Abstract
Keywords
Introduction
Geriatric dysphagia is a disease frequently misunderstood, with symptoms often blurred. In addition, the dysphagia prevalence varies according to age, estimated at 15% in subjects over 65, while it exceeds 30% over 80 years old.1-3
The elderly patient commonly presents a reduced swallowing reserve, a paraphysiological condition defined as presbyphagia.4,5
Aging through central and peripheral mechanisms involves a reduced muscle tone and a sensorineural defect of taste and smell.6,7
However, if elderly subjects without further comorbidities do not necessarily correlate with the genesis of swallowing disorders, cognitive deficits could favor dysphagia onset.8-10 Indeed, elderly patients with cognitive impairment seem to have an increased risk of swallowing disorders, frequently involving the oropharyngeal phase.11,12 However, this topic is low investigated in the literature.
As discussed by Alagiakrishnan et al., dysphagia prevalence differs according to the cognitive impairment subtypes and the onset timing of the cognitive disease. 13
Recently, Michel et al. hypothesized that buccofacial apraxia in elderly patients with neurodegenerative diseases as Alzheimer’s disease or frontotemporal cognitive impairment might contribute to oropharyngeal dysphagia disorders. 14
To confirm limited evidence reported in the literature, we carried out a case-control study to evaluate the possible correlation between cognitive impairment and oropharyngeal dysphagia in elderly subjects.
Materials and Methods
We performed a retrospective study at the ENT Unit of the University Hospital of Catania from 2010 to 2020. We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). We initially considered for the study inclusion elderly patients (≥65 years) sent to our ENT Unit to perform a swallowing evaluation. The study design is summarized in Figure 1. Flow Chart study design.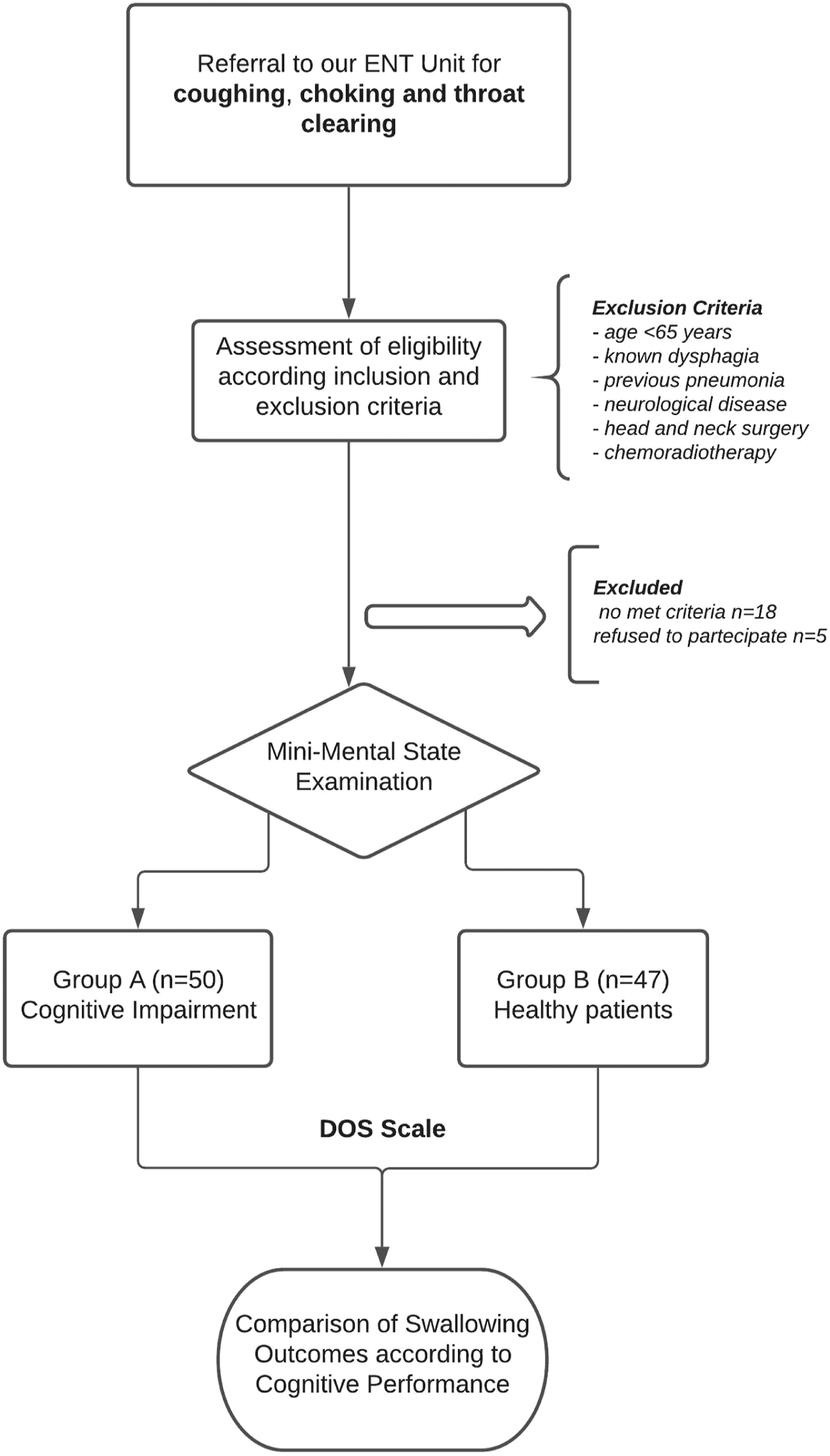
All subjects were outpatients reporting choking or throat clearing symptoms after eating, coughing, and difficulty swallowing certain foods.
