Abstract
We present a case of delayed vocal process granulomas and anterior glottic web after treatment of vocal fold papillomas with a laryngeal microdebrider and CO2 laser ablation. This case highlights a risk of vocal fold granuloma in the setting of recurrent respiratory papillomatosis and microdebrider use, warranting close follow-up and consideration of prophylactic voice therapy, anti-reflux medications, and steroid injection in cases of violation of vocal cord epithelium.
Clinic Case Report
The patient is a 28-year-old male who was referred for evaluation of 9 months of dysphonia. He reported moderately rough voice, limited range, and occasional odynophonia. He denied dysphagia, odynophagia, and postnasal drip. Although he denied subjective sensation of reflux, he was trialed on ranitidine for several months without benefit. He was otherwise healthy. He denied tobacco use and reported infrequent alcohol use. His profession had high voice demands. History was negative for human papillomavirus (HPV) vaccination. On physical examination, his voice was severely harsh and breathy with poor projection. His GRBAS (grade, roughness, breathiness, asthenia, strain) score was 10 (3,2,3,0,2). Flexible laryngoscopy with videostroboscopy revealed bilateral, asymmetric, exophytic lesions studding the medial aspects of each true vocal fold, resulting in reduced vibratory amplitude of the vocal folds, reduced propagation of the mucosal wave, and incomplete glottic closure during sustained phonatory tasks. A large, vascular, exophytic lesion was also noted on the left laryngeal surface of the epiglottis. Examination suggested recurrent respiratory papillomatosis (RRP), and the patient was scheduled for surgery.
The patient underwent microdirect laryngoscopy with biopsy, bronchoscopy, and excision of vocal fold lesions. The lesions were removed using a laryngeal microdebrider, and CO2 laser was used to ablate remnant tissue. Excision was carried to the epithelium, with care taken to avoid trauma to the lamina propria. Pathology confirmed squamous papilloma of the vocal folds. He was doing well at postoperative follow-up 1 week later, with improvement in voice (GRBAS 5; 2,1,1,0,1) and improved glottic closure with no evidence of recurrent epithelial lesions.
Four months later, he re-presented with dysphagia, mild throat discomfort, decreased projection, and mild roughness. GRBAS was rated as 3 (l,1,0,0,1). Flexible laryngoscopy revealed multiple recurrent large supraglottic papillomas. No glottic lesions were present.
He returned to the operating room one month later for treatment and found to have progression, with bilateral false vocal fold and epiglottal lesions, as well as distinct large, symmetric, inflammatory lesions overlying both vocal processes, and an anterior glottic web (Figure 1 and Figure 2). These were excised and ablated with a combination of the laryngeal microdebrider and CO2 laser, and triamcinolone was injected into the supraglottic areas of scar (Figure 3). Pathology confirmed polypoid granulation tissue, consistent with superimposed granuloma, in addition to recurrent squamous papilloma. At most recent follow-up, one month after surgery, the patient’s voice was improved and stable (GRBAS 5; 2,2,1,0,1), with strobocoscopy showing normal amplitude and propagation of the mucosal wave, and no recurrent papillomatous or granulomatous lesions.
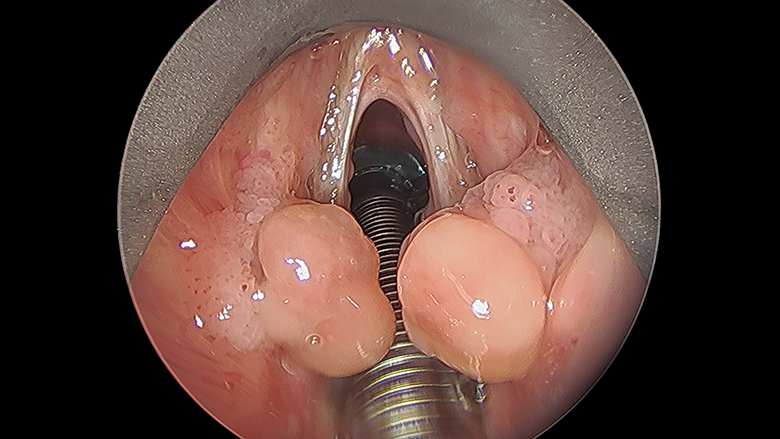
Microdirect laryngoscopy during revision RRP surgery revealed bilateral false vocal fold and epiglottal papillomatous lesions, as well as distinct, large, symmetric inflammatory lesions overlying both vocal processes, and an anterior glottic web.
