Abstract
Objective
To test the applicability of a fresh tissue model for teaching facial plastic techniques and approaches to the eyelids and orbit.
Design
Observational animal experiments.
Subjects
Ten prepubescent sheep heads harvested following humane euthanasia at the completion of unrelated live animal research.
Methods
Young sheep were saline perfused at the end of an in vivo protocol. Head and neck tissues were harvested and refrigerated for 3–7 days. An experienced oculoplastic surgeon and an otolaryngologist explored the feasibility of common oculoplastic procedures in the ovine model.
Results
The model has potential for teaching basic principles in eyelid surgery including upper lid blepharoplasty, aponeurotic ptosis repair, upper lid gold weight lid loading for facial paralysis, lateral canthotomy and inferior limb cantholysis, lower lid tightening, and transconjunctival approach to the orbital floor. Eye muscle advancement, optic nerve sheath fenestration, and enucleation also accurately simulated human surgery. Anatomic variations limit the sheep model for orbital floor reconstruction and lacrimal drainage procedures.
Conclusions
The sheep head and neck provide an inexpensive, safe model for developing skills in several oculoplastic procedures. Formal simulation testing is needed to confirm these expert opinions.
Introduction
Developing skills in eye and facial plastic surgery poses a special challenge in otolaryngology resident training. Facial plastic surgery patients are often awake and aware during surgery and demand near-perfect results. In such a setting, hands-on resident surgical training is limited.
Simulation has gained wide acceptance in medical student and resident education—providing trainees with first-hand exposure to emergency situations and complex procedures while minimizing the risk to patients. This is particularly true in surgical training, where simulation allows students to acquire mechanical skills and teachers to document technical competence in a standardized fashion. 1 Fresh animal globes are a widely used model for teaching intraocular surgery in ophthalmology training. 2 No similar, standard animal model exists for surgery of the eyelids and orbit.3-5 Fresh human cadavers can be used for resident oculoplastic training but are expensive and pose substantial risks of infectious disease transmission. 6
Our group described the use of a fresh, saline-perfused sheep head and neck model for surgical simulation in Otolaryngology—Head & Neck Surgery. 7 This article explores the potential and limitations of this model in surgery of the eyelids, lacrimal system and orbit based on study of comparative anatomy and a series of dissections by experienced facial plastic surgeons.
Methods
Tissue was collected from prepubescent sheep (n = 10; mean age: 4 months; mean mass: 28 kg) following humane euthanasia (100 mg/kg sodium pentobarbital solution) at the end of an in vivo, protocol approved by the Lewis Katz School of Medicine of Temple University through its Institutional Animal Care and Use Committee (IACUC). These sheep were obtained from a Temple University and United States Department of Agriculture approved supplier, tested for Coxiella burnetii according to Centers for Disease Control and Prevention (CDC) protocol, accepted into the University Laboratory Animal Resource facility of Temple University after full inspection by a licensed veterinarian to assure good health. No live animals were used in this study. Post-euthanasia, as part of Temple University’s tissue sharing program, the sheep vascular system was perfused with cold saline to aid in tissue preservation and slow autolysis. The head and neck of the sheep were disarticulated 4–6 cm above the sternal notch. The tissue was stored at 5°C until ready for dissection—usually 1–5 days.
Prior to working with fresh ovine tissue, study personnel were enrolled in the Temple University’s Occupational Health program and screened for Q-Fever antibodies. They wore appropriate personal protective clothing during experimentation. Sheep tissues were transported in double plastic disposable bags, and all carcass material was treated as biomedical waste through a commercial waste management company.
Operative procedures were performed in the animal facility using a veterinary operating table. Standard oculoplastic and strabismus surgical sets were used. Monopolar electrosurgical instruments functioned well with a grounding pad beneath the moist tissue. The dissectors wore magnifying loupes appropriate for fine work.
The effectiveness of the model was subjectively assessed by the two experienced surgeons who were both present during all of the dissections. The model was judged as useful when sheep anatomy and tissue quality were similar to that in human surgery. The model was judged inappropriate when there were major anatomic differences or marked variations in tissue quantity or quality compared to the human orbit. In cases of disagreement between the experts, both opinions were documented.
Results
Applicability of the sheep head and neck model for several oculoplastic simulations.
Aponeurotic advancement for upper eyelid ptosis.
Canthotomy and cantholysis.
