Abstract
Objectives:
The role of autoimmune diseases on the risk for acute epiglottitis remains uncertain. This study aimed to delineate the association between epiglottitis and autoimmune diseases using population database.
Methods:
A population-based retrospective study was conducted to analyze claims data from Taiwan National Health Insurance Research Database collected over January, 2000, to December, 2013.
Results:
In total, 2339 patients with epiglottitis were matched with 9356 controls without epiglottitis by sex, age, socioeconomic status, and urbanization level. The correlation between autoimmune diseases and epiglottitis was analyzed by multivariate logistic regression. Compared with controls, patients with epiglottitis were much more likely to have preexisting Sjögren syndrome (adjusted odds ratio [aOR]: 2.37; 95% CI: 1.14-4.91; P = .021). In addition, polyautoimmunity was associated with increased risk of epiglottitis (aOR: 2.08; 95% CI: 1.14-3.80; P = .018), particularly in those aged >50 years (aOR: 2.61; 95% CI: 1.21-5.66; P = .015).
Conclusions:
Among autoimmune diseases, we verify the association between epiglottitis and Sjögren syndrome in Taiwan. Furthermore, we present the novel discovery that patients with epiglottitis have an increased risk of polyautoimmunity, particularly those aged >50 years.
Introduction
Epiglottitis is a life-threatening medical emergency, with the potential risk of sudden airway obstruction secondary to the extensive inflammatory response in the epiglottis, aryepiglottic folds, and arytenoid regions. Bacterial infection is the most common etiology; other causes are viral or fungal infection, chemical ingestion, and inhalation burns. 1 Acute onset of epiglottitis symptoms in children usually occurs in those who were formerly healthy, whereas in >50% of adult cases, epiglottitis occurs in those with underlying comorbidities such as alcohol abuse and diabetes mellitus (DM), which can compromise their immunity and increase susceptibility to various infections. 2 -4 Although epiglottitis is a severe inflammatory disease, it also occurs in patients with compromised immune system or impaired inflammatory response. 5,6 Epiglottitis in immunocompromised patients is reported together with various underlying conditions and is associated with higher mortality than epiglottitis in immunocompetent patients, which makes early diagnosis and treatment crucial. 5
A primary defect in the immune system, the long-term use of immunosuppressive therapy, complement abnormalities, alterations to the innate and adaptive immune response, and splenic dysfunction may all contribute to patients with autoimmune diseases having increased susceptibility to encapsulated bacteria such as Haemophilus influenza as well as to meningococcal, salmonella, and pneumococcal infection, which are major pathogens of the infectious epiglottitis. 5,7,8 Moreover, mucosa ulcer and edema of epiglottic structures, cricoarytenoid arthritis, and necrotizing vasculitis have been reported in noninfectious epiglottitis caused by autoimmune disease with laryngeal involvement. 9 -11 Studies have highlighted the high prevalence of supraglottic involvement in patients with Sjögren syndrome (SS), Behcet disease, rheumatoid arthritis (RA), and systemic lupus erythematosus (SLE), 10,12 -16 and case reports have indicated that epiglottitis might present as the initial or flare manifestation of SLE. 11,17
Although the aforementioned studies have discovered crucial connections between the 2 rare disease entities, the lack of a control group and small samples have limited the research. Considering expansion of these research findings, an association can be reasonably hypothesized to exist between the autoimmune diseases and epiglottitis; however, clear clinical advice cannot yet be formulated because of the lack of robust evidence. In the current nationwide population-based case–control study, we investigated the association between epiglottitis and autoimmune diseases—particularly SS, RA, SLE, and ankylosing spondylitis (AS)—so as to determine whether this association is substantial and thereby to identify early risk factors for acute epiglottitis in the Taiwanese population.
Methods
Study Design and Data Source
We performed this retrospective nationwide case–control study using data from Taiwan National Health Insurance (NHI) Research Database (NHIRD). Taiwan government established NHI in 1995, which was covering >99.6% of residents in 2018, 18 and NHIRD details on all the medical information of NHI beneficiaries, including surgery and intervention procedure types, prescribed drugs, residence location, monthly income level, and disease diagnoses at clinic visits or hospitalization according to the International Classification of Diseases, Ninth revision, Clinical Modification (ICD-9-CM) codes. 18
In the present study, the Longitudinal Health Insurance Database 2005 (LHID2005) data were employed to generate case and control groups for the formal analysis. Longitudinal Health Insurance Database 2005 is a representative subdatabase of NHIRD and comprises the entire medical claims of 1 million insured people, who were randomly selected in 2005 through systematic sampling of NHIRD. 19 According to the reports from the National Health Research Institutes, all individuals in LHID2005 have nonsignificant differences in their age, sex, and health care costs compared with all enrollees in NHIRD. 19 This study was approved by the Institutional Review Board of Chang Gung Memorial Hospital (201901186B1), which waived the need for obtaining informed consent because NHIRD data are made suitable for public research through anonymization and deidentification by scrambling identification codes. This study was also conducted in compliance with the Declaration of Helsinki principles.
