Abstract
Objectives:
To analyze the impact of steroids on postoperative tonsillectomy recovery and implement findings for improvement in postoperative management.
Methods:
Institutional review board approved prospective study with retrospective analysis of private practice setting tonsillectomy patients (November 2015 to January 2017). A questionnaire was provided postoperatively to patients undergoing tonsillectomy with or without adenoidectomy. The study population was separated into 2 groups: patients who received steroids (3 days of either dexamethasone or prednisolone), postoperative steroid (POS), versus patients who did not receive steroids (PONS).
Results:
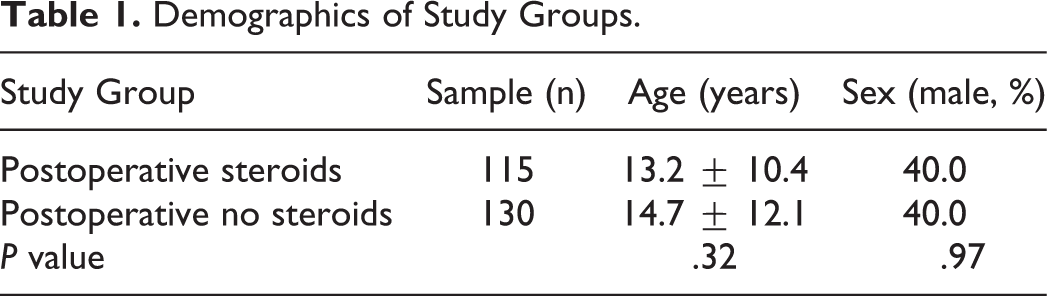
The questionnaire had a return rate of 27.3% (254/931). Nine of the 254 responses were disqualified for lack of information; therefore, the total number of responses was 245. Of these, 115 were POS and 130 were PONS. The groups were similar in mean age (POS: 13.2 ± 10.4 years, PONS: 14.7 ± 12.1 years, P = .32) and sex (POS: Male 40.0%, PONS: Male 40.0%, P = .97). There was an overall decrease of pain and nausea/vomiting (N/V) in the steroid group (P = .0007). There was reduction in pain (P < .05) from postoperative day (POD) 2, 3, 4, and 6 in the POS group. Otherwise, there was no significant reduction in pain from POD 7 to 14, day-by-day rate of N/V, bleeding, or rate of emergency department (ED) or clinic visit (P > .05).
Conclusion:
Postoperative steroid reduced overall pain and N/V, as well as daily pain on POD 2, 3, 4, and 6. Pain from POD 7 to 14, rate of ED or clinic visit, or daily N/V and bleeding rate were not significantly different between cohorts.
Introduction
Tonsillectomy with or without adenoidectomy is one of the most common ambulatory procedures performed by otolaryngologists, with more than 737,000 performed annually. 1 Indications for the procedure include, but are not limited to, sleep-disordered breathing, obstructive sleep apnea, chronic tonsillitis, history of peritonsillar abscess(es), and PFAPA (periodic fever, apthous stomatitis, pharyngitis, and adenitis). 2 Several studies have shown significant quality of life improvements in patients who have undergone tonsillectomy for both sleep-related and infectious etiologies.3-6
In counseling patients and their caregivers prior to the procedure, the surgeon must not only describe the benefits but also the risks, complications, and morbidities one might encounter postoperatively. Complications and morbidities include throat pain, postoperative nausea and vomiting, delayed feeding resulting in dehydration, voice changes, hemorrhage, and rarely death. 2 Arguably the most common morbidity is pain, which typically remains unchanged within the first 4 days postoperatively. 7
Current guidelines strongly recommend that children should receive a single dose of intravenous dexamethasone perioperatively to reduce postoperative nausea and vomiting, along with pain and swelling. 2 This has been supported with several meta-analyses and systematic reviews. However, there is a relative paucity of data investigating administration of oral steroids in the postoperative period. Therefore, we sought to analyze the postoperative relationships that oral steroids have on complications and morbidity of tonsillectomy with or without adenoidectomy.
