Abstract
Background
Velopharyngeal dysfunction (VPD) in head and neck cancer is frequently clinically reported, affecting both speech and swallowing function. This review sought to identify the tumor subsites and treatment modalities reported in association with VPD and summarize the current reporting methodology of VPD-related speech and swallowing outcomes in patients following head and neck cancer treatment.
Methods
A literature search was conducted through December 2020 using electronic databases and a total of 15 studies were included in review.
Results
Reported VPD was largely secondary to palate resections. Large variability in reporting methodology was noted with heavy reliance on speech-related perceptual measures and swallowing-related patient-reported outcomes over imaging and instrumental evaluations.
Conclusions
This review revealed inconsistencies in evaluating and reporting VPD, which likely translates into inconsistencies in clinical management. Further attention to VPD secondary to other head and neck malignancies would provide a broader perspective on VPD through head and neck cancer treatment.
Introduction
Speech and swallowing disorders are largely prevalent in patients with head and neck cancer. 1 A systemic understanding of the various levels of breakdown is critical to the clinical management of the overall dysfunction. One prevalent deficit that affects both speech and swallowing is velopharyngeal dysfunction (VPD).
The velopharyngeal (VP) port, constructed by the velum and the surrounding pharyngeal walls, is a critical junction for both speech and swallowing tasks. The velum typically remains lowered with the patent nasopharyngeal airway during breathing. In speech, adequate VP closure, through the combined action of superoposterior movement of the velum and anteromedial movement of the pharyngeal walls, allows for sound energy and airflow to be directed through the oral cavity. Depending on the relative contributions of the VP structures, different patterns (coronal, sagittal, and circular) of VP closure have been described in the literature. 2 -6 Deficits in VP closure during speech manifests as hypernasality (i.e., excessive nasal resonance perceived in speech) and/or nasal airflow deviation errors (i.e., nasal air emission). On the swallowing front, VP closure is necessary to restrict entry of food and liquids into the nasal cavities. Closure of the VP port during swallow appears to occur with significantly greater closure force and muscular activity with fast-twitch motor units dominantly fired in comparison with VP closure during speech. 7,8 Functionally, VPD may result in reduced speech intelligibility and dysphagia with social disturbance and consequently reduced social interactions. 9 The negative impact of VPD on quality of life (QOL), including social/emotional disturbance, is well documented in the literature. 10 In fact, while swallowing dysfunction was rated to be the most reliable predictor of health-related QOL outcomes in patients treated for nasopharyngeal cancer, 11 nearly 60% of the patients with nasopharyngeal cancer exhibit VPD. 12
Head and neck cancers are steadily rising in recent years, partially attributed to the rise in human papillomavirus (HPV)-related tumors. 13 Treatment effects are often drastic, leading to significant impacts on speech and swallowing function. Based on site and extent of tumor, treatment modalities typically include a combination of surgery, radiation, and chemotherapy. 14 For instance, certain oral cavity tumors are primarily treated with surgical management followed by adjuvant radiotherapy with or without chemotherapy, whereas other tumors of the oropharynx are often treated with primary radiotherapy with or without chemotherapy.
The impact on functional outcomes has been studied widely with various degrees of focus. Nguyen and colleagues 15 specifically studied the impact of dysphagia on QOL in a retrospective analysis of patients treated with definitive single modality radiotherapy, chemoradiotherapy, and surgery followed by adjuvant radiotherapy. Their results showed that patients with documented moderate-to-severe dysphagia had worst outcomes on overall QOL. Swallowing outcomes were immediately followed by speech outcomes, which weighed heavier than other physical and aesthetic dysfunctions. Swallowing function after chemoradiation therapy was investigated by Eisbruch et al., 16 in which VPD was identified, along with other deficits, such as poor airway protection and swallowing inefficiency at post-therapy. According to Wu et al., 17 greater than one fourth of patients with nasopharyngeal cancer treated with radiation therapy presented with late-radiation effects (e.g., poor pharyngeal contraction, airway protection, and vocal cord palsy) with greater than 58% of patients demonstrating VPD. Given the critical role of the VP function in both speech and swallowing, more attention and understanding of these deficits are warranted.
