Abstract
Background:
Chronic rhinosinusitis (CRS), as an inflammatory airway disease, could be a risk factor for COVID-19 patients. This study aimed to investigate the frequency and severity of symptoms of COVID-19 in patients with CRS and to assess the association between the status of CRS symptoms and the quality of life (QoL) of the patients.
Methods:
In this observational and cross-sectional study, 207 adult CRS patients participated. The patients, who presented the symptoms of COVID-19, were examined by taking the reverse transcription–polymerase chain reaction test. A questionnaire was completed by each patient, regarding their demographic and clinical data. In addition, the GA2LEN and Sino-Nasal Outcome Test (SNOT-22) standard questionnaires were used to identify the comorbid allergic condition and QoL of CRS patients.
Results:
The frequency of patients with COVID-19 was 25 (12.1%) of which 22 were treated as outpatients, 2 of them admitted in wards and 1 at intensive care unit. The severity of hyposmia in the patients was 2 (8%) as mild, 5 (20%) moderate, and 11 (72%) as anosmia. The most common allergic and underlying comorbid diseases were allergic rhinitis (88%) and thyroid disorders (28%). Further, the average SNOT-22 score in 4 SNOT-22 domains (nasal, otologic, sleep, and emotional symptoms) was significantly decreased in CRS patients after a period of one year since the pandemic started (40.1 ± 18.0 vs. 46.3 ± 17.7; P < .0001).
Discussion:
This study showed a low frequency of COVID-19 in patients with CRS and about the same rate of infection positivity in the general population; therefore, we concluded that CRS could not be considered as a risk factor for COVID-19. Interestingly, the lower average score of SNOT-22 after one year of the pandemic in the patients with CRS confirmed the necessity for performing the standard health protocols by the patients.
Introduction
The coronavirus disease 2019 (COVID-19) is a new strain of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and at the present, it is one of the most public health emergencies throughout the world. Noticeably, since the onset of the pandemic, Iran has been one of the first countries being affected by the virus. 1,2
According to the reports from different studies carried out throughout the world, the most prevalent symptoms of patients afflicted with COVID-19 are fever, cough, fatigue, headache, dyspnea, sputum production, arthralgia, diarrhea, rhinorrhea, and sore throat. 3,4 Moreover, loss of smell, due to olfactory dysfunction, is a common symptom in patients, and it is associated with concomitant subjective gustatory dysfunction (loss of taste). Further, olfactory dysfunction has a significant impact on the quality of life (QoL), and the prevalence of the disorder among COVID-19 patients was reported to be as high as 85.6%. 5,6 In fact, SARS-CoV-2 binds to 2 protein receptors, angiotensin-converting enzyme 2 (ACE2) and transmembrane serine protease 2 (TMPRSS2), both of which are required for host cell entry. 7 Furthermore, considering the high expression of these receptors in the respiratory epithelium and subepithelial glands, SARS-CoV-2 possibly induces conductive olfactory dysfunction through hypersecretion and goblet cell hyperplasia. 8 Actually, chronic rhinosinusitis (CRS) is an inflammation of the nose and the paranasal sinuses which are characterized by the presence of symptoms including nasal blockage (obstruction or congestion), anterior or posterior nasal discharge (PND) with or without facial pain (or pressure), and reduction or loss of smell for more than 12 weeks. Basically, according to the nasal endoscopic findings, CRS is phenotypically classified as CRS with nasal polyps (CRSwNP) and without nasal polyps (CRSsNP). 9 In fact, the nasal cavity plays an important role in COVID-19, and concerning the site of viral entry and replication, nasal symptoms of the disease showed a lower prevalence (less than 10%). 10 Sinonasal symptoms in an acute phase of upper respiratory tract infection (URTI) such as COVID-19 are often due to generalized mucosal edema. 11
This study aimed to investigate the frequency and severity of symptoms of COVID-19 in patients with CRS. In addition, we assessed the severity of CRS symptoms and the QoL of the patients involved with COVID-19 for a period of one year after the onset of the pandemic by using the Sino-Nasal Outcome Test (SNOT-22) questionnaire.