Selection of Cases and Controls Through Matching
A flowchart showing how patients were selected is presented in Figure 1. To form the case group, we enrolled from LHID2005 all patients with new otolaryngologist-made diagnosis of epiglottitis (ICD-9-CM codes 464.3, 464.30, and 464.31) in an inpatient claim or at least 3 outpatient claims during January 1, 2000, to December 31, 2013. Only cases of epiglottitis diagnosed by otolaryngologists were included, and patients with concomitant deep neck infection were excluded to increase the accuracy of diagnosis. Finally, 2339 patients with epiglottitis comprised the case group. To ensure that our statistical power would be high, 1:4 matching of cases with randomly selected controls without epiglottitis was performed. The control group was obtained from LHID2005 according to the case index date. In total, the control group comprised 9356 patients, frequency-matched to the case group in age, sex, degree of urbanization level, and socioeconomic status.
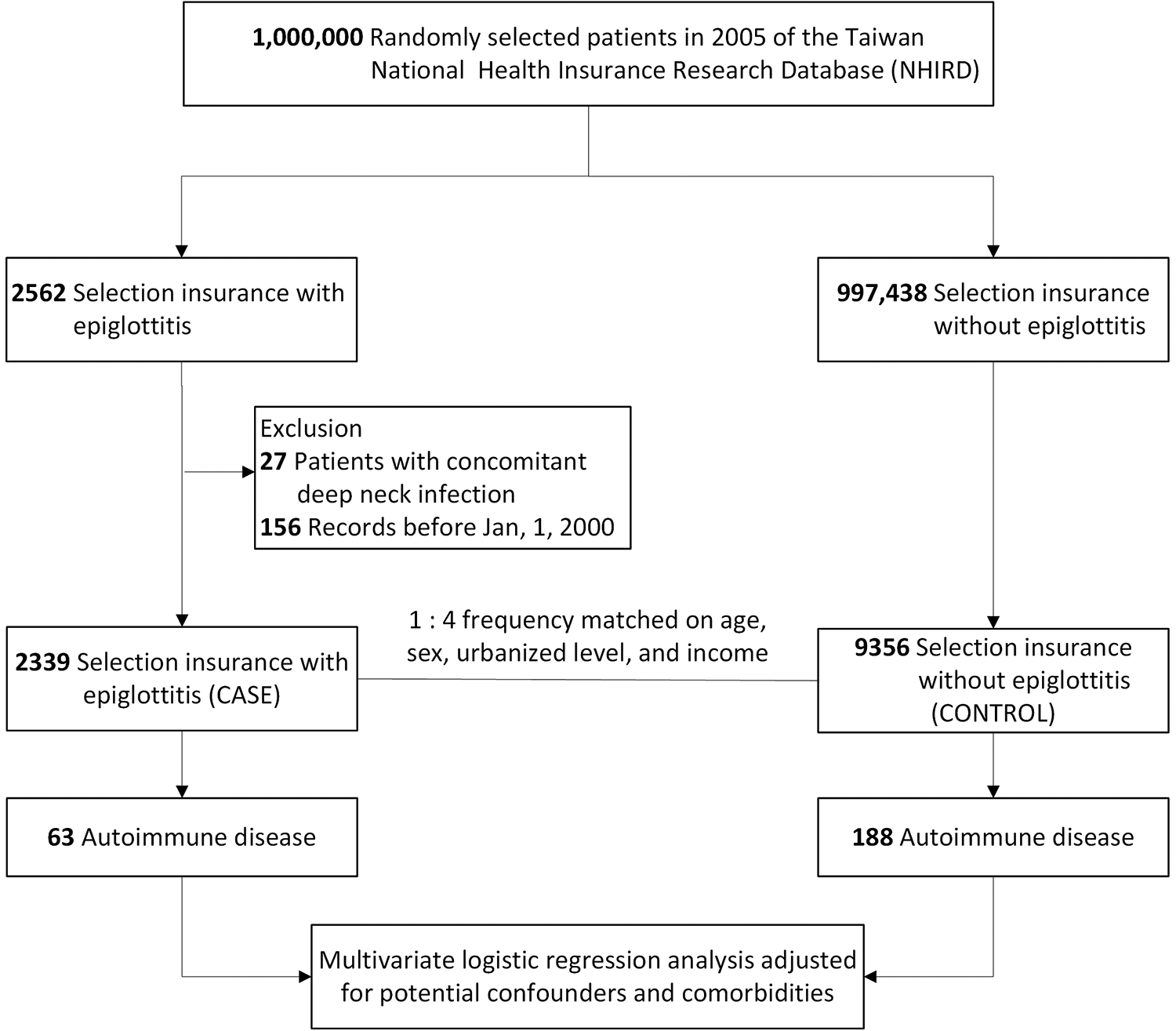
Flow chart of the case and control group selections.