Of specific interest to this review are VPD issues in individuals who underwent head and neck cancer treatment. Although VPD is a common clinical manifestation following head and neck cancer treatment, its management has yet to be systematically reviewed. Thus, the purpose of this scoping review was 2-fold: (1) to identify and describe the tumor subsites and treatment modalities reported in association with VPD in the available literature and (2) to summarize the current reporting methods of VPD-related speech and swallowing outcomes in patients following head and neck cancer treatment. Guided by this scoping review, we will propose some guidelines for clinicians to improve appropriate testing of VP function.
Methods
Literature Search
First, inclusion and exclusion criteria were established to guide our literature search. Inclusion criteria was broad, including all peer-reviewed journal publications that investigated VPD and any head and neck cancer site. All available studies until December 2020 were included in the analysis. Lower limits on time were not set due to the anticipation of a small number of studies. Inclusion criteria and exclusion criteria were set based on etiology. Inclusion criteria were set at head and neck cancer and exclusion criteria included studies that investigated VPD as a result of other etiologies (e.g., cleft palate or other neurogenic disorders). Three major databases were utilized in the search process: PubMed, Google Scholar, and ScienceDirect. Additional articles were obtained through other sources, including textbooks and reference lists.
The first level of search included PubMed with the terms “velopharyngeal dysfunction AND head and neck cancer,” which yielded 22 results. Nine articles were selected due to the abstracts specifically reporting on VP function. The search terms, “hypernasality AND head and neck cancer,” yielded 39 results. Three were selected based on the abstract that specifically listed speech, swallowing, and nasality information. On ScienceDirect, the search terms “velopharyngeal dysfunction AND head and neck cancer,” and “hypernasality AND head and neck cancer” yielded 79 and 54 results, respectively; however, 0 were selected due to the articles not specifically addressing VPD and often including other etiologies in the search results. The same search terms on Google Scholar yielded 99 results out of which conference abstracts, presentations and book chapters were excluded and a total of 20 articles that met the inclusion criteria were selected. Following removal of duplicates and articles that were clinical reports and instructional in nature, a total of 15 articles were selected for review.
Data Extraction
