Abstract
Kimura’s disease (KD) is currently considered a rare chronic inflammatory disease of unknown etiology. It is more common in the Asian population, especially in young and middle-aged men, and can involve the lymph nodes, salivary glands, and subcutaneous tissues. It has been reported in adults and children, and is often accompanied by elevated peripheral blood eosinophils and elevated serum immunoglobulin E (IgE). Herein, we report a case of KD in a 46-year-old man with bilateral masses behind the ears since childhood that had gradually enlarged over 40 years. The patient’s peripheral blood eosinophils were elevated, and interestingly, homocysteine levels were also elevated. After surgical resection of the bilateral posterior auricular masses, follow-up over 5 years indicated good recovery and no signs of recurrence.
Introduction
Kimura’s disease (KD), also known as eosinophilic lymphogranuloma, is a rare disease that primarily involves the lymph nodes, salivary glands, and subcutaneous tissues. 1 It clinically manifests as painless lymphadenopathy, elevated peripheral blood eosinophils, and elevated IgE, and is more common in the Asian population, especially in young and middle-aged men. 2 Relevant research is lacking; therefore, its etiology remains poorly understood, and it is often missed or misdiagnosed in patients. Herein, we report a patient with Kimura’s disease with masses behind both ears that had developed in childhood and gradually enlarged over 40 years. In addition to elevated peripheral blood eosinophils, the peripheral blood homocysteine level was also elevated, typically associated with cardiovascular disease and thrombosis; however, the patient had neither. Five years after undergoing resection of the bilateral posterior auricular masses, he exhibited good recovery and no signs of recurrence.
Case Report
A 45-year-old man presented with bilateral posterior auricular masses behind the ears that had been present since childhood and gradually grown over 40 years. The patient usually did not pay attention to the masses; however, 2 weeks before admission, the patient developed bilateral swelling and pain of the ears after a “cold.” There was occasional itching, although no history of hearing loss, tinnitus, fever or night sweats, skin ulceration, bleeding, contact with farms, coronary heart disease, vascular embolism, diabetes, hepatitis, tuberculosis or other infectious diseases, surgery, blood transfusion, drug allergy, or genetic disease. The patient had good dietary and sleep habits, normal bowel movements, and no weight loss. The patient smoked about 2–-3 cigarettes per day and has no history of alcohol abuse. There was a raised oval mass with a maximum diameter