Patients with the following features were excluded from the study: - Age <65 years; - Patients with a previously diagnosed dysphagia; - Previous pneumonia; - Subjects with a clinical history positive for several neurological diseases such as stroke, Alzheimer’s, or Parkinson’s disease, myopathy, myasthenia gravis, amyotrophic lateral sclerosis, or brain-stem lesions; - Previous head and neck surgery or chemoradiation treatment.
The first phase of the study involved a neurological evaluation of all patients considered to diagnose cognitive impairment. Subsequently, subjects underwent swallowing assessment through history, clinical examination, and fiberoptic swallowing evaluation (FEES). Diagnosis of cognitive impairment was made with a neurological evaluation and a Mini-Mental test examination (MMET).
Cognitive Assessment With MMET
The MMET is a 30-point test usually administered to measure thinking ability and evaluates patients’ cognitive impairment risk. 15 The test consists of thirty items referring to seven different cognitive areas: orientation over time and in space, words recording, attention and calculation, re-enactment, language, and constructive praxia. We administered the validated Italian version. 16
According to the MMET score, patients were divided into the following groups: - Normal status, MMET score >23—mild cognitive impairment, MMET score from 23 to 20—moderate cognitive impairment, MMET ranging from 19 to 11—severe cognitive impairment, MMET score <11.
After the MMET evaluation, we divided the patients into two different groups: - Group A, patients diagnosed with cognitive impairment under the results of the MMET—Group B or control, made up of elderly patients without cognitive impairment at the initial assessment.
Swallowing Assessment
Oropharyngeal dysphagia was established by irregular swallowing physiology estimated by a clinical, bedside screening test, and consequently confirmed by FEES assessment in all patients included in this study. Oropharyngeal dysphagia screening was performed by a trained Speech-Language–Pathologist (SLP) through a clinical evaluation of the patient’s features before and after water intake. Thus, bedside examination was performed via the dysphagia risk score (DRS). 17 Consequently, two different otolaryngologists experienced in the swallowing evaluation assessed dysphagia severity through flexible endoscopic swallowing evaluation (FEES) (C.S., G.A).
