Abstract
Objective:
Early detection of hypopharyngeal squamous cell carcinoma (SCC) is important for both an improved prognosis and less-invasive treatment. We retrospectively analyzed the detection rates of early hypopharyngeal SCCs according to the evaluation methods and the clinical management of early hypopharyngeal SCCs.
Methods:
Sixty-eight patients with early hypopharyngeal SCC who were diagnosed were reviewed.
Results:
The number of early hypopharyngeal cancer patients with asymptomatic or synchronous or metachronous esophageal cancer examined by upper gastrointestinal endoscopy with narrow-band imaging (NBI) was significantly higher than those examined by laryngopharyngeal endoscopy with NBI. The 3-year disease-specific survival rates according to T classification were as follows: Tis, 100%; T1, 100%; T2, 79.8%; and overall, 91.2%, respectively.
Conclusions:
Early-stage hypopharyngeal SCC can be cured by minimally invasive transoral surgery or radiotherapy. Observation of the pharynx using NBI in patients with a history of head and neck cancer, esophageal cancer, gastric cancer, or pharyngeal discomfort is very important, and routinely examining the pharynx with NBI, even in patients undergoing endoscopy for screening purposes, is recommended.
Introduction
Hypopharyngeal squamous cell carcinoma (SCC) comprises approximately 10% of all head and neck SCCs. 1 It is usually diagnosed at an advanced stage and therefore has a relatively poor prognosis. Indeed, the 5-year overall survival (OS) rate is reported to be only 15% to 45%. 2 Surgical resection of advanced lesions may cause a drastic reduction in the quality of life. Thus, early detection is important for both an improved prognosis and less-invasive treatment. However, the detection of hypopharyngeal SCC in patients with asymptomatic early lesions can be difficult for otolaryngologists, and it was reported that stages I and II disease were diagnosed in only 4% and 9% to 13% of patients, respectively. 3,4 Approximately 60% to 80% of patients have clinically metastatic cervical lymph nodes at the diagnosis. 5
In terms of head and neck cancer (HNC), it is well known that multiple SCCs frequently arise in the esophagus and pharynx, a phenomenon known as field cancerization. 6 Patients with esophageal SCCs therefore require careful monitoring of the pharynx to detect early pharyngeal SCCs. Recently, magnifying gastrointestinal (GI) endoscopy with narrow-band imaging (ME-NBI) was shown to provide high-resolution images and was proven useful for allowing GI physicians to detect early superficial laryngopharyngeal SCCs, which are difficult to detect by standard laryngopharyngeal endoscopy. 7,8 However, there have been only a few reports regarding the detection rates of early hypopharyngeal SCCs among different evaluation methods (GI endoscopy vs laryngopharyngeal endoscopy) 9 or the clinical management of early hypopharyngeal SCCs.
In this study, we retrospectively analyzed the number of early cancer lesions detected by an otolaryngologist or GI physician and the clinical course, management, and survival outcomes of patients with early hypopharyngeal cancer lesions.
Materials and Methods
Patients
Sixty-eight patients with early hypopharyngeal SCC (Tis, T1, and T2N0) who were diagnosed at the Department of Otolaryngology in Kyushu University Hospital between 2010 and 2018 were reviewed. The patients’ clinical characteristics are summarized in Table 1. There were 62 men and 6 women, with a mean age of 68.0 years. The TNM classification and staging were performed according to the UICC classification (eighth edition). The proportion of Tis hypopharyngeal cancer was 10% (7 of 68), that of T1 was 52% (35 of 68), and that of T2 was 38% (26 of 68). Among the tumors, 43 (63%) were in the pyriform sinus (PS), 5 (7%) were in the postcricoid (PC), and 20 (29%) were in the posterior wall (PW). The mean follow-up period was 48 (range 3-110 months) months. Forty-one (60%) of the 68 patients had synchronous or metachronous esophageal cancers (EC).
Patients Characteristics.