Following removal of duplicates, a total of 15 articles met the inclusion and exclusion criteria. As the major goal for this review was to identify gaps in the literature, the scope was kept broad in order to account for all possible measures of functional outcomes. Therefore, case studies and studies with low n-size were also included in this analysis. PRISMA guidelines were used for abstracting data (Figure 1). In order to facilitate data analysis, key information, including study objectives, sample size, study design, tumor subsite, treatment modality, and detailed speech and swallowing outcome measures reported, was extracted and summarized in table form. PRISMA flow diagram depicting the flow of study search and selection.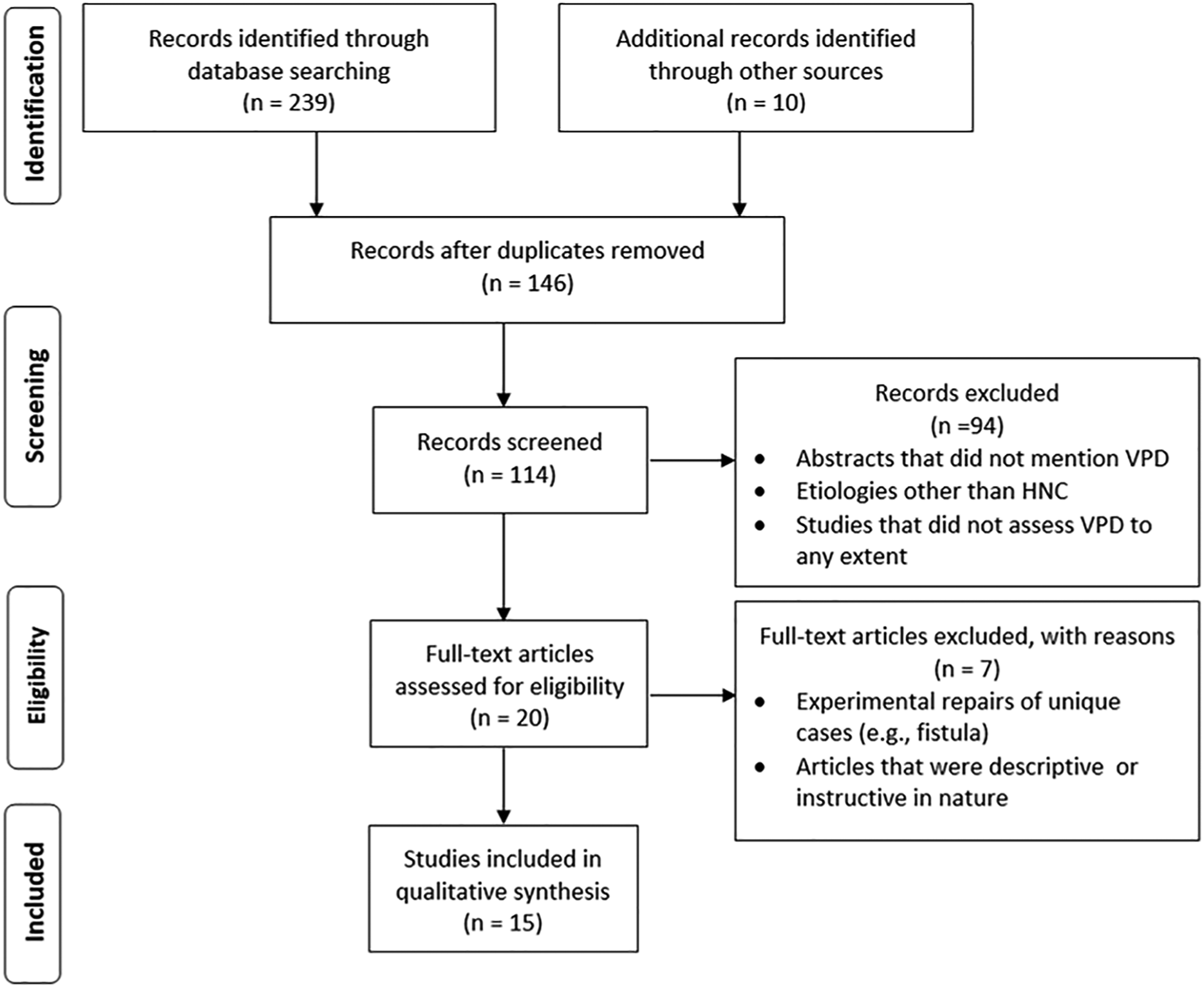
Results
Tumor Subsite and Treatment Modality
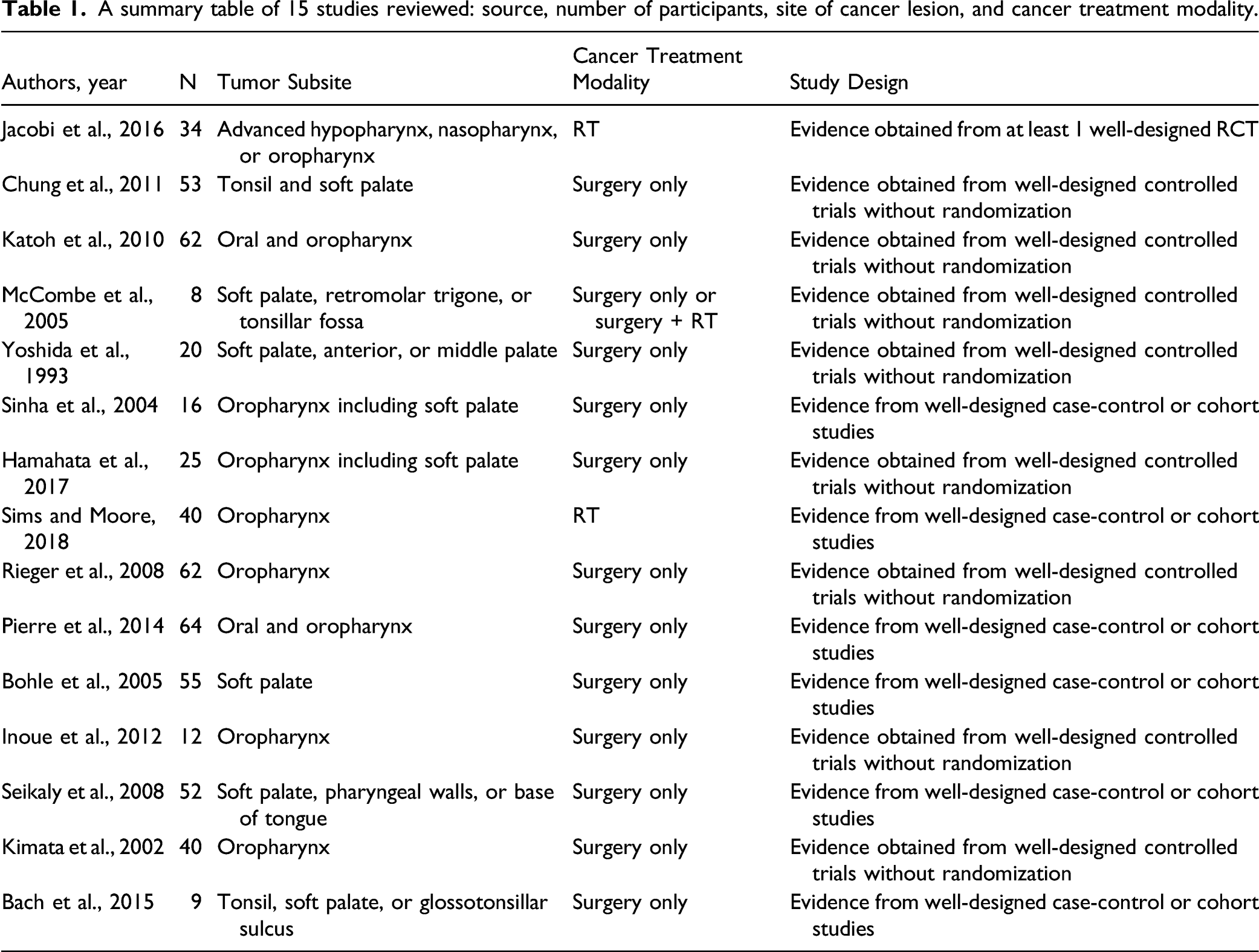
A total of 542 patients from 15 studies were included and reviewed with the sample size ranging from 8 to 64 patients per study. Tumor subsites identified in the reviewed studies included the soft palate, hard palate, base of tongue, tonsil, retromolar trigone, and pharynx. Specifically, 5 studies indicated soft palate ± other regions; 12,18 -21 1 study indicated soft palate, hard palate, or both; 22 and the remaining 9 studies indicated oral and/or oropharyngeal regions that may include the soft palate. 23 -31
A summary table of 15 studies reviewed: source, number of participants, site of cancer lesion, and cancer treatment modality.
Speech Assessment and Outcome Measures
Summary of results of the 15 studies that reported speech and/or swallowing assessment/outcome measures.