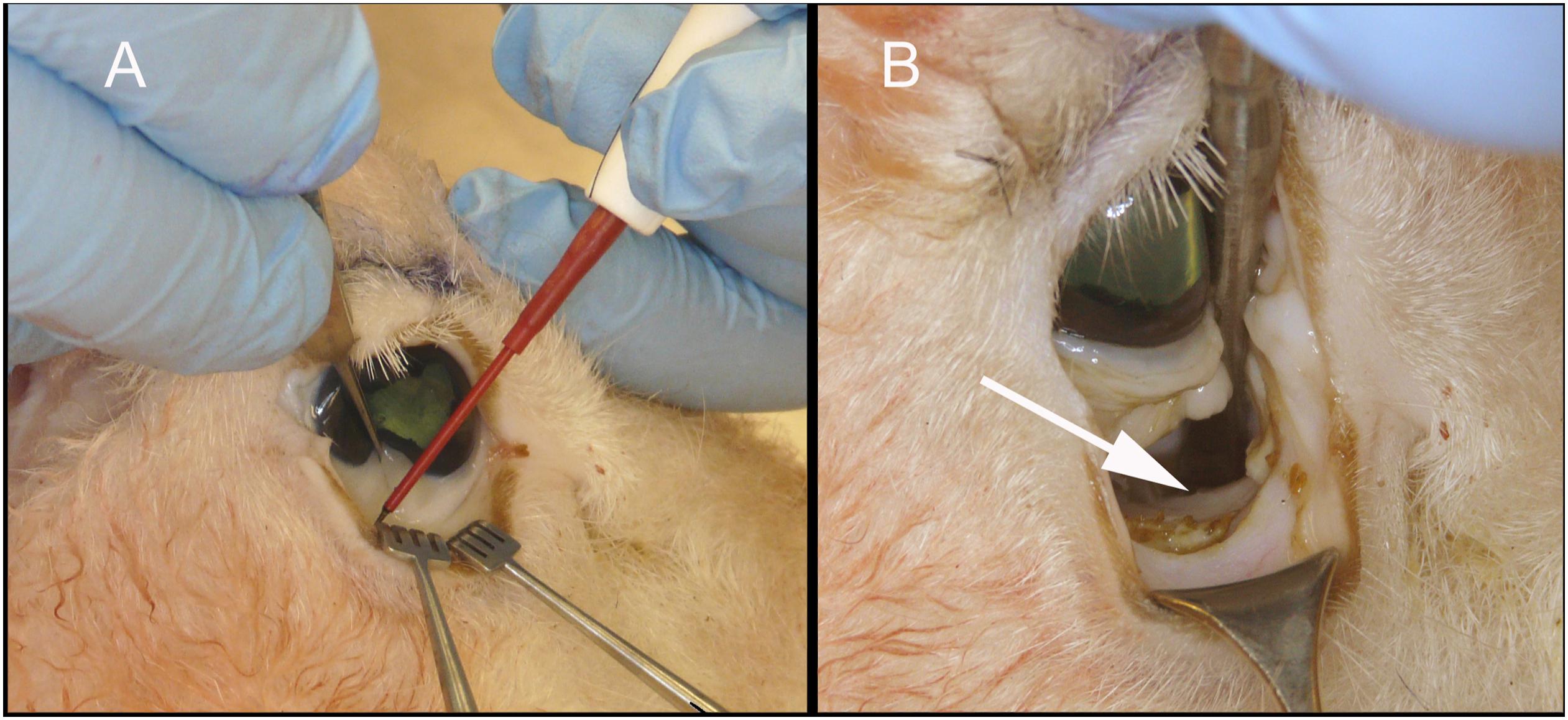
Transconjunctival approach for lower eyelid blepharoplasty or for orbital rim or floor fracture repair.
Description of individual procedures
Upper blepharoplasty
As sheep have fur on their eyelids, the cilia of the upper eyelid skin should be trimmed to facilitate surgical design and incisions. An eyelid crease incision site is outlined along with an elliptical strip of skin to mimic an upper blepharoplasty. The skin is injected with saline to hydraulically dissect skin from the underlying orbicularis muscle. The orbital septum is located high toward the superior orbital margin and transected. Gentle pressure on the globe aids in localization of the nasal fat pad. Fat in sheep upper eyelids is sparse in the upper eyelids in both the nasal and central fat pads compared to that in human surgery. Fat removal can be practiced using standard blepharoplasty techniques, and the skin closed with interrupted or absorbable sutures.
