Abstract
Hearing loss in older people can cause communication impairments, decreased quality of life, social isolation, depression, and dementia. Cochlear implant surgery is an effective treatment for older patients with hearing loss who cannot achieve satisfactory audiologic outcomes with hearing aids. However, older people have an increased risk of heart disease and often take medications that affect heart rhythm. Herein, we report a case of an 80-year-old woman who underwent cardioversion at 50J after cochlear implant surgery. Electrical impedance before and after cardioversion showed only minor changes without abnormality, and the cochlear implant functioned well. We believe that the electronic circuits of the cochlear implant may have been relatively tolerant to the electrical shock from the external defibrillator. Typically, cardioversion should be avoided in cochlear implant recipients because it may damage the implant. If cardioversion cannot be avoided, we strongly recommend starting cardioversion at the lowest energy level (50 J) and removing the sound processor of the implant during the procedure.
Introduction
The cochlear implant (CI) is an electronic device that restores hearing in people with hearing impairment. Cochlear implant surgery can be effective for people who have severe to profound hearing loss despite the use of hearing aids. Unlike hearing aids, CIs bypass the hair cells of the cochlea via a surgically implanted intracochlear electrode. As such, electric signals can directly stimulate the spiral ganglion cells of the cochlear nerve and are recognized as sound through the cerebral auditory cortex.
It was previously believed that CIs for older people may not be beneficial due to the age-related degeneration of the central and peripheral auditory pathways. 1 However, recent studies have shown that CIs in older people can result in excellent improvements in speech reception, which are comparable to improvements observed in younger people with CIs.2,3 Additionally, CIs have been shown to significantly improve the quality of life, cognitive function, depression, and dementia in older people. 4 Therefore, several countries have strongly recommended using CIs for older people with profound hearing loss.
However, older CI recipients may experience unexpected and life-threatening medical events. Herein, we report a rare case involving a patient who underwent cardioversion after receiving a CI, and we review the literature regarding the impact of cardioversion on CIs.
Case Report
An 80-year-old woman with a history of hypertension and diabetes visited our hospital. She lost hearing in her right ear many years prior and had been wearing a hearing aid in her left ear for 10 years. Pure tone audiometry revealed profound left-sided hearing loss and contralateral deafness. The patient’s brainstem auditory evoked response showed that wave V at 90 dB was present on the left side but absent on the right. We evaluated the patient’s operative risk based on underlying diseases before CI surgery. Electrocardiography (ECG) showed normal sinus rhythm and a heart rate of 86 beats per minute (bpm).
The left ear was successfully implanted with a Cochlear Nucleus CI622 (Cochlear; Figure 1A). No major medical problems related to general anesthesia occurred. The patient’s electrode impedance was measured on postoperative day 3. The impedance of the individual electrodes was within the normal range (1-20 kΩ), and short and open circuits were not present (Figure 2A). On postoperative day 4, the patient experienced chest discomfort and intermittent dyspnea. Electrocardiography revealed atrial flutter with variable atrioventricular block (Figure 1B), but the patient’s cardiac marker levels were normal. Transthoracic echocardiography (TTE) indicated mild left ventricular systolic dysfunction with an ejection fraction (EF) of 45.8%. We performed electrical cardioversion at 50 J after explaining to the patient’s daughter that the procedure was necessary, even if the CI malfunctioned. After DC cardioversion, the patient’s ECG revealed sinus bradycardia with a heart rate of 57 bpm, and her symptoms improved. At 2 days postcardioversion, the electrical impedance of the CI electrode was remeasured, and we confirmed that the electronic circuit of the implant was preserved (Figure 2B). When turning on the device at postoperative week 4, the CI functioned well. Presently, she is undergoing mapping.