Autoimmune Diseases and Other Covariates
The present study’s primary outcome of interest was the prevalence of preexisting autoimmune diseases during the study period. Patients in both groups who had a diagnosis of specific comorbid autoimmune disease were identified by searching for ICD-9-CM codes 720 and 720.0 for AS; 714 and 714.xx for RA; 710.0 for SLE; 710.2 for SS; 710.3 and 710.4 for dermatopolymyositis; 245.2 for Hashimoto thyroiditis; 136.1 for Behcet disease; 242.0, 242.00, and 242.01 for Graves disease; 340 for multiple sclerosis; 710.1 for systemic sclerosis; 696, 696.1, and 696.0 for psoriasis; 360.01–360.03, 360.1, 360.11, 360.12, 360.14, 360.19, 362.18, 363.0x, 363.1x, 363.2x, 364.0, 364.00, 364.01–364.05, 364.1x, 364.2x, and 364.3 for incident uveitis; and 358.0 for Myasthenia gravis. Given that study has highlighted the occurrence of distinct autoimmune diseases within an individual, 20 the polyautoimmunity is defined as at least 2 autoimmune diseases being recorded in a single patient during the study period. Whether each case patient had an autoimmune disease before epiglottitis and whether each control patient had an autoimmune disease before the matched index date were determined. As well as for age and sex, adjustments were made for degree of urbanization of residence, monthly income, and the following concomitant covariates related to epiglottitis 21,22 : liver cirrhosis (LC, ICD-9-CM 571.xx), chronic obstructive pulmonary disease (COPD, ICD-9-CM 490-496), coronary artery disease (CAD, ICD-9-CM 410-414), hypertension (HTN, ICD-9-CM 401-405), DM (ICD-9-CM 250.xx, A-code A-181), and chronic kidney disease (CKD, ICD-9-CM 582, 582.xx, 585, and 586). In order to enhance the accuracy of the diagnosis, a diagnostic code of above-mentioned diseases in an inpatient care claim or at least 3 outpatient claims prior to the index date was used to indicate that one patient had the comorbid disease. 23 We analyzed the covariates as binomial variables.
Statistical Analysis
Intergroup differences in baseline characteristics were determined using Pearson chi-square testing for nominal data. In order to investigate the association of autoimmune diseases with acute epiglottitis, conditional logistic regression analysis was performed with control for potential confounders—including sex, age, urbanization level, monthly income, COPD, DM, HTN, LC, CAD, and CKD—to calculate the adjusted odds ratio (aOR) and 95% CI. Logistic regression was performed in subgroup analysis to identify the associations between epiglottitis risk and the number of preexisting autoimmune diseases when adjusting for age, sex, income, degree of urbanization of residence, and covariates. All analyses were performed on SAS (version 9.4; SAS Institute). We considered a 2-sided P of <.05 to indicate the significance of the associated result.
