Abstract
Objective:
Rhinosinusitis is a global health problem affecting millions of people around the world. Baicalin is a bioactive compound isolated from medicinal plant Scutellaria baicalensis Georgi. The present study aims to investigate potential effects of baicalin on clinicopathological changes in nasal/sinus mucosa in a mouse model.
Methods:
A mouse model of sinonasal inflammation induced by high dose of ovalbumin was applied to evaluate effects of baicalin. Rhinosinusitis symptoms, histopathological features, levels of histamine, immunoglobulin E (IgE), IL-17A, IL-10, and balance of regulatory T cell (Treg)/T-helper 17 (Th17) responses were examined.
Results:
Baicalin significantly relieved rhinosinusitis symptoms in mice, reduced histopathological changes, and suppressed serum levels of histamine and IgE in a dose-dependent manner. In lymphocytes of mice, baicalin modulated balance of Treg/Th17 proportions by attenuating Th17 cells and enhancing Treg cells, respectively. The serum IL-17A was decreased and IL-10 was increased in mice treated by baicalin. In addition, baicalin promoted levels of Smad protein 3 (p-Smad3) and forkhead box P3 (FOXP3) to promote Treg cells while suppressed levels of p-Stat3 and retineic-acid-receptor-related orphan nuclear receptor γt (RORγt) to inhibit Th17 cells.
Conclusions:
These data demonstrate that baicalin effectively ameliorates sinonasal inflammation in a mouse model by recovering the immunological balance of Treg/Th17 responses. Our finding highlights the potential value of baicalin for the treatment of rhinosinusitis.
Introduction
Rhinosinusitis is a common disease affecting 5% to 10% of the general adult population worldwide. 1 According to European Position Paper on Rhinosinusitis and Nasal Polyps 2020, acute rhinosinusitis has a 1-year prevalence of 6% to 15%, and chronic rhinosinusitis (CRS) affects 5% to 12% of the general population. 2 The overall prevalence of rhinosinusitis in China was 8.0%. 3 Rhinosinusitis symptoms can have significant negative impact on the patients’ quality of life, thus it is becoming a global health problem to cause substantial economic burden. For example, total €7160 were cost per patient/year, considering the outpatient department visiting, hospitalization, and the productivity losses. 4 Chronic rhinosinusitis is considered as an inflammatory condition affecting the mucus membranes of nose and paranasal sinuses. The pathogenesis of rhinosinusitis is complicated and affected by multiple factors. Various etiologies such as alterations in microbiome composition, defects in innate immunity, and epithelial barrier dysfunction, may contribute to the progress of rhinosinusitis. Therefore, successful treatment of rhinosinusitis is still challenged, and the developments of safe, efficient, and convenient medicines are needed urgently.
Allergic diseases, especially immunoglobulin E (IgE)-mediated airway inflammation like allergic asthma and allergic rhinitis, are commonly thought to be associated with the CRS disease state. The allergy-induced mucosal inflammation may lead to sinus obstruction and secondary infection. 5 The immunological balance of regulatory T cell (Treg)/T-helper 17 (Th17) cell plays a critical role in atopic inflammation including allergic asthma and allergic rhinitis.6,7 Interestingly, in patients with allergic fungal sinusitis, total serum IgE level, cytokines IL-17, IL-21, IL-1β, and TGF-β were comparatively elevated, while IL-2 and IL-10 were reduced along with lower percentages of CD4+CD25+ Treg cells. 8 These observations may provide a new strategy for effective therapy of CRS via targeting on immunological balance of Treg/Th17 cells.
Baicalin (Figure 1A) is a major flavonoid compound isolated from the medicinal plant Scutellaria baicalensis Georgi. Studies have shown baicalin has multiple biological functions, including antitumor, antioxidant, and anti-inflammation activities.9-11 It was found that baicalin prevented allergic response effectively in allergic rhinitis guinea pigs and inhibited inflammatory response via blocking JAK2-STAT5 and NF-kB pathways. 12 Another study indicated that baicalin could regulate arachidonic acid metabolism, the citric acid cycle, histidine metabolism, and linoleic acid metabolism in allergic rhinitis mice to improve the symptoms. 13 Xu et al reported the regulatory effect of baicalin on the imbalance of Treg/Th17 responses in mice with allergic asthma. 14 However, the potential effects of baicalin on immunological balance of Treg/Th17 responses in rhinosinusitis is still largely unknown. In this study, we aim to evaluate efficacy of baicalin on sinonasal inflammation (SI) as well as the effect on regulating balance of Treg/Th17 responses using a mouse model of SI induced by high dose of ovalbumin (OVA). Moreover, the underlying mechanisms involved will be clarified.