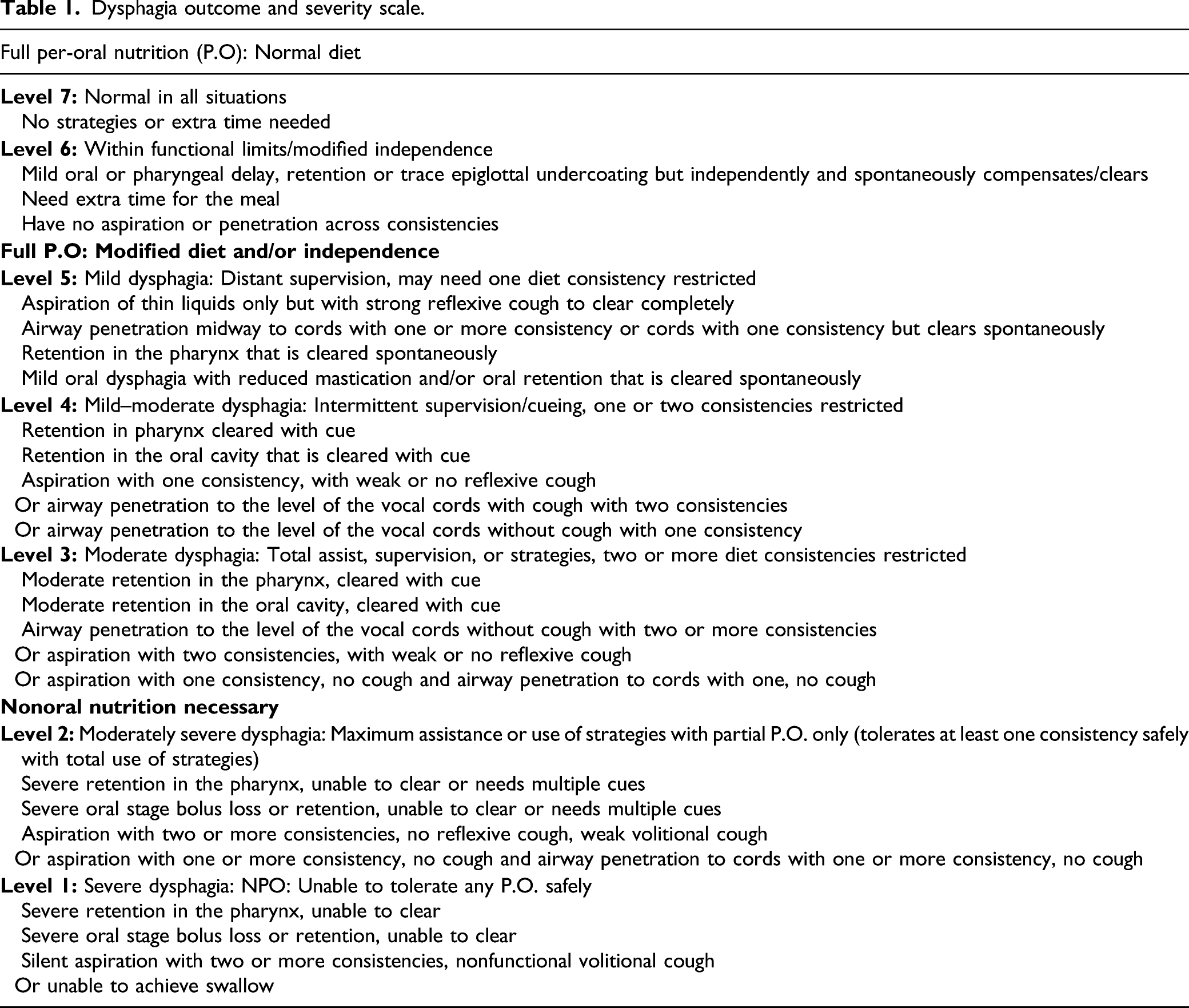
Dysphagia outcome and severity scale.