Patients and Methods
Study Design and Setting
This is an institutional review board (IRB No. 261892) approved study of a retrospective analysis of a prospectively collected deidentified dataset including all patients undergoing tonsillectomy with or without adenoidectomy in an outside private practice group, in an outpatient surgery center. All patients with medical comorbidities that would require further monitoring (ie, diabetes and obstructive sleep apnea) were not included in this study. As such, the side effects of steroid usage were not studied. All data were collected as part of standard clinical care and a quality improvement initiative. A deidentified dataset was provided to the investigators for analysis. The procedures were completed by 10 American Board of Otolaryngology–certified surgeons. No otolaryngology trainees performed any procedures. Procedures were performed using diathermy cautery. The patients or their families were provided a questionnaire at the time of discharge from their procedure. They were instructed to provide the patient’s pain level at the time of recovery and before bedtime from postoperative days (PODs) 1 to 14 along with what type of pain medicine they took that day; if the patient needed a postoperative emergency department (ED) or clinic visit; and whether or not there was nausea/vomiting (N/V), or bleeding. The completed surveys were mailed to the practice secretary or returned in person during the initial postoperative visit. The data were collected from November 1, 2015, to January 1, 2017. The patients were separated into 2 groups: those who received postoperative steroids (POS) and those who did not (PONS). Five of the 10 surgeons prescribed postoperative oral steroids, and their patients were placed into the POS group. Oral steroids consisted of either prednisolone or dexamethasone dosed at 2 mg/kg/d and 0.1 mg/kg/d, respectively. The duration of treatment was 3 days for both medications. Outcome measures were symptoms requiring ED or clinic visit, N/V, bleeding, and pain based on the Wong-Baker face scale. The face scale has been shown to be appropriate for the use in assessment of the intensity of children’s acute pain from age 4 or 5 onward. 8
Data Acquisition
Demographics for age and sex were compiled for all patients. The following data were extracted from each patient’s survey starting from recovery room to POD 14: pain rating, type of pain medicine used, presence or absence of N/V, and presence or absence of bleeding. This dataset was deidentified in accordance with HIPAA regulations and stored in a password-protected, encrypted repository to prevent disclosure of any protected health information.
Statistical Analysis
Statistical comparisons consisting of t test assuming unequal variance were employed to compare the age and pain scale between the 2 groups. Statistical comparisons consisting of χ2 testing were employed to compare sex, rate of postoperative ED or clinic visit, rate of postoperative N/V, and rate of postoperative bleeding. A P value of <.05 was used as a measure for statistical significance in situations where this was appropriate.
Results
A total of 931 surveys were distributed, with a total of 254 responses. Eight responses were excluded because the patient did not answer if they had received POS. One response was excluded because the patient did not specify their sex. This resulted in a total (n) of 245 responses, for a response rate of 27.3%. The 2 groups were almost identical in sample size, and there was no significant difference between the groups for sex and age. Table 1 summarizes the demographics of the study.
Demographics of Study Groups.
A comparison of the groups for all days combined was conducted for pain, rate of bleeding, and rate of N/V. There was a significant difference between the POS and PONS for both pain and N/V, but not for bleeding. Table 2 summarizes the overall comparisons between the 2 groups.
Comparison of Overall Rates.
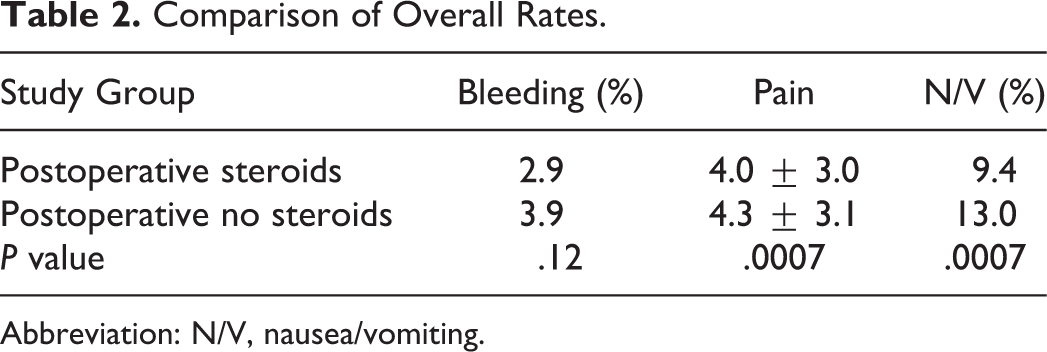
Abbreviation: N/V, nausea/vomiting.
Postoperative pain was measured using the Wong-Baker face scale from recovery to POD14. The general trend for both groups was that pain was the worst within the first postoperative week and stayed relatively constant during that time. It was not until POD 6 to 7 that pain started to steadily decline. There was a significant difference between the 2 groups each day during POD 2, 3, 4, and 6 (P < .05 for all days), with the POS group being up to 1.4 points less than the PONS group. The difference between the 2 groups approached significance in POD 1 and 5, with a P = .06. Figure 1 shows the postoperative pain scale for both groups.