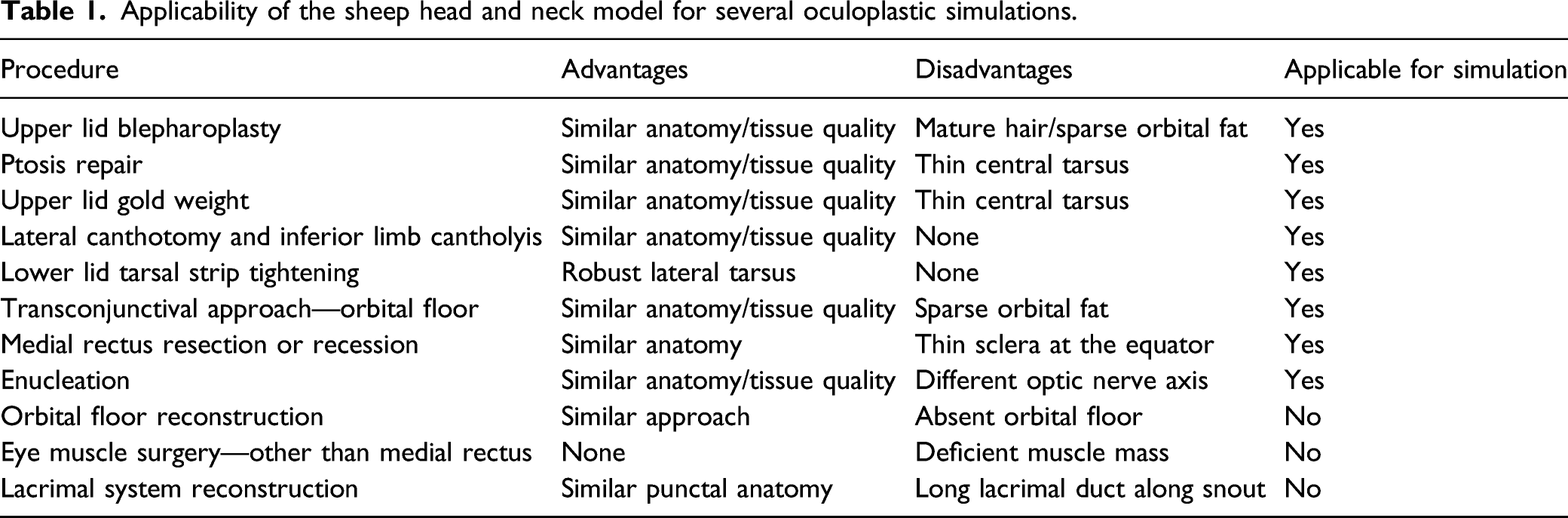
Ptosis repair (Figure 1)
A lid crease incision is made and deepened through orbicularis. Using two forceps to elevate the orbicularis at the junction of the pretarsal and preseptal regions, the orbicularis is divided and dissection is carried superiorly to expose the orbital septum almost as high as the arcus marginalis, until preseptal fat is visualized beneath the orbital septum. In the case illustrated, a Colorado needle (Stryker, Kalamazoo, MI) is employed. The septum is now opened across the upper eyelid and the levator is located. Dissection is also carried out to the tarsus. The tarsus in the sheep is thinner centrally and more closely resembles the human tarsus at its lateral and medial margins, but can be isolated and the levator muscle advanced and sutured using a single or multiple sutures. Lid crease formation can also be practiced using standard oculoplastic techniques, either with supratarsal fixation or by attaching the levator to preseptal orbicularis or skin.
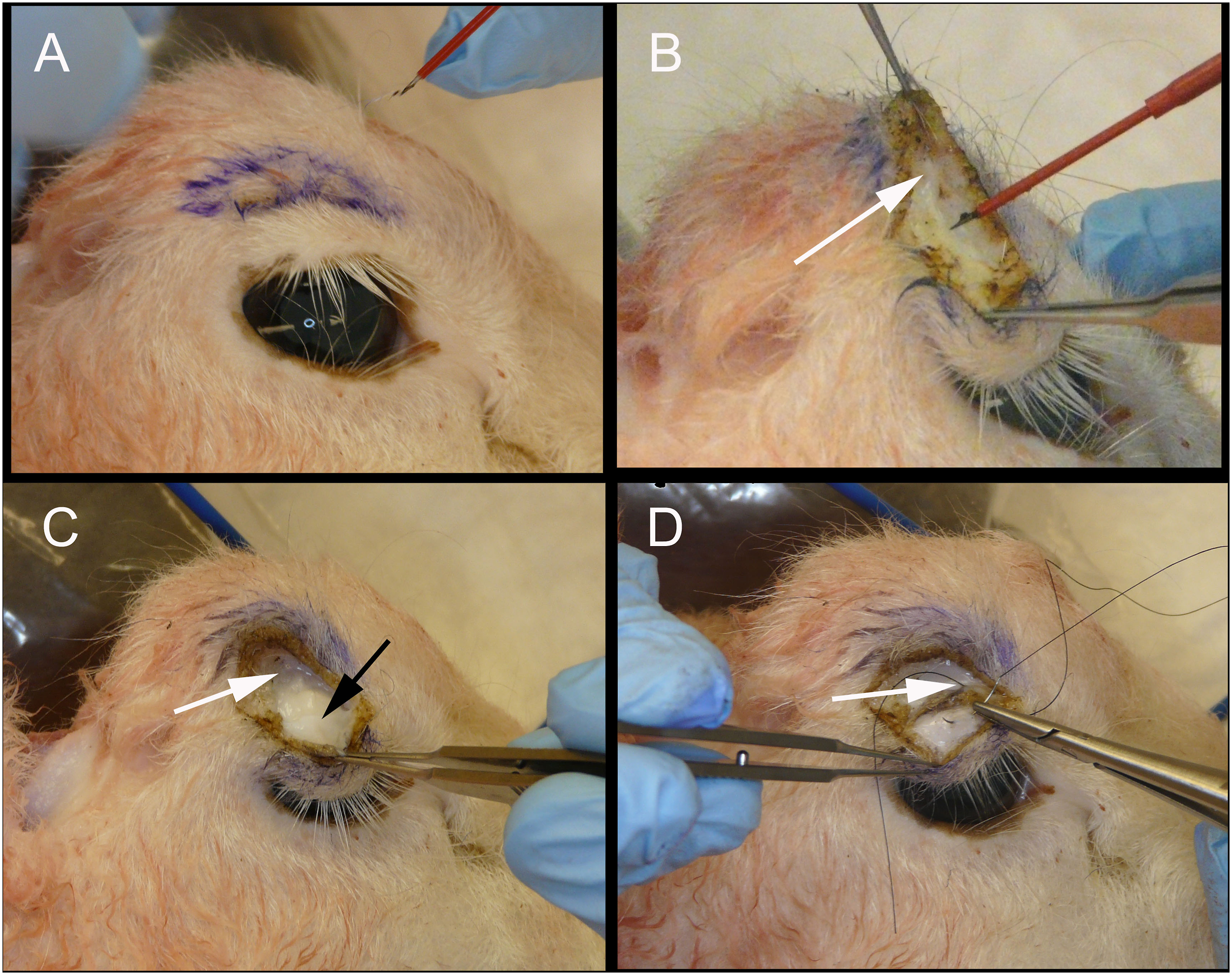
Lateral canthotomy/cantholysis (Figure 2)
The standard lateral canthal procedure with division of the inferior limb of the lateral canthal tendon can be practiced, using an iris scissor, while directly palpating the lower eyelid to experience the “release” of the inferior limb that indicates the success of anterior decompression achieved with successful cantholysis. The tarsus and lateral ligaments are robust and readily apparent in the sheep and allow the neophyte to gain a clear tactile sense of liberation of the lower eyelid from its attachments to the lateral canthal tendon.
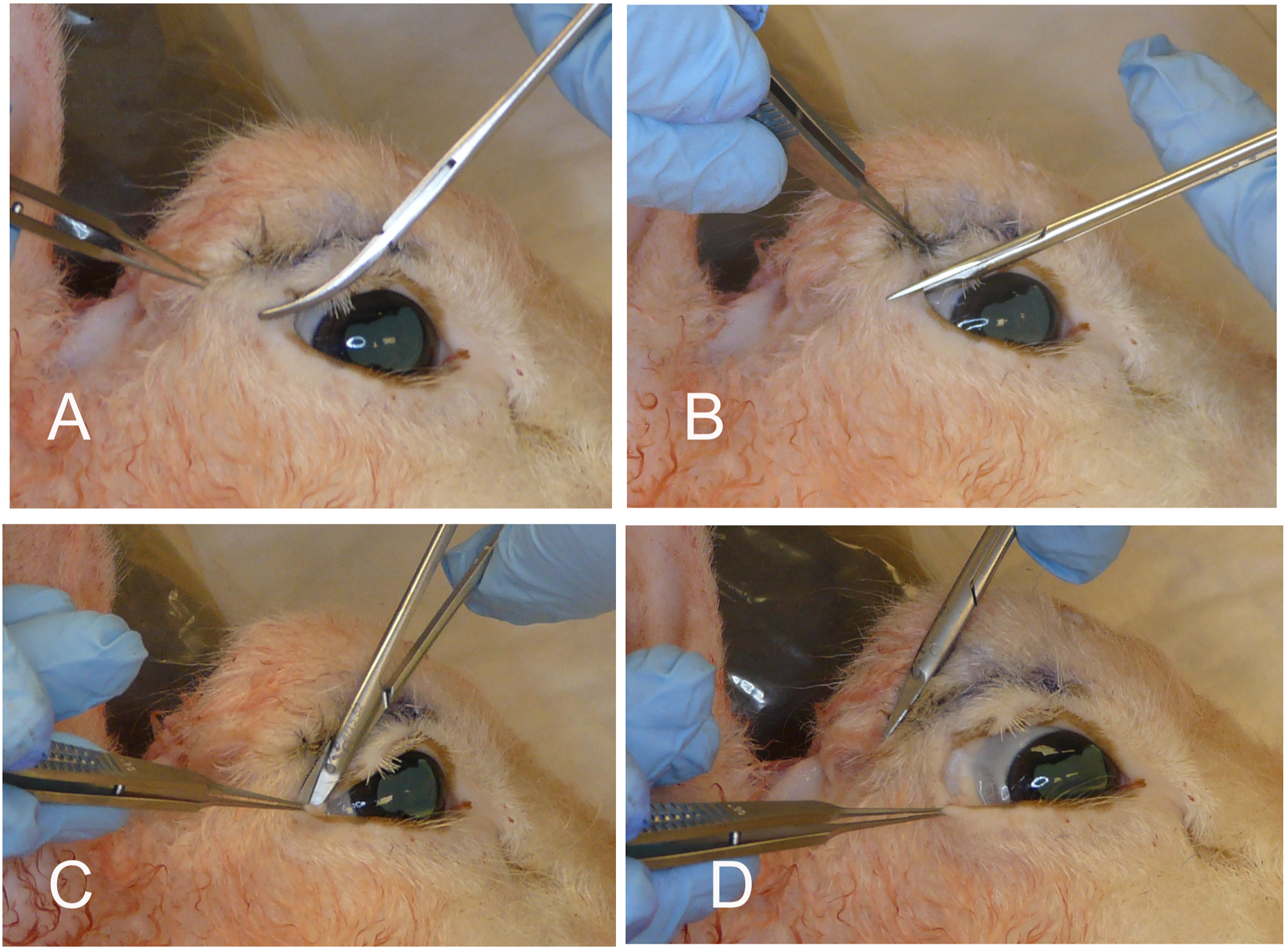