Flexible Endoscopic Swallowing Evaluation Protocol
Participants were offered three tests of thin liquid and three tests of thick liquid. In each test, we administered 10 cc of water with thin liquid and 10 cc with thick liquid of aquagel (Nestlè Nutricia Nutilis®), dyed with Methylene Blue for food. The hard and soft solid textures were evaluated in the absence of signs of dysphagia with thin or dense liquids. We rated solids ingestion with a banana as a soft food and a cracker as a hard one. The same food consistencies were administered in all patients analyzed.
Statistical Analysis And Data Comparison
Data analysis was performed using IBM SPSS Statistics for Windows, IBM Corp Released 2017, Version 25.0. Armonk, NY: IBM Corp. Descriptive statistics were reported on average ±standard deviation or proportion. The t-test for paired samples was used to determine the difference between observations for normally distributed data. The Mann–Whitney U test was performed to analyze group differences for continuous skewed data.
The ANOVA test assessed the differences in MMET and DOSS levels between the groups. Moreover, we also evaluated the different dependent variables such as age, sex, and marital status that could influence the swallowing function of the patients enrolled.
Linear regression was used to estimate the linear relationship between a dependent and an independent variable. The ethics committee approved our study protocol. Participants were informed and gave written informed consent of the study’s purpose and procedures, which were conducted according to the Declaration of Helsinki.
Results