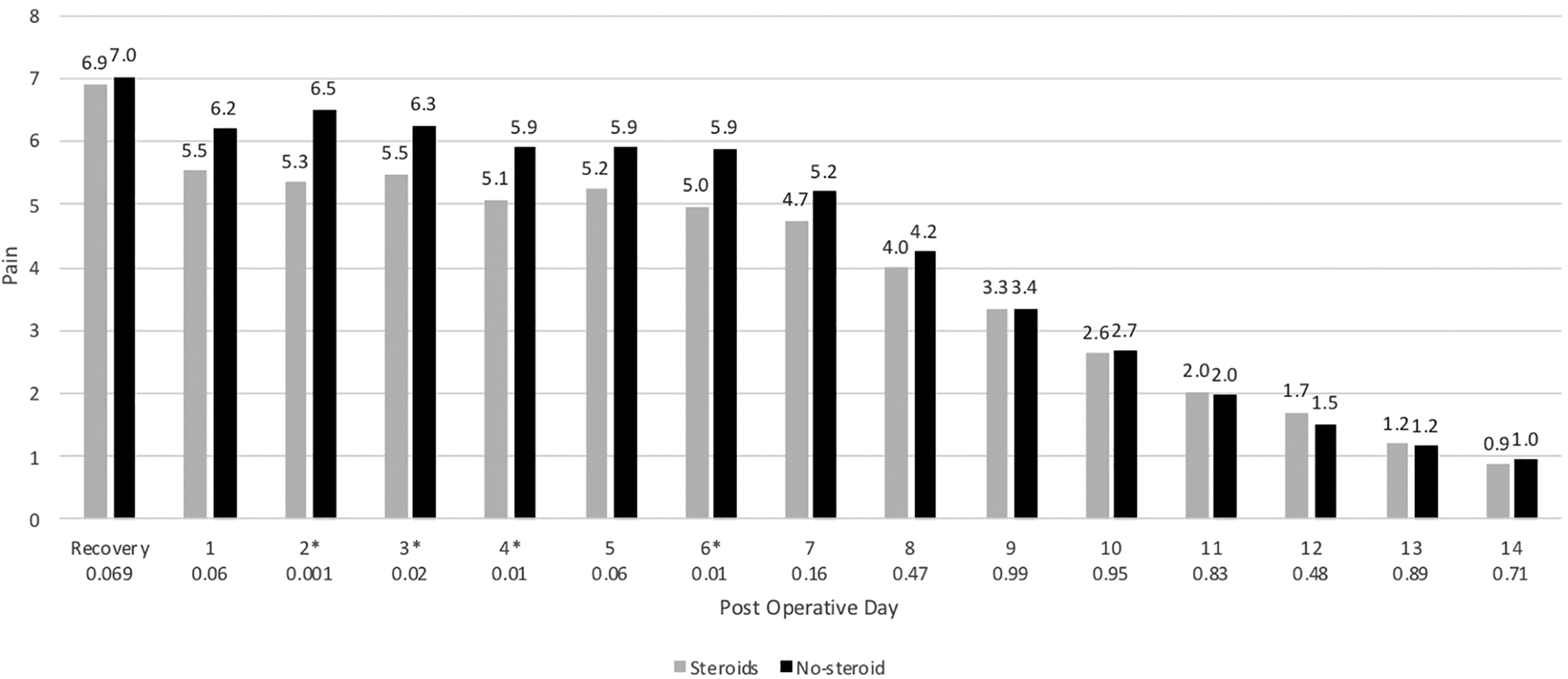
Postoperative pain scores using the Wong-Baker Scale. Asterisk (*) indicates significance (P < .05).
Emergency department or clinic visit for bleeding, pain, or dehydration was also measured. In the POS group, 6 patients required a visit, 102 patients did not, and 7 patients did not respond, resulting in a rate of 5.5%. In the PONS group, 3 patients required a visit and 127 patients did not, resulting in a rate of 2.3%. There was no significant difference between the 2 groups (P = .19).
Postoperative N/V was measured as a positive or negative question for each day until POD14. The highest rates of N/V occurred within the first 2 PODs and declined slowly over the following 2 weeks. The POS group reached its lowest N/V rate of 0.9% by POD 14, whereas the lowest rate for the PONS group was 0.8% at POD 12. The only significant difference between the 2 groups was POD 3 and 4 (P = .02 and .002, respectively). Figure 2 shows the rates of N/V for both groups.