Availability of Data and Material
The data underlying this study is from the NHIRD, which has been transferred to the Health and Welfare Data Science Center (HWDC). Interested researchers can obtain the data through formal application to the HWDC, Department of Statistics, Ministry of Health and Welfare, Taiwan (http://dep.mohw.gov.tw/DOS/np-2497-113.html).
Results
Characteristics of Case and Control Cohorts
Between January 1, 2000, and December 31, 2013, the case group inclusion criteria were met by 2339 patients (1203 males and 1136 females) with a new diagnosis of acute epiglottitis coded by otolaryngologist. The matched control group comprised 9356 patients without epiglottitis. Approximately half of both groups were male (51.4%), and the majority of patients were 50 years or younger (72.7%). The median age of all 11 695 study enrollees was 34 (interquartile range: 10-51) years. The case group had higher incidences of preexisting SLE (P = .043) and SS (P = .005, Table 1). The prevalence of other 2 major autoimmune diseases, AS and RA, was higher in the case group, but not statistically significant (P = .167 and .067, respectively). For some diseases, such as dermatopolymyositis, Behcet disease, systemic sclerosis, and myasthenia gravis, the number of patients was too small to be analyzed. Significant intergroup differences were discovered in the percentage of numbers of polyautoimmunity (P = .016), COPD (P < .001), DM (P = .004), HTN (P = .031), LC (P = .002), and CAD (P = .008).
Demographic Characteristics of Patients With Epiglottitis and Matched-Control Group.
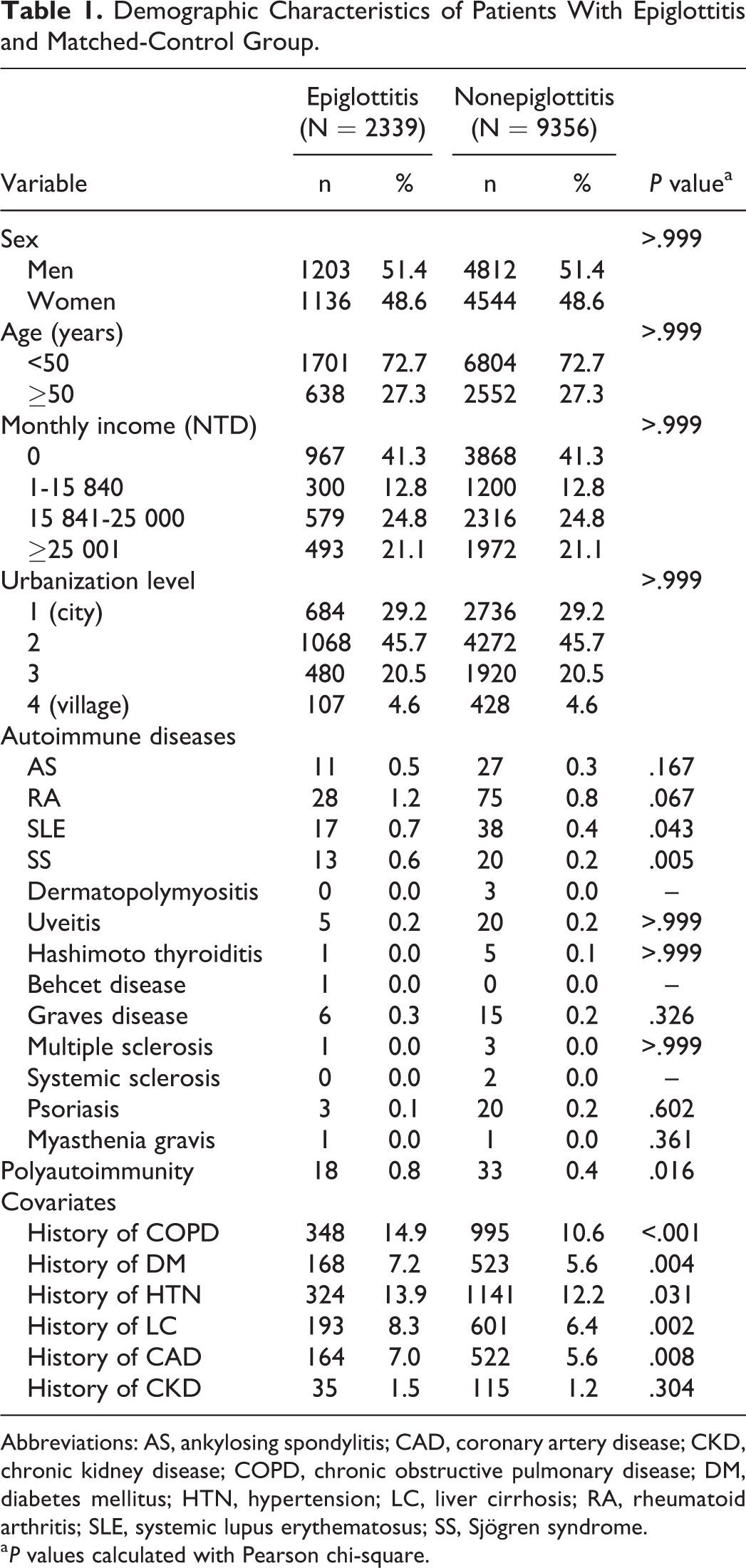
Abbreviations: AS, ankylosing spondylitis; CAD, coronary artery disease; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; HTN, hypertension; LC, liver cirrhosis; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SS, Sjögren syndrome.
a P values calculated with Pearson chi-square.
Characteristics of Patients With Epiglottitis
The case group’s baseline characteristics are presented in Table 2, which showed that the diagnosis was made at similar ages in the male and female patients with epiglottitis (P = .734). Hashimoto thyroiditis, multiple sclerosis, and myasthenia gravis were only found in the female patients with epiglottitis, and polyautoimmunity was more common in the female patients with epiglottitis (P = .016, Table 2).
Characteristics of Patients With Epiglottitis.
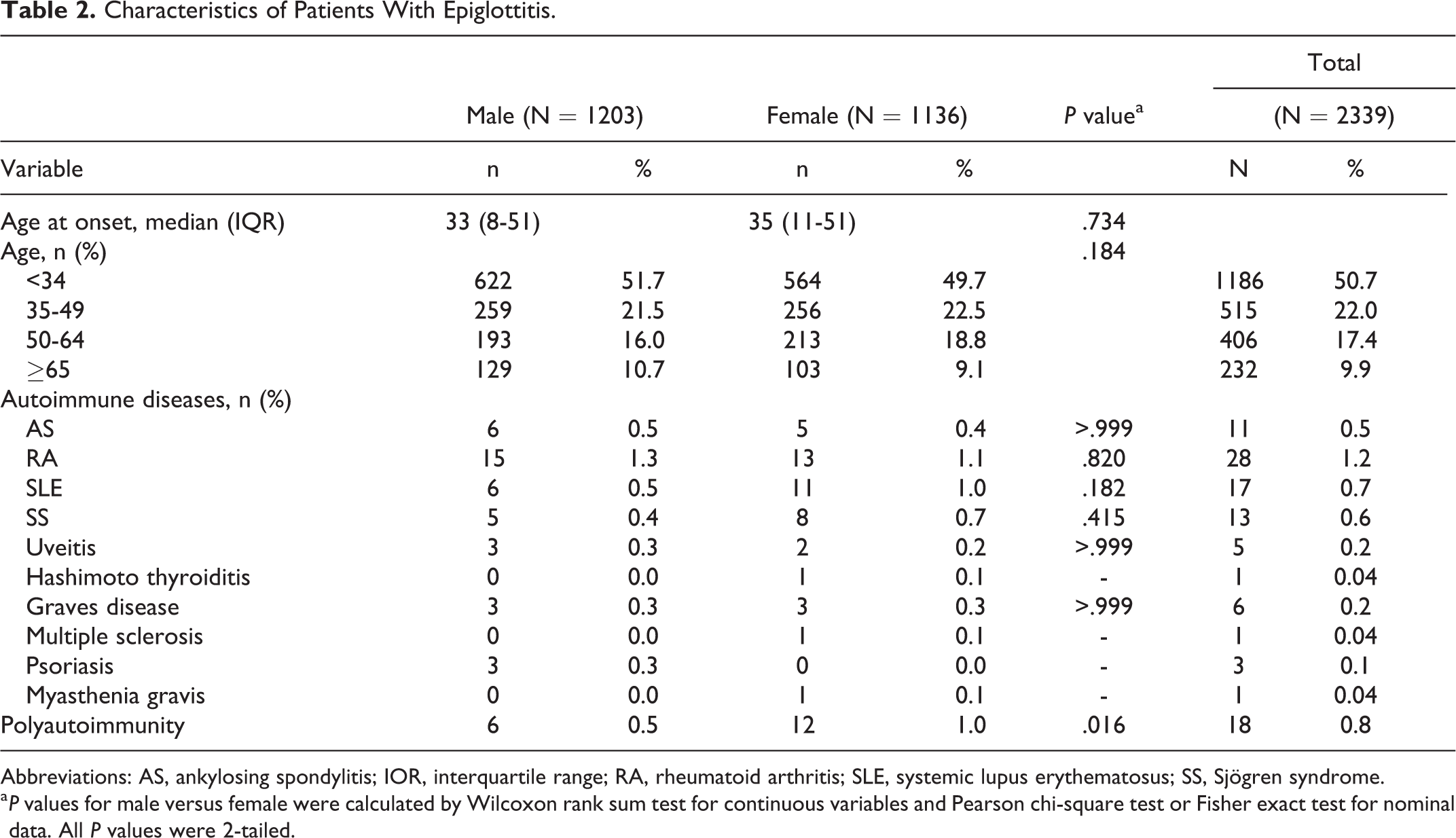
Abbreviations: AS, ankylosing spondylitis; IOR, interquartile range; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SS, Sjögren syndrome.
