Abstract
Objectives:
Balloon eustachian tuboplasty (BET) is a promising therapeutic option for eustachian tube (ET) dysfunction. However, data are lacking on the effect of BET in adults with symptoms of chronic ET dysfunction but without a contributing pathology. This study investigated the effect of BET in adult patients with only symptoms of chronic ET dysfunction.
Methods:
This prospective clinical trial included adult patients with aerated physiological middle ears and symptoms of ET dysfunction for more than 6 months. Compliance with follow-up was 93.3%. We evaluated the effects of BET with tympanometry, assessment of the Valsalva or Toynbee maneuver with tympanometry verification, a Eustachian Tube Dysfunction Questionnaire (ETDQ-7), and pure-tone audiometry. Data were recorded 1 day before surgery and 2, 6, and 12 months after BET. Therapy was considered successful when the patient exhibited a newly acquired ability to perform the Valsalva or Toynbee maneuver or when the ETDQ-7 score improved by 20% or more.
Results:
We included 14 ears in the analysis. After 2, 6, and 12 months, therapy was successful, according to the ETDQ-7, in 11/14 (78.6%; 95% CI: 48.8-94.3), 13/14 (92.9%; 95% CI: 64.2-99.6), and 12/14 (85.7%; 95% CI: 56.2-97.5) ears, respectively. These results were statistically significant. The ETDQ-7 scores also significantly decreased at 2, 6, and 12 months after the BET, when any change was observed. All patients experienced improvement. Only 1 patient reported temporary deterioration after 2 months. Treatment was more frequently successful in patients without nasal polyps or pollinosis.
Conclusions:
Adults with only symptoms of chronic ET dysfunction benefitted more and had longer lasting results from BET, compared to patients with pathologies caused by ET dysfunction. Balloon eustachian tuboplasty could be recommended for these patients.
Introduction
Balloon eustachian tuboplasty (BET) is the dilatation of the cartilaginous part of the eustachian tube (ET) with a transnasal balloon. Balloon eustachian tuboplasty is becoming increasingly popular due to its promise as a therapeutic option for ET dysfunction. Current studies have demonstrated a 47% to 100% success rate for BET therapy, depending on the evaluated parameter and length of follow-up. 1 -6 However, results are difficult to compare between studies, due to the lack of standardized evaluation protocols. In addition, most studies were conducted in large but significantly inhomogeneous groups of patients. Patients with an aerated physiological middle ear that had only symptoms of ET dysfunction (patients with symptoms of ET dysfunction “without pathology” on examination or audio testing) were evaluated together with patients with acute/chronic otitis media with effusion, patients with tympanic membrane adhesion, patients with chronic suppurative otitis media, and patients after or during middle ear surgery. 1 -6 Therefore, for any given patient, it is difficult to estimate an expected outcome and provide reliable advice.
Recently, a few studies focused on adults with chronic middle ear effusion. This was the first subgroup studied among patients with chronic ET dysfunction. 7 -9 However, data are lacking on how BET might affect other subgroups, including patients that only have symptoms of chronic ET dysfunction.
Therefore, this prospective clinical trial aimed to provide data on the effect of BET in adult patients that only had symptoms of chronic ET dysfunction (patients with symptoms of ET dysfunction “without pathology” on examination or audio testing). To our knowledge, this study was the first to report the effects of BET in this group of patients.
Methods
Ethical Considerations
This study was approved by the ethics committee of our institution. It was performed in accordance with the Declaration of Helsinki, applicable regulatory requirements, and the principles of good clinical practice. The study was registered at ClinicalTrials.gov under the identifier NCT03556215. Written informed consent was obtained from the patients before initiating any procedure. All authors reviewed and approved the final manuscript.
Design and Setting
This prospective clinical trial was performed between May 2017 and August 2020 in a tertiary referral hospital.
Participants
This study included adult patients with an aerated physiological middle ear and symptoms of ET dysfunction for more than 6 months (patients with symptoms of ET dysfunction “without pathology” on examination or audio testing). All patients underwent conservative treatment with no effect. They reported no improvement for at least 6 months and was referred to tertiary referral hospital. The diagnosis of ET dysfunction was confirmed with the Eustachian Tube Dysfunction Questionnaire (ETDQ-7; a score cutoff of >14.5; Figure 1). 10 A higher ETDQ-7 score indicated greater ET dysfunction. All patients underwent computed tomography, which focused on the integrity of the bony canal of the internal carotid artery. This examination provided confirmation of no middle ear secretions. Exclusion criteria were an obstruction of the ET in the nasopharynx, acute or chronic otitis media, middle ear secretions, type B tympanometry, conductive hearing loss, cleft palate, history of middle or inner ear surgery, history of BET, head and neck cancer, pregnancy, consumption of anticonvulsants or antipsychotic medications, or incomplete integrity of the bony canal of the internal carotid artery, based on computed tomography.