Patients were divided into 5 groups based on the reason for laryngopharyngeal endoscopy or upper GI (UGI) endoscopy: screening UGI examination (UGI screening group), pharyngeal discomfort (pharyngeal discomfort group), surveillance after treatment of HNC (HNC group), pretreatment examination or posttreatment surveillance for EC (EC group), and pretreatment examination or posttreatment surveillance for gastric cancer (GC group). All UGI endoscopy procedures were performed using a transoral standard endoscope (GIF-H260Z, HQ290; Olympus Optical) or ME (GIF-RQ260Z, GIF-H290Z; Olympus Optical) with an NBI system (Evis Elite; Olympus Optical). The oral cavity, oropharynx, hypopharynx, and larynx were observed by GI physicians. Laryngopharyngeal endoscopy was performed using a standard procedure with a transnasal flexible fiber scope (ENF-VQ, ENF-VH; Olympus Optical) and an NBI system. The nasopharynx, oropharynx, hypopharynx, and larynx were observed by otolaryngologists.
This study protocol was approved by the institutional review board of Kyushu University (reference number: 29-43). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Statistical Analyses
All calculations were performed using the JMP 14 software program (SAS Institute). Statistical analyses were performed using the Mann-Whitney U test or Fisher exact probability test. The OS rate, disease-specific survival (DSS) rate, and progression-free survival rate were calculated using the Kaplan-Meier method and evaluated with the log-rank test.
Results
Forty-nine (72%) patients were examined by UGI endoscopy with NBI, whereas 19 (28%) patients were examined by laryngopharyngeal endoscopy with NBI (Table 1). Figure 1 shows the details concerning the detection of hypopharyngeal cancer according to the reason for endoscopy. There were 7 (10%) patients in the UGI screening group, 13 (19%) in the pharyngeal discomfort group, 6 (9%) in the HNC group, 38 (56%) in the EC group, and 4 (6%) in the GC group. The EC group had a significantly higher rate of hypopharyngeal SCC than the other groups, followed by the pharyngeal discomfort group and UGI screening group. The number of early hypopharyngeal cancer patients with asymptomatic or synchronous or metachronous EC examined by UGI endoscopy with NBI was significantly higher than those examined by laryngopharyngeal endoscopy with NBI (P = .001, P = .0001). Figure 2 shows representative images of superficial hypopharyngeal SCC. Upper GI endoscopy with NBI showed the superficial cancerous lesion as a brownish area more clearly than laryngopharyngeal endoscopy with NBI.
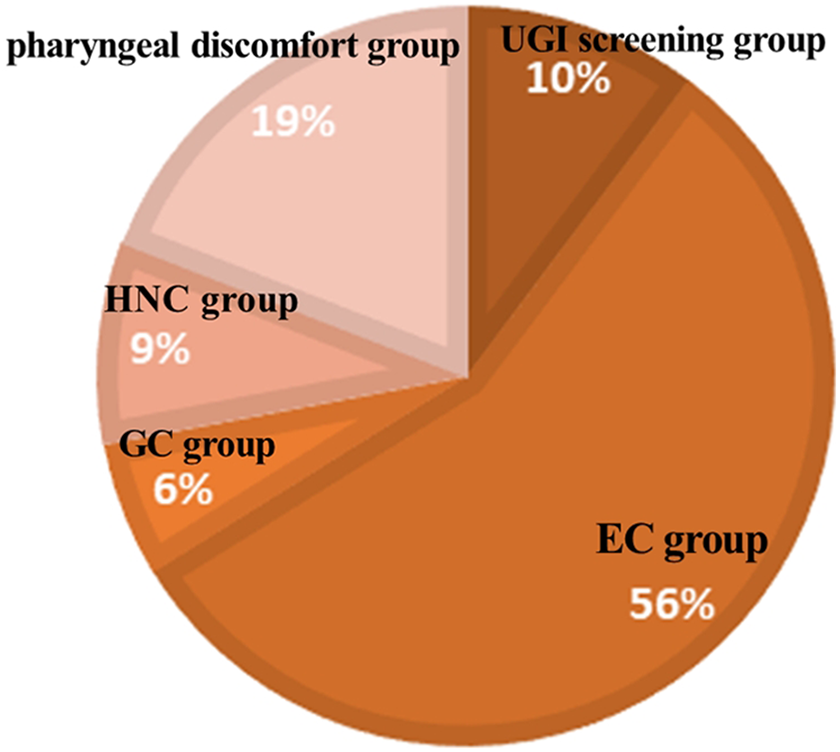
Number of early hypopharyngeal SCCs according to the reason for endoscopy. SCC indicates squamous cell carcinoma.
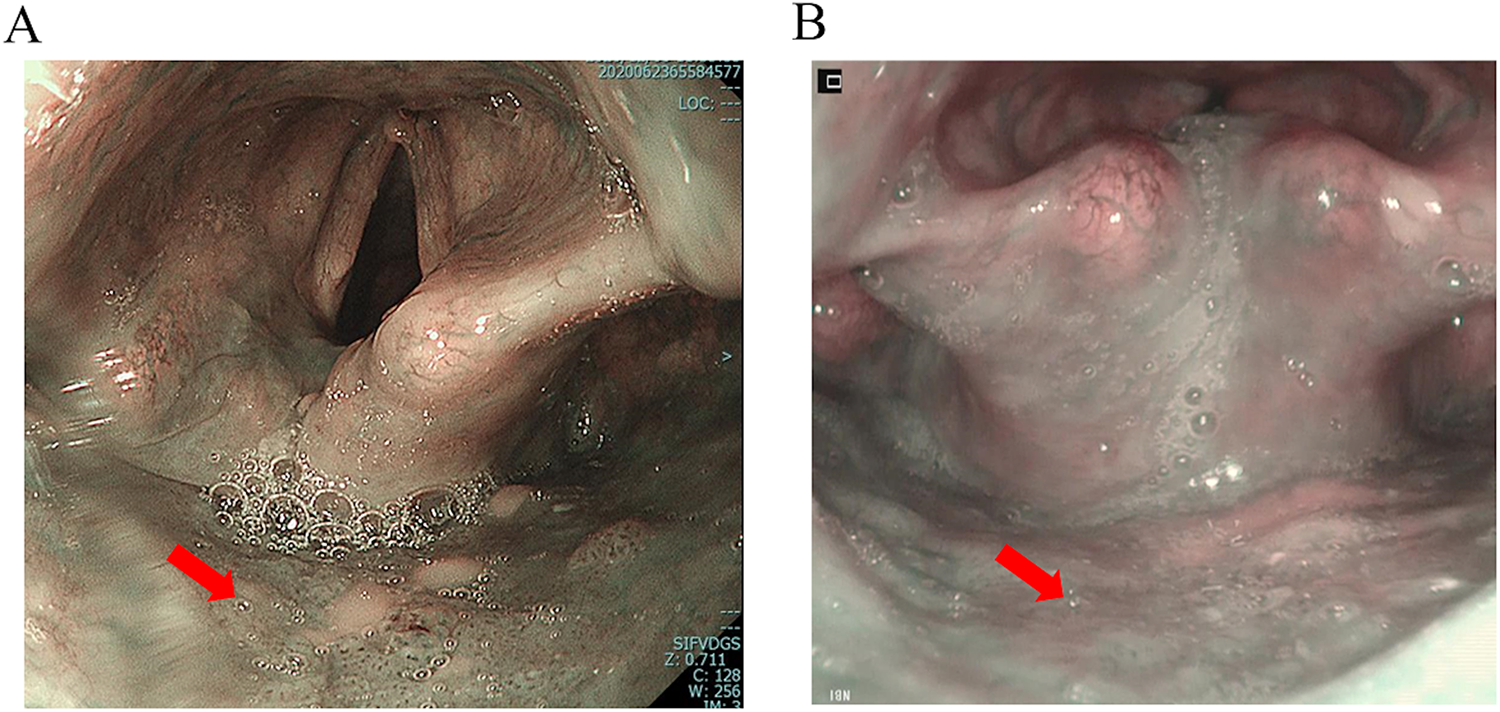
Representative images of superficial hypo pharyngeal SCC (posterior wall). A, Transoral flexible GI endoscopy with NBI system. B, Transnasal flexible laryngopharyngeal endoscopy with NBI system. NBI shows a brownish area (red arrow) in the right arytenoid. GI indicates gastrointestinal; NBI, narrow-band imaging; SCC, squamous cell carcinoma.
Of the 68 patients, 28 (41%) were treated with transoral surgery (TOS), either with or without radiotherapy (RT) for their primary tumor. Transoral surgery was performed as follows: a curved rigid pharyngo-laryngoscope (Nagashima Medical Instruments Company, Ltd) was inserted under general anesthesia, then NBI endoscopy was inserted transorally to visualize the surgical field. The extent of the lesion was determined based on findings from NBI endoscopic examination and iodine staining. A mixed solution of epinephrine (0.02 mg/mL) and saline was injected into the subepithelial layer beneath the lesion to elevate it, then the lesion was resected with 1 to 2 mm margins using transorally inserted curved forceps and a curved electrosurgical needle knife (KD-600; Olympus Medical Systems). Patients with positive surgical margins received postoperative RT (60 Gy).
Twenty-five of these patients had primary tumors Tis-T1, and the other 3 had primary tumors T2. Thirty-five (51%) received chemoradiotherapy (CRT) or RT alone. Fourteen of these patients had primary tumors T1, whereas 21 had primary tumors T2. Thus, patients with Tis or T1 primary tumors tended to be treated with TOS, while those with T2 primary tumors tended to undergo CRT or RT (Table 2, Figure 3). Five (8%) patients received best supportive care alone because of their poor health condition.
Treatment Method According to T Classification.
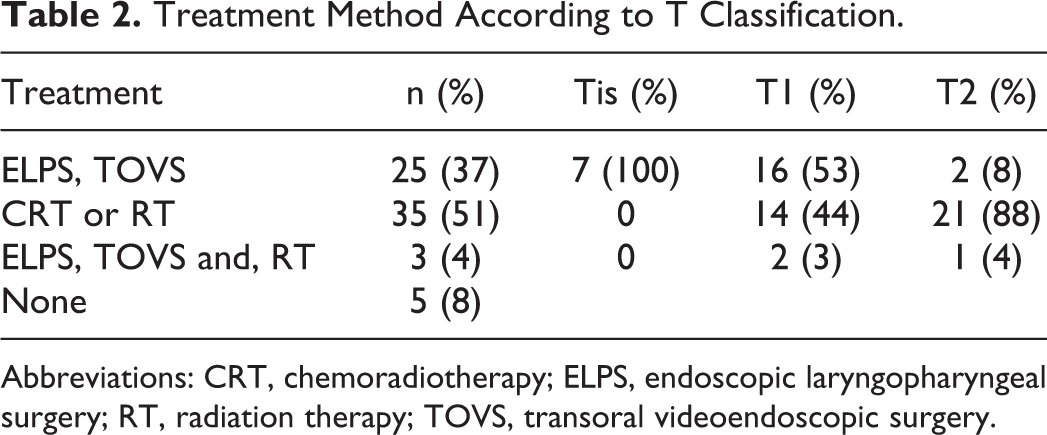
Abbreviations: CRT, chemoradiotherapy; ELPS, endoscopic laryngopharyngeal surgery; RT, radiation therapy; TOVS, transoral videoendoscopic surgery.
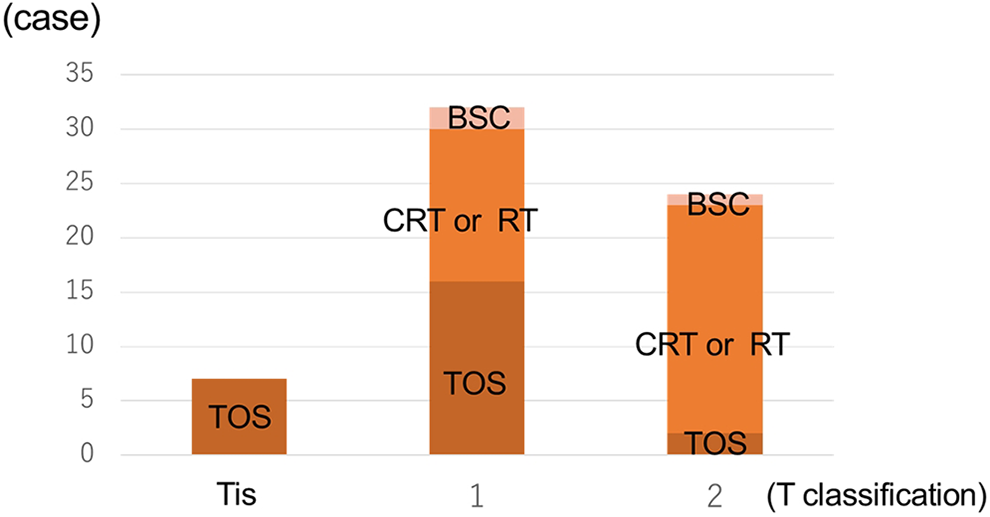
Treatment methods according to T classification.
The 3-year OS rate for 63 patients treated with TOS and/or CRT or RT was 80.7% (Figure 4A). The 3-year DSS rates according to T classification were as follows: Tis, 100%; T1, 100%; T2, 79.8% (Figure 5A); and overall, 91.2% (Figure 4B). The 3-year DSS rates in the TOS ± RT and CRT or RT groups were 89% and 91.1%, respectively (Figure 5B). There was no significant difference (P = .52) between TOS ± RT and CRT or RT groups. The 3-year DSS rates according to primary subsite were as follows: PS, 95.1%; PW, 78.6%; and PC, 100% (Figure 5C). There was no significant difference (P = .90) among them.
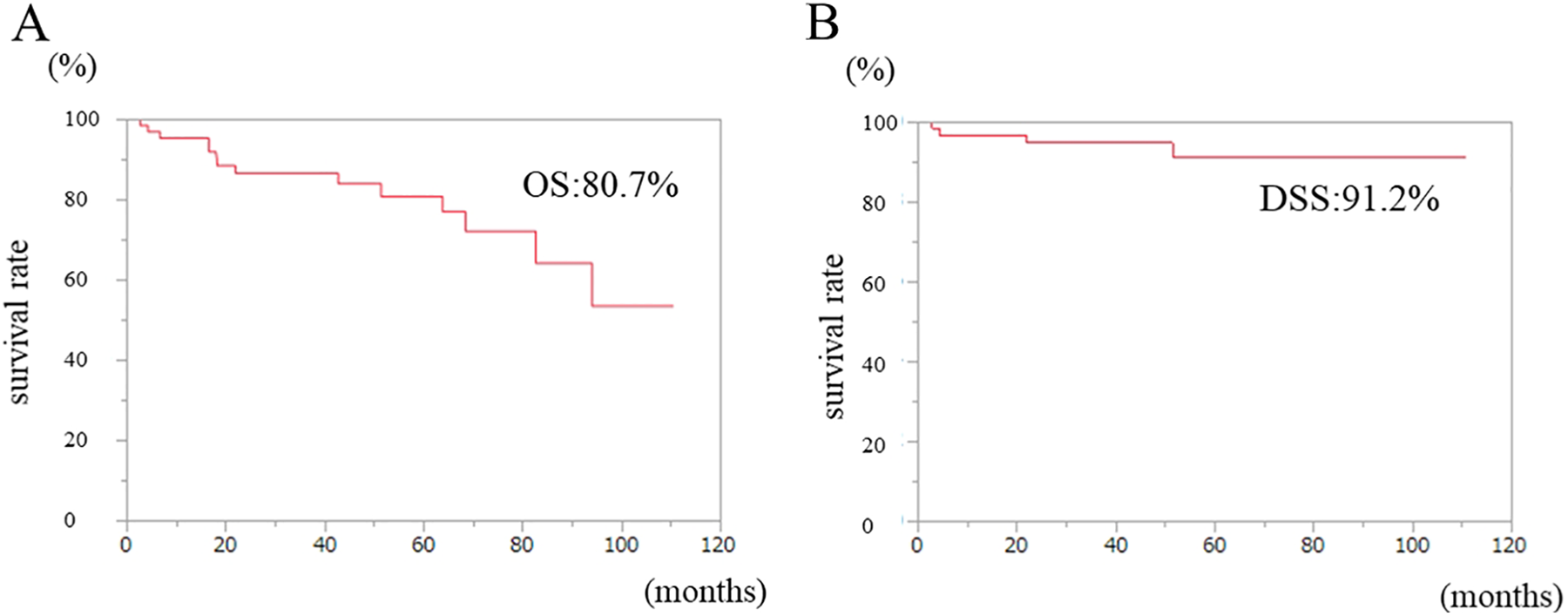
Kaplan-Meier curves of the (A) OS and (B) DSS for early-stage hypo pharyngeal SCC. DSS indicates disease-specific survival; OS, overall survival; SCC, squamous cell carcinoma.
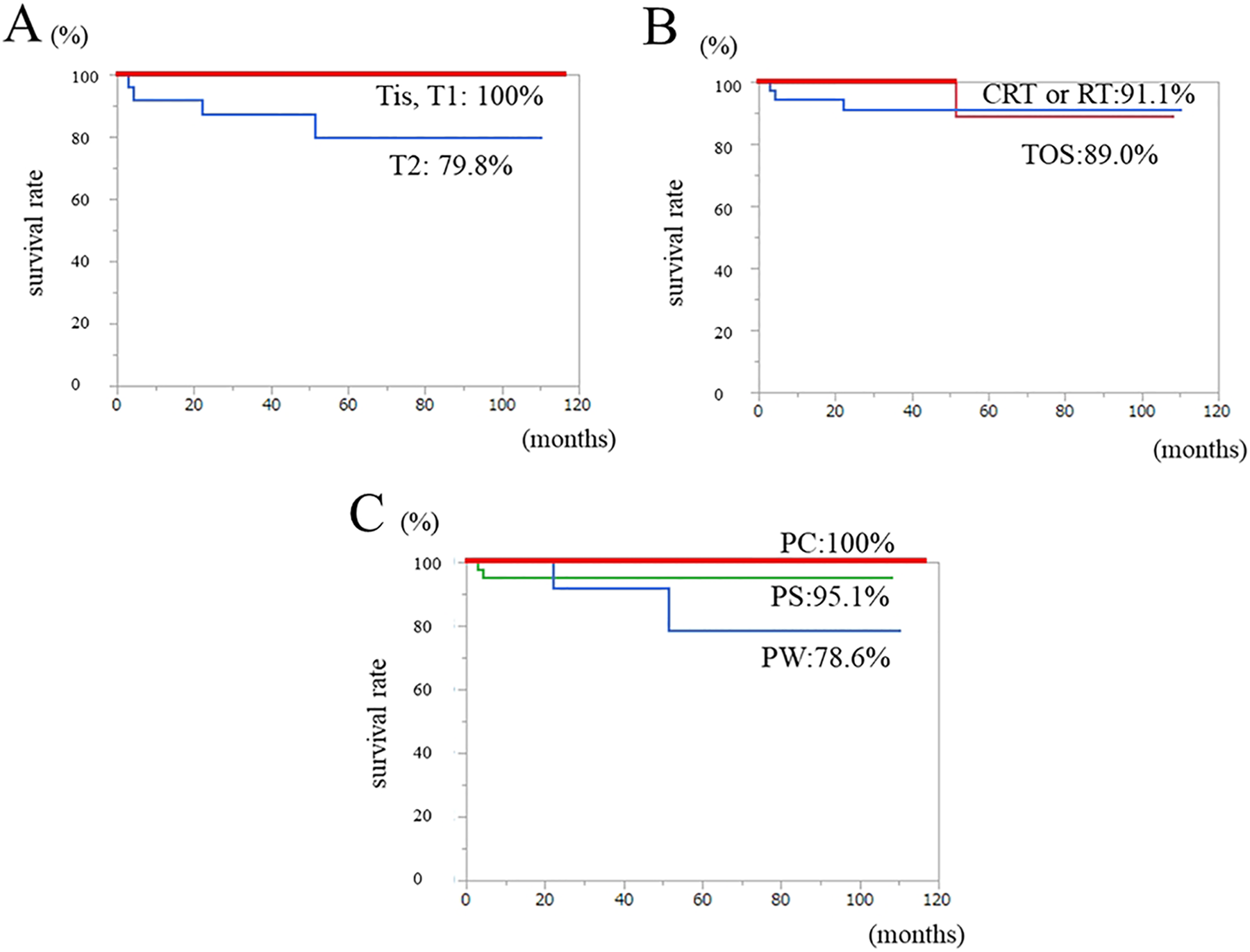
Kaplan-Meier curves of the DSS according to the (A) T classification, (B) treatment method, and (C) subsites. DSS indicates disease-specific survival.
Discussion
Hypopharyngeal SCC is the rare malignancy that contributes to 10% of head and neck carcinomas, 1 being mainly observed in men over 50 years. Patients with hypopharyngeal SCC tend to have a high prevalence of second primary SCC in the esophagus (ESCC), and patients with ESCC are at a high risk of the development of HNC. 10 Thus, the ability to detect a second primary cancer at the earlier stage would be of great benefit for patients at high risk of superficial Head and neck squamous cell carcinoma (HNSCC).
Recently, NBI has enabled the detection of superficial SCC that were previously difficult to discover in the head and neck region. 11 As a result, the number of early T-stage patients has increased following the advent of NBI endoscopy and an increased awareness of HNC among GI physicians in other fields. 12 Indeed, UGI endoscopy with NBI showed the superficial cancerous lesion as a brownish area more clearly than laryngopharyngeal endoscopy with NBI. In addition, our results clearly demonstrate that significantly more cases with early hypopharyngeal SCC were examined by UGI endoscopy with NBI (72%) than by laryngopharyngeal endoscopy (28%). Kumai et al also reported that 82 (75.2%) of 109 cases of hypopharyngeal cancer were detected by GI physicians with UGI endoscopy with NBI, and only 27 (24.8%) were detected by otolaryngologists with laryngopharyngeal endoscopy. 9 Before performing UGI endoscopy, GI physicians prepare the patient by using pronase, sodium bicarbonate, dimethylpolysiloxane, and lidocaine. They were generally used in Japan to remove gastric mucus and improve visualization of the gastric wall during UGI endoscopy. They may assist in removing pharyngeal mucus and thus improve visualization of the pharyngeal mucosa.
In terms of the reasons for endoscopy, the pharyngeal discomfort group had the highest rate (19%) of hypopharyngeal SCC following the cancer groups (HNC, EC, and GC) and was higher than the screening group (10%). The most common area of lesions was the PS, which accounted for 64% of lesions. Although hypopharyngeal SCC most commonly develops around the PS, cancerous lesions were also detected in other areas. These results underscore the importance of observing the pharynx with NBI in patients with a history of HNC, EC, GC, or pharyngeal discomfort. Furthermore, a thorough examination of all parts of the hypopharynx should be performed in these patients.
According to the treatment results, actuarial 5-year DSS rates of 70% to 96% are reported in stage I-II SCCs treated with CRT or RT and/or transoral laser microsurgery (TLM). 13,14 Weiss et al also found that the 5-year OS rate was 77.6% in pT1 and 52.8% in pT2, and the 5-year CSS rate was 96.3% in pT1 and 96.7% in pT2 by TLM. 15 Our findings indicating a 3-year OS of 80.7% and 3-year DSS of 91.2% are similar to those of previous reports on early-stage hypopharyngeal SCC. Recently, TOS is also considered a definitive therapy with comparable outcomes to RT for early T-stage hypopharyngeal cancer, showing a consistent organ and function preservation. 16 Although patients who received CRT or RT may have had more extensive lesions in the present study, there were no significant differences between the TOS ± RT and CRT or RT groups. These results suggest that early-stage hypopharyngeal SCC can be cured by minimally invasive TOS or RT.
In conclusion, this study showed that the number of early hypopharyngeal cancer patients with asymptomatic or synchronous or metachronous EC examined by UGI endoscopy with NBI was significantly higher than those examined by laryngopharyngeal endoscopy with NBI. Furthermore, observation of the pharynx using NBI in patients with a history of HNC, EC, GC, or pharyngeal discomfort is very important, and routinely examining the pharynx with NBI, even in patients undergoing endoscopy for screening purposes, is recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by the following grant and foundation: JSPS KAKENHI 20K09757.
