Abstract
Perineurioma (PN) is an uncommon benign peripheral nerve sheath tumor. For the rarity of this tumor in the oral cavity, otolaryngologists and oral surgeons might not be familiar with this entity. Perineuriomas are typically benign and complete excision is deemed adequate management. Thus, their histological recognition is mandatory to avoid unnecessary overtreatment. We report the clinicopathologic findings of an uncommon variant, the Extraneural Sclerosing PN, in an unusual and never described site, the tongue.
Perineurioma (PN) is an uncommon, often underrecognized and nearly always benign peripheral nerve sheath tumor (PNST) composed entirely of neoplastic cells that show ultrastructural and immunohistochemical features similar to those of their normal cell counterpart. 1,2 Two main variants of PN have been distinguished. 1,2 The intraneural PN is typically restricted to peripheral nerve boundaries, enlarges the affected nerve, and can cause motor and/or sensory dysfunction. In contrast, the extraneural PN (EPN) variant is not associated with peripheral nerves, most often presents as asymptomatic subcutaneous nodule and based of the clinicopathologic features includes different variants among which the soft tissue and the sclerosing (S-EPN) ones are the most common. In the tongue, PNs are rare. Indeed, only 13 cases (8 intraneural and 5 extraneural) were reported in a recent review of the literature. 3
Herein, we present a 51-year-old Caucasian male with a 6-month history of painless lump involving the posterior third of the tongue. He was healthy and denied trauma, history of tongue tumors, or previous local surgery. The lesion was moveable and approximately 6 mm in greatest dimension. The overlying surface was smooth and pink. The rest of the ENT examination was normal. No cervical lymphadenopathies were clinically evident. An excisional biopsy was performed under local anesthesia.
Histologic examination revealed a circumscribed but unencapsulated mucosal-based nodular proliferation small spindle to oval epitheliod cells with indistinct cell borders forming ill-defined fascicles in a dense sclerotic collagenous matrix (Figure 1). Cell pleomorphism, mitosis, and necrosis were absent. The overlying epithelium was unremarkable. Immunohistochemical analysis (Figure 2), performed as previously, 4 -6 revealed immunoreactivity of the