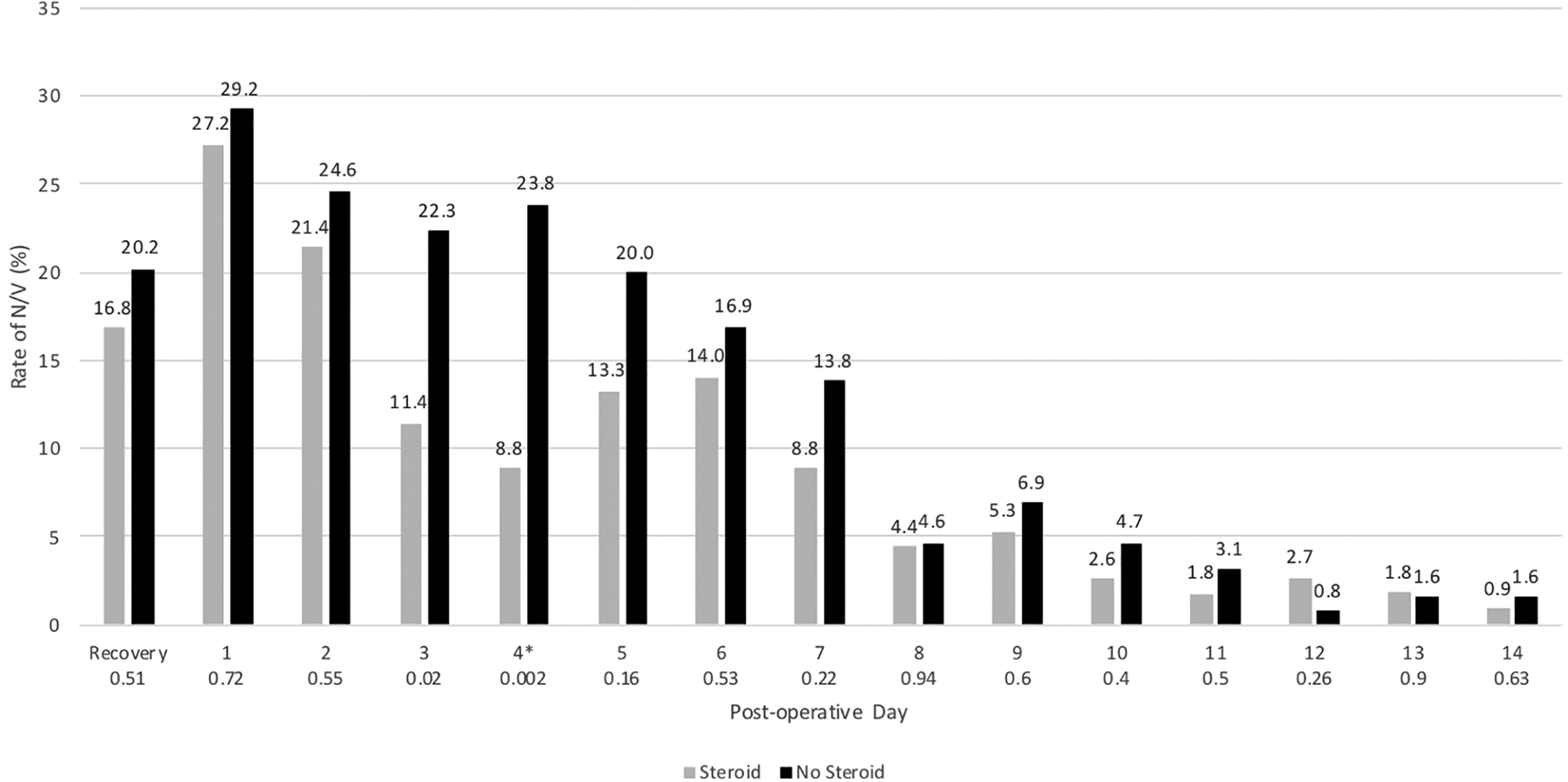
Rates of postoperative nausea and vomiting (N/V). Asterisk (*) indicates significance (P < .05).
Postoperative bleeding was also measured as a positive or negative question for each day until POD 14. There were no obvious trends in bleeding rates between the 2 groups. The only significant difference between the groups was seen on POD 8. Figure 3 illustrates the postoperative bleeding rates.