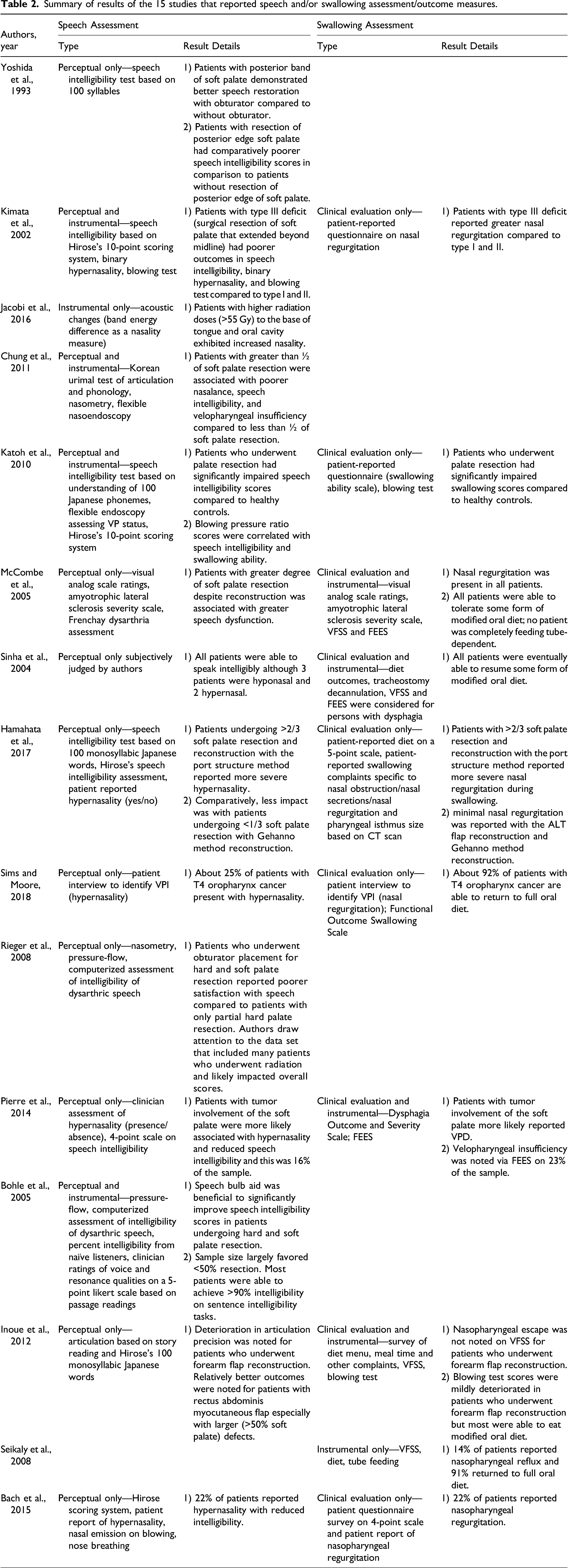
Speech intelligibility was assessed in various ways across studies. For example, 3 studies employed formal tests for evaluating speech intelligibility: Frenchay Dysarthria Assessment 19 and Computerized Assessment of Intelligibility of Dysarthric Speech. 12,21 Four studies 25,26,30,31 consistently utilized Hirose’s 10-point scoring system as a speech intelligibility measure, which involves a combined scoring of the patient’s speech understandability by family and by others on a 5-point rating scale (5 points, clearly understood; 4 points, occasionally misunderstood; 3 points, understood only when subject is known; 2 points, occasionally understood; 1 points, never understood). Yoshida et al. 22 estimated speech intelligibility by having patients produce 100 Japanese syllables and 10 unknown listeners transcribe the sounds; the mean percentage of correct responses from the listeners served as an index of speech intelligibility. A similar approach was used by Katoh et al., 24 in which speech intelligibility was estimated by having 5 speech therapists score the patient’s production of 100 Japanese phonemes. Chung et al. 18 assessed speech intelligibility based on the percentage of consonants correct calculated from a formal articulation/phonology test (Korean Urimal Test of Articulation and Phonology). Inoue et al. 27 reported “speech clarity” through analyzing the patient’s articulatory function during a story reading and during the production of 100 monosyllabic Japanese words. While all patients were found to be able to “speak intelligibly,” no detailed information was provided on how speech intelligibility was assessed in the Sinha et al. study. 20 Katoh et al. 24 and McCombe et al. 19 additionally included patient-reported questionnaire on speech ability/intelligibility/severity in addition to the perceptual speech evaluation.
Seven studies described the patients’ resonance characteristics, although varying in detailed approaches. According to Bohle et al., 12 a speech-language pathologist rated the patient’s voice and resonance characteristics during a passage reading using a 5-point Likert scale. Two studies 28,30 included a binary rating system (presence vs absence) of hypernasality as part of perceptual evaluations. Sinha et al. 20 described some patients exhibiting hypernasality or hyponasality without any details on the resonance assessment approach. Bohle et al. 12 and Hamahata et al. 25 included the presence or absence of hypernasality based on patient-reported questionnaires and complaints. Sims and Moore 26 listed VP insufficiency (VPI) as one of the long-term complications among patients with head and neck cancer in their retrospective study, and yet, no details were provided on the nature of VPI or its clinical manifestations.
Seven out of 14 studies incorporated instrumental evaluation to assess VP function. Conventional instrumental measures included nasometry, 18,21 pressure-flow technique, 12,21 and nasendoscopy. 18,24 Somewhat unconventional instrumental measures were also reported in the reviewed literature, which included the relative band energy of the formants as an estimate of nasality, 23 soft blowing/blowing ratio to estimate the extent of nasal emission, 24,27,30 and manometry-based oropharyngeal pressure measures. 24
Swallowing Assessment and Outcome Measures
Swallowing outcomes were reported in 10 out of 15 studies. Outcome measures utilized for VP deficit in swallowing ranged from patient reports of food or liquid escape into the nasal cavity, diet outcomes questionnaires and surveys, manometry, blowing test, and conventional imaging techniques such as FEES and VFSS.
Eight out of 10 studies utilized patient-reported outcomes that included patient interview, 26 Swallowing Ability Scale, 24 patient questionnaire survey, 19,27,30,31 Amyotrophic Lateral Sclerosis Scale (ALSS), 19 patient-reported diet outcomes. 20,25 Sims and Moore 26 utilized Functional Outcome Swallowing Scale (FOSS) and presence or absence of percutaneous endoscopic gastrostomy (PEG) tube as outcome measures. The ALSS is a patient-reported outcome scale utilized to assess the patient’s view on speech and swallowing changes, originally developed for use with the ALS population and does not have specific questions related to VPD. McCombe et al. 19 incorporated a combination of perceptual (ALSS, visual analog scale) and instrumental (VFSS and FEES) to assess VP function.
Six studies utilized instrumental evaluation of swallowing via VFSS and/or FEES to assess swallowing. 19,20,24,27 -29 All 6 studies reported some level of VPI ranging from mild to gross nasopharyngeal escape. Katoh et al. 24 additionally reported oropharyngeal pressure measures based on manometry.