a P values for male versus female were calculated by Wilcoxon rank sum test for continuous variables and Pearson chi-square test or Fisher exact test for nominal data. All P values were 2-tailed.
Association Between Autoimmune Diseases and Epiglottitis
Compared with the controls, for the patients with epiglottitis, the odds of preexisting autoimmune diseases were greatest for SS (aOR: 2.37; 95% CI: 1.14–4.91; P = .021, Table 3) after adjustments for age, sex, monthly income, degree of urbanization of residence, and covariates. Because the epiglottitis may not be associated with the autoimmune disease diagnosed shortly before it attacked, we repeated the analysis by excluding patients with autoimmune disease diagnosed within 6 and 12 months before epiglottitis to ensure a lag time between preexisting autoimmune disease and epiglottis. The results were consistent and presented in the Supplement File. Other concomitant covariates including COPD and LC also had significant associations with epiglottitis. Risk of epiglottitis was not found to be significantly associated with the history of RA, SLE, AS, uveitis, Hashimoto thyroiditis, Graves disease, multiple sclerosis, or psoriasis (P = .170, .775, .261, .829, .778, .461, .808, and .465, respectively).
Association Between Epiglottitis and Autoimmune Diseases.
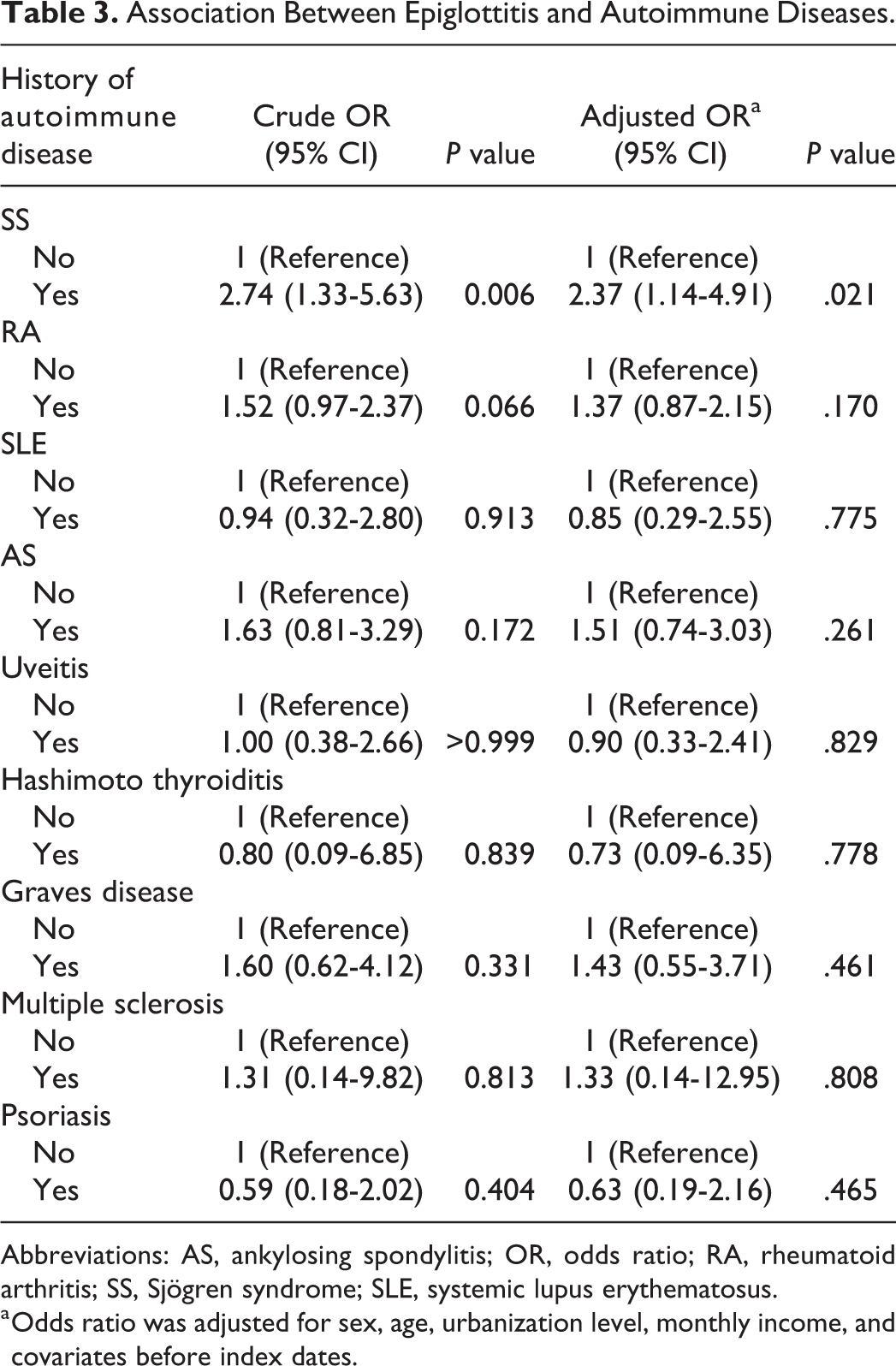
Abbreviations: AS, ankylosing spondylitis; OR, odds ratio; RA, rheumatoid arthritis; SS, Sjögren syndrome; SLE, systemic lupus erythematosus.
a Odds ratio was adjusted for sex, age, urbanization level, monthly income, and covariates before index dates.
Associated Autoimmune Disease Counts Stratified by Age and Sex
Table 4 details the subgroup analysis on the association between the risk of epiglottitis and the polyautoimmunity. Compared with no autoimmune disease, patients with polyautoimmunity were susceptible to epiglottitis (aOR: 2.08; 95% CI: 1.14-3.80; P = .018). We then performed age-group stratification with the cutoff age of 50 years, 24 which demonstrated a significant association between epiglottitis and polyautoimmunity among those who are aged 50 years or older (aOR: 2.61; 95% CI: 1.21-5.66; P = .015); this association was absent in the younger age-group. In both sexes, having more than one autoimmune disease was associated with elevated epiglottitis risk, but the result was nonsignificant (aOR: 2.39; 95% CI: 0.84-6.81; P = .103 for men, and aOR: 1.98; 95% CI: 0.94-4.15; P = .072 for women).
Subgroup Analyses for Associations Between Epiglottitis and Autoimmune Disease Counts.
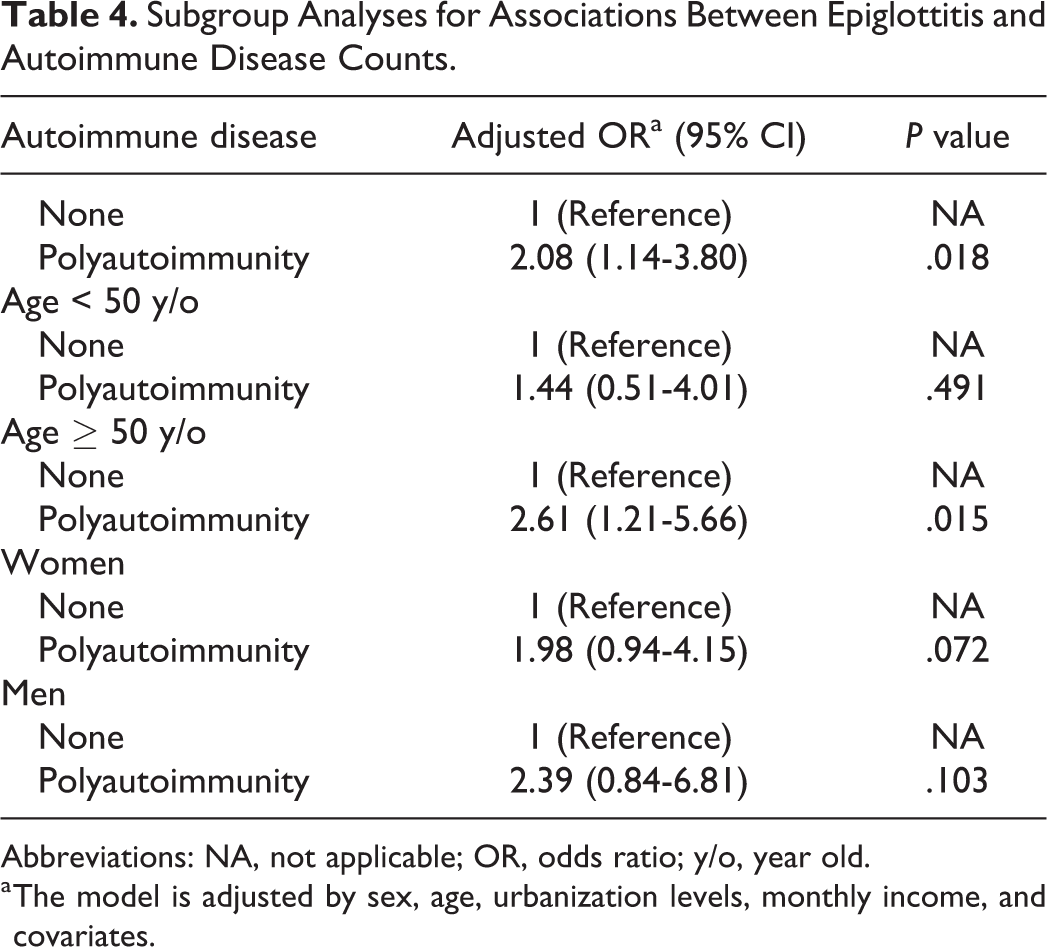
Abbreviations: NA, not applicable; OR, odds ratio; y/o, year old.