Methods and Materials
Study Design
In this observational and cross-sectional study, 207 adult patients (15-70 years old) who were diagnosed with CRS and registered in Sinusitis Registry Center, affiliated to the Medical University of Bushehr (SRCB), Iran, were participated. Generally, CRS is clinically diagnosed by using the EPOS2020 criteria. 9 The present study was approved by the Ethics Committee of Bushehr University of Medical Sciences, Bushehr, Iran (ethics committee approval code: IR.BPUMS.REC.1399.177), and the informed written consent for the study protocol was obtained from all the participants.
Patient’s Participants
Of the total participants, those who were presented with the symptom of COVID-19 were invited to do the reverse transcription–polymerase chain reaction (RT-PCR) test for SARS-CoV-2. The diagnosis of the patients for COVID-19 was done according to the World Health Organization guideline. 12
Data Collection
The specific questionnaire designed for the study was completed for the patients who had a positive RT-PCR test for SARS-CoV-2 at the end of the second week. Also for those who had a negative test result, this was done by using a well-trained staff of SRCB. The questionnaire included demographic and clinical data of the participants, considering the COVID-19 symptoms, the severity of the disease according to visual analog scale (VAS), the hyposmia or anosmia, length of hospitalization in intensive care unit (ICU) or ward, the pulmonary involvement, hypoxemic condition (SpO2<93%), and other comorbidities. In addition, we identified the allergic diseases in the patients by using the GA 2 LEN questionnaire. Furthermore, we used the SNOT-22 questionnaire to examine the physical impairments, functional and social limitations, disabilities, and QoL effects caused by CRS.
Statistical Analysis