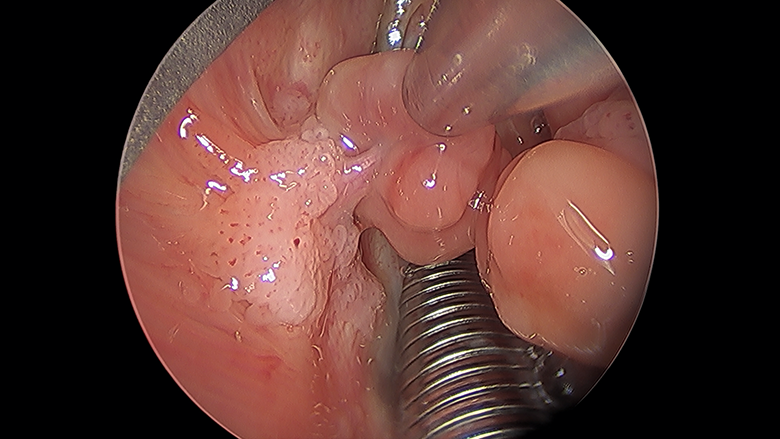
A laryngeal suction is used to retract the right vocal process granuloma, revealing a pedunculated stalk attached to a papillomatous base.
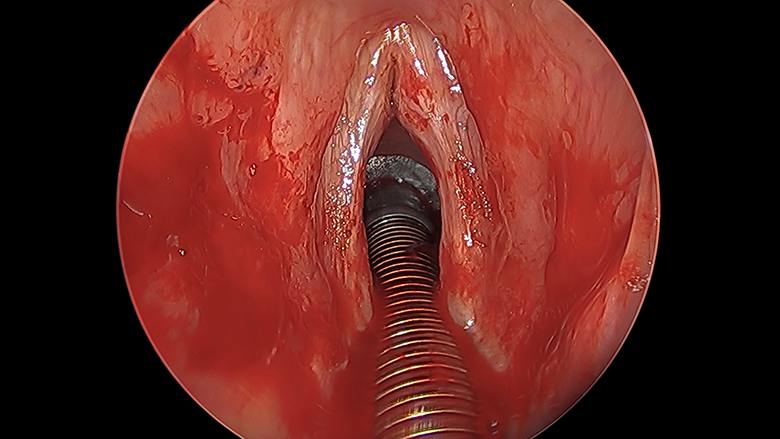
One month posttreatment, the appearance of the glottis after treatment with laryngeal microdebrider, CO2 laser, and supraglottic triamcinolone injection, with care taken not to violate the vocal fold epithelium.
Although recurrence of respiratory papillomas is common, as evidenced by the high rates of surgery required for management (with one study showing 5.1 surgeries on average annually in the first 5 years after diagnosis), delayed granuloma formation after microlaryngeal surgery is rare. 1 Granulomas and anterior glottic web were not present in this patient 4 months postoperatively. They developed between 4-month clinic follow-up and subsequent surgery one month later, suggesting a delayed trigger. Only one other case of granuloma with anterior glottic web after laryngeal microdebrider use has been reported in the literature. 2 In this case, a microdebrider was used to excise an anterior vocal fold polyp, but the excision was carried into the lamina propria with exposure of the vocalis muscle.
The combination of trauma from intubation and surgical manipulation, postoperative inflammation, laryngopharyngeal reflux (LPR), and phonotrauma from compensatory voice misuse likely precipitated granuloma formation, although reflux was not ruled out with pH manometry and could have contributed. Furthermore, the presence of HPV in the respiratory epithelium may also drive ongoing inflammation. We opted to excise the granulomas with the CO2 laser followed by triamcinolone injection, with no evidence of recurrence 1 month later, although others advocate a more conservative approach with medication (including injection of botulinum toxin) or observation only, with the thought that reoperation could worsen trauma and inflammation of the tissue.
This case highlights a risk of vocal fold granuloma in the setting of recurrent respiratory papillomatosis. These patients have a number of factors placing them at increased risk for granuloma formation, including mechanical trauma from frequent laryngeal surgery with intubation and poor compensatory voice use, inflammation from HPV, and LPR. We agree with previous authors that violation of the vocal fold epithelium should be minimized or avoided. Additionally, sources of mechanical and inflammatory trauma should be addressed prophylactically, with appropriate measures taken including voice therapy, anti-reflux medications, and consideration of steroid injection in cases of violation of vocal cord epithelium.
Footnotes
Authors’ Note
Institutional Review Board: This manuscript is exempt from IRB review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