a The model is adjusted by sex, age, urbanization levels, monthly income, and covariates.
Discussion
Inconsistent conclusions have been drawn regarding whether patients with autoimmune disease are predisposed to epiglottitis, 25 and studies investigating the autoimmune disease–epiglottitis relationship are limited. 12 -14,16,17,26 Based on our literature review, the present research is the first population-based study on the epiglottitis–autoimmune disease association. In working to this end, obtaining a sufficiently large sample with long observation period from only one medical institution would be difficult given the rarity of the diseases investigated. Accordingly, we used a nationwide database to recruit enough patients with epiglottitis while minimizing the selection bias. In addition, we adopted the aOR to compare the case and control groups to minimize the effects of potential covariates. Once various covariates had been accounted for—including COPD, CKD, DM, HTN, CAD, and LC—associations were found of preexisting SS and polyautoimmunity with elevated risk of epiglottitis in this case–control study involving 11 695 people. According to the age-stratified analysis, the polyautoimmunity–epiglottitis association was strongest in the 50 years and older age-group, and this may be explained by an overlap of the age distributions of adult patients with epiglottitis and those with more than one autoimmune disease. 24,27 This study extends the literature on epiglottitis and can raise awareness regarding the association of SS and polyautoimmunity with high epiglottitis risk. For patients with epiglottitis and repeated episodes or a refractory treatment course, underlying comorbid SS or polyautoimmune diseases should also be considered, particularly in those more than 50 years old.
Patients with autoimmune disease are susceptible to higher risk of respiratory tract infection caused by Streptococcus pneumoniae and H influenzae, 28,29 common pathogens involved in epiglottitis. The clinical presentation of epiglottitis in patients with autoimmune or immunocompromised disorders follows a highly variable course, ranging from a mild symptomatology to fulminant, life-threatening airway compromise. 5,26 Odynophagia, dysphonia, stridor, and dysphagia should alert primary care physicians to the presence of epiglottitis, 30 which warrant appropriate differential diagnosis, timely referral, and thorough radiologic or fiberoptic laryngoscopic evaluation for early diagnosis and intervention. 10,31 Epiglottitis treatment, particularly in the case of patients with comorbid autoimmune diseases, is centered on airway management, given that laryngeal involvement in autoimmunity may exacerbate the upper airway obstruction. 25,32 An 8-year retrospective review revealed that 13.2% and 3.6% of patients admitted for epiglottitis underwent intubation and tracheostomy, respectively. 21 In patients with both epiglottitis and autoimmune disease, stridor and respiratory distress may result either from supraglottic inflammation and edema or from cricoarytenoid arthritis, both of which are robust predictors of airway intervention. 13,32,33 Subtler signs and symptoms, such as rapid symptom onset, tachypnea, and tachycardia, which may mislead clinicians, were also reported to predict airway compromise in a retrospective review. 33 The use of corticosteroid as an adjunct in epiglottitis treatment 21 may not only control the underlying autoimmune comorbidities but also shorten the intensive care unit stay and overall hospital stay. 33,34 However, steroid use in the management of epiglottitis is controversial. 35 Further prospective studies are warranted to probe the effects of steroid in patients with epiglottitis comorbid with autoimmune disease.
