Abstract
We present a case of an 80-year-old male who developed skull base osteomyelitis after nasopharyngeal cyst removal. A review of the literature regarding complications after nasopharyngeal cyst removal was performed. We describe the difficulty of diagnosing an osteomyelitis infection and the best approach to recognizing osteomyelitis before complications worsen.
Introduction
Nasopharyngeal cysts are usually benign and asymptomatic, found during routine imaging and endoscopies. Asymptomatic cysts may not require treatment, while large, symptomatic growths close to the Eustachian tube may require surgical resection. 1 Postsurgical skull base osteomyelitis is a rare complication in both adults and children that can occur after any nasopharyngeal surgery owing to the close proximity of the clivus and skull base. 2 We now report an individual who developed skull base osteomyelitis after nasopharyngeal cyst removal.
Case
An 80-year-old male was seen in an otolaryngology clinic with an incidental finding of a nasopharyngeal lesion on magnetic resonance imaging (MRI) ordered for the evaluation of his chronic headaches and imbalance (Figure 1A). A benign nasopharyngeal cyst was found on endoscopy (Figure 1B), and a review of his previous imaging showed that the cyst was not present on previous MRIs performed in 2014. The patient was offered the choice of elective excision or observation as the cyst appeared benign and was unlikely to be causing his symptoms.
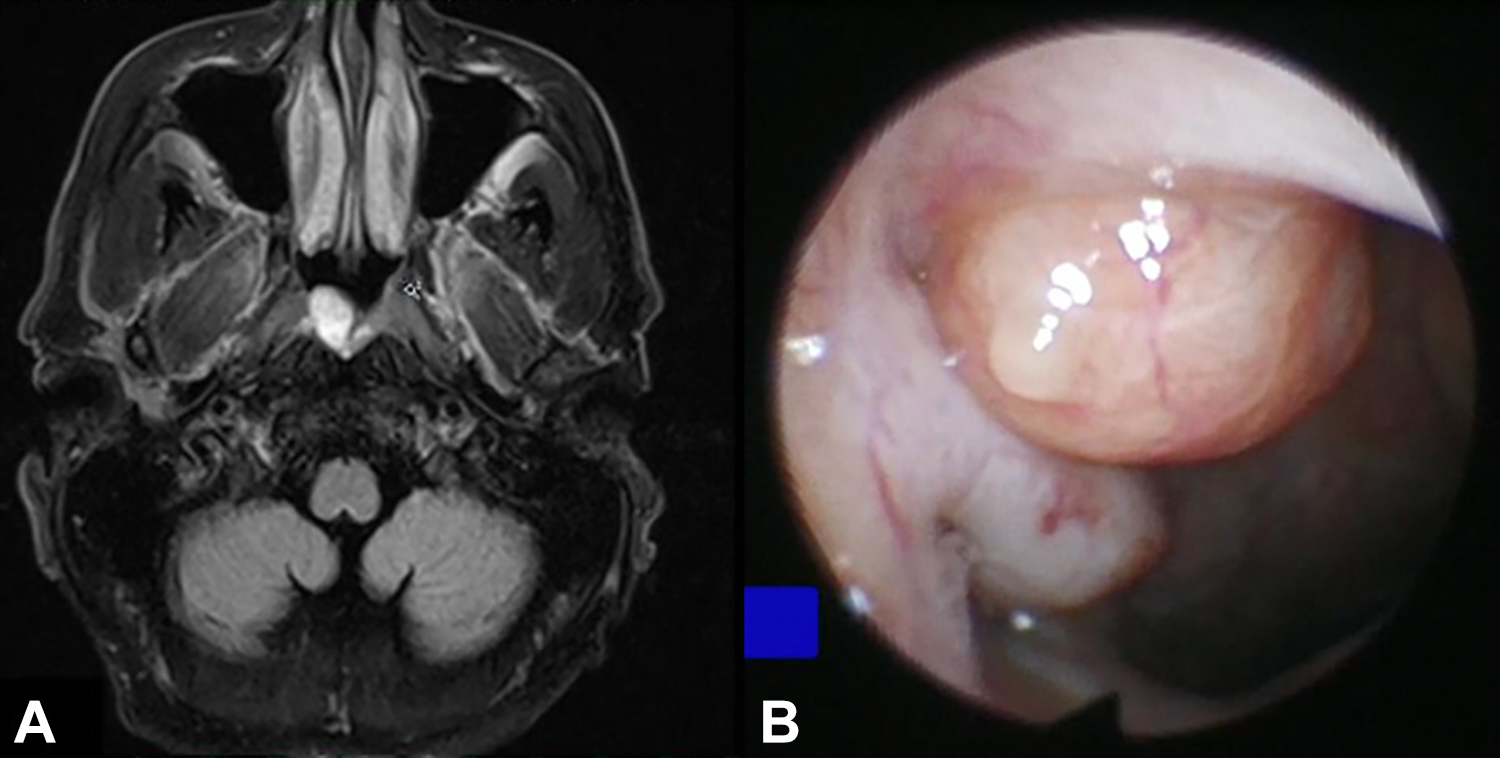
A, Preoperative T1 magnetic resonance imaging (MRI) of nasopharyngeal cyst. B, Preoperative endoscopic view of a nasopharyngeal cyst.
