Abstract
Introduction
Chronic Rhinosinusitis (CRS) and Chronic Rhinitis (CR) are highly prevalent conditions in adults, affecting roughly 10% of the general population, 1 that have significant impacts on morbidity and quality of life (QoL), constituting major consequences for patients, their families, and society altogether. To add insult to injury, the prevalence is increasing annually throughout the world.2-6
One of the main mechanisms suggested for CRS development is a formation of biofilm in the nasal and sinus cavities.7,8 The EPOS 2020 steering group has emphasized doubt regarding the use of systemic antibiotics in CRS patients due to a rise of bacterial resistance occurring worldwide, which in turn compromises the efficacy of antibiotics. Consequently, this recommendation led to the emergence of other treatment possibilities,9-11 one of which suggests topical antibiotics as an alternative for systemic treatment. It has been proposed that this method allows deliverance of high concentrations of antibiotics onto the sinus surfaces where they may penetrate a bacterial biofilm. 12 Topical antifungals represent another appealing alternative for similar reasons, also due to the observation that CRS may result from an exaggerated allergic response to fungi in nasal mucus. 13 Nevertheless, distribution of topical treatments to a non-operated sinus was found to be rather limited, accounting for less than 2–3% of the total irrigation volume or nebulized solution attaining sinus penetration in the setting of CRS with mucosal edema.14,15
Pomegranates have a long history of antibacterial use dating back to biblical times. Punica granatum L. (Punicaceae) is a shrub native to Asia and Mediterranean Europe, popularly referred to in English as pomegranate. According to Ebers Papyrus, one of the oldest medical writings, circa 1500 BC, the plant was used by Egyptians as a treatment for tapeworm and other parasitic infestations. 16
Several studies investigated the bactericidal effects of pomegranates on a number of highly pathogenic and drug-resistant strains, determining bactericidal potency of different pomegranate plant extracts against a range of different bacteria by utilizing disc diffusion assays or minimum inhibitory concentration.17,18
Pomegranate extract was found to have antibacterial activity against S. aureus S. epidermidis, L. acidophilus, S. mutans, and S. salivarius. 19 Moreover, it has also been shown to inhibit the formation of biofilms as well as to disrupt preformed biofilms. 20 In one study, ethanolic extracts of pomegranate were effective at inhibiting 35 hospital isolates of MRSA, and scanning electron microscopy of the bacteria showed that the pomegranate extract caused alterations in the bacterial cell walls after 2 hours of treatment. 21
Several other studies have been conducted regarding antiviral activities associated with pomegranate and its extracts. The fruit’s antiviral effects have been reported against clinically relevant viruses: influenza virus, herpes virus, poxviruses, human immunodeficiency (HIV-1) virus, FCV-F9, MNV-1, and bacteriophage MS2.22-27
Pomegranate, with its dual contribution, both as an anti-biofilm and an antimicrobial agent, has directed us to evaluate its clinical efficacy utilizing topical nasal Pomegranate Fruit Extract (PFE) in patients with Chronic Rhinitis, Chronic Rhinosinusitis with Nasal Polyposis (CRSwNP), and Chronic Rhinosinusitis without Nasal Polyposis (CRSsNP) in the setting of a double-blinded randomized study.
Materials and Methods
Study Participants and Data Collection
Data was prospectively collected on 111 consecutive patients afflicted by CRS and CR during the study period of April 2012-January 2017 at the Department of Otorhinolaryngology, Head and Neck Surgery, Hillel Yaffe Medical Center, Hadera, Israel.