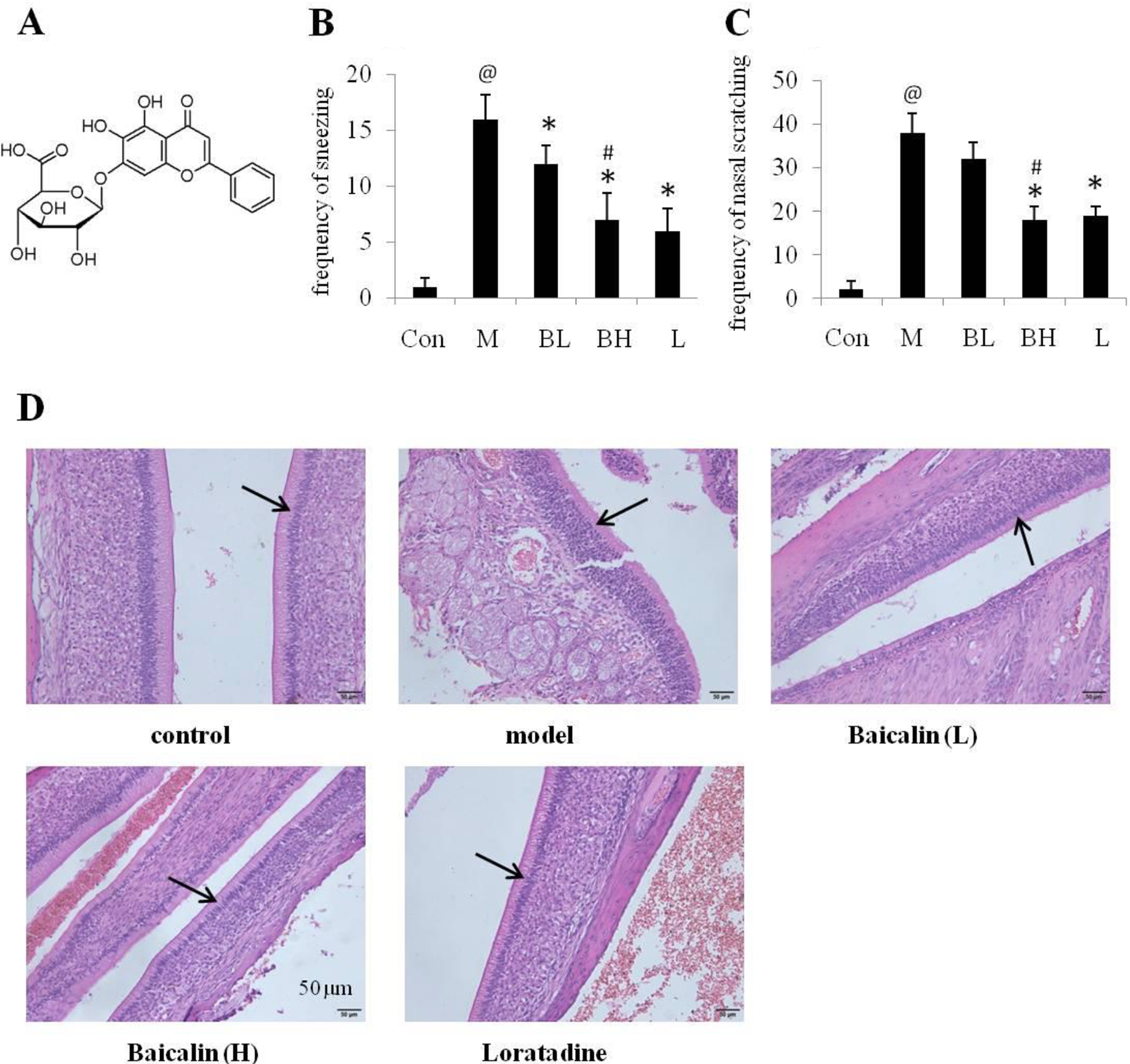
Effects of baicalin on nasal symptoms and histopathological changes in SI mice. A, Chemical structure of baicalin. Effects of baicalin (50 or 200 mg/kg/d) on the frequencies of sneezing (B) and nasal scratching (C) in SI model mice. Loratadine (2 mg/kg/d) as positive control. Con: control group; M: SI model; BL: baicalin low dose (50 mg/kg); BH: baicalin high dose (200 mg/kg); L: loratadine. D, The sinonasal mucosa samples of mice were collected and stained with hematoxylin and eosin (HE). (@ P < .05, SI model to control; * P < .05, treated group to SI model; # P < .05, baicalin high dose to baicalin low dose). Data are shown as mean ± SE, n = 6. SI indicates sinonasal inflammation.
Materials and Methods
Reagents
Ovalbumin was purchased from Sigma-Aldrich. Imject alum adjuvant was obtained from Thermo Scientific. Baicalin (purity >95%, tested by HPLC) was from Aladdin Biochem (Shanghai, China). Loratadine was from Bayer HealthCare AG.
Animal Experiments Design
All animal experiments were approved by the Research Animal Care Committee of Nanjing University of Chinese Medicine and were conducted in accordance with Provisions and General Recommendation of Chinese Experimental Animals Administration Legislation. Male C57BL/6 mice were obtained from SLAC Laboratory Animals. Mice were housed in a temperature-controlled room with a 12-hour light/dark cycle and were allowed free access to food and water during the course of experiments.