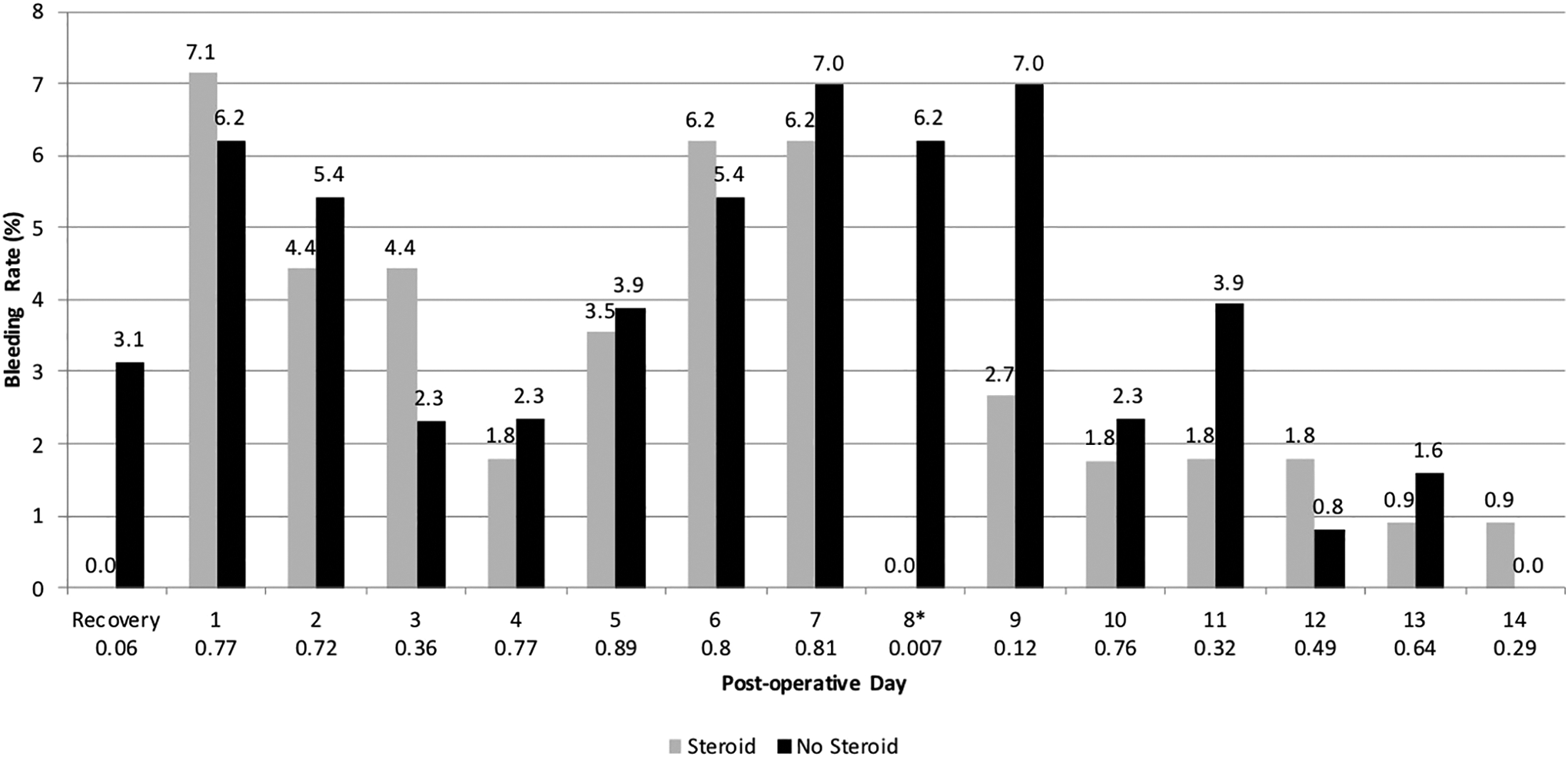
Rates of postoperative bleeding. Asterisk (*) indicates significance (P < .05).