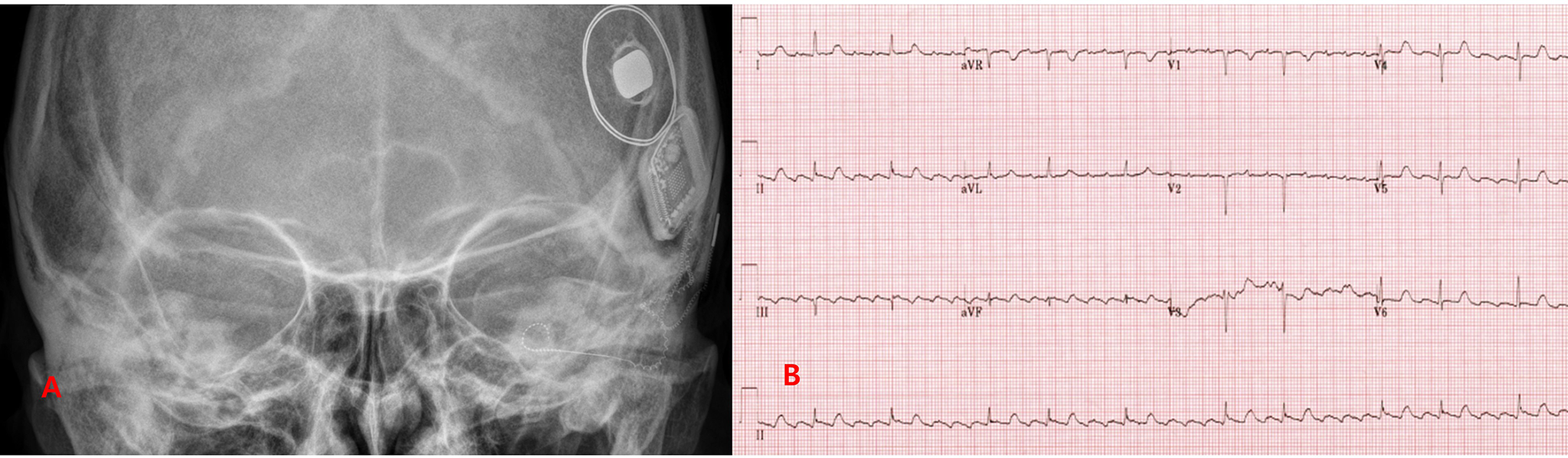
A, Postoperative transorbital x-ray showing a cochlear implant electrode array successfully inserted into the cochlea (spiral-shaped inner ear). B, Patient’s 12-lead electrocardiogram revealing atrial flutter with variable atrioventricular (AV) block.
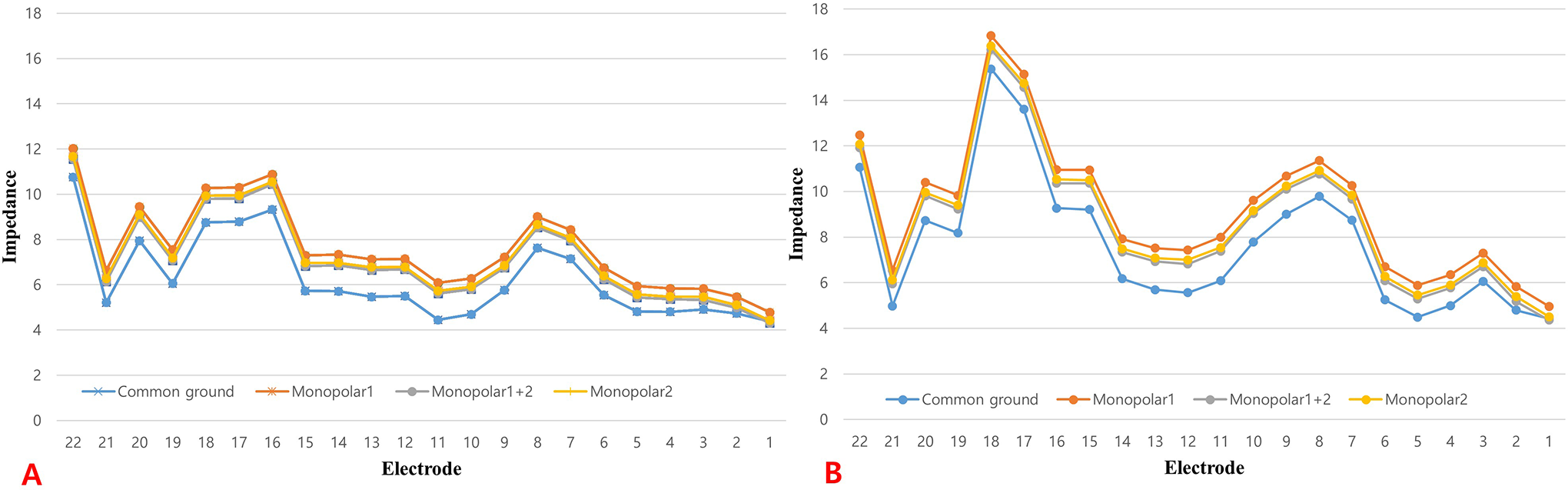
Graphs depicting the electrical impedance of the cochlear implant electrodes before (A) and after (B) cardioversion. The electrodes exhibited only minor changes in electrical impedance after cardioversion, and low impedance (indicating a short circuit) and high impedance (indicating an open circuit) did not occur. The implanted device functioned well 4 weeks after cochlear implant surgery.