The SI mouse model induced by high dose of OVA was established as described before.15,16 In brief, on day 0, mice were sensitized by intraperitoneally (IP) injected with suspensions of OVA and alum adjuvant (for each mouse, 0.9 mg OVA and 77-µL alum adjuvant suspended in 0.1-mL PBS). On days 5 and 10, these mice received additional IP injections of approximately 0.45 mg of OVA and 77 µL of alum adjuvant suspended in 0.1-mL PBS. On days 15, nasal sensitization of mice were induced by intranasal challenges of 1.5 mg of OVA in 20 µL of PBS distributed equally between the 2 nostrils. Intranasal challenges were administered daily for 10 days.
Mice with SI were divided into 4 groups randomly (8 mice per group): SI treated with saline, SI treated with low dose of baicalin (50 mg/kg/d), SI treated with high dose of baicalin (200 mg/kg/d), and SI treated with loratadine (2 mg/kg/d). Baicalin and loratadine were prepared in 0.5% CMC-Na. The control group was given vehicle (saline). All these reagents were delivered by oral gavage daily for 10 days. Meanwhile, to maintain the intranasal stimulation, the SI mice were still challenged with 0.5 mg OVA solution daily.
Evaluation of Nasal Symptoms of SI Mice
After 10-day treatment, the mice were placed in observation cages to evaluate nasal symptoms of rhinosinusitis. The frequencies of sneezing and nasal scratching in the mice were counted for 30 minutes immediately after the last challenge of OVA.
Histological Analysis
The sinonasal mucosa samples of mice were isolated, fixed in 4% formaldehyde, and embedded in paraffin. Four-µm sections of nasal tissues were deparaffinized, rehydrated, then stained with hematoxylin and eosin (HE), and analyzed using light microscopy.
Enzyme-Linked Immunosorbent Assay Assays
Serum samples of mice were collected and stored at −80 °C. Levels of IgE, histamine, IL-17A, and IL-10 in the serum were determined using ELISA kits according to the manufacturer’s instructions. (ELISA kits of IgE and histamine were from Jiancheng Biotech. ELISA kits of IL-17A and IL-10 were from Neobioscience Biotech).
Flow Cytometry Analysis
Lymphocytes were isolated from spleens of mice using a commercial lymphocyte separation solution (Solarbio) according to the instruction. For analysis of Treg cells (CD4+/CD25+/forkhead box P3[FOXP3]+) or Th17 cells (CD3+/CD4+/IL-17A+), 5 × 106 lymphocytes were washed twice with PBS and resuspended in 90-µL PBS, then added antibody of CD4-APC/CD25-FITC or CD4-APC/CD3-FITC (Biolegend), mixed well and incubate at 37 °C for 20 minutes, then fixed cells with Fixation Buffer and permeabilized with permeabilization Wash Buffer (Biolegend). The cells were further stained with antibody of FOXP3-PE or IL-17A-PE (Invitrogen). After washing twice with PBS and resuspending in 500-µL PBS with 10% FBS, the stained cells were analyzed by flow cytometry (FACS Calibur; Becton-Dickinson). The number of Treg cells (CD4+/CD25+/FOXP3+) or Th17 cells (CD3+/CD4+/IL-17A+) was quantified using a BD CellQuest software.
Western Blot Analysis
Lymphocytes isolated from spleens were washed in PBS and lysed (Beyotime Biotech). A 20-µg proteins of each sample were used for Western blotting analysis. The primary antibodies as follows were used at 1:1000 dilution: anti-actin, anti-p-Smad protein 3 (Smad3; Ser423/425), anti-Smad3, anti-signal transducer and activator of transcription 3 (Stat3), anti-p-Stat3 (Tyr705; all from cell signalling), anti-retineic-acid-receptor-related orphan nuclear receptor γt (RORγt) and ant-FOXP3 (all from Abcam), followed by incubation with horseradish peroxidase–linked IgG peroxidase (1:5000, Vazyme Biotech). The bands were visualized and densities of the bands were analyzed using Tanon ChemImaging Systems (Tanon Tech, Co).
Statistical Analysis
Data are presented as means ± SE and were analyzed by paired, Student t test or by analysis of variance with a Bonferroni correction for multiple group comparisons. P values < .05 were considered statistically significant.
Results
Baicalin Relieved Symptoms and Histopathological Changes in Sinonasal Mucosa of SI Mice
In this study, an SI mouse model induced by high dose of OVA was established as described before to evaluate baicalin efficacy.15,16 After the last challenge with OVA solution, the frequencies of nasal scratching and sneezing in mice within 30 minutes were recorded to evaluate effects of baicalin on allergic symptoms. As presented in Figure 1B and C, the frequencies of sneezing and nasal scratching in SI model mice were hugely increased, compared with the control mice (P < .05). After treating by baicalin, the nasal allergic symptoms in SI mice were relieved significantly, compared to SI model mice (P < .05). The frequencies of sneezing and nasal scratching in high dose of baicalin (200 mg/kg)-treating mice were reduced, respectively. Obviously, high dose of baicalin showed better efficacy on relieving nasal allergic symptoms compared to low dose of baicalin. The anti-allergic effects of high dose of baicalin are comparable to loratadine (Figure 1B and C).