of about 3 cm on physical examination with poor mobility and intact surface skin behind each ear. Chest X-ray and electrocardiography were normal. Ultrasound indicated echoes of multiple lymph nodes behind the ears; the largest one on the right side was about 3.2 × 0.7 cm, and the largest one on the left was about 2.0 × 0.5 cm. The margins of the masses were clear, full in morphology, and cortical and medullary structures could be distinguished (Figure 1). The patient has no history of hepatitis B, HIV infection, or syphilis. Urine and bowel habits were normal, and renal function, and transaminase, and blood sugar levels were normal. However, the homocysteine level was 25.54 μmol/L (reference range: 0–15 μmol/L), the WBC count was 6.52 × 109/L (reference range: 3.5–9.5 × 109/L), the eosinophil count was 0.68 × 109/L (reference range: 0.02–0.52 × 109/L), and the eosinophil percentage was 10.4% (reference range: 0.4–8.0%). Ultrasonic appearance of the mass behind the ears: the margins of the masses were clear, full in morphology, and cortical and medullary structures could be distinguished. (A): The largest one on the left was about 2.0 × 0.5 cm. (B): The largest one on the right side was about 3.2 × 0.7 cm.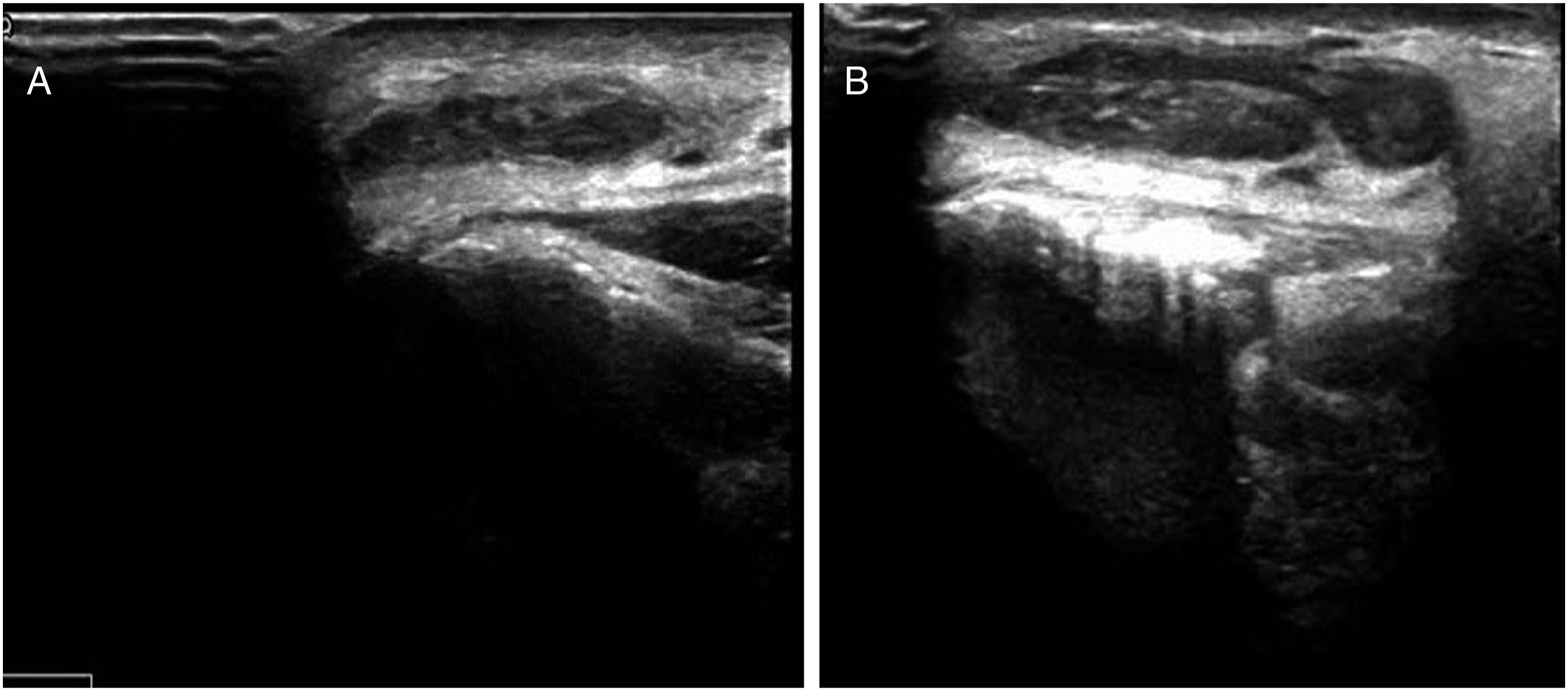
During the operation, bilateral auricular masses were found that were thick, tough, yellowish-brown in color, and having a clear boundary with the surrounding tissues. The surface was smooth, and the maximum diameter was approximately 3 cm. Postoperative pathology indicated lymph node lymphoid tissue hyperplasia (behind the left and right ears) with eosinophil infiltration. Hyperplastic germinal centers were consistent with the morphology of KD (Figure 2). The patient was given prophylactic antibiotic treatment; no hormone therapy or radiotherapy was administered. The patient recovered well after the operation and has been followed up for 5 years without any signs of residual mass or recurrence to date. Postoperative pathology. (A): The germinal centers of hyperplasia in the lymph nodes are consistent with the shape of KD (HE stain, ×40). (B): Lymph node lymphoid tissue hyperplasia with eosinophilia infiltration and angiogenesis (HE stain, ×200).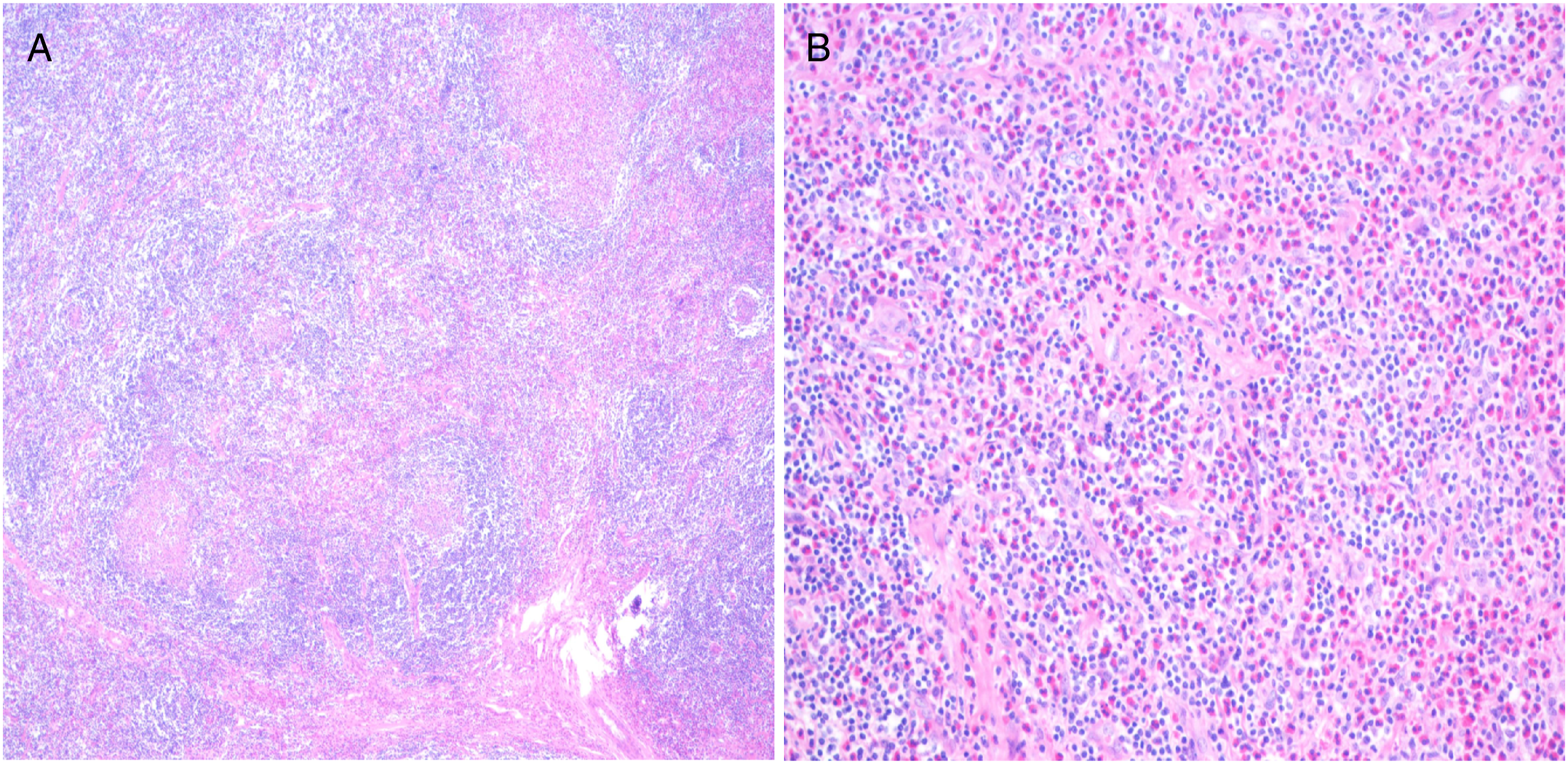
Discussion
Kimura’s disease, also known as eosinophilic lymphogranuloma, is a rare disease that primarily involves the lymph nodes, salivary glands, and subcutaneous tissues, and clinically manifests as painless lymphadenopathy, elevated peripheral blood eosinophils, and elevated IgE. It is more common in the Asian population and especially in young and middle-aged men. The disease can occur at any age, although incidence among men is much higher than among women 2 ; the ratio of male to female patients is 5–14:1,3,4 and in children, it can reach 28:1. 