Transconjunctival blepharoplasty, approach to the orbital floor, and the correction of eyelid malpositions (Figure 3)
Fat is sparse in the lower eyelid compared to mature humans. An incision is made, either with a blade or a Colorado needle, above the inferior fornix but below the lower eyelid tarsus, retracting the lower eyelid with 2 small rakes. The lower eyelid retractors and conjunctiva are grasped firmly and the septum is dissected with a cotton-tipped applicator, a peanut, or a Kittner until the arcus marginalis is exposed. The surgeon can now excise fat, as in transconjunctival blepharoplasty, or to sharply incise periosteum and to reflect periorbita using a Cottle elevator to expose the orbital floor. The inferior orbital floor itself plunges beyond the rim, and we did not feel it would be useful for practicing plating for floor fractures.
This approach would also be used in fat transposition procedures, and any fat found in the above dissection can be brought forward and sutured pre- or subperiosteally through the lower eyelid and cheek skin.
Quickert sutures can also be placed to reposition the eyelid in horizontal mattress fashion, creating both an inturned eyelid or an externally rotated eyelid by varying the position of the mattress sutures as they pass from low on the posterior eyelid surface to high on the skin surface (creating ectropion, and thus correcting entropion), or from high on the posterior eyelid surface to low on the skin surface (creating entropion, and thus correcting ectropion). The placement of these sutures clearly illustrates to the resident the role of Quickert sutures in the repair of eyelid malpositions. The conjunctiva can be closed, but is quite diaphanous when compared to the human.