The association between autoimmune disease and epiglottitis has been investigated by few researchers. Bizaki et al retrospectively investigated 308 patients with epiglottitis to discover that 20.8% of the patients had concomitant autoimmune disease. 2 They indicated that unusual pathogens may lead to epiglottitis in immunocompromised patients with increased risk of death, but their study lacked a control group, making further analysis of the association between 2 disease entities impossible. Although the exact pathogenesis between epiglottitis and autoimmune disease remains unclear, our study obtained the novel finding that epiglottitis risk is much higher in patients with polyautoimmunity than in those without autoimmune disease, particularly in patients aged >50 years. This novel observation may be partially due to the prevalence of antinuclear antibodies, the most common biomarker of autoimmunity, is higher in older adults (age ≥50 years) 24 and patients with polyautoimmunity having more complicated underlying immune dysregulation and receiving more intense treatment, such as immunosuppressant or immunomodulatory drugs, leading to vulnerable respiratory immunity and higher susceptibility to epiglottitis after a long disease course. Polyautoimmunity is also a frequent condition in patients with SS, 36 and our study results showed that among autoimmune diseases, SS had the strongest association with the risk of epiglottitis. Increasing studies suggest that SS is associated with several lower respiratory tract manifestations, such as chronic interstitial lung disease and tracheobronchial disease, and recurrent respiratory infections are reported in 10% to 35% of patients with SS. 37 However, both upper aerodigestive tract and the lower airway may be involved in the disease course of SS. From a population-based study, Chang et al reported SS to be a risk factor for chronic sinusitis, with the risk being increased 2.5-fold. 38 Thick secretion in the upper respiratory tract may lead to impaired mucociliary function 39 and exacerbated upper airway obstruction. 40 The decreased amylase and carbonic anhydrase secretions may further cause a deficit of supraglottic innate immunity in patients with SS. 41 In addition, Belafsky and Postma discovered that individuals with SS were predisposed to laryngopharyngeal reflux, which may lead to chronic mucosal destruction and inflammation of the supraglottic area. 12 In the histopathological examination, patients with SS had severe glandular atrophy and marked lymphocytic infiltration in the supraglottic airway, suggesting that SS may be essential to the pathogenesis of noninfectious inflammatory epiglottitis. 42 These study results may explain the findings in our study and provide crucial clues for elucidating the pathogenesis of epiglottitis from an immunologic perspective. Nevertheless, our results do not appear to be completely consistent with those of prior studies, 9,10 which were mainly case reports or series, because we did not observe significant association between epiglottitis and RA or SLE, possibly due to the ethnic differences and the lack of statistical power. In addition, studies have reported laryngeal involvement in patients with dermatopolymyositis, Behcet disease, systemic sclerosis, and myasthenia gravis. 43 -46 However, determining whether these diseases were associated with epiglottitis was difficult in our study because of the small sample size.
The present study has various strengths. First, it included a large sample (2339 patients with epiglottitis) from a nationwide population database and had long study duration. Epiglottitis studies performed in tertiary referral centers usually lack generalizability because of referral bias; this population-based study broadens the general applicability of this issue. Second, we considered epiglottitis to have been confirmed only when the diagnosis had been specifically coded in claims data by an otolaryngologist in addition to an ICD-9 code being present in the database. In addition, we did not include patients with concomitant deep neck infection; therefore, our results should not be biased by the presence of these diseases. There are several limitations in this study. First, despite the use of a national database in the present study, the sample size was small because of the rarity of epiglottitis. Even the lower number of patients with autoimmune comorbidities precluded us from analyzing their associations with epiglottitis. Second, detailed laboratory data indicating inflammation, infection, and imaging reports are not currently available in the database; hence, we could not examine whether the severity of autoimmune disease was correlated with the severity of epiglottitis. Third, no data are available in NHIRD regarding certain potential risk factors of epiglottitis and impaired immunity, such as cigarette smoking and alcohol consumption. 47 However, a study identified COPD as an independent factor associated with cigarette smoking 48 ; adjustment for COPD likely reduced the potential impacts of smoking on the present results. Fourth, the overlapping nature of autoimmune diseases, particularly the SS, 49 may lead to a degree of bias in favor of SS and polyautoimmunity as contributing factors of epiglottitis. In view of this, we performed subgroup analysis across both sexes and patients younger or older than 50 years, and more large-scale prospective studies are warranted to further clarify our observations. Finally, the lack of definite etiology of epiglottitis in NHIRD is another limitation that precluded us to compare diseases, such as immunoglobulin G4-related disease, 50 with SS in causing epiglottitis. Although the study results supported our hypothesis and reached statistical significance, the reader should consider the aforementioned limitations when interpreting the findings.
In conclusion, this is the first study to investigate the association between autoimmune disease and epiglottitis in Taiwan using a real-world database. We confirm the association between epiglottitis and SS. A novel finding is that patients with epiglottitis, particularly those older than 50 years, are at higher risk of polyautoimmunity. The present results serve as a reference for clinicians with regard to the early diagnosis and treatment of epiglottitis in patients with autoimmune comorbidities. The mechanism underlying SS, polyautoimmunity, and the development of epiglottitis warrants further investigation.
Supplemental Material
Supplemental Material, sj-pdf-1-ear-10.1177_01455613211033689 - Epiglottitis in Patients With Preexisting Autoimmune Diseases: A Nationwide Case–Control Study in Taiwan
Supplemental Material, sj-pdf-1-ear-10.1177_01455613211033689 for Epiglottitis in Patients With Preexisting Autoimmune Diseases: A Nationwide Case–Control Study in Taiwan by Cheng-Ming Hsu, Ming-Shao Tsai, Yao-Hsu Yang, Ko-Ming Lin, Yun-Ting Wang, Shu-Yi Huang, Meng-Hung Lin, Ethan I. Huang, Geng-He Chang, Chia-Yen Liu and Yao-Te Tsai in Ear, Nose & Throat Journal
Footnotes
Authors’ Note
CMH and YTT wrote the main manuscript text. MST, YHY, and KML designed the study. SYH, CYL, and MHL analyzed data and prepared tables. EIH, GHC, and YTW prepared figures. YTT supervised the study. All authors reviewed the manuscript.
Acknowledgments
We extend our thanks to the Health Information and Epidemiology Laboratory (CLRPG6G0043) for its data analysis support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding through a grant from the Chiayi branch of Chang Gung Memorial Hospital (CFRPG6J0101) and utilized NHIRD, which the Department of Health and Central Bureau of the NHI established and which the National Health Research Institutes maintain. The inferences and opinions herein are not reflective of those of any of these organizations.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