Descriptive analyses of the variables were expressed as mean ± standard deviation for the quantitative variables, and as a percentage for the qualitative variables. We used χ2 tests to identify the presence of hypoxemia and pulmonary system involvement among patients with COVID-19 and CRSwNP. Moreover, we used paired t test for the assessment of the correlation of SNOT-22 outcomes. Finally, we used the software SPSS (version 22.0, IBM Corp) for analysis of the statistical variables. P values less than .05 were set as a significant level.
Results
In the present study, we found PND (91.3%) as the most common symptom among the patients. The most frequent allergic and underlying diseases in the patients were allergic rhinitis (AR) (85.5%) and migraine (13.5%). Of the total patients, those having regular consumption of corticosteroid nasal spray, oral consumption of antihistamines, and having nasal surgeries within the past one year were 31.9%, 33.3%, and 3.1%, respectively. Demographic and clinical data of the patients are shown in Table 1.
Demographic and Clinical Data of the Participants.
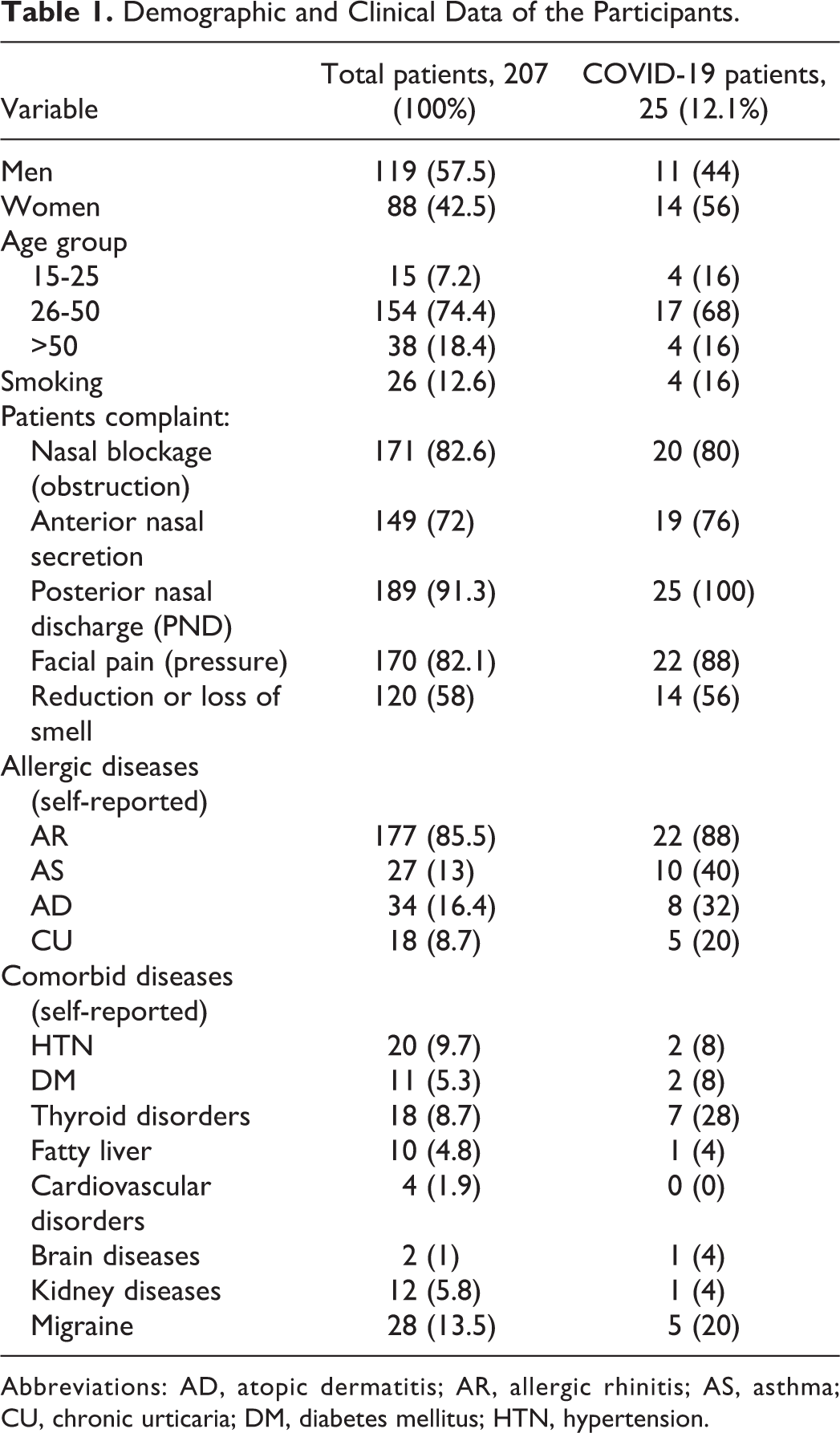
Abbreviations: AD, atopic dermatitis; AR, allergic rhinitis; AS, asthma; CU, chronic urticaria; DM, diabetes mellitus; HTN, hypertension.
In addition, the total frequency of COVID-19 in the patients with CRS was 25 (12.1%) of whom, 22 were treated as outpatients, 2 admitted in wards, and 1 in ICU. A significant association was observed between having thyroid disorder and COVID-19 (P = .02). The frequency of hyposmia symptoms in the patients was 2 as mild, 5 moderate, and 11 as severe (anosmia). In addition, the severity of COVID-19 based on VAS was 10 as mild, 5 as moderate, and 10 as severe. Furthermore, the symptoms and other clinical presentations of COVID-19 in the patients with CRS and CRSwNP showed in Table 2. Additionally, the SNOT-22 test outcomes in the patients with and without COVID-19 are shown in Table 3. The average SNOT-22 score in the patients involved with COVID-19 for a period of one year after the onset of the pandemic in patients with CRS is shown in Table 4. Finally, our results showed that the average SNOT-22 score in patients with CRSwNP was significantly higher than compared with the CRSsNP (P < .05).
Symptoms and Findings of COVID-19 in the Patients With CRSwNP in Comparison With CRSsNP.