Tarsal strip procedure
As in the standard procedure or its variants, a lateral canthotomy inferior limb cantholysis precedes the creation of a lateral tarsal strip. The tarsus is robust in the sheep. The tarsal strip is anchored to the lateral orbital rim using a double armed horizontal mattress suture. The lateral canthal angle can be reformed with a buried suture through the gray line and the skin can be closed.
Medial rectus resection/recession (Figure 4)
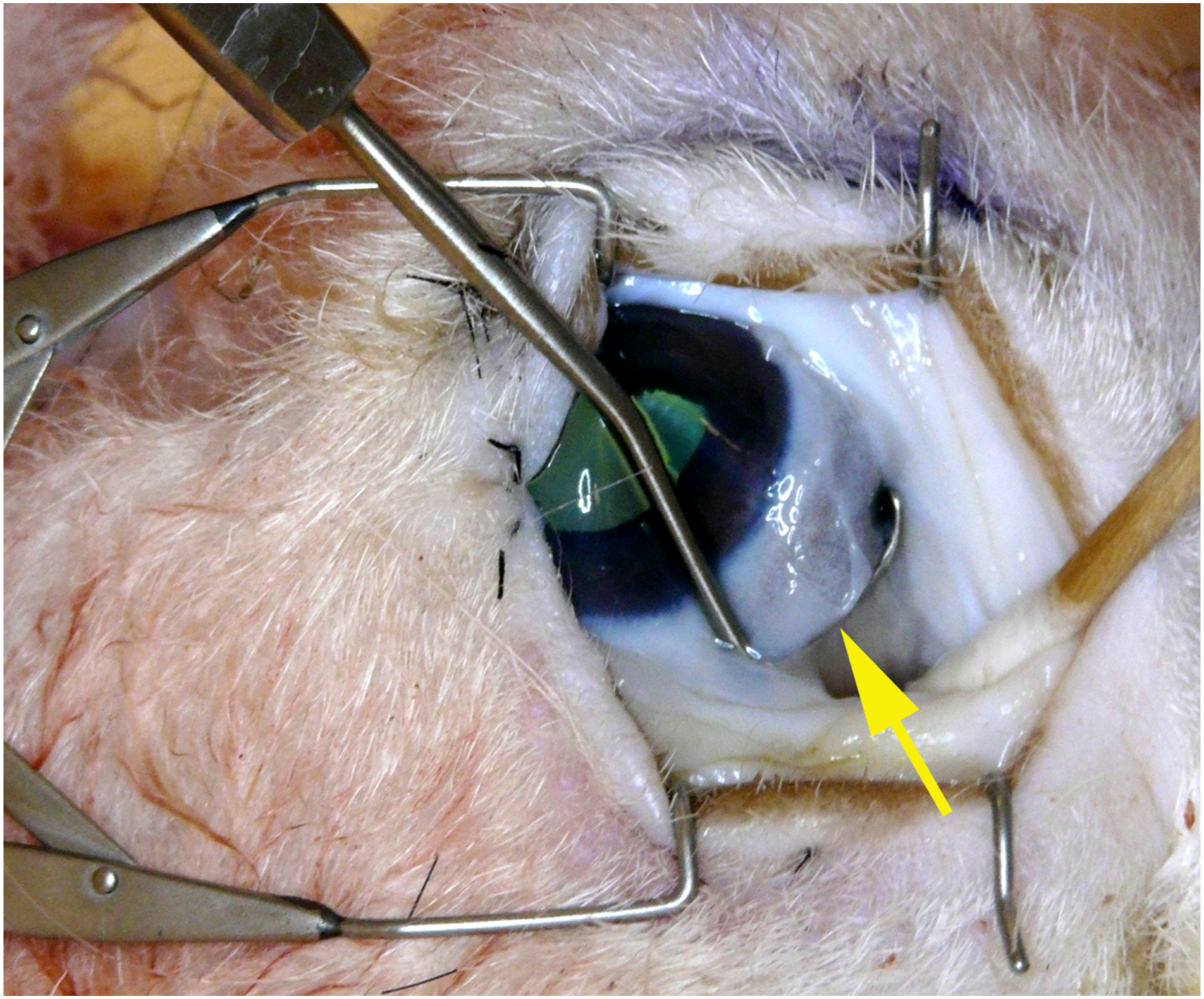
The medial rectus muscle is isolated with a Green eye muscle hook (yellow arrow).
A medial limbal peritomy or a conjunctival forniceal incision is made and thickened Tenon’s fascia (thicker than in the human) is dissected down to the scleral surface. The medial rectus muscle is isolated using standard muscle hooks, and is thinner anteriorly in the sheep than in the human, but easily recognized and isolated. Resection and recession can be practiced using standard techniques. The sclera of the sheep is thinnest in the equatorial region and thus suture passage without penetration in recession can prove to be challenging. The sclera thickens more anteriorly, and resection may be less challenging.
Enucleation
Enucleation is performed using conventional techniques. After limbal periotomy, and dissection to the sclera between the extraocular muscles, the muscles are isolated and either sutured through their tendinous insertions prior to division using a cross swords technique or one of its modifications, or allowed to recess freely within the orbit. The inferior and superior obliques must also be isolated and cut. The retractor bulbi, and posterior Tenon’s fascia, are thicker in the sheep and must be dissected to free the globe. The optic nerve can be directly visualized and the globe removed.