Meanwhile, HE staining were conducted to test effects of baicalin on the histopathological changes of the sinonasal mucosa in mice (Figure 1D). The sinonasal respiratory epithelium disruption and leucocytes infiltration were observed in the sinonasal mucosa sections of SI model mice. On the contrary, sinonasal mucosa injuries were alleviated significantly in mice treated by baicalin and loratadine, compared to SI model mice (P < .05). Histological studies indicated that baicalin attenuated OVA-induced inflammatory responses in sinonasal mucosa of mice.
Baicalin Decreased Serum Levels of IgE, Histamine, and Th17 Cytokine IL-17A but Increased Level of Treg Cytokine IL-10 in SI Mice
The total IgE levels and histamine levels in serum of mice were analyzed by ELISA kits. As shown in Figure 2A and B, levels of IgE and histamine in SI model mice were increased significantly compared to the control mice, whereas the increases of IgE and histamine were strongly suppressed by baicalin, respectively, in a dose-dependent manner.
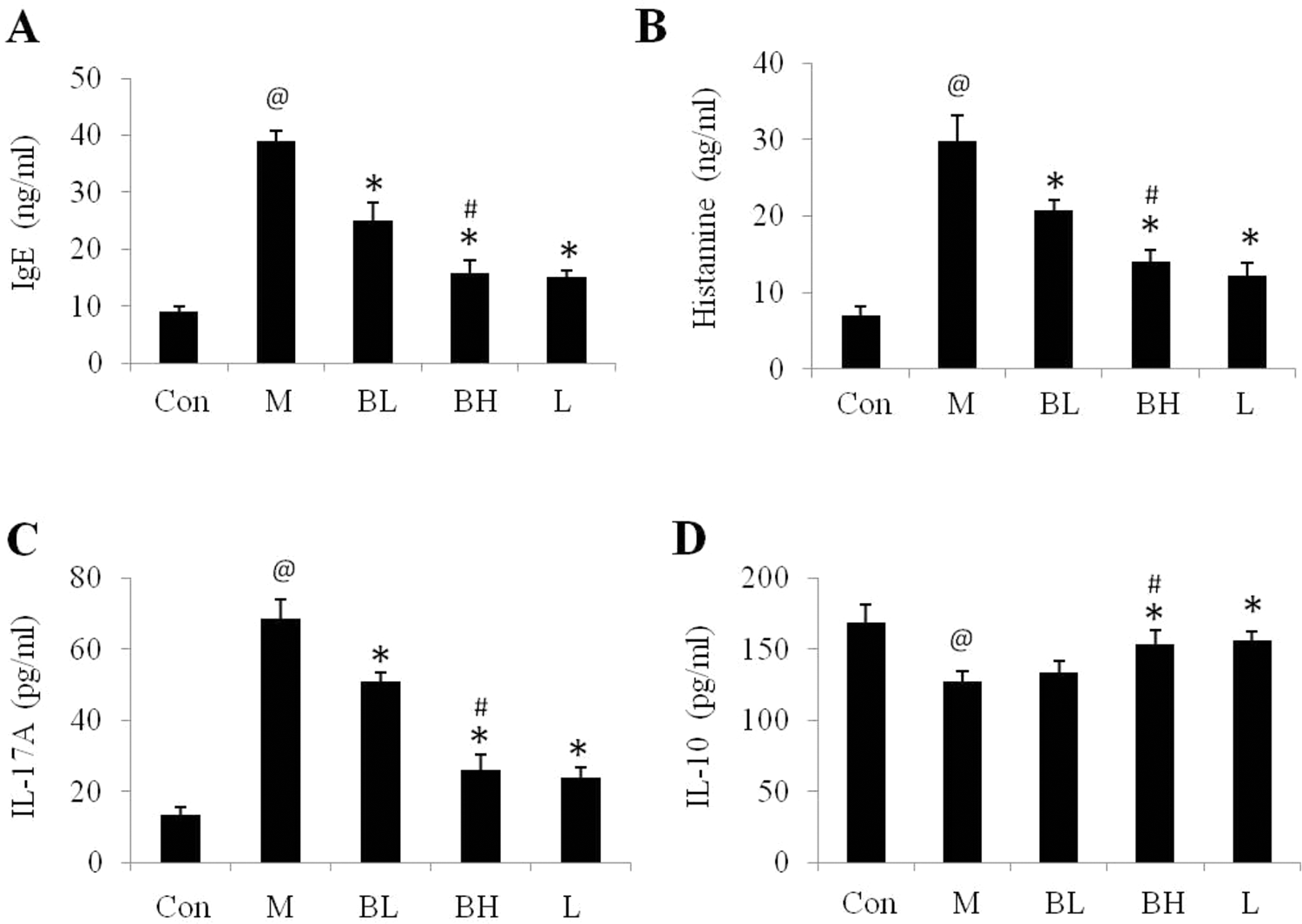
Effects of Baicalin on serum levels of IgE (A), histamine (B), IL-17A (C), and IL-10 (D) in SI mice. Con: control group; M: SI model; BL: baicalin low dose (50 mg/kg); BH: baicalin high dose (200 mg/kg); L: loratadine. (@ P < .05, SI model to control; * P < .05, treated group to SI model; # P < .05, baicalin high dose to baicalin low dose). Data are shown as mean ± SE, n = 6. IgE indicates immunoglobulin E; SI, sinonasal inflammation.