Discussion
The use of steroids for the control of postoperative complications and morbidity in tonsillectomy has been a subject of study for several decades. Papangelou published in 1972 that betamethasone was effective for controlling pain and minimizing edema without causing an increase in bleeding. 9 The vast majority of literature in the past 15 years pertaining to steroids and postoperative tonsillectomy with or without adenoidectomy focus on 2 specific aspects of its administration: immediate (within 24 hours postoperatively) timing10-14 and route (intravenous).10-14 The results of these examinations have led to the strong recommendation by the American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS) that children undergoing tonsillectomy should receive a single dose of steroid to prevent postoperative N/V. 2
Clearly, the evidence for perioperative intravenous steroids has been thoroughly examined. However, there is a paucity of data examining the effects of oral steroids on post-tonsillectomy morbidity. In our literature search, we could only find 5 published trial for oral steroids post tonsillectomy. The data were conflicting about its efficacy and recommended use.
In a randomized, controlled trial of 200 patients examining an 8-day course of dexamethasone in adults, Stewart et al concluded that dexamethasone and a nonsteroidal anti-inflammatory drug (NSAID) significantly reduced pain on all days except the day of surgery and second day after surgery compared to the 2, dexamethasone or NSAID, separately. 15 The most recent randomized controlled trial in 2015 of both pediatric and adult patients showed a 7-day course of prednisolone significantly decreased pain scores at 7 days but not 14 days after the procedure. 16
In the same year as Stewart et al, another randomized, double-blinded, placebo-controlled trial in both children and adults showed a 7-day course of prednisolone decreased postoperative N/V, but had no effect on pain. 17 Furthermore, a randomized, double-blinded, placebo-controlled trial of 102 adult patients saw no significant difference in pain scores out to POD 7 with a 3-day course of dexamethasone. 18 Macassey et al also showed in a randomized, double-blinded, placebo-controlled trial of a 5-day course of oral prednisolone in 193 children no significant difference in postoperative pain or N/V. 19
The results of our investigation follow the formerly described studies which showed a significant decrease in postoperative pain in the steroid group, mainly during the first postoperative week. Similar to Park et al, we did not show any difference in pain scores during the second postoperative week. It can be difficult to draw a definitive conclusion from these studies since there are many variables to consider. First, there was no standardized length of treatment for the steroids. The lengths varied from 3 days such as in our study to 8 days. Also, both dexamethasone and prednisolone were used. Lastly, the trials included both pediatric and adult patients, which can theoretically lead to variability in the quantification of pain.
The effects of steroids on postoperative hemorrhage rates after tonsillectomy were questioned after its recommended use in the perioperative stage of the procedure. Several meta-analyses of dexamethasone administration have shown either a decrease, no overall association, or no significant increase of postoperative hemorrhage.11,20-23,24-26 It has been shown that the rate of post-tonsillectomy bleeding did not increase after publication of the AAO-HNS guidelines despite a modest increase in the rate of dexamethasone use. 27 A single retrospective cohort study did find that dexamethasone administration was associated with a higher standardized rates of primary postoperative hemorrhage, but not secondary postoperative hemorrhage. 28 It is worth noting that these studies reviewed intravenous administration of dexamethasone. Of the oral steroid trials published, only Park et al investigated postoperative hemorrhage as an outcome measure. They, like us, found that there was no statistical difference in bleeding rates for patients who received oral steroids.
A strength of our study was ensuring consistent surgical technique and experience. No trainees performed any of the procedures included in this study. This is significant since it has been shown that experience is more important than technique in preventing postoperative morbidities, at least, postoperative hemorrhage. 29 Limitations of our study include lack of randomization or having a placebo-controlled group. Additionally, this study was limited by the lack of monitoring for complications due to steroid use in this patient population. Future studies on the possible complications caused POS use would be beneficial, especially in a patient population with known comorbidities that are likely to be affected.
Conclusion
This study showed that oral corticosteroids significantly decrease pain scores during the majority of the first postoperative week following tonsillectomy with or without adenoidectomy. They also decrease the overall pain score and rate of N/V during the first 2 postoperative weeks. There was no significant difference in pain scores during the second postoperative week, in the rate of ED or clinic revisit, or in the daily rates of bleeding and N/V. Further research can aim for a standardized dosage and length of treatment for the oral steroids, along with including a broader range of outcome measures in hopes that a meta-analysis can be performed.
Footnotes
Authors’ Note
Presented at: 2017 AAO-HNS Annual Meeting & OTO Experience; Chicago, IL. This study was approved by the UAMS Institutional Review Board (IRB No. 261892). All procedures in this study were conducted in accordance with the UAMS institutional review board (IRB No. 261892) approved protocols. Informed consent for patient information to be published in this article was not obtained because human subjects were not interacted with during the course of analysis of the deidentified dataset provided to the investigators.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