5 Currently, the pathogenic mechanism of the disease is not well understood. Because it is often accompanied by elevated peripheral blood eosinophils and elevated IgE, it is considered an IgE-mediated type I hypersensitivity reaction not caused by viruses, parasites, cancer, or autoimmune reactions.6-8 In 1948, the Japanese researcher Kimura first described this disease in detail. 9 With increased awareness of the disease in recent years, its diagnosis rate has also increased significantly, and signs of KD have been discovered in some uncommon sites as well. Ranjith et al. reported a case of KD in the ileocecal region of a child. 10 KD is easily confused with other diseases such as angiolymphoid hyperplasia with eosinophilia (ALHE) and cancer. Pitak-Arnnop et al. reported on the major differences between KD and ALHE, 11 citing differences in the pathogenic mechanism and clinical characteristics, and stating that the kidney may be involved in patients with KD. Researchers have indicated that relapse is common when eosinophil counts > 50%, serum IgE levels > 10 000 IU/mL, or multifocal lesions are found outside the salivary glands. 12 In our case, the patient had bilateral posterior auricular lesions that were confirmed pathologically as KD. The percentage of eosinophils in the patient was less than 50%, indicating that it may be of a type that is less prone to relapse. To our knowledge, this is the first time a case of KD with elevated homocysteine has been reported. Homocysteine is not present in food though it is an important intermediate product in the normal methionine metabolism in mammals. High homocysteine levels are considered a major risk factor for thrombotic vascular disease. The patient had an elevated homocysteine level without thrombotic lesions, and it is unclear whether the elevated homocysteine level was related to KD. However, the relationship between homocysteine and KD warrants further study.
The diagnosis of KD is difficult 13 and must be based on medical history, hematology, ultrasound, CT, MRI, or other findings, and the final diagnosis is based on pathological examination. KD is generally considered benign and can be accompanied by nephrotic syndrome, asthma, and other diseases. Currently, KD is not believed to be associated with a tendency for malignant transformation, although some reports have suggested the possibility. 14 Akhavan et al.15 reported a case of a patient with synchronous unilateral renal cell carcinoma and KD of the kidney, suggesting that KD may also lead to severe disease. Thus, patients with KD should be followed up closely, as these studies suggest a malignant transformation process associated with KD. In addition, it has been speculated that KD may change the immune regulation of T lymphocytes in conjunction with certain viral infections or induce IgE-mediated type I hypersensitivity reactions, leading to lymphokine release and characteristic changes in the lymph nodes. 16
