Abstract
Salivary Bypass Tube is an important tool to prevent or treat some complications of laryngeal and hypopharyngeal surgery and its placement may prove difficult. In this article, we propose a new technique to simplify its management by using an Oral/Nasal Tracheal Tube Cuffed-Reinforced that allowed us to reduce operating times, complications related to prolonged general anesthesia, and the traumas on the tissues incurred during the forced positioning of the device with standard techniques.
Surgical resection of laryngeal malignancies and reconstruction of the subsequent defects in the aerodigestive tract are among the most challenging of oncological procedures. Pharyngocutaneous fistula (PCF), tracheoesophageal fistula (TEF), and pharyngoesophageal stenosis (PES) are among the most common complications following total laryngectomy; they have adverse effects on post-operative rehabilitation, adjuvant therapy, and overall survival. 1 It is known the importance of preventative application of a salivary stent in selected patients at high risk of fistula development.2,3 Conservative treatment is considered in most of the cases as the first option; when these measures fail, surgical treatment and contextual use of salivary devices like the Montgomery® Salivary Bypass Tube (MSBT) is indicated. Sometimes, due to the flexibility and softness of the materials used, the positioning of the device can be particularly complex and risky, especially in patients suffering from the anatomical alterations described above. In this article, we propose a new technique to simplify the placement of a MSBT by using a Mallinckrodt™ Oral/Nasal Tracheal Tube Cuffed-Reinforced.
We tested this new technique in patients with history of squamous cell carcinoma of the larynx subjected to total laryngectomy who developed a TEF or a PES after surgery and cyclically undergoes device replacement. During the following replacements, the procedure proved to be rather difficult with the standard insertion technique due to a progressive formation of a fibrous ring with partial stenosis of the hypopharyngeal tissues. Therefore, we decided to attempt a new method using a MSBT (Boston Medical Products) with a Mallinckrodt™ Oral/Nasal Tracheal Tube Cuffed-Reinforced (Medtronic-Covidien) as introducer (Figure 1) to overcome resistance and to reduce the risk of incorrect positioning. The size of the tracheal tube was calibrated guaranteeing the possibility of easy introduction/removal from the device and the solidity of the anchorage by cuffing appropriately (Figure 2).

The figure shows a Montgomery® Salivary Bypass Tube n°12 (Boston Medical Products) and a n°4.0 Mallinckrodt™ Oral/Nasal Tracheal Tube Cuffed-Reinforced (Medtronic-Covidien).

The tracheal tube is inserted inside the MSBT: it is important to evaluate the smoothness of the tube inside the device to avoid problems during the disarming phase (A). A sample is colored to highlight the cuff and the detail of its anchorage after the insufflation (B). MSBT indicates Montgomery® Salivary Bypass Tube.
The tool obtained is positioned under general anesthesia through direct laryngoscopy with visualization of the esophagus. The device is maneuvered as during a tracheal intubation using the bevel at the tip of the tube in order to reduce trauma and overcome stenosis.
In fact, one of the main positioning problems that prompted us to try this new technique is the need for compression of the device which can easily be solved by folding up the distal end and grasping it with alligator-type endoscopic forceps as descripted by Montgomery.4,5 However, this procedure assumes that the folded distal part must slide with the forceps into the esophagus, risking its rupture or enlargement of the TEF/PCF. On the other hand, if the distal part is released from the forceps first, the lower edge of the device could get stuck in the fistula and compromise the sliding of the device into the esophagus as well as risking again an enlargement of the fistula.
The advantages of the proposed insertion method, also in comparison with other techniques reported in the literature, 6 are as follows:
– The Mallinckrodt™ Oral/Nasal Tracheal Tube is soft and flexible with a spiral-wound reinforcing wire which allows the operator to direct the salivary device into which it is inserted.
– Using the portion found at the tip of the tube and positioning the insufflated cuff astride the distal edge of the device, it is possible to overcome obstacles by blunt way.
– Deflating the cuff, as we have inserted an endotracheal tube of a smaller caliber than the device, it will be simple to remove it from the device itself.
According to our experience with n°12 and n°14 MSBT, we can estimate the caliber couplings between endotracheal tubes and MSBT considering the difference between the external diameters of the tubes (OD), the cuff diameters, and the caliber of the salivary devices (Figure 3). The use of this new tool and the related device positioning technique allowed us to reduce operating times, the complications related to prolonged general anesthesia, and the traumas incurred during the standard techniques. Furthermore, we also verified the feasibility of this procedure avoiding general anesthesia by repositioning the device assisted by superficial sedation. The operation was in fact performed with the consent expressed by the patient, in a protected environment, using a slight hyperextension of the patient’s head and ensuring a prompt conversion to general anesthesia in case of complications. The procedure performed in this way required a few seconds of compliance by the patient, thus avoiding more serious anesthetic complications.