Cooperative healthy non-smoker subjects aged ≥18 years were enrolled in the study as per inclusion criteria. Patients under 18 years of age, history of prior sinonasal surgeries or chemoradiation, concurrent medical conditions, allergy to pomegranates/yeast were excluded from the study. Corresponding to the pathology, subjects were divided into 3 groups: CR, CRSwNP, and CRSsNP. Chronic Rhinitis patients were diagnosed according to the World Health Organization and the Allergic Rhinitis and its Impact on Asthma (ARIA) Classification and were distinguished from CRS patients who were diagnosed based on the 2012 European position paper on rhinosinusitis and nasal polyps
28
using major and minor criteria, for patients suffering symptoms for more than 12 weeks, backed by endoscopic test showing purulent discharge or nasal polyposis, or by CT findings of presence/absence of sinonasal polyposis. Since allergic and non-allergic patients were not distinguished separately and were allocated to their group based on symptomatology alone, the presence of aeroallergy was not considered as mandatory. Abovementioned 3 groups were further randomly assigned into either PFE treatment or Placebo groups. The study design is displayed in Figure 1. Study design.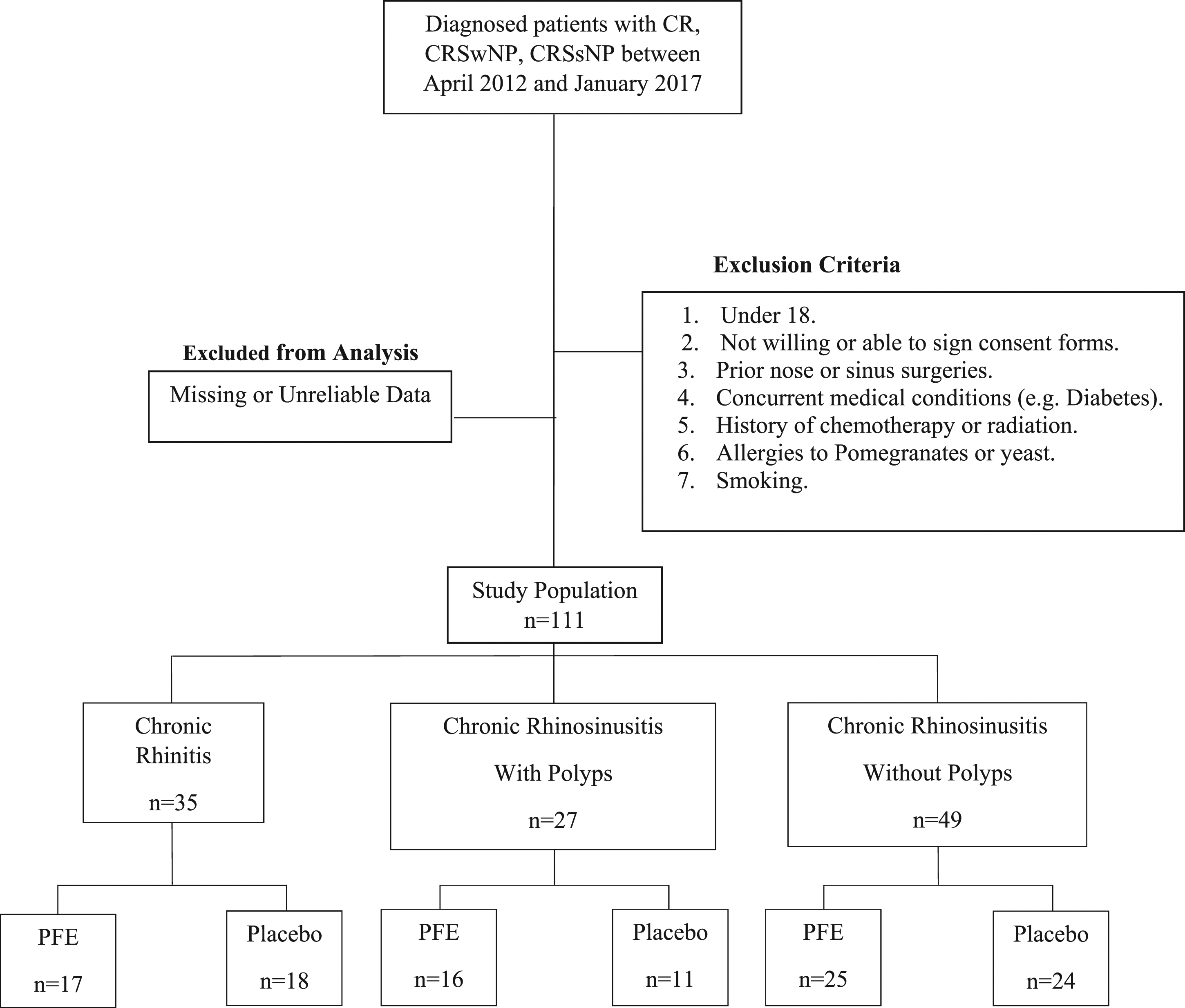
Detailed documentation of the relevant parameters before and 1 month after the treatment was made. Inferior turbinate hypertrophy, discharge from inferior and middle meatus, and nasal polyposis were appreciated by anterior rhinoscopy and fiberoptic exam. Otoscopy was done using otologic microscope. In addition, Lund-Mackay scores, eosinophilic counts, and C-Reactive Protein values were recorded along with Sino-Nasal Outcome Test-20 (SNOT-20) questionnaire responses.
Treatment Protocol
Each group received either PFE or Placebo spray twice daily for 30 days. The solutions were packaged in 2 amber glass bottles, each containing 15 mL. Each bottle was sealed with a nasal spray device, made of polypropylene (PP), Polyoxymethylene (POM), and Ethylene-vinyl acetate (EVA), capable of delivering .15 ± .01 mL per dose (ie, a total of 200 doses).
Statistical Analysis
Data was analyzed using SPSS software. To assess the difference of improvement between each of the study groups with the placebo group, before and after the treatment, the paired t-test was applied for continuous variables and chi-square test categorical variables. Eta squared was applied to measure association, as appropriate. A two-sided P-value of ≤.05 was considered statistically significant. Mean values are presented with their standard deviations.