The immunological imbalance of Treg/Th17 cell contributes to the pathogenesis of allergic diseases. Moreover, IL-17A which is a cytokine secreted by Th17 cells and IL-10 which is a cytokine to promote differentiation of Treg cells were examined by ELISA assays. It was observed that IL-17A was hugely upregulated in SI model mice compared to the control mice (Figure 2C); on the contrary, IL-10 was downregulated in SI model mice significantly (Figure 2D). Baicalin showed regulating effects on these 2 cytokines to decrease the IL-17A level and increase the IL-10 level notably (Figure 2C and D).
Baicalin rebalanced the Treg/Th17 Cell Proportions in Spleens of SI Mice
To further confirm the effects of baicalin on regulating the balance of Treg/Th17 cells, the percentages of Treg cells and Th17 cells in spleens of mice were analyzed by FACS respectively. As presented in Figure 3, the percentage of Treg cells in spleens of SI model mice was 2.05%, which was significantly lower than that of control mice (3.12%). In contrast, as shown in Figure 4, the percentage of Th17 cells in spleens of SI model mice was 3.69%, which was much higher than that of control mice (2.01%). Interestingly, here we noticed that baicalin could modulate the balance of Treg/Th17 cells via upregulating the percentage of Treg cells (2.64%, high dose of baicalin) and downregulating the percentage of Th17 cells (1.93%, high dose of baicalin; Figures 3 and 4). These findings suggested that baicalin could rebalance the Treg/Th17 cell responses to relief the symptoms of SI mice.
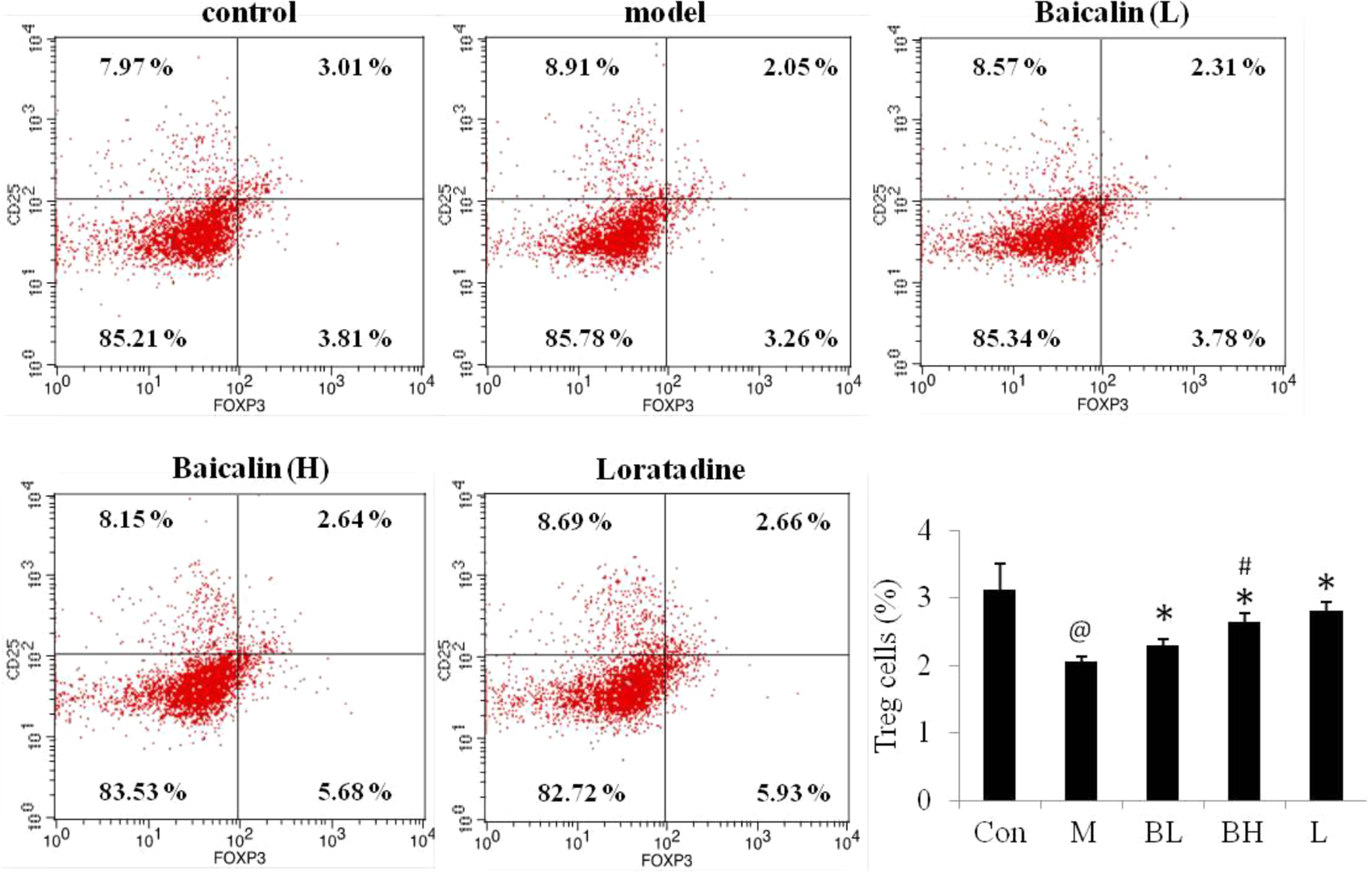
Regulating effects of baicalin on the percentage of CD4+/CD25+/FOXP3+ Treg cells in spleens of SI mice. Representative flow cytometry dot plots for each groups were shown, and ratios of Treg cells were calculated and analyzed. Con: control group; M: SI model; BL: baicalin low dose (50 mg/kg); BH: baicalin high dose (200 mg/kg); L: loratadine (@ P < .05, SI model to control; * P < .05, treated group to SI model; # P < .05, baicalin high dose to baicalin low dose). Data are shown as mean ± SE, n = 4. Treg, regulatory T cell. FOXP3indicates forkhead box P3; SI, sinonasal inflammation.