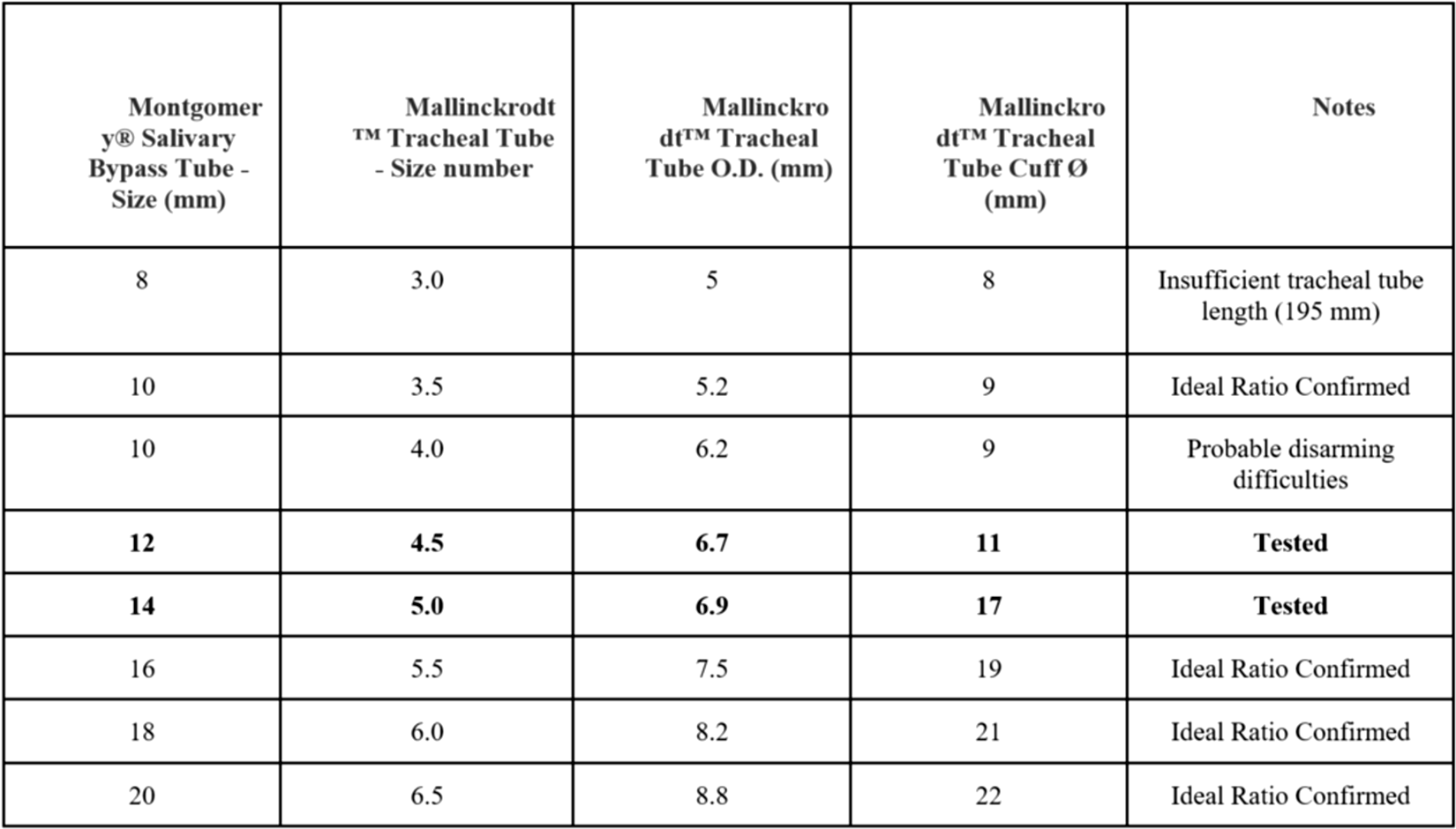
The figure shows a hypothetical guide to the correct choice of the MSBTs in relation to the external diameters of the tracheal tubes and the relative cuffs. In addition, the ideal ratios to be taken into consideration for the various devices not yet tested and any problems relating to the tool obtained with certain couplings were noted. MSBT indicates Montgomery® Salivary Bypass Tube.
Footnotes
Authors’ Note
All the clinicopathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