Ethical Issues
The current prospective, double-blinded, randomized study followed the tenets of the Declaration of Helsinki and was approved by the institutional ethical committee (Hillel Yaffe Medical Center Committee, No. 0089-11-HYMC). Informed consent was obtained from all participating patients.
Results
Male to Female Ratio and Mean Age among Study Groups, (n = 111).
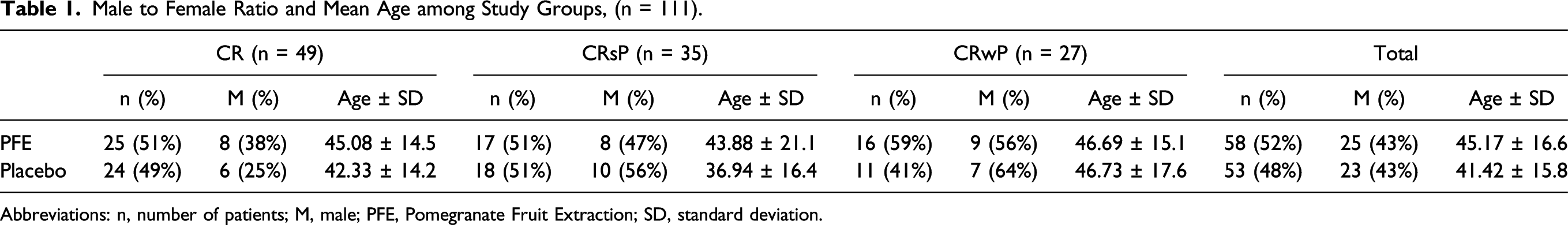
Abbreviations: n, number of patients; M, male; PFE, Pomegranate Fruit Extraction; SD, standard deviation.
Pre-, Post-Treatment SNOT-20 Score and Improvement among the Different Groups.
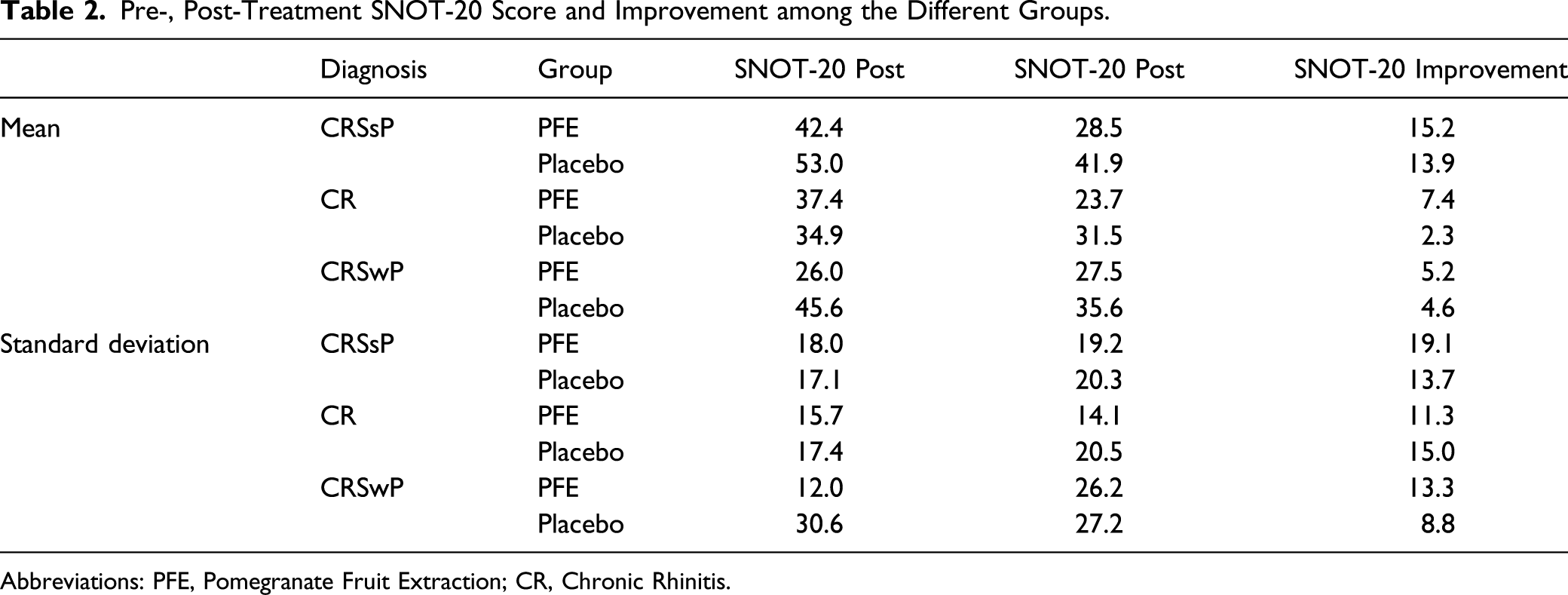
Abbreviations: PFE, Pomegranate Fruit Extraction; CR, Chronic Rhinitis.