Discussion
An ideal model for simulation in surgical training should: (1) accurately reflect normal human anatomy; (2) be readily available to most training programs; (3) be inexpensive; (4) have a low risk of disease transmission. We will discuss how the sheep model satisfies each of these requirements and where it does not.
Comparative Anatomy
The sheep orbit is very different from that of humans and other primates. As with most grazing animals, the orbital is located on the side of the ovine head, allowing a huge visual field (350°) with limited binocular field (25°). The optic nerves enter the sides of cranial cavity forming an angle of 120° between the orbital axes. The eye sockets themselves are deep with a sharply angled, complete rim. As with all ruminants, the orbital floor and lateral walls are incomplete, with large openings that communicate with the temporal fossa and coronoid process of the mandible (Figure 5).The optic foramen is at the apex of the orbit with 10 surrounding smaller foramina. There is a sturdy periorbita—being thicker in areas where the orbital bone is absent. Sheep orbit viewed from above, dried bone. OF = optic foramen. Mandibular coronoid process visible through the dehiscent orbital floor.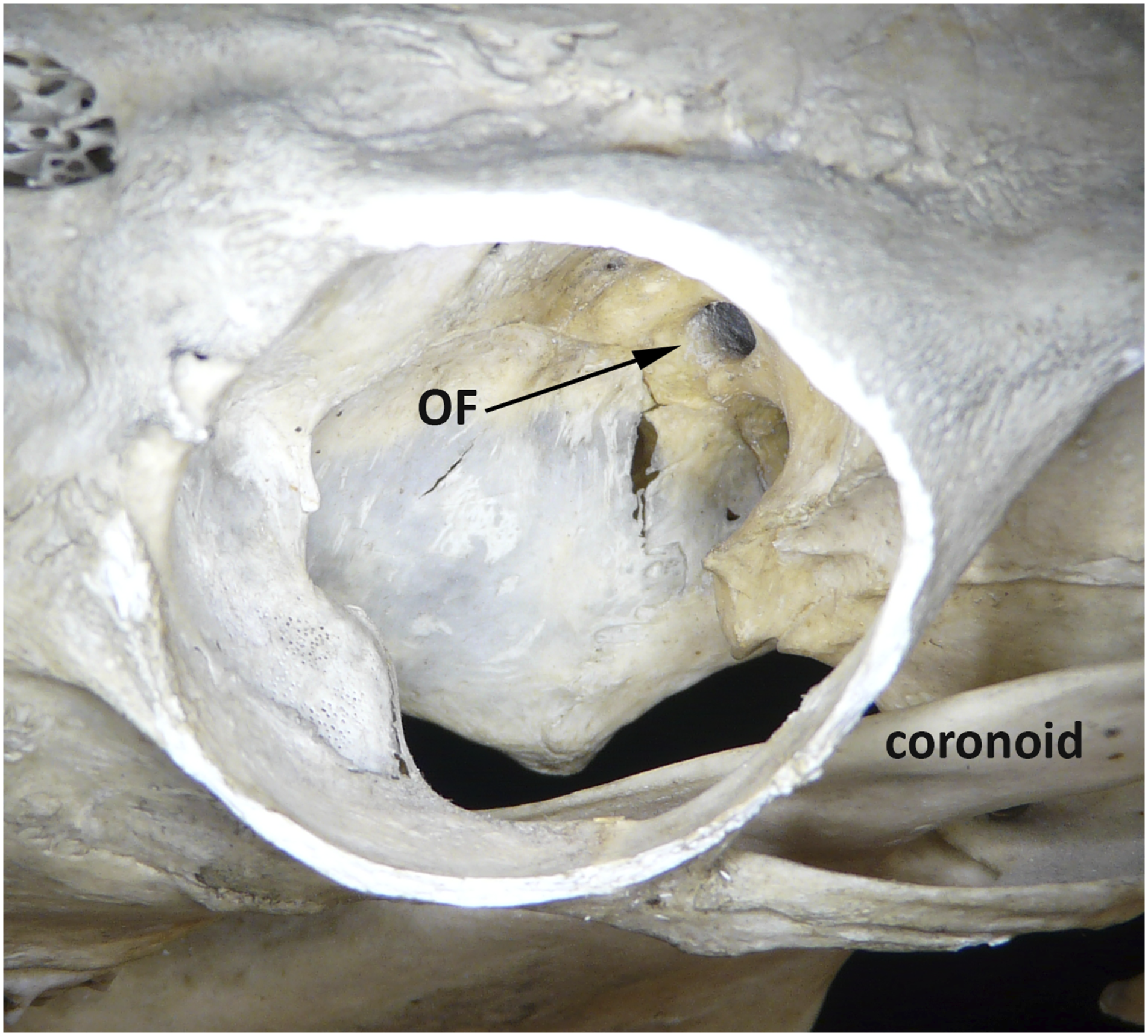