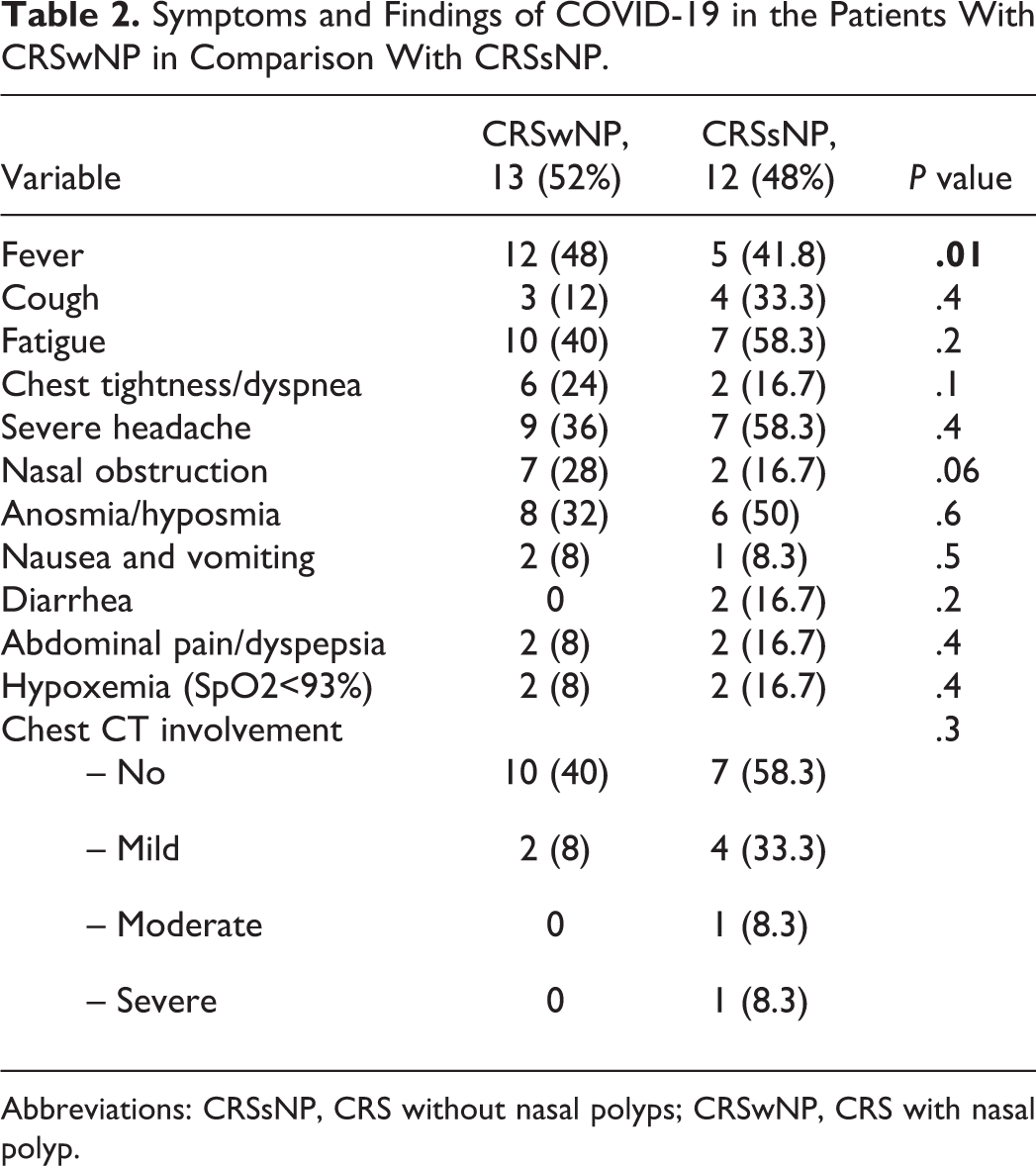
Abbreviations: CRSsNP, CRS without nasal polyps; CRSwNP, CRS with nasal polyp.
SNOT-22 Test Outcomes in the Patients With and Without COVID-19.
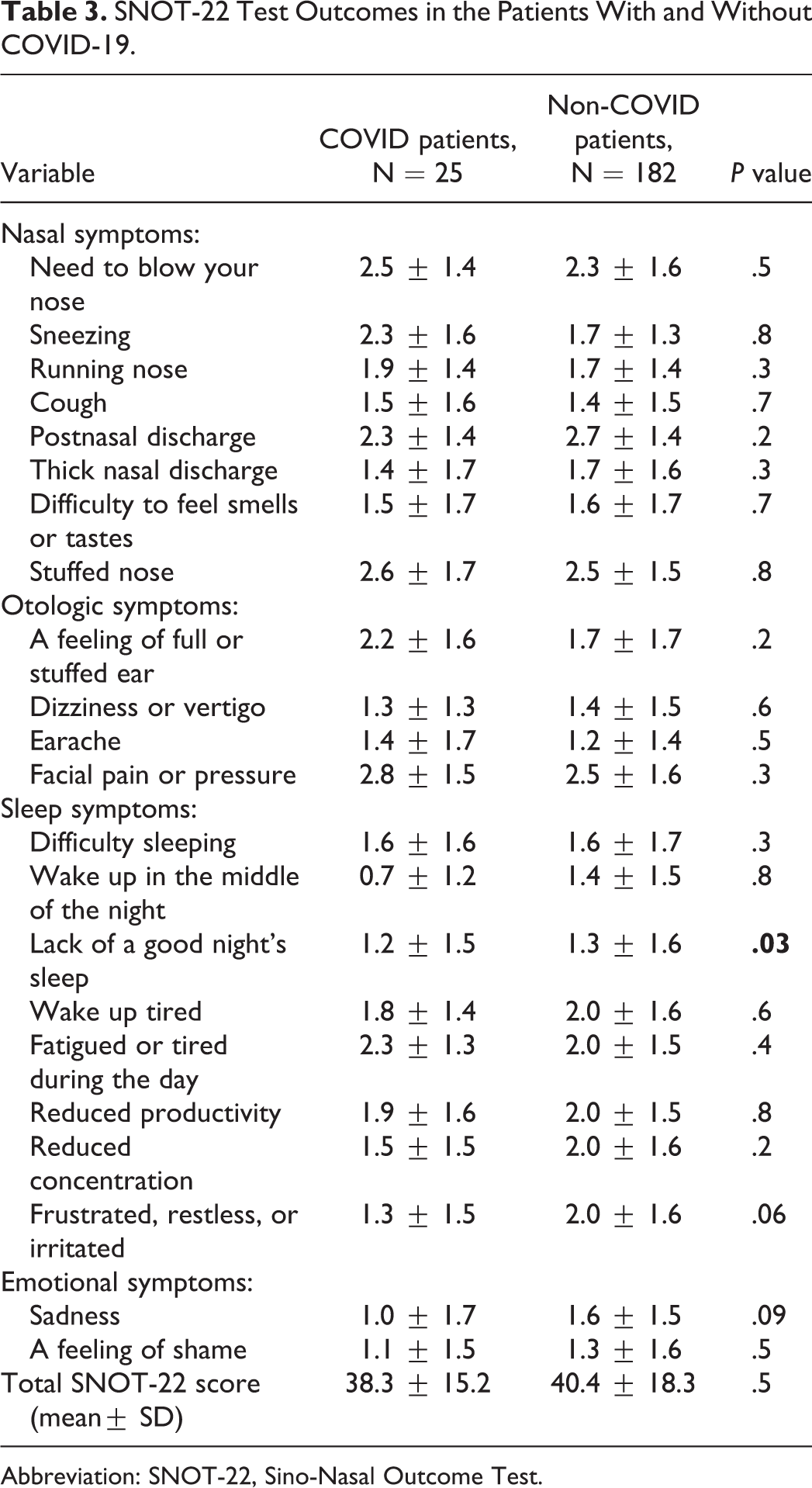
Abbreviation: SNOT-22, Sino-Nasal Outcome Test.
SNOT-22 Test Outcomes in the Patients With CRS Before and After One Year of COVID-19 Pandemic in the Total Patients With CRS.
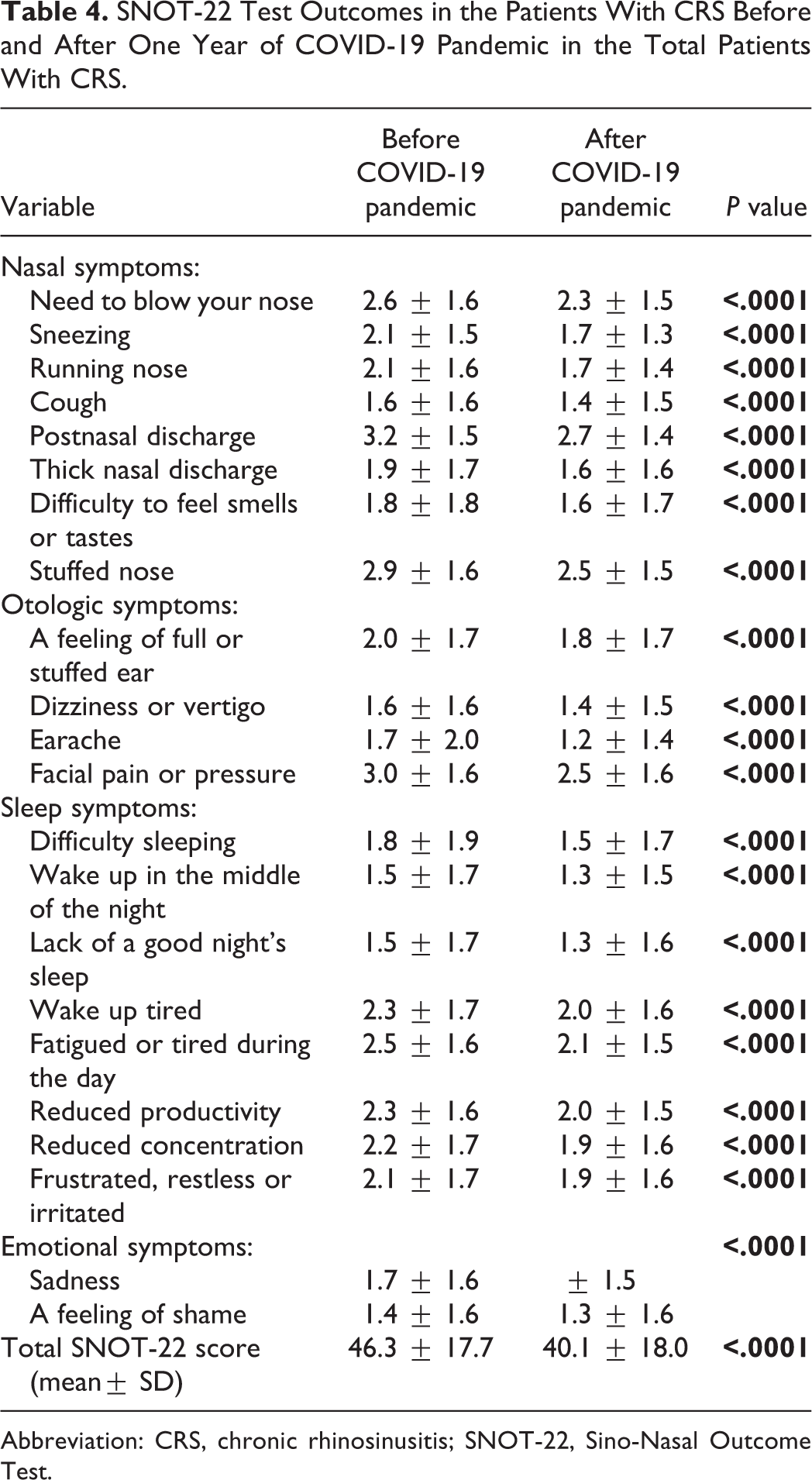
Abbreviation: CRS, chronic rhinosinusitis; SNOT-22, Sino-Nasal Outcome Test.
Discussion
In the present study, we have assessed the frequency and clinical symptoms of COVID-19. Moreover, we evaluated the impact of the pandemic on the QoL of the patients with CRS. Actually, up to date, a few epidemiological studies are being carried out in the world for determining the prevalence of COVID-19 in patients with CRS.
The results of the present study indicated that the prevalence of COVID-19 in patients with CRS was 12.1%. In addition, the findings of our previous study (under review), conducted on 400 COVID-19 patients revealed that the prevalence of asthma, AR, and CRS, was 7.3%, 16%, and 1.8%, respectively. Further, a seroepidemiological study carried out on 11 256 Iranian participants reported that the prevalence of COVID-19 (estimated until August 20, 2020) was 14.2%. 