findings might suggest that CG represents a relatively good prognosis in patients with decreased vision. This is likely related to the fact that it takes time to catabolize cholesterol crystals and promote foreign body giant cell reaction, differing from ischemia or acute inflammation, which can cause a rapid and significant decline in visual acuity (e.g., sudden vision loss to no light perception in a few hours). Even with immediate surgical intervention, the postoperative visual acuity still did not recover in patients with ischemia or acute inflammation. 12
Zhang et al. previously examined the correlations between middle ear CG and dyslipidemia. 14 They analyzed blood lipid results in cases of middle ear CG, comparing these to age- and sex-matched cases of chronic otitis media without CG, and found that patients with middle ear CG showed higher lipid levels. However, this association was not observed in our case series. This may be related to the limited number of cases studied here and the inability to carry out an age- and sex-paired t-test. We look forward to conducting further relevant research in the future with additional age- and sex-paired cases.
To the best of our knowledge, this is the first report on Onodi cell mucocele with CG. Among the 24 cases in the literature review, eight had pathological results of inflammation or mucocele; however, CG was not mentioned. Although the small size prevents definitive associations, the observations from this case series are noteworthy.
The diagnosis of mucocele depends on imaging examination. A paranasal sinuses CT scan shows expansive space-occupying bone around paranasal sinuses absorbed, thinned, or defective, which is a typical but nonspecific manifestation of mucocele. If necessary, MRI should be performed to differentiate a mucocele from a tumor. Mucocele appears differently on MRI because of changing protein concentration over time. The mucocele’s initially high water content leads to hypointense T1 and hyperintense T2-W images, while the gradual increase in protein concentration will result in a reverse intensity 15 .
As most mucoceles are asymptomatic at early stages, some patients may experience sinusitis-like symptoms over time. When lesions affect the orbit, vision loss, exophthalmos, and ophthalmalgia may occur. We found that symptom severity was related to the extent and period of compression. In our study, the incidence rate of ocular complications in our case series was 3/8 (37.5%). Unlike in the literature review, all patients had different degrees of vision loss, except one patient who was diagnosed with trochlear nerve palsy. 16 This may be related to case-selection bias in case reports. If a patient with a mucocele has no orbital or intracranial complications, the diagnosis and treatment process are similar to those of chronic sinusitis. Thus, physicians may not choose to report the case. Other researchers found that many patients with sinus mucocele had a history of sinus surgery or head injury. 17 However, in our study, all patients denied having experienced nasal surgery or trauma. This may be related to the deep location of the Onodi cell, which makes this area less affected by external forces or traumas, and more affected by chronic inflammation due to anatomical variation and poor drainage.
Endoscopic sinus surgery is a simple, safe, and appropriate procedure for Onodi cell mucocele, as the sinuses provide a natural route to approach the sino-orbital interface. 18 This procedure involves dissecting the natural sinus drainage pathway and decompression of the lesion(s), with preservation of the original mucosa of the nasal sinus to promote postoperative healing. In particular, the cystic wall should not be removed if the mucocele is associated with a bone defect to avoid local adhesions and potentially serious complications. When combined with sphenoid sinus opacification, sphenoidotomy should be performed at the same time. On the premise of ensuring the safety of patients, the earlier the operation, the better. In our study, patients who were prepared for more than two days before operation were delayed due to referral or to imaging and consultation appointments in different departments. Immediate emergency surgery may be performed if the patient has rapid vision loss or massive bleeding, but it did not appear in our series.
Conclusion
Given the risk of vision loss, mucocele and CG diagnoses should not be ignored if Onodi cell opacity is observed. Coexisting mucocele and CG can be more likely to promote bone destruction and cause serious optic neuropathy than simple mucocele. Early intervention may prevent ophthalmological complications and increase the likelihood of vision recovery. Endoscopic sinus surgery is appropriate. A multidisciplinary team should perform all diagnoses, treatments, and follow-up. Moreover, we should advocate multi-center cooperation, expand the number of cases, improve the data of patients with Onodi cell mucocele with or without optic neuropathy, pay more attention to the pathological results, and explore the role of CG in the occurrence and development of the disease.
Footnotes
Author contributions
Conceptualization: Min Wang and Zhimin Xing;
Data curation: Congli Geng and Shien Huang;
Formal analysis: Qizhe Tong, Yi Wang, and Zhimin Xing;
Funding acquisition: Min Wang;
Investigation: Shien Huang and Kunkun Sun;
Project administration: Min Wang;
Software: Kunkun Sun;
Writing – original draft: Congli Geng and Qizhe Tong;
Writing – review and editing: Yi Wang and Min Wang.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Capital Health Development Foundation (No: 2020-1-2051) and Peking University People’s Hospital Scientific Research Development Funds (RDL2021-05) supported this study.
Institutional review board statement
This study was approved by the Clinical Research Ethics Committee of Peking University People’s Hospital and was conducted following the Helsinki Declaration.
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