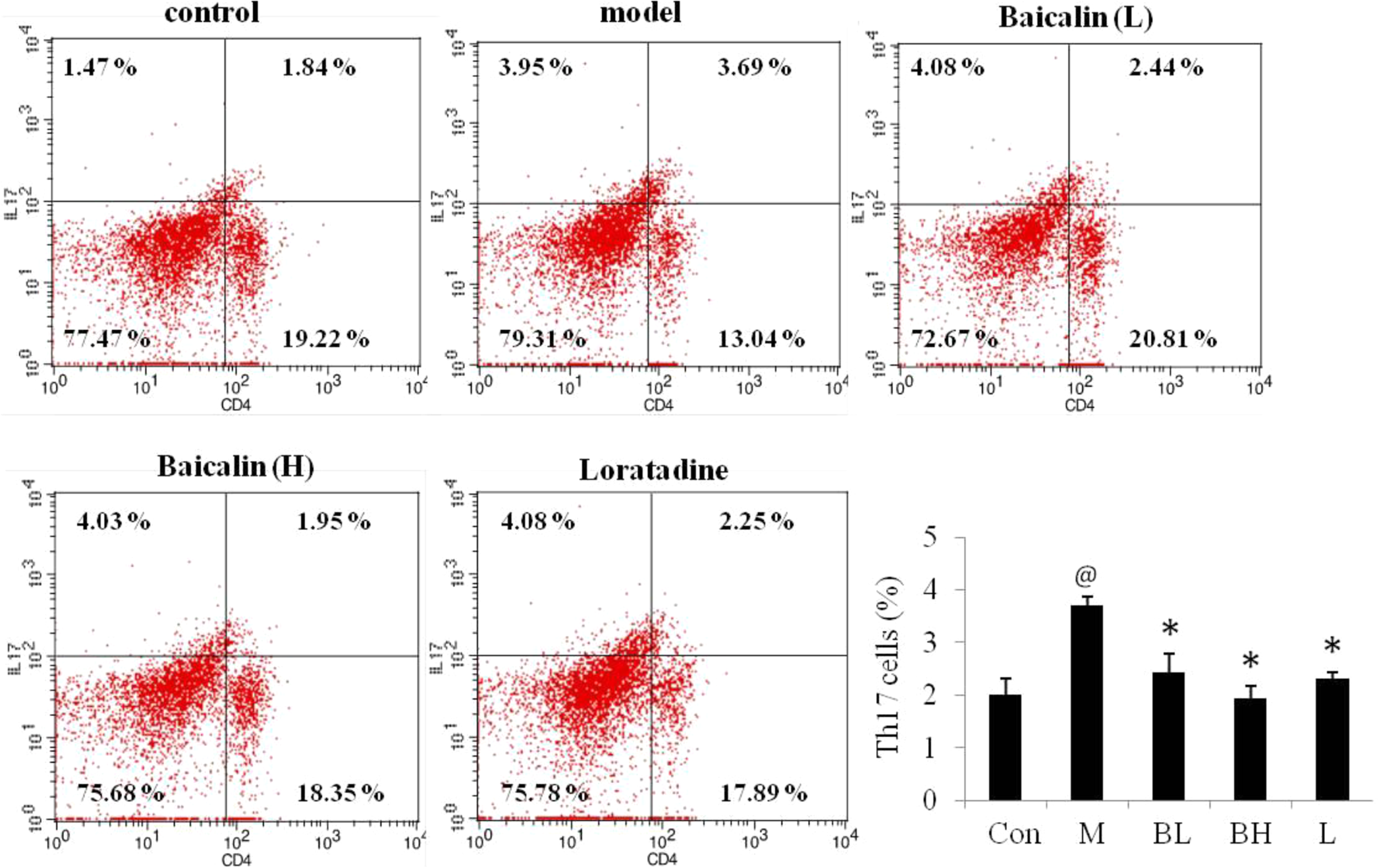
Regulating effects of Baicalin on the percentage of CD3+/CD4+/IL-17A+ Th17 cells in spleens of SI mice. Representative flow cytometry dot plots for each groups were shown, and ratios of Th17 cells were calculated and analyzed. Con: control group; M: SI model; BL: baicalin low dose (50 mg/kg); BH: baicalin high dose (200 mg/kg); L: loratadine. (@ P < .05, SI model to control; * P < .05, treated group to SI model; # P < .05, baicalin high dose to baicalin low dose). Data are shown as mean ± SE, n = 4. Th17, T-helper 17. SI, sinonasal inflammation.