13 Moreover, according to the official reports from the Ministry of Health in Iran, by June 20, 2021, 22 321 457 PCR tests were performed, of which 3 086 974 were positive. On the other hand, the positivity rate of SARS-CoV-2 infection was 13.8% in the general population (http://ird.behdasht.gov.ir/). Meanwhile, the positivity rate of the tests in the region was 11.8% (https://health.bpums.ac.ir). Ultimately, concerning the same rate of infection positivity in the general population and the data obtained from our study, perhaps, CRS could not be a risk factor for COVID-19. A recent study from South Korea reported that the positivity rate for SARS-CoV-2 in the patients with CRS was 3.1% against 2.5% for the group without CRS. Furthermore, they reported that 21% of the CRS patients had severe COVID-19 compared to the group without CRS (13.3%). On the contrary to our study, they concluded that patients with CRS had an increased risk of COVID-19 susceptibility and severity. Moreover, they confirmed that CRSsNP, CRS treated with intranasal corticosteroid, and nonatopic CRS were greater risk factors for SARS-CoV-2 infection and severe COVID-19 than compared to the patients with CRSwNP or atopic CRS. 14
Generally, CRS is a persistent inflammation of the mucous membrane of the nose and paranasal sinuses. 9 Meanwhile, with the use of anti-inflammatory therapies with the topical corticosteroid administration as the baseline therapeutic golden standard for the disease, perhaps, CRS would have a promoting effect on SARS-CoV-2 infection. 15,16 However, studies reported that type 2 immune inflammations are being seen in asthmatic patients. Furthermore, corticosteroid consumption by downregulating the ACE2 and TMPRSS2 receptors in the nose decreases the risk of SARS-CoV-2 infection in patients with respiratory allergic diseases. 16 -18 Therefore, the low frequency of COVID-19 in patients with CRS reported in our study showing that CRS same as asthma and allergic diseases are not risk factors for SARS-CoV-2 infection. This might be because of the immunopathology and the treatment of CRS for which intranasal corticosteroid is the main drug. Furthermore, a recent study found that asthmatic patients with preexisting eosinophilia had a protective effect from COVID-19-related admissions and mortalities. 18 Importantly, these studies suggest that a prominent type 2 immune response caused by nasal polyps may contribute to the low SARS-CoV-2 infectivity and COVID-19 severity compared with CRSsNP. Therefore, we conclude that CRS especially CRSwNP is related to type 2 inflammation and, most probably, could not be an underlying disease for the high prevalence of SARS-CoV-2 infection.