There is identifiable orbital fat deep to the orbital septa of both lids. As in humans, it invests the globe, extraocular muscles, orbital vessels, and nerves. It is firmer than adult human fat and does not protrude easily when the orbital septum is incised. The extraocular muscles are similar in number and position to those in the human. They are less robust, as sheep look about principally by head turning, rather than through extraocular movements. Within the cone of extraocular muscles is the circular retractor bulbi. This strong muscular ring draws the globe inward allowing the nictitating membrane to passively sweep across the cornea to protect it during grazing (Figure 6). Sagittal drawing of anatomy of the sheep orbit. Note insertion of the retractor bulbi muscle.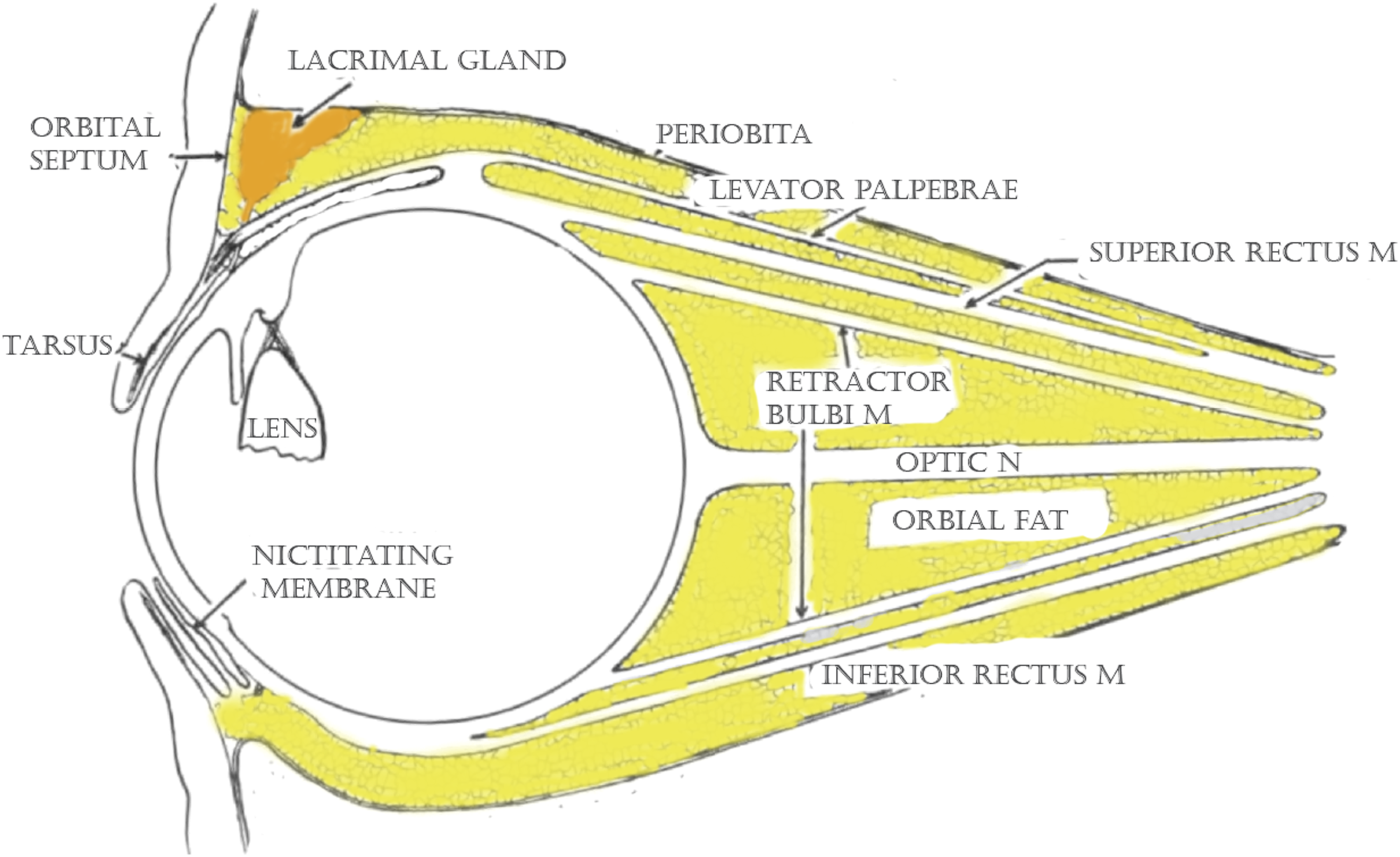
The eyelids of the sheep are thinner than in the pig or goat, but thicker than in man and covered by mature hair. All the expected layers and glandular structures are present. The tarsal plate is well-defined, though lesser in height than in humans. The lacrimal system is similar in structure, but different in proportion. Single puncta are present at the anterior lid margins. They communicate with a rudimentary lacrimal sac. The nasolacrimal duct extends several centimeters along the long ovine snout, making it a poor model for surgery of the lacrimal system. 8
Availability of the model
Sheep are widely available in the North America and around the world. We were fortunate to have access to a regular supply of sheep head and neck preparations through a shared tissue program at Temple University. These tissues were nearly ideal as they were cooled by saline perfusion immediately after euthanasia and refrigerated for only a few days prior to dissection.