Ultimately, he chose to undergo excision, and the cyst was endoscopically excised using needle tip bovine cautery and blunt dissection in an uneventful surgery. Three days post-surgery, the patient called to clinic and reported that he had severe headaches and felt that he had a sinus infection. Although he was given amoxicillin–clavulanate postoperatively, he had stopped taking the medication after only one dose due to nausea. He reported good efficacy and tolerance to azithromycin, so he was switched to this medication. An endoscopic examination at his first postoperative visit, 8 days after surgery showed him to be appropriately healing.
At 11 days after surgery, the severe headaches were still a problem, so a computed tomography (CT) scan was ordered due to the severity of the headache, but showed only mild inflammation in the sphenoid sinuses. At his second postoperative visit 18 days after surgery, the severe headaches had not resolved, and the patient had other symptoms that pointed toward a potential nonotolaryngologic etiology. For example, he complained that the skin of his scalp was extremely sensitive and painful to light touch. However, the surgical site did have some purulent mucus and granulation tissue. A culture was taken and came back positive for Pseudomonas Aeruginosa, and oral levofloxacin therapy was initiated. At the time, the complete blood count (CBC) showed white blood cell (WBC) 13.5 with a left shift, and the patient’s blood pressure was highly elevated during his clinic visits, so he was urged to see both his primary care physician (PCP) and neurologist.
At the third postoperative visit 24 days after surgery, the infection appeared to be resolved, and by the fourth visit 38 days after surgery, the surgical site was completely healed, but the patient was still complaining of headaches and imbalance. He was urged again to see his neurologist and PCP since these were complaints that he had prior to surgery and led to him having the initial MRI that diagnosed with nasopharyngeal lesion.
His neurologist evaluated him and eventually referred him to rheumatology for potential temporal arteritis. His workup by the neurologist and rheumatologist revealed an elevated erythrocyte sedimentation rate at 80, positive antinuclear antibody and anti-double-stranded DNA antibodies, and a normal WBC at that time. He was subsequently started on high-dose prednisone for a presumptive diagnosis of temporal arteritis, but a biopsy was performed of the temporal artery which was ultimately normal. Steroids were initially tapered off, but the patient’s headache would recur whenever the steroids were reduced, so rheumatology and neurology kept him on oral steroids. Additionally, the patient saw an orofacial pain and temporomandibular joint disorder specialist.
Our patient presented 3½ months postoperatively to the initial surgeon’s neuro-otology colleague with decreased hearing and mucoid effusions. He was started on fluticasone nasal spray and oral decongestants and was instructed to return if symptoms did not resolve so a myringotomy tube could be placed. He was still on 20 mg of prednisone at this time despite not having a firm rheumatologic diagnosis. He presented 4 days later to the emergency department with dysphagia and right-sided facial weakness. Flexible laryngoscopy showed normal vocal cord movement and no sign of any nasopharyngeal infection. Radiographic workup showed bilateral noncoalescent mastoiditis on CT. Bilateral myringotomy was performed and pressure equalization tubes were placed emergently, and the culture grew Pseudomonas.
When he developed aphasia a day into his hospital course, a lumbar tap was performed that showed clean cerebrospinal fluid. A CT demonstrated mild enhancement and lytic destructive changes of the clivus and skull base centrally involving the sphenoid bone suggesting skull base osteomyelitis, shown in Figure 2A. The technetium-99m (Tc99m) bone scan confirmed this diagnosis, shown in Figure 2B.