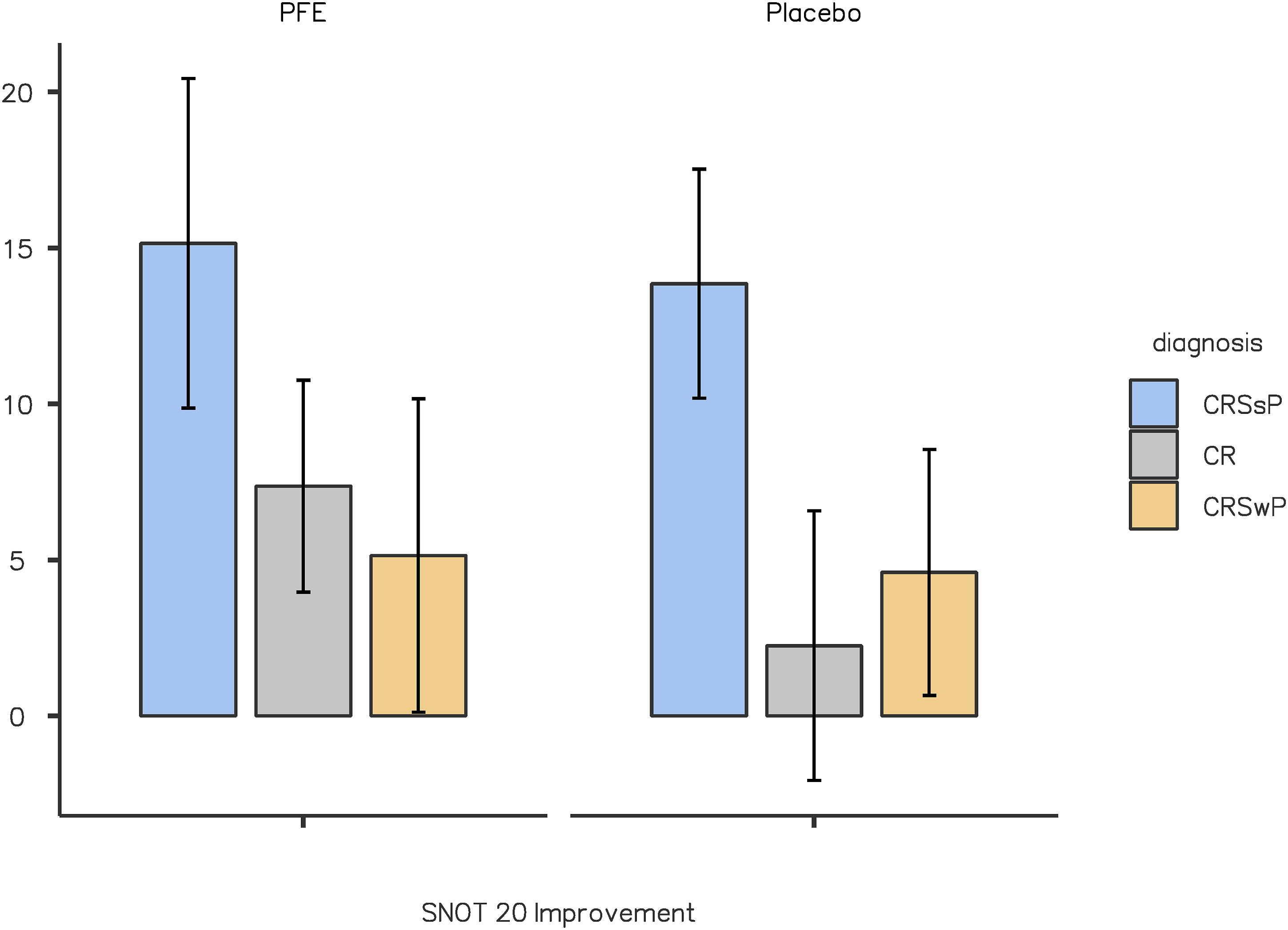
SNOT-20 mean improvement among the different groups.
To compare pre-and post-treatment signs and symptoms between the PFE and Placebo groups, paired t-tests were employed, exhibiting no significant difference between groups regarding physical exam, laboratory values, and Lund-Mackay score. To capture the culprit of discrepancies in the SNOT-20 questionnaire, its separate components were further analyzed using estimate independent mean difference. The questions pertaining to thick nasal discharge, difficulty falling asleep, reduced productivity, reduced concentration, and sadness variated significantly among disease groups as follows.