Main demographics features of both groups included, * DOSS level comparison, swallowing disorder prevalence was higher in cognitive group (normal vs dysphagic; P-value = .001).
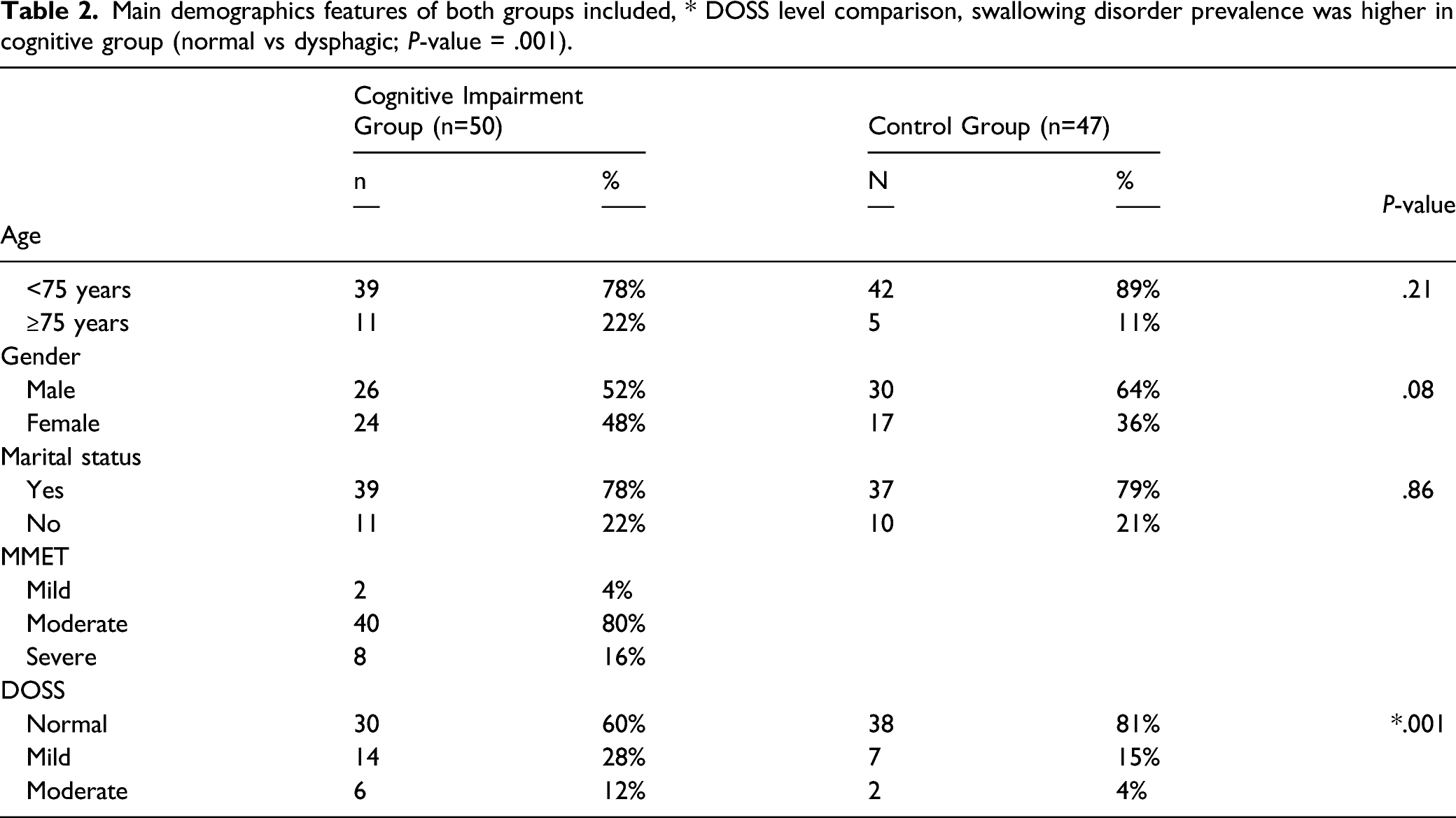
In patients with cognitive impairment, the mean MMET score was 14.6 ± 3.6 (range 6-21), while the control group reported a mean score of 26.2 ± 1.4 (range 24-29) with a significant difference between groups (P < .001). Patients with cognitive impairment on MMET presented the following severity score distribution: 2 (4%) mild, 40 (80%) moderate, and 8 (16%) severe.
The cognitive impairment group reported a significantly higher rate of swallowing disorders than controls (40% vs 19%; P = .001). Furthermore, when comparing DOSS outcomes, among patients in the cognitive group were identified 30 (60%) healthy subjects, 14 (28%) mild dysphagia, and 6 (12%) moderate; instead, in the control group, we found 38 (81%) healthy subjects, 7 (15%) mild dysphagia, and 2 (4%) moderate.
Furthermore, patients with cognitive impairment subdivided according to swallowing functions demonstrated statistically significant differences in MMET scores (P < .001; P < .001; P = .01) (Figure 2A). a) Box plot according Mini-Mental test examination (MMET) and Dysphagia Outcome Severity Scale (DOSS) outcomes in cognitive impairment group found statistical difference among levels (P < .001; P < .001; P = .01). b) Box plot according MMET scores and DOSS levels in control group. No statistical significance was recorded among different subgroups (P = .27; P = .73; P = .77).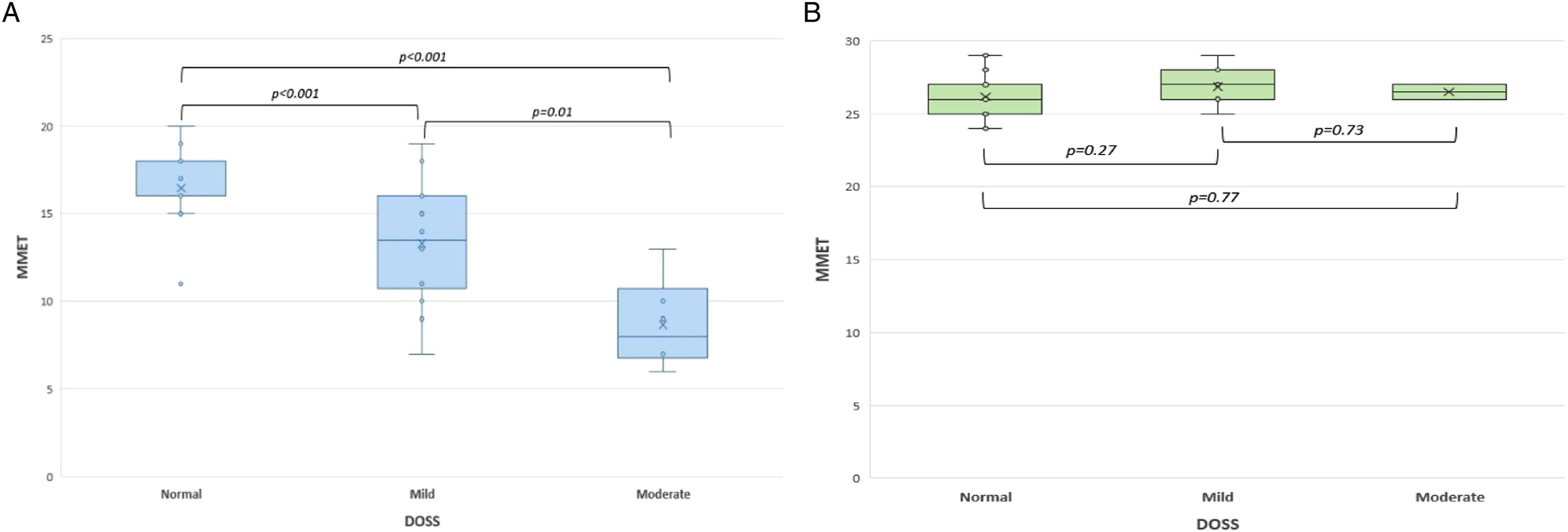