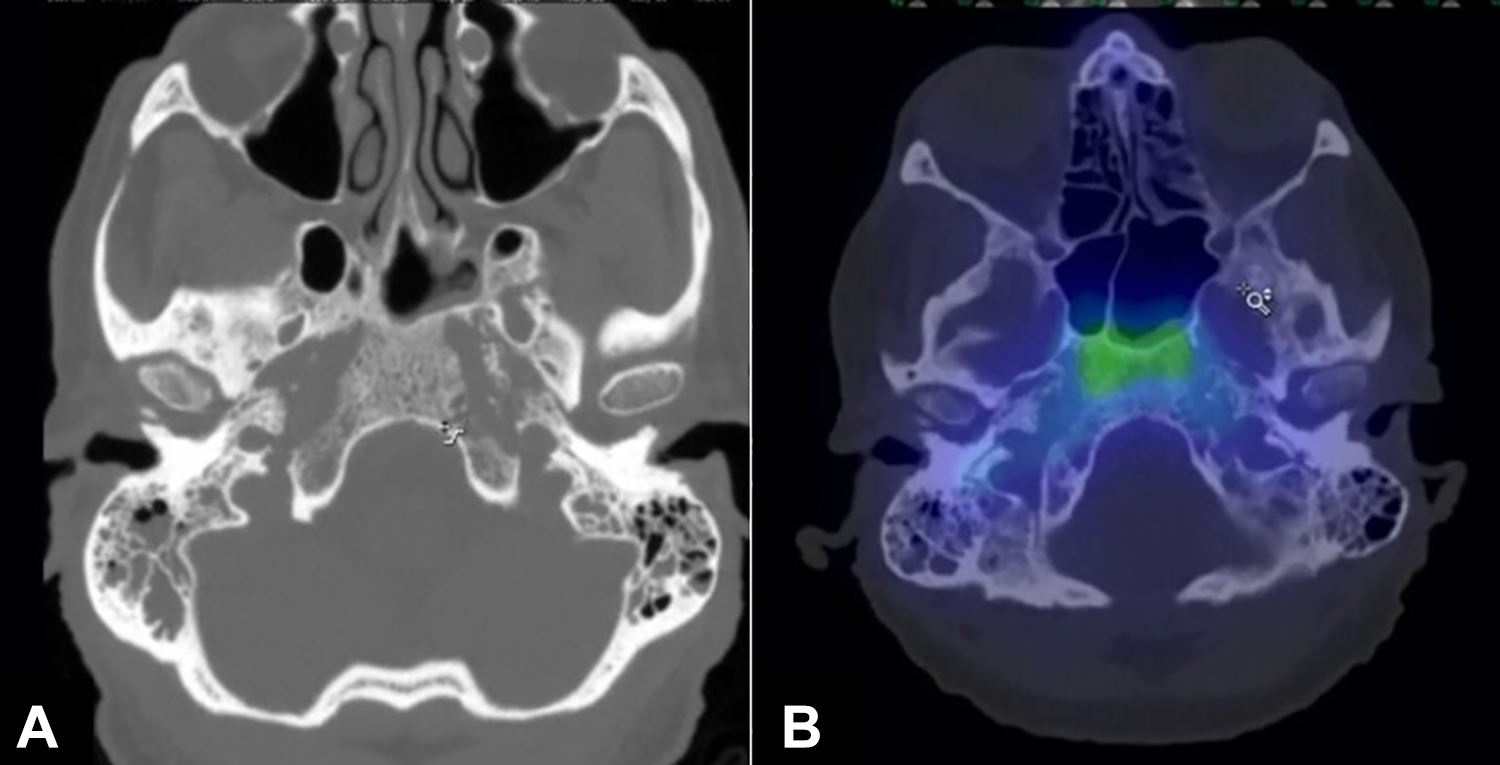
A, Computed tomography (CT) demonstrating lytic changes in skull base and clivus with sphenoid inflammation. B, Technetium-99m bone scan demonstrating hyperactivity in the clivus and skull base.
Under the direction of infectious disease specialists, he was treated with intravenous antibiotics for 3 months (initially piperacillin/tazobactam but later switched to meropenem during his initial hospital stay) and followed with gallium-67 scans. After 3 months, the final gallium scan was negative and antibiotics were stopped. At his most recent follow-up, our patient had a CN VII HB 1/6 bilaterally, but unfortunately, his hearing diminished over the course of the infection despite treatment.
Discussion
The central skull base is an important passage for many cranial nerves and neurological structures, and skull base osteomyelitis can be a debilitating infection. Skull base osteomyelitis is an extremely rare complication of nasopharyngeal surgery. 3 Mindfulness and early diagnosis are crucial due to the high potential morbidity that can be caused when evaluating a postoperative patient presenting with unusual symptoms. According to Muranjan et al, imaging plays a crucial role in the diagnosis of skull base osteomyelitis; specifically the use of MRI with gadolinium contrast can allow for the visualization of poorly demarcated soft tissue that is surrounding bone and is currently touted as the most sensitive modality for detecting osteomyelitis. 3 When high-resolution MRI with contrast is unavailable or contraindicated, Tc99m scans are very sensitive in detecting high osteoblastic activity and making the initial diagnosis of osteomyelitis. However, Tc99m scans will remain positive long term even after the infection is fully treated, so gallium-67 scans are often used to follow and document resolution of the infection as the radionuclide binds acute phase reactants in infected or inflamed tissue. In our case, the prior nasopharyngeal surgery and postoperative infection with Pseudomonas provided the likely culprit for the skull base osteomyelitis, but in certain situations, a biopsy may be required to make a diagnosis or provide tissue for culture and sensitivities.
Skull base osteomyelitis is an extremely dangerous entity that can result in temporary and potentially permanent cranial neuropathies if there is repeated failure of medical treatment. 4 According to the comprehensive treatment review done by Khan et al, early aggressive culture-guided administration of broad-spectrum antibiotics decreases complications that may be associated with this aggressive infection. 5 In this case, it is likely that long-term treatment with steroids led to immunosuppression and spread of an initially localized infection since our patient had no other medical conditions that would make him more prone to this disease such as diabetes mellitus, human immunodeficiency virus, autoimmune disease, or malignancy.
Footnotes
Authors’ Note
This manuscript is original and has not been submitted elsewhere in part or in whole.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