Regarding thick nasal discharge, both CRSwNP and CRSsNP cases showed a decrease within PFE and Placebo groups. However, it was found to be significant only among CRSsNP cases (P = .001 and P = .001, respectively), and utilizing estimate independent mean difference showed no superiority to either group. Conversely, within the CR group, PFE exhibited superiority over placebo, with a significant decline of thick nasal discharge after treatment (P = .005). Similarly, superiority was demonstrated amongst the CR group for difficulty falling asleep (P = .04), reduced productivity (P = .04), and sadness (P = .04). Regarding Reduced productivity, a decrement was observed also in the CRSsNP group (P = .02), but no superiority over the placebo group was observed.
In CR patients, solely the PFE group showed Improved concentration (P = .007), while no significant differences within CRSwNP and CRSsNP patients were observed. Additional analysis showed tendency to favor the CR group, but no significant superiority was demonstrated using estimated means difference (P = .06).
Figure 3 demonstrates a comparison of means for the aforementioned SNOT-20 questions in all disease types for PFE and Placebo, before and after treatment; Figure 4 displays estimate independent mean difference for the discussed questions. Comparison of mean values for thick nasal discharge, difficulty falling asleep, decreased productivity, decreased concentration, and sadness. Estimate independent mean difference within CR patients for selected SNOT-20 questions.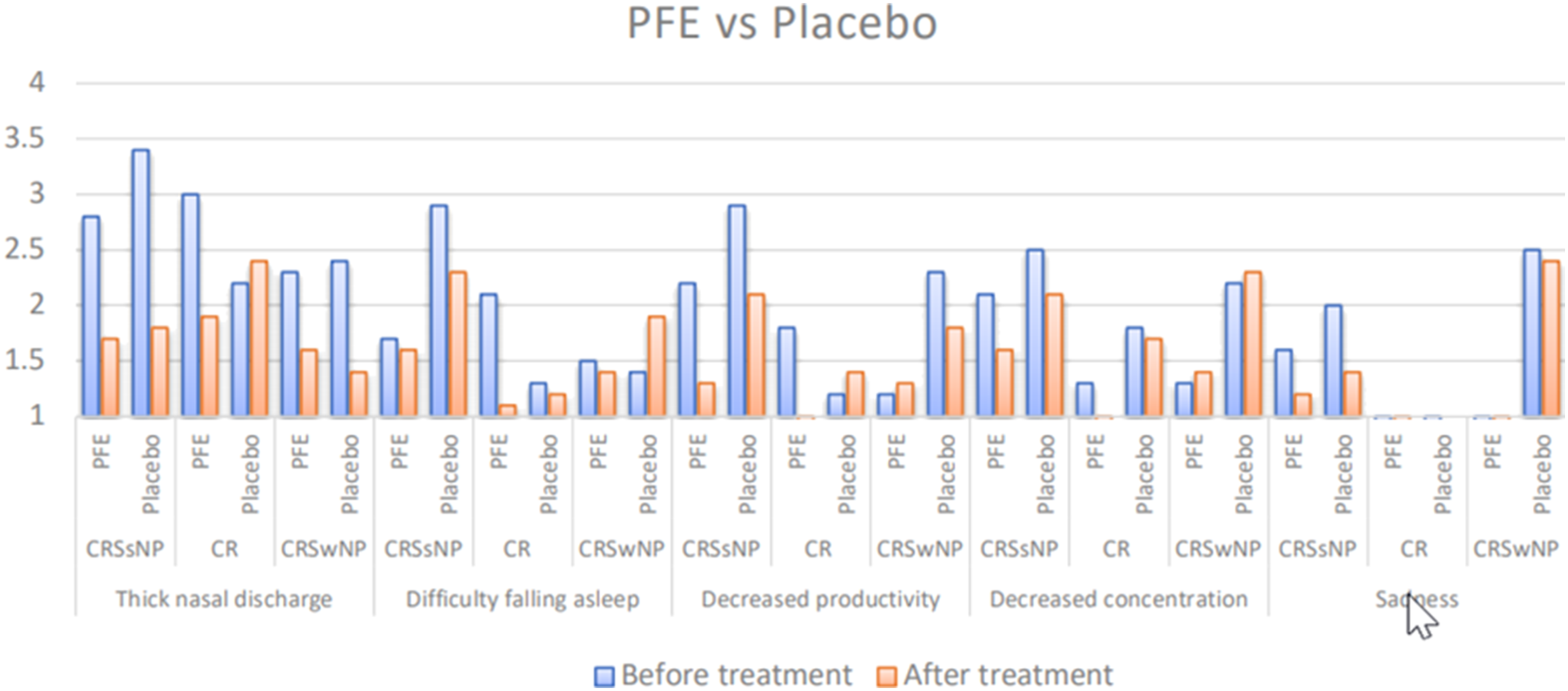
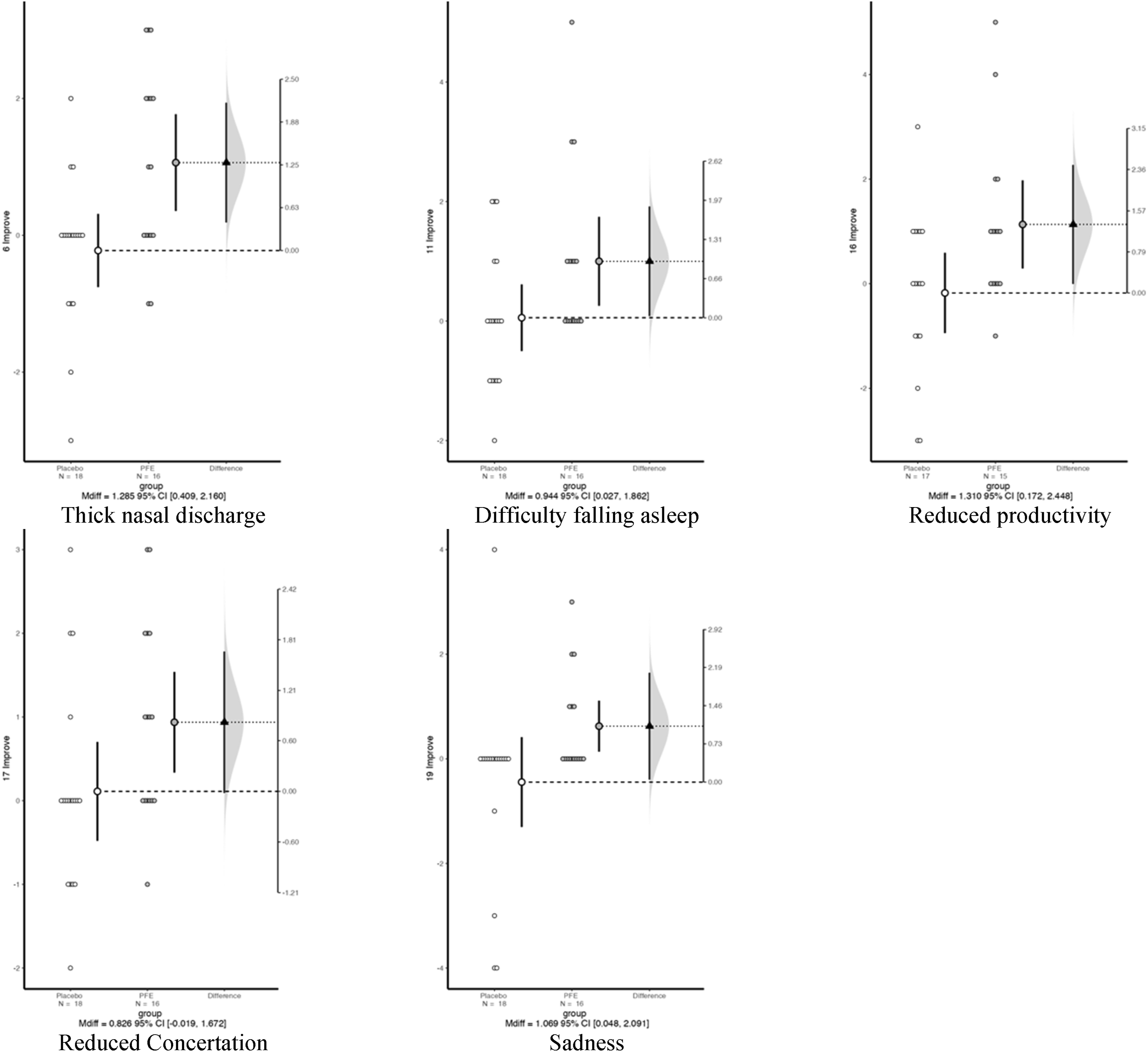
Broadly, CRSwNP and CRSsNP patients did not show noteworthy differences between PFE and Placebo groups. However, within CR patients, significant recovery in symptoms according to the SNOT-20 questionnaire in PFE vs the Placebo group