On the contrary, in the control group of healthy elderly patients, no significant differences in MMET scores were reported according to the swallowing performances subgroups (P = .27; P = .73; P = .77) (Figure 2B).
At ANOVA test among patients with cognitive impairment age>75 and MMET<10 were significant dependent variables (F=3.862, P = .028; F=17.49, P = .000). Other factors as marital status or gender were not significantly associated with dysphagia severity (P > .05 for all). Instead, among the control group, marital status was found significantly associated with swallowing disorders (F=4.722, P = .014).
Moreover, a linear correlation was found in the presence of cognitive impairment, with better swallowing function in aPatient With Higher MMET Scores (P < .001) (Figure 3) Linear regression model comparing MMET and DOSS. A significant correlation was found between the two parameters, with better swallowing function in patients with higher MMET scores (P<.001).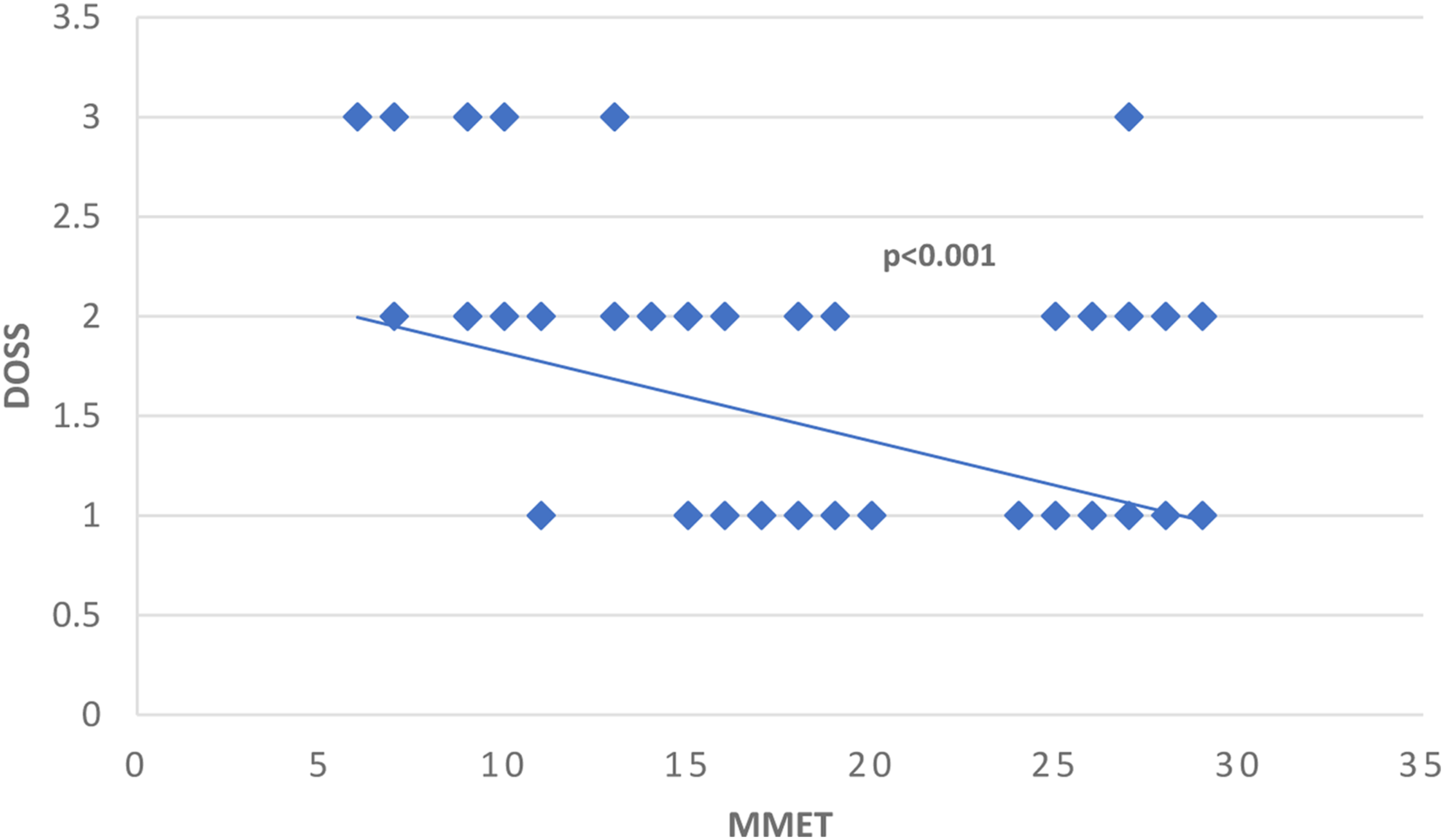
Discussion
Elderly patients have a physiological reduction in muscle mass, including that critical for chewing and swallowing, affecting the ability to manage fluids and increasing the risk of aspiration.19-21 Swallowing disorders in older subjects are frequent and often result from the higher prevalence of chronic diseases or part of the so-called geriatric syndrome. 22 Shimazaki et al. demonstrated poor swallowing function in over 60% of prefrail and frail older patients (OR = 6.4; OR = 10.2, respectively). 23 Besides, as demonstrated by de Lima Alvarenga et al., within a healthy population, some elderly subjects may suffer from swallowing disorders. 