Baicalin Modulated Specific Transcriptional Factors Related to Differentiations and Functions of Treg and Th17 Cells
Next, expressions of the specific transcriptional factors FOXP3 and RORγt, which are essential for differentiations and functions of Treg and Th17 cells, respectively,17,18 were examined.
FOXP3 expression was decreased by 2.38-fold in lymphocytes of SI model mice compared to that of control mice, while RORγt expression was increased by 2.34-fold in lymphocytes of SI model mice compared to that of control mice (Figure 5A and B). The downregulation of FOXP3 and upregulation of RORγt were both reversed by baicalin. Baicalin significantly restored the expression of FOXP3 and suppressed the expression of RORγt (Figure 5A and B).
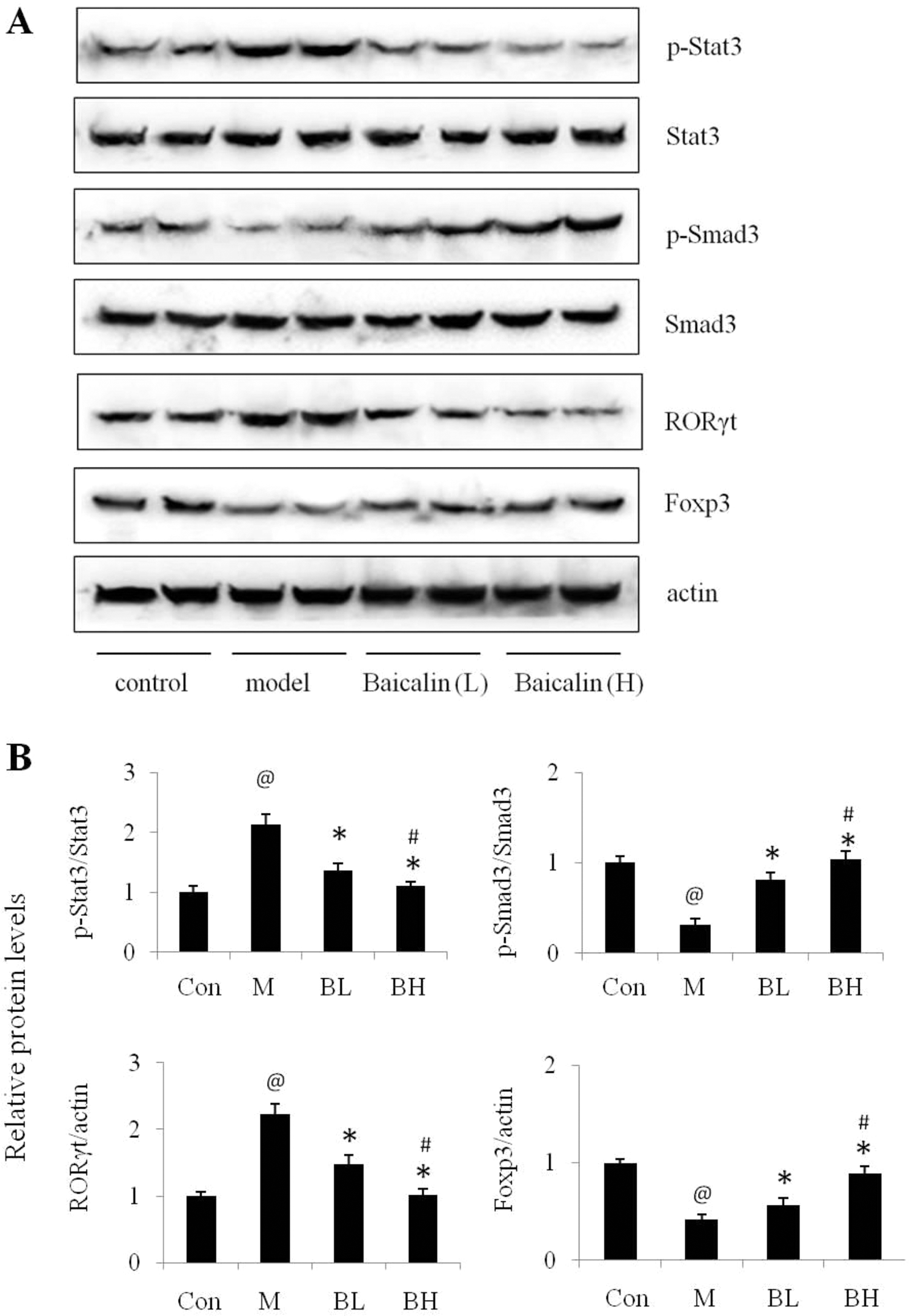
Diverse effects of Baicalin on transcriptional factors Stat3, Smad3, FOXP3, and RORγt in lymphocytes of SI mice. A, Representative pictures of Western blot analysis were presented. B, The densitometric analyses of 3 independent experiments are shown. Con: control group; M: SI model; BL: baicalin low dose (50 mg/kg); BH: baicalin high dose (200 mg/kg; @ P < .05, SI model to control; * P < .05, baicalin-treated group to SI model; # P < .05, baicalin high dose to baicalin low dose). FOXP3 indicates forkhead box P3; RORγt, retineic-acid-receptor-related orphan nuclear receptor γt; SI, sinonasal inflammation; Smad3, Smad protein 3; Stat3, signal transducer and activator of transcription 3.