Eustachian Tube Dysfunction Questionnaire-7.
Data Acquisition
The effects of BET were evaluated with tympanometry, verification of the ability to perform a Valsalva or Toynbee maneuver with tympanometry, the ETDQ-7, and pure-tone audiometry. Tympanometry and audiometry were performed by an audiology assistant. Tympanometry was performed with a Madsen Zodiac tympanometer (Otometrics). Regular tympanometry was performed first, then tympanometry was performed after the Valsalva (first) and Toynbee (second) maneuvers. Audiometry was performed in a soundproof cabin with a Madsen Orbiter 922 audiometer (Madsen Ltd), compliant with ISO 389 standards. Pure-tone audiometry was performed according to international standards (ISO 8253-1).
Procedure
Balloon eustachian tuboplasty was performed in patients under general anesthesia with orotracheal intubation. Patients were placed in the supine position with the head slightly inclined and slightly bowed to the right. The procedure was initiated by decongesting the nasal mucosa with nasal swabs soaked in adrenaline at a dilution of 1:10 000. Balloon eustachian tuboplasty was performed with a commercial TubaVent set (Spiggle & Theis Medizintechnik GmbH), which consisted of a single-use insufflation pump and a catheter with a dilatation balloon at the end. The catheter was inserted into a metal inserter, bent at 30° at the end (Spiggle & Theis); then, the instrument was introduced transnasally into the nasopharyngeal orifice of the ET and the cartilaginous part of the ET. The introduction was visualized with a transnasally inserted, rigid endoscope (bent at 30°, 4-mm diameter) with high resolution. Next, the balloon was filled with saline to a target pressure of 10 bar with an insufflation pump. This pressure was applied for 2 minutes. Then, the saline was drained, and the balloon was pulled out of the nasal cavity, together with the inserter. In the case of bilateral ET dysfunction, the same procedure was followed on the other side during anesthesia. A new single-use material was used.
Follow-Up
Data were recorded 1 day before surgery and 2, 6, and 12 months after the BET.
Statistical Analysis
Data were processed with descriptive statistics, expressed as the arithmetic mean, SD, and absolute and relative frequencies. Treatment outcomes were evaluated with the Wilcoxon paired test, and the results are expressed as the success rate and CI. The statistical tests were assessed with a significance level of 5%. Therapy was considered successful when the patient exhibited a newly acquired ability to perform the Valsalva or Toynbee maneuver or when the ETDQ-7 score improved by 20% or more. All statistical analyses were performed with Stata 13 software (Stata Corp).
Results
The study included 15 (8 right-sided and 7 left-sided; n = 11 patients) ears with chronic ET dysfunction alone. Compliance with follow-up was 93.33%. Immediately after BET, 1 ear was lost to follow-up and, subsequently, was excluded. Thus, 14 ears were analyzed.
The final cohort included 6 men and 4 women; the average age was 46.6 years. Only 1 man was a smoker. Five (8 ears) patients had minor nasal polyps, which did not reach the common nasal meatus or the nasopharynx. Two (4 ears) patients had pollinosis. All patients were taking local nasal corticosteroids and had performed nasal irrigation and politzerization for at least 3 months, with no effect, prior to study inclusion.
All ears demonstrated a type A tympanogram and no conductive hearing loss before the BET. These features remained stable throughout the 12-month follow-up.
After 2, 6, and 12 months, >20% reductions in the ETDQ-7 scores indicated successful therapy in 11/14 (78.6%; 95% CI: 48.8-94.3), 13/14 (92.9%; 95% CI: 64.2-99.6), and 12/14 (85.7%; 95% CI: 56.2-97.5) ears, respectively. The patients served as their own control group because none of the patients responded to any conservative treatment before study inclusion. Results are statistically significant. There was also significant reduction of ETDQ-7 score after 2, 6, and 12 months after BET when any change was evaluated (without application of >20% score reduction criteria for successful therapy; Figure 2). All patients showed improvement after the BET compared to baseline. Only 1 patient reported a temporary deterioration after 2 months.
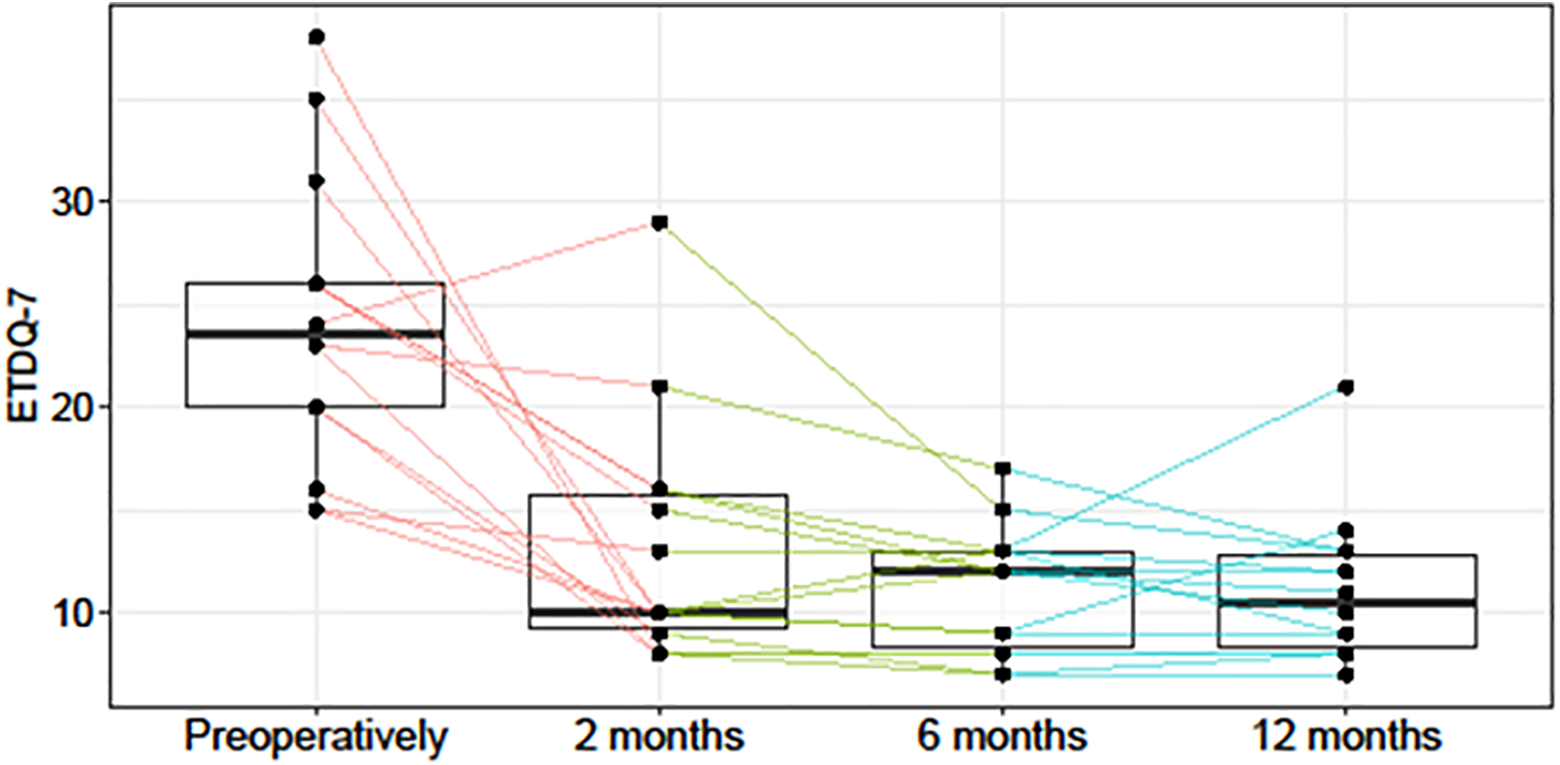
Questionnaire scores (y-axis) during follow-up (P values for changes compared to baseline were evaluated with the Wilcoxon rank-sum test; 0-2 months [red], P = .001; 0-6 months [green], P < .001; 0 12 months (blue), P = .001).
Treatment success was more frequently observed in patients without nasal polyps or pollinosis, compared to patients with nasal polyps or pollinosis (Table 1). However, these groups were too small and too dissimilar to evaluate the significance of these differences.
Treatment Success, According to ETDQ-7 Scores, in Patients With or Without Nasal Polyps or Pollinosis That Underwent BET for Symptoms of Chronic ET Dysfunction.a