Discussion
Cardioversion is a medical procedure that restores normal heart rhythm for people with arrhythmia. The most common indications for cardioversion are unstable atrial fibrillation, atrial flutter, atrial tachycardia, and supraventricular tachycardia. Typically, it is performed by applying electrical shocks to the heart via electrodes attached to the chest. Cardioversion delivers electrical currents with lower energy than in the defibrillation used in a patient with pulseless ventricular tachycardia or ventricular fibrillation. The recommended electrical energy levels for cardioversion vary from 50 to 200 J. The safest and least complicated method for implementing cardioversion is to start at the lowest energy level (50 J). Then, if the initial shock is unsuccessful, the amount of electrical energy should be doubled. 5
Cardioversion is not recommended for patients with CIs due to the potential risks of damage to cochlear tissue and permanent damage to the implant. However, if we hesitated to perform cardioversion for our patient, the result could have been fatal. Moreover, it remains unclear whether cardioversion causes the loss of CI function.
Our patient had persistent atrial flutter, chest discomfort, and intermittent dyspnea. Moreover, TTE showed that the patient’s EF (45.8%) was decreased (normal, ≥55%). Therefore, we performed cardioversion at 50 J, the safest and lowest energy level, considering the possible impact on the electrode of the CI.
Little is known about the effect of external defibrillators on CIs, and only 2 cases involving this issue have been published. Busse et al reported the case of a 61-year-old man with a CI who underwent electrical defibrillation at 500 J after cardiac arrest. As in our case, they concluded that electrical defibrillation did not significantly affect CI function, even though the electrical threshold and comfort level of the CI electrodes exhibited minor changes. 6 Schild et al reported that a CI malfunctioned in a patient who underwent cardioversion at 100 J for atrial flutter and intermittent atrial fibrillation. However, unlike in our case, their patient wore a sound processor during the cardioversion, and they speculated that this may have had an effect on the electronic circuit of the CI. 7
An implantable cardioverter defibrillator (ICD), which is inserted into the body, can also provide cardioversion for patients with arrhythmia. An ICD stimulates the heart via bipolar electrodes and delivers approximately 40 J of electrical energy. 8 An ICD is not considered to have any significant effect on the electrodes of cochlear devices. Green et al and Kaneshiro et al reported that CIs continued to function well in patients with Jervell and Lange-Nielsen syndrome, despite undergoing defibrillation with an ICD.9,10
Our patient with a CI underwent cardioversion to restore a normal heart rhythm. Nevertheless, the electrical impedance of the CI electrodes after cardioversion only exhibited minor differences compared to before cardioversion, and low impedance (indicating a short circuit) and high impedance (indicating an open circuit) did not occur. Further, the patient’s CI functioned well after cardioversion. We believe that the electronic circuits of CIs may be relatively tolerant to not only the electrical shock of an external defibrillator but also to the shock delivered by an ICD.
Conclusion
It is unclear whether cardioversion poses a risk of severe damage to the electrodes of CI devices and cochlear tissue. Typically, cardioversion should be avoided in CI recipients because it may damage the implanted devices. If cardioversion cannot be avoided, we strongly recommend starting at the lowest energy level (50 J) and removing the sound processor during the procedure. If the initial shock is unsuccessful in normalizing heart rhythm, the amount of electrical energy applied should be doubled.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Bio & Medical Technology Development Program of National Research Foundation of Korea (NRF) funded by the Korean government MSIT (NRF-2019M3E5D1A02067955) and a grant (HCRI20015) Chonnam National University Hwasun Hospital Institute for Biomedical Science.