24 Interestingly, the authors reported at fiber optic endoscopic swallowing evaluation a pharyngeal residue in 39% (CI 29.4-48.6), laryngeal penetration in 9% (CI 3.4-14.6) up to laryngeal tracheal aspiration in 2% of the subjects enrolled (CI 0-4.7). Moreover, aging interacts in multiple swallowing processes, from the first oral phase with a reduced muscle activity of the tongue, the delay in the swallowing reflex triggered by pressure at the faucial’s pillars up to the gradual sensorineural loss of taste and smell. 25 The physiological swallowing function also requires an adequate cognitive awareness, characterized by the visual recognition of the food, the planning, and the subsequent triggering of the swallowing act. 26 Consequently, the cognitive decline might play a role in the dysphagia genesis of elderly patients, frequently associating with poor nutritional status and increased aspiration pneumonia risk. 27 However, the correlation between dysphagia and cognitive impairment remains debated, and insufficient data are reported in the literature.8-12
In this regard, Park et al. found in 395 older adults ranging from 65 to 103 years old a dysphagia prevalence rate of 52.7% (208), with 41.1% (162) patients at high risk while 11.6% (46) moderate. 28
Later, Maeda et al. reported that the Mini-Mental State Examination score had no impact on hospital-associated dysphagia (beta = .063, P = .378) in a retrospective study on 249 geriatric patients. 29
In contrast, our study demonstrated a linear correlation between mild to moderate oropharyngeal dysphagia and cognitive impairment, with worse swallowing function when the MMET scores were lower (Figure 2A) (P < .001).