Individualized treatment for KD should be adopted based on the different conditions of patients. A single localized mass can be completely resected by surgery; however, if the margins of the mass are unclear and difficult to remove or multiple masses are present, surgery alone is prone to relapse, and surgery in conjunction with radiotherapy or hormone therapy may be needed to reduce the recurrence rate. 17 Of course, the levels of eosinophils and IgE in the patient must be considered to determine the best treatment method. In our case, the patient developed bilateral masses behind the ears, although there was a clear margin between the mass and the surrounding tissue, and the mass was completely resected. In addition, the eosinophil percentage was less than 50%, and there was no recurrence after 5 years of follow-up. Systemic hormone therapy is the first choice for patients with renal involvement, particularly nephrotic syndrome. For patients with recurrence or hormone resistance, cytotoxic drugs and immunosuppressive therapy should be considered; some curative efficacy with leflunomide, 18 cyclosporine, 19 and mycophenolate mofetil 20 has been reported in the literature.
A review of clinical experience found that the addition of low-dose radiotherapy or combination glucocorticoid therapy after surgery could significantly improve the cure rate. KD is sensitive to radiotherapy, and most reports have indicated high efficacy. In addition to postoperative recurrence, radiotherapy should be considered in cases of large lesions, multiple foci, unclear margins, or local infiltration. Repeat radiotherapy is still effective for recurrent lesions following surgery or radiotherapy. 21
Conclusion
Kimura’s disease is rare, and our understanding of it is still lacking. Its pathogenic mechanism, pathophysiological process, diagnosis, and treatment remain to be further investigated. Additionally, whether homocysteine plays a clear role in the development and progression of KD and how to better determine individualized treatment through laboratory testing remain to be further studied. Although most patients have a good outcome after treatment, long-term follow-up is still needed to prevent more serious complications and inhibit the cancerous progression of KD.
Footnotes
Author’s Note
The patient’s permission was obtained.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