Furthermore, another 2 crucial transcriptional factors Stat3 and Smad3 involved in differentiations of Treg and Th17 cells were measured as well.19,20 In lymphocytes of SI model mice, the activation of Smad3 (p-Smad3) was inhibited, and the activation of Stat3 (p-Stat3) was enhanced compared to that of control mice respectively (Figure 5A and B). Similarly, baicalin presented diverse modulating effects on activations of Stat3 and Smad3 to rebalance the Treg/Th17 cell responses (Figure 5A and B).
Discussion
Rhinosinusitis is one of the most common chronic inflammatory diseases of the upper airway. Even though rhinosinusitis is not a life-threatening disease, symptoms still have significant negative impact on the quality of life in patients. 21 Moreover, CRS is associated with several comorbid disorders such as nasal polyposis, atopic dermatitis, and bronchial asthma. 22 A large population-based cohort study demonstrated that patients in Taiwan with previous CRS are at greater risk of developing the adenocarcinoma subtype of lung cancer. 23 However, due to the multifactorial etiology and complex pathophysiology of CRS, the effective medical treatment of CRS is still challenging.
Complementary and alternative medicine plays an important role in healthcare worldwide. According to the Taiwan National Health Insurance Research Database, 29% of patients with CRS used traditional Chinese medicine in conjunction with Western medicine. 24 Notably, Chinese herbal medicine have been evaluated for efficacy in rhinosinusitis treatment in clinic. For example, a clinical study evaluated the effect of Chinese traditional medicine lianhuaqingwen granules on CRS. Lianhuaqingwen granule has been demonstrated it could control the inflammation in nasal mucosa and can improve CRS via regulating the ratio of CD4+ and CD8+ T cell subpopulations, 25 which indicated the potential impacts of Chinese herbal medicine on modulating immune balance.
In the present study, baicalin, a bioactive component isolated from the Chinese medicinal plant Scutellaria baicalensis Georgi, was shown to relieve rhinosinusitis symptoms, reduced histopathological changes, and suppressed serum levels of histamine and IgE in a dose-dependent manner in OVA-induced SI in mice. Moreover, baicalin modulated balance of Treg/Th17 responses by attenuating Th17 cells and enhancing Treg cells, respectively. Mechanism studies revealed that baicalin promoted levels of p-Smad3 and FOXP3 to promote Treg cells, while suppressed levels of p-Stat3 and RORγt to inhibit Th17 cells.
There is evidence showing the immunological imbalance of Treg/Th17 cell involved in the pathogenesis of allergic diseases like allergic asthma and allergic rhinitis6,7 as well as in allergic fungal sinusitis. 8 Besides Th1 and Th2 cells, Th17 cell is the third subset of CD4+ T cells by characterized as IL-17-producing effector T helper cells.26,27 T-helper 17 cells have critical roles in mucosal defense and are major contributors to inflammatory disease. On the contrary, Treg cells promote self-immune tolerance and maintenance of immune balance. The specific transcriptional factors FOXP3 and RORγt are essential for differentiations and functions of Treg and Th17 cells, respectively.17,18 Smad3 is another key transcriptional factor involved in functions of both FOXP3 and RORγt. 28 In naive T cells, the induction of FOXP3 expression by TGF-β was significantly reduced in the absence of Smad3. On contrast, Smad3 deficiency led to enhanced Th17 differentiation. Moreover, Smad3 was found to interact with RORγt and decrease its transcriptional activity.
The balance of Tregs cells and Th17 cells plays a key role in maintaining immune homeostasis, while the imbalance of them is related to many diseases such as autoimmune diseases, allergy, infection, and cancer. The plasticity between Tregs and Th17 cells has been proved that Treg and Th17 cells can be regulated reciprocally during differentiation. 29 Therefore, the therapeutic strategies by targeting regulators of Treg/Th17 balance or regulators of the expression and activity of FOXP3 and RORγt are promising and should be paid more attention.
Conclusions
Taken together, our findings demonstrate that baicalin effectively relieves symptoms in a mouse model of SI by recovering the immunological balance of Treg/Th17 responses. Our study provide evidence that baicalin might be a potential candidate for rhinosinusitis therapy via regulating Treg/Th17 balance.
Footnotes
Authors’ Note
YM, ZXY, FFD, and GQH performed experiments; ZXP, XY, and WX helped perform statistical analysis of results and wrote related parts of the manuscript. YXT, HXY, and WX designed the work and wrote the final manuscript. All authors gave input to the manuscript. All animal experiments were approved by the Research Animal Care Committee of Nanjing University of Chinese Medicine, and were conducted in accordance with Provisions and General Recommendation of Chinese Experimental Animals Administration Legislation. The reference number is IACUC-001-10.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Nanjing Science and technology development projects, 201803039.