was observed, (Figure 5, bolded), exhibiting improvement regarding thick nasal discharge, difficulty falling asleep, reduced productivity, reduced concentration, and sadness (P = .004, P = .02, P = .03, P = .007, and P = .02, respectively). Table 3 displays the changes in the CR group. SNOT-20 Questionnaire. Red colored are symptoms relieved in CR patients with Topical Nasal PFE treatment. PFE, Pomegranate Fruit Extract; CR, Chronic Rhinitis. Chronic Rhinitis SNOT-20 Symptoms, Before and After Treatment with Pomegranate Fruit Extract and Placebo. Abbreviations: PFE, Pomegranate Fruit Extraction.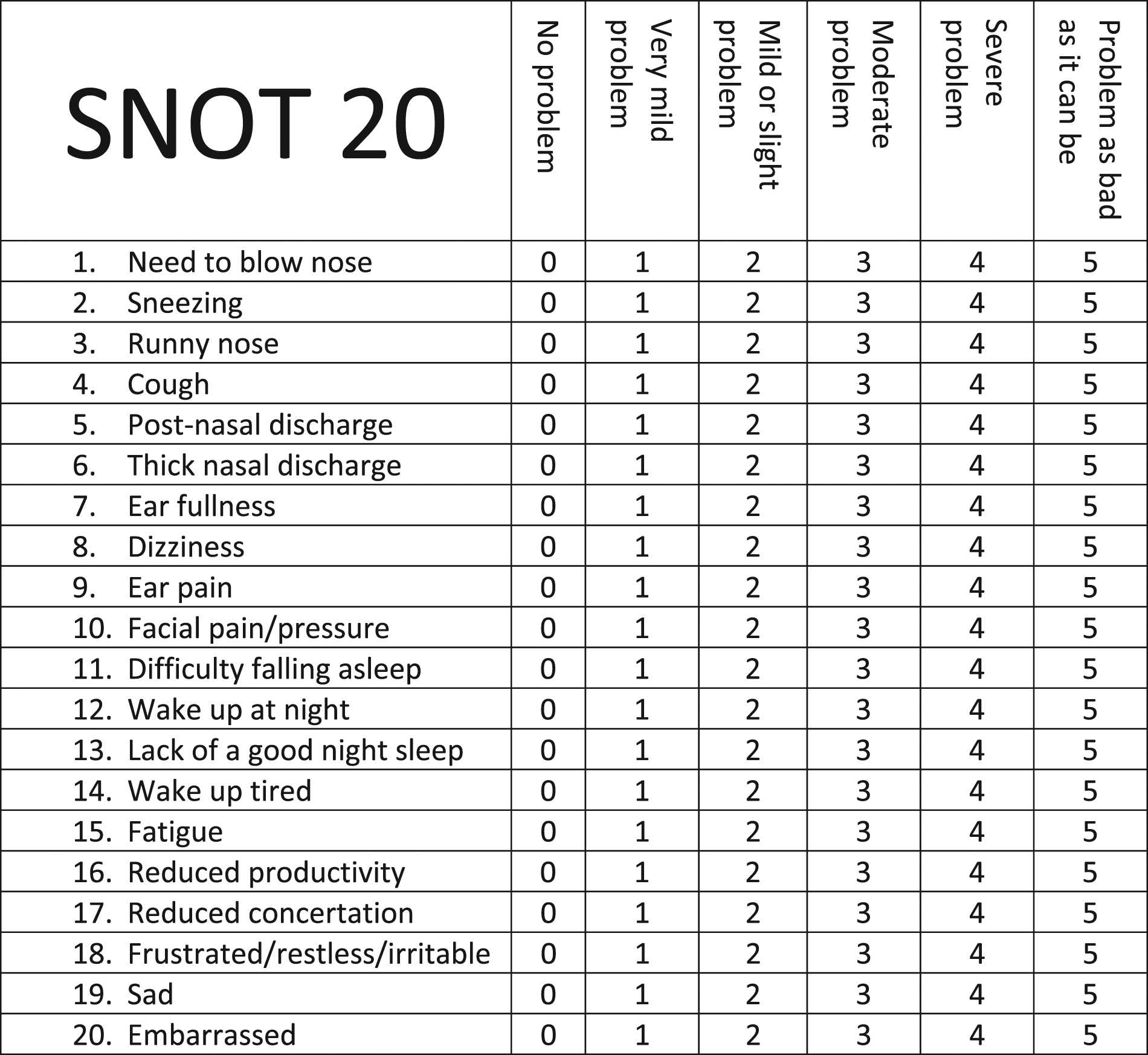

Discussion
In the present prospective, double-blinded, randomized study, we assessed and analyzed the clinical efficacy of topical nasal Pomegranate Fruit Extract for Chronic Rhinitis, Chronic Rhinosinusitis with Nasal Polyposis, and Chronic Rhinosinusitis without Nasal Polyposis.