Discussion
Velopharyngeal function is an important element associated with speech and swallowing outcomes in head and neck cancer. The multiple aspects involved in VP movement make it a challenging area to study especially when multi-modality treatment methods have very different effects on function. The current review revealed a total of 15 studies that included speech outcomes, swallowing outcomes, or both in relation to VP function in head and neck cancer. Revisiting the purpose of this review, VPD is anticipated in patients with an obvious palate resection and/or those who undergo radiotherapy wherein the field of radiation encompasses soft tissue, including the underlying muscles, of the VP region. However, VPD was also reported when treatment sites include neighboring structures such as base of tongue and posterior pharyngeal wall, making it important to include VPD assessment when evaluating patients with tumors of the posterior oral cavity as well. Radiotherapy-related ramifications on VPD, while noted clinically, were not systematically reported. It would seem apparent that VPD would be more prevalent in patients treated for cancers of the nasopharynx, hard and/or soft palate, and lateral pharyngeal wall. However, it is essential to note that certain oropharyngeal tumors (e.g., palatine tonsil) may require surgical resection of the palate to obtain clear margins. Clear resection margins (i.e., complete resection) are associated with improved curative disease free survival in head and neck cancer and influence the decision regarding adjuvant radiotherapy with or without chemotherapy. 32,33 While adding radiation or chemoradiation therapy can offer superior curative outcomes, the consequent connective tissue fibrosis can result in drastic functional changes. 34 In fact, adjuvant radiotherapy has been identified as an independent predictor of speech and swallow dysfunction. 35 Taken together, it is vital to extend VPD assessment when evaluating patients with head and neck cancer with tumor location beyond the palatal and nasopharyngeal structures and treatment modalities beyond surgical resection.
Reported speech and swallowing outcomes varied across studies, ranging from patient-reported and perceptual outcomes to objective/instrumental assessment, but were largely skewed towards speech-related perceptual measures and swallowing-related patient-reported outcomes. There is value in understanding the use of certain outcome measures. Clinical impressions of perceptual assessments and patient reports are often what guides appropriate referrals in a clinical setting and therefore the use of speech intelligibility tests is worthwhile. However, its value in estimating VP function is limited unless coupled with instrumental measurements. The established standard of care for VP function among patients with cleft palate and craniofacial disorders includes a combination of perceptual resonance and speech intelligibility assessments and instrumental assessment with the use of nasometry and VP imaging. 36,37 Unfortunately, no such standardized clinical protocol evaluating VP function in patients with head and neck cancer was observed in the reviewed studies, rendering it difficult to draw equal comparisons across studies. It is the understanding of pathophysiology that carries the clinician forward towards treatment and management. The paucity in understanding the physiologic underpinning is likely a contributing factor to the lack of clarity among clinicians in evaluating and treating VPD in head and neck cancer.
Additionally, novel evaluation methods add another layer of variability in understanding the deficit. For instance, the efficacy of a “blowing test” was examined in one study, 24 where the authors attempted to determine if this clinical test could predict VP function for speech. The blowing test requires the individual to blow air into a cup filled with water to a particular level under 2 conditions: nares closed and nares open. A blowing time ratio is determined by dividing blowing time with open nose condition over closed nose condition. This measure, without any validation in relation to an external criterion procedure (i.e., established instrumental assessments of VP function), was then correlated with a perceptual speech intelligibility test. On the one hand, a nonspeech blowing test provides little insight into the VP function for speech as nonspeech static oral movement is different from the highly coordinated and precise gestures required for speech. 7,38,39 On the other hand, sole reliance of syllable or word intelligibility tests may not translate into overall speech intelligibility of connected speech. Despite the obvious site of resection, the salient feature (VPD) affecting speech intelligibility was not comprehensively evaluated in a large portion of the literature.
Transitioning to swallowing outcomes, the complexity of the musculature involved in achieving VP closure for swallowing as well as changes in oral cavity dynamics with respect to sensory feedback would demand a more comprehensive understanding. However, no study included in this review reported specific information regarding VP function on swallowing with the use of imaging studies such as VFSS or FEES. While most utilized clinical report of nasal regurgitation during meals, the details of cranial nerve assessments, details of surgical changes and bolus trials utilized in clinical swallow evaluation that can illustrate the mechanism of deficits were also limited. The use of VFSS in the assessment of these patients is necessary in order to understand the pathophysiology.