For those who do not have such access, it is possible to use farm-raised animals for the same purpose. 9 Sheep heads are not prized in the United States and are available at slaughterhouses for only a few dollars. We experimented with heads obtained from a Philadelphia-area abattoir. Most tissues behaved very similarly to the saline-perfused animals’ but the globes lost some of their turgor if dissection was delayed by more than a few days.
If use of fresh, refrigerated tissue is not practicable, alternative methods of tissue preservation might be considered. Classic tissue infusion with formalin renders tissues stiff and unnatural in dissection and should be avoided. 10 Newer techniques of “light” preservation have shown promise in human tissue simulation. 11 Fresh-frozen and thawed tissues have been used for the study of human facial muscles 12 and might serve as an alternative to refrigerated animal heads for orbital dissection.
Infection risk
A discussion of zoonotic disease is relevant when considering the handling of fresh ovine tissue. Sheep rarely transmit infections to humans.
13
The main concern with using mature farm animals is Query Fever (Q fever), a treatable but potentially dangerous disease
Conclusion
Despite variations in proportion and structure, the authors find that the sheep head and neck preparation has potential as a simulation model for several oculoplastic procedures. It is inexpensive, safe, and readily available. Formal simulation testing using a standardized assessment tool is needed to confirm this preparation’s place in teaching facial plastic surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Supported in part by Department of Defense/Office of Naval Research grants N000141210810 and N000141210597.