According to our results, Yatabe et al. noted in both dentulous and edentulous patients with higher MMET scores a significantly lower probability of risk of dysphagia (OR = .87, 95% CI = .80–.96; OR = .92, 95% CI = .83–1.00 respectively). 30 The correlation between oropharyngeal dysphagia and the degree of cognitive impairment has been investigated by Michel et al. 14 The authors demonstrated that patients with buccofacial apraxia presented both more frequent oropharyngeal dysphagia (P = .04) and a significantly lower MMET score (P = .01) than the control group.
Differently, other authors reported that in hospitalized patients undergoing rehabilitation with dysphagia, the severity of cognitive impairment at the Mini-Mental State Examination had no impact. The cognitive decline also removes all of these mechanisms, often frequent in dysphagia patients trying to adapt to reduced swallowing reserves, such as changing food quality or reducing food during meals.27-30 Additionally, the cognitively impaired patient may not be aware but exhibit significant in-direct signs of dysphagia, such as an irritating cough, choking, or clearing.
Numerous additional agents in the literature are expressed as to potential determinants of swallowing disorders' severity. Szu et al. in 2017 analyzing the determinants of dysphagia in stroke patients, identified as significant factors age, marital status, stroke site, and cognitive status (MMET). 31 Conversely, our study found marital status or gender not significantly associated with dysphagia severity in elderly patients (P > .05 for both).
The different significance of the reported dependent variables might be due to numerous confounding factors in the study protocols, inadequate patient selection criteria, small study samples, and low-evidence study design.
As described in the literature, elderly patients exhibit an increased risk for unrecognized swallowing disorders, with a prevalence of mild to moderate forms. Furthermore, this risk may be associated with poor cognitive performance, as reported by our study. Yet, the main limitation of our investigation is precisely due to the challenging identification and missing assessment silent dysphagia subjects. Therefore, it would be necessary to establish new evaluation protocols and diagnostic algorithms by integrating FEES as a first-line method.
Another obvious limitation is the use of WST to rule out overt aspiration, not offering sufficient screening accuracy. An interesting metaanalysis carried out by Brodsky et al. suggested that individual sips may improve the WST efficacy, especially when clinical signs of aspiration are present. 32 Moreover, the combination of the airway with the vocal response could enhance the diagnostic precision of WST.
An additional procedural limit may be inherent in choosing the best tracer in the evaluation by FESS. As performed in our study, the use of boluses of water colored with blue dye could sometimes make it difficult to identify thin liquid dysphagia on FEES. Indeed, blue water may only appear in the presence of copious amounts on the vocal cords, reducing the effectiveness of endoscopic evaluation.
Conclusions
Elderly patients have a high risk of mild to moderate dysphagia. As assessed by MMET scores, impaired cognitive function may be related to unrecognized swallowing disorders, with a prevalence of mild to moderate forms. Greater clinical awareness must be provided in subtle dysphagia symptoms, especially if related to impaired cognitive functions. Therefore, assessing cognitive performance through MMET could be a useful tool for clinicians and geriatricians to direct patients to in-depth diagnostic and swallowing evaluation.
Footnotes
Authors’ Note
Conceptualization, A.M. and S.C.; methodology, G.I.; software, A.M., E.P., S.C.; validation, C.V., G.M. and A.P.; formal analysis, JR. L, G.C.; investigation, G.A.; resources, A.M.; data curation, C.S.; writing—original draft preparation, A.M.; writing—review and editing, I.L.M.; visualization, C.G.; supervision, S.F.; project administration, S.C.; All authors have read and agreed to the published version of the manuscript.
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained from all subjects involved in the study.