Both Sinha et al. 20 and Hamahata et al. 25 utilized diet outcomes post-operatively as a measure of swallowing outcome. Patients were merely asked to report on their oral intake and a report of eating a regular diet would be scored as “no difficulty.” Taking a step back from VPD, it is important to realize that subjective report of diet outcomes and patient reports of difficulty swallowing may not accurately portray intact or impaired swallowing physiology. There appears to be lack of correlation between patient reported outcomes in swallowing and the true physiologic swallowing outcomes noted under imaging . 40,41 For example, Arrese et al. 40 examined the relationship between patient-reported outcomes on the Eating Assessment Tool-10 (EAT-10) and VFSS physiologic findings among patients who underwent head and neck cancer treatment. The findings relayed that these patients often under-reported their deficit. Similarly, Kirsh and colleagues 41 reported reduced patient awareness of compromised swallowing function following chemoradiation.
Despite the variability in assessment of VPD across the various studies, some general conclusions can be drawn from the results relevant to the scope of this paper. Over 2/3rd of the reviewed studies reported that greater tumor burden along the soft palate with >50% resection and reconstruction are more often correlated with poorer outcomes for both speech and swallowing. 18,19,21 -28,30 The greater presence of soft palate is associated with improved intelligibility. Studies that assessed functional oral intake outcomes reported a high number of patients who were able to return to oral diet 19,20,26,27 with 4 studies reporting between 14 and 23% of patients reporting or identifying some form of VPI. 28,29,31 With respect to radiation associated VPD, it appears a radiation dose of >55 Gy is associated with increased nasality on careful acoustic analysis. 23
The deficit in the use of standard perceptual and instrumental evaluation of VPD is enlightening and helps clinicians understand barriers in VPD assessment and management for patients undergoing treatment for head and neck cancer. In the grand scheme of the review, the need for physiology-based outcome measures with respect to speech and swallowing functions is apparent. There was and continues to remain a heavy reliance on patient-reported outcomes alone to account for “improvement.” In addition, the variability in measurements and lack of general standard of care in the assessment of VP function make it challenging to compare outcomes across studies. Despite the limited number of studies, it is evident that patients may present with VPD at any stage in cancer treatment (i.e., post-surgery, post-(chemo)radiotherapy, late radiation effects). Identifying the unique impact of surgery, radiation therapy, and combination of both effects through establishing a standard-of-care evaluation methodology for VPD can help in guiding clinical and multi-disciplinary treatment to result in effective measurable gains post-cancer treatment for this patient population.
For future direction and for clinicians seeking to improve evaluation, we recommend a combination of perceptual (e.g., patient-reported outcomes and speech intelligibility) and instrumental evaluations (e.g., pressure-flow testing or nasometry and manometry for speech and swallowing, respectively, along with VP imaging) would be ideal to create a profile of speech/swallowing function for this unique population. At the authors’ institution, a combination of Speech Handicap Index, Assessment of Intelligibility of Dysarthric Speech, and nasometry is utilized to complete a comprehensive assessment of speech for patients with head and neck cancer. VFSS along with the standardized MBSImP rating scale is utilized to assess impact of VPD on swallow function. As is the case with head and neck cancer, for patients who undergo radiotherapy, recent statistics report approximately 5% of all patients treated will present with late radiation effects in 5 years post-treatment and approximately 12% in 12 years. 42 A salient feature of late radiation effects includes VPD in the setting of multiple cranial nerve neuropathies. Optimizing function for patients undergoing late radiation effects includes a prosthodontic evaluation for devices such as palatal lift. The efficacy of VPD management in patients undergoing late radiation changes is limited due to the limited understanding of changes and methods to manage the overall oropharyngeal anatomic changes. While exercise-based therapies are not well supported for targeting VP function, preliminary work by Hutcheson et al. 43 revealed increased soft palate activation following a course of expiratory muscle strength training. An extended scope would be to create a physiologically based profile of VPD across the continuum of head and neck cancer treatment: a profile that includes objective speech and swallowing outcomes post-treatment with attention to location, size of tumor and sets a standard for assessment of VP function.
Footnotes
Author Note
Part of this study was presented at the American Speech-Language-Hearing Association (ASHA) annual convention, Orlando, Florida, November 2019.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