Both CRS and CR constitute a significant impact on the healthcare system. In the United States, Allergic Rhinitis (AR) alone results in 3.5 million lost workdays and 2 million lost school days annually. 29 Most patients with CRS and CR seek medical treatment only when they experience significant QoL impairments. Thus, improvements in the SNOT-20 questionnaire, corresponding to QoL in these conditions, can reasonably serve as reliable indicators for treatment success. 30
Whether it stems from allergic (AR) or non-allergic (NAR) origin, Chronic Rhinitis has a variety of proposed treatments. One option is intranasal steroids; however, safety of long-term use is questionable31,32 as they may result in adverse effects such as mucosal atrophy, 33 hypothalamic-pituitary-adrenal axis suppression, and other systemic effects. 34 Prior studies have shown superior results with the addition of azelastine hydrochloride to intranasal steroids over solely intranasal steroids use, 35 while others have shown significant QoL improvement with anti-histaminergic therapy. 36 Last, surgical treatment with radiofrequency turbinoplasty or immunotherapy may be suggested for patients who failed to respond to conservative lines of treatment. 37
Likewise, topical intranasal and systemic oral corticosteroids constitute the first line of therapy in treating CRS. However, they must be given with caution in particular populations such as children, pregnant women, and elderly patients and especially in those with comorbid conditions such as asthma, due to possible high steroid intake. 38
Presence of bacterial biofilm species has been demonstrated in several in vitro trials for CRS patients,8,39 though according to the International Consensus Statement on Allergy and Rhinology: Rhinosinusitis Executive Summary, there is insufficient clinical evidence to determine their role. 40
Topical antimicrobial agents might be preferable due to their ability to localize delivery to the sino-nasal mucosa and minimize systemic effects found with oral agents. This could play an important role in treating biofilms, where higher concentrations of antibiotics usually required. However, until now, topical antibiotics have not shown consistent results for CRS treatment. While mupirocin reduced S. aureus biofilm mass and topical macrolides showed significant improvement in CRS patients, topical ciprofloxacin and vancomycin were both largely ineffective in reducing biofilms.41,42 Overall, several randomized control trials and systemic reviews have been performed on this topic. 40 While the antibiotic mechanism of delivery (standard vs non-standard delivery, ie, sprays and drops vs irrigation and nebulizers, respectively) and patients’ selection (CRSwNP vs CRSsNP) varied significantly, these studies and reviews have failed to show additional benefit of topical antibiotics. 40
Previous studies described a significant improvement in SNOT-22 results for CRS patients using intranasal chemical surfactants as agents to disrupt biofilm integrity, 43 while others demonstrated destabilization of P. aeruginosa with the use of topical furosemide under in vitro experiment. 44
In addition to being proven to be a safe therapeutic option, 45 Pomegranate Fruit Extract and its biologically active compounds have been studied extensively. 46 Many studies have shown its various pathophysiologic anti-inflammatory effects, including inhibition of PMACI-induced proinflammatory cytokines assembly by inhibiting gene expression through the blockage of JNK and ERK-MAPK and NF-κB activation in human KU812 cells, 47 a reduction of Prostaglandin E2 (PGE2) levels in colonic mucosa by downregulating overexpressed COX-2 and Prostaglandin E synthase (PTGES) level through the action of ellagic acid, 48 inhibition of numerous single transduction pathways and downstream pathogenic cellular response, 49 and disrupting biofilms, 20 possibly through alterations in bacterial cell walls by ethanolic extracts. 21 Interestingly, PFE may have a therapeutic role in other disease states. A randomized controlled trial 50 demonstrated that daily oral pomegranate consumption may lead to enhanced protection from UV photodamage, possibly through abundance of skin bacteria containing pigments absorbing UV irradiation that might contribute to UVB skin protection.
The current analysis shows that topical nasal PFE impacts QoL in patients with CR, being effective in reducing thick nasal discharge, difficulty falling asleep, reduced productivity, reduced concentration, and sadness. Despite this, in the present study, PFE did not prove effective for the treatment of CRS. Nonetheless, we believe that it cannot yet be concluded that PFE is not an applicable antimicrobial and anti-biofilm agent, as, in this study, we used a standard irrigation method in the form of nasal spray. Thus, further research and development of different delivery methods, enabling higher concentration levels of PFE into the nasal sinuses, may yield more favorable outcomes. Moreover, it is possible that PFE will be effective for patients who have undergone ESS, as it has been shown to enhance the delivery of nasal solutions into the sinuses.51-54
This study has several limitations. First, albeit a relatively large sample size, the sub-divided groups are more modest. Second, CR was treated without division into sub-groups and separate etiologies, which might obscure the results. Third, our study was conducted prior to the publication of the new EPOS 2020; therefore, data regarding the proportion of patients with type 2 and non-type 2 CRS is lacking, which may have influenced contemporary interpretation. Future studies integrating this division are required and may shed a brighter light on the topic.
Conclusion
The current study showed that topical nasal PFE could offer some clinical benefits for treating patients affected by Chronic Rhinitis, specifically for thick nasal discharge, difficulty falling asleep, reduced productivity, reduced concentration, and sadness. However, in patients afflicted by CRSwNP or CRSsNP, this substance presently displayed no significant treatment role, possibly due to inability to reach the sinus mucosa. Future studies are required concerning development of an optimal delivery device that may ameliorate some of the abovementioned results.
Footnotes
Author’s note
The authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