neoplastic cells for Vimentin, EMA, Claudin-1, CD34, and CD99 and their negativity for S100, CD117, Desmin, smooth muscle actin, muscle-specific actin, and progesterone receptor. Many mastocytes were highlighted by CD117. The proliferative index (Ki67) was less than 1%. The pathological findings were consistent with S-EPN. As EPNs have been occasionally reported in patients with neurofibromatosis type 1 7 and type 2, 8 a clinical workup for both diseases was performed but it was negative. The postoperative course was unremarkable and recurrences were not appreciated for 8 months after surgery.
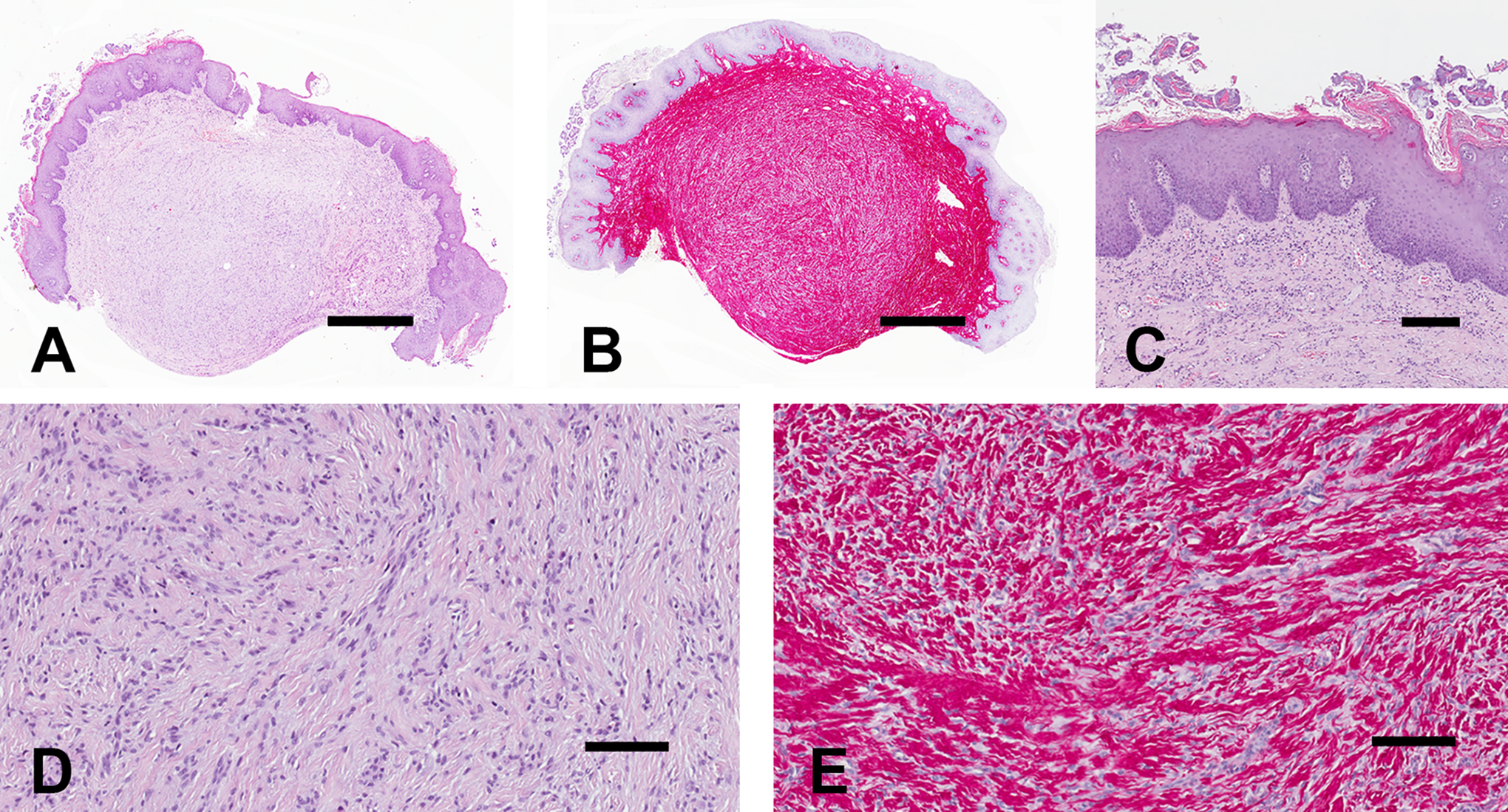
Low power magnifications of the excised sample on sections stained with hematoxylin-eosin and picrosirius red are illustrated in A and B, respectively. The image in C details the squamous epithelial lining with focal evidence of filiform papillae surrounded by bacterial colonies. High power magnifications of the tumor on sections stained with hematoxylin-eosin and picrosirius red are illustrated in A and B, respectively. The lesion is composed of spindle to oval cells devoid of atypia in a dense sclerotic collagenous matrix. Bars: 1 mm in A and B, 200 µm in C and 100 µm in D and E.
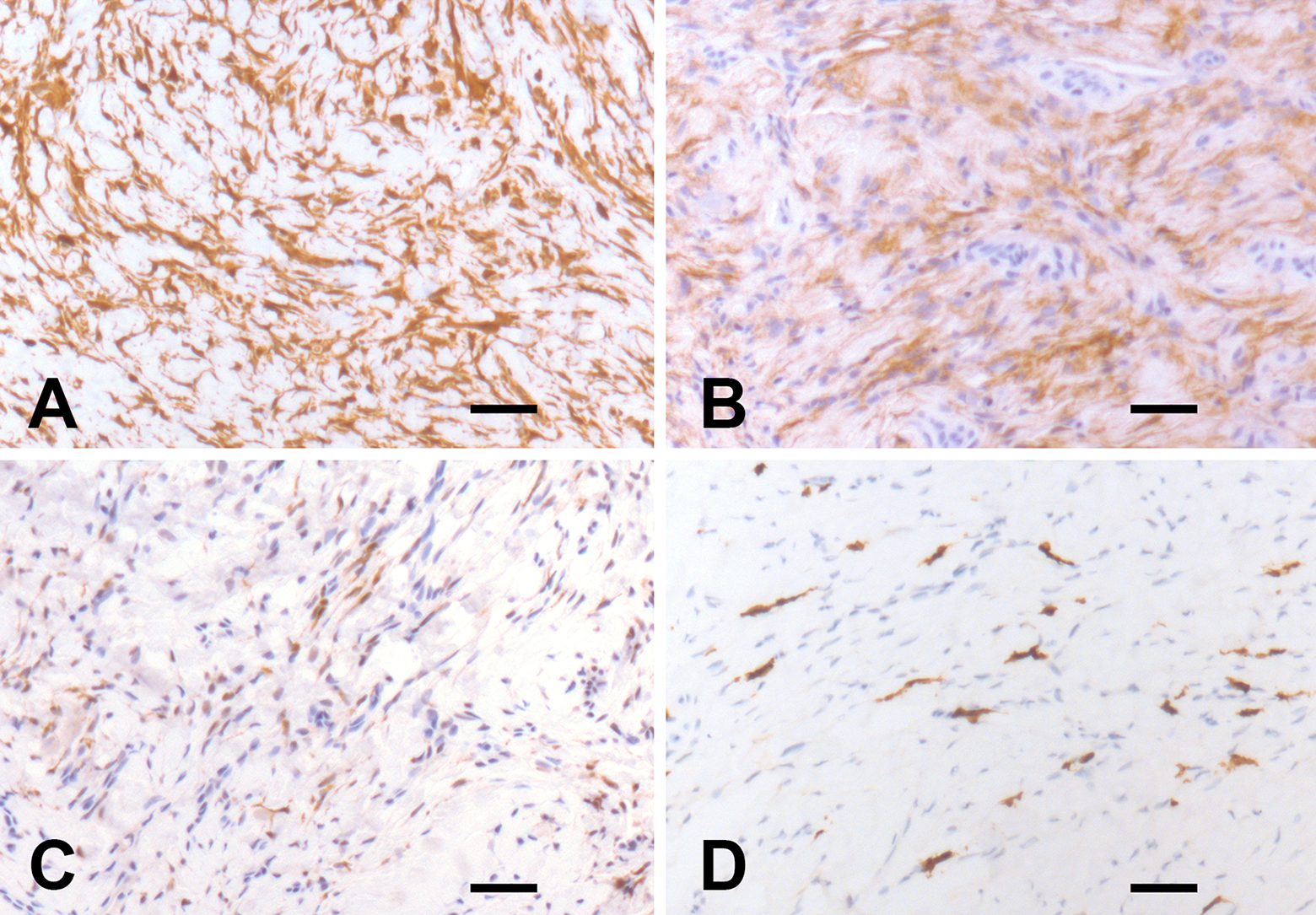
Immunohistochemical profile of the tumor. A: Vimentin. B: EMA. C: Claudin-1. D: CD117. Neoplastic cells are positive for vimentin and EMA and, focally, for Claudin-1 and negative for CD117. The CD117 immunoreactive cells are mastocytes. Bars: 80 μm.
S-EPN was first described in 1997 by Fetsch and Miettinen 9 who reported 19 tumors, all arising in the hands mainly of young male adults. Since then only 2 S-EPNs have been reported in the oral cavity, one in the lower lip mucosa of a 26-year-old male 10 and the other in the right buccal mucosa of a 17-year-old male. 11 In both cases, recurrence was not appreciated for 8 to 9 months after surgery.
For the rarity of PN in the oral cavity, 3 otolaryngologists and oral surgeons might not be familiar with this tumor. When encountered in the oral cavity/tongue, presentation of PNs is not specific and soft tissue tumors most common at this site, as granular cell tumor and other benign PNSTs, are usually clinically considered. 