Moreover, the results of the present study were in line with other studies. 11,19,20 showing that 18 (72%) patients with COVID-19 of 25 presented the loss of smell. Further, according to the information obtained from the questionnaire, there were 2 patients as mild, 5 as moderate, and 11 as severe hyposmia (anosmia). One of the limitations of this study was that we were not able to perform the olfactory tests, because of the contagious nature of the disease, and the median long duration of the symptoms (about 10 days). Therefore, this study confirmed that the loss of smell and taste could be 2 important parameters for the clinical diagnosis of COVID-19. 19,21 However, loss of smell and taste due to olfactory dysfunction can occur following the wide range (18%-42%) of URTIs. Furthermore, we suggest that severe inflammation of the nasal mucosa and the obstruction due to prominent mucosal edema and the subsequent obstruction in the middle and the lower parts of the nasal cavities block the passage of olfactory molecules to the clefts. However, the SARS-CoV-2 is a neurotropic virus that can spread to the central system from the peripheral olfactory. The hypothesis of direct damage of the olfactory pathways by the SARS-CoV-2 is highly probable and could explain the smell loss in patients without sinonasal symptoms or with the persistent olfactory loss after the acute phase of the infection. 11,22,23
In fact, the SNOT-22 represents the reference questionnaire to assess the health status and health-related QoL in patients with CRS. 24 However, patient-specific factors may affect the degree of SNOT-22 changes after the intervention modalities. 25 Noticeably, our data revealed that there was no difference in the average SNOT-22 score in the 4 domains (nasal, otologic, sleep, and emotional symptoms) of CRS patients with COVID-19. Ultimately, COVID-19 did not aggravate sinonasal symptoms in patients with CRS which might affect the QoL. Although, our study showed that the SNOT-22 scores obtained after one year of the onset of the pandemic were significantly lower in CRS patients compared with the pre-pandemic, but the sum of mean differences of SNOT-22 scores was less than the minimal clinically important difference (MCID). This could be due to the effect of the period of the pandemic on CRS patients, while MCID measures the effects of medical management on the patients. Moreover, our study showed that the MCID was less than 9; however, the cutoff point for MCID is approximately 9. Importantly, the MCID may be quite specific but poorly sensitive for the patients with the improvement in CRS symptoms. 26 Certainly, the performance of the health protocols such as masking and frequent washing of hands and face helps in preventing the occurrence of respiratory viral infections, without interfering with the effectiveness of the treatment.
Finally, our data showed that the symptoms of COVID-19, hypoxemia, and pulmonary system involvement in the patients with CRSwNP were not different from the CRSsNP.
Conclusion
In the present study, we conclude that the low frequency of COVID-19 in patients with CRS and its similarity with infection positivity in the general population confirms that CRS could not be considered as a risk factor for COVID-19. Moreover, the most frequent symptoms were shown to be hyposmia/anosmia. Furthermore, the low average score of SNOT-22 observed after one year from the onset of the pandemic in the patients with CRS suggests the necessity for performing the health protocols by the patients.
Footnotes
Acknowledgment
This study was funded by Bushehr University of Medical Sciences (BPUMS), Iran.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