3,10,11 As PNs are typically benign and complete excision is deemed adequate management, their correct recognition, that requires correlation of the histologic findings with immunohistochemical evidence of perineurial differentiation, is mandatory to avoid unnecessary overtreatment. However, many soft tissue tumors may be confused with PN 1,2 . Although not specific, the immunoreactivity of the neoplastic cells for markers of perineurial cells, including EMA, Claudin-1, and human erythrocyte glucose transporter-1 is worthwhile for the diagnosis of PN. 1 -3 Cytogenetic and molecular studies are still of limited value for the diagnosis of PN but may play a fundamental role in excluding relevant mimics as low-grade fibromyxoid sarcoma. 12
Perineuriomas may also recur and uncommonly may be malignant. 1,2,13 Recurrent 3 and malignant 14 PNs have also been described in the tongue. Although recurrences may be expected when the tumor has not been completely excised, malignancy has to be considered when hypercellularity, nuclear atypia, necrosis, and high mitotic rate are present. Malignant PNs are known to behave less aggressively than conventional malignant PNST. 13 The unique case of malignant PN reported to date in the tongue was in a 24-year-old Japanese man treated by partial glossectomy who failed to develop recurrence and metastasis for more than 2 years after surgery. 14 Finally, PNs of uncertain malignant potential have been also described. 1 These cases are devoid of the overtly malignant histological features and characterized by nuclear atypia, abrupt transition from typical cell morphology and storiform architecture to high cellular areas with fascicular growth pattern, and diffuse infiltration of adjacent skeletal muscle. 1 As far as we know, PNs of uncertain malignant potential have never been reported in the oral cavity.
In conclusion, we reported here the clinicopathologic findings of an uncommon benign tumor, the S-EPN, in an unusual site, the tongue. To date, this variant of EPN has never been reported at this site.
Footnotes
Authors’ Note
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request. All the clinicopathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards. Written informed consent for publication of data and images was obtained from the patient.
Acknowledgments
The authors wish to thank Claudia Bonomo (Department of Pathology, IRCCS Regina Elena National Cancer Institute, Rome, Italy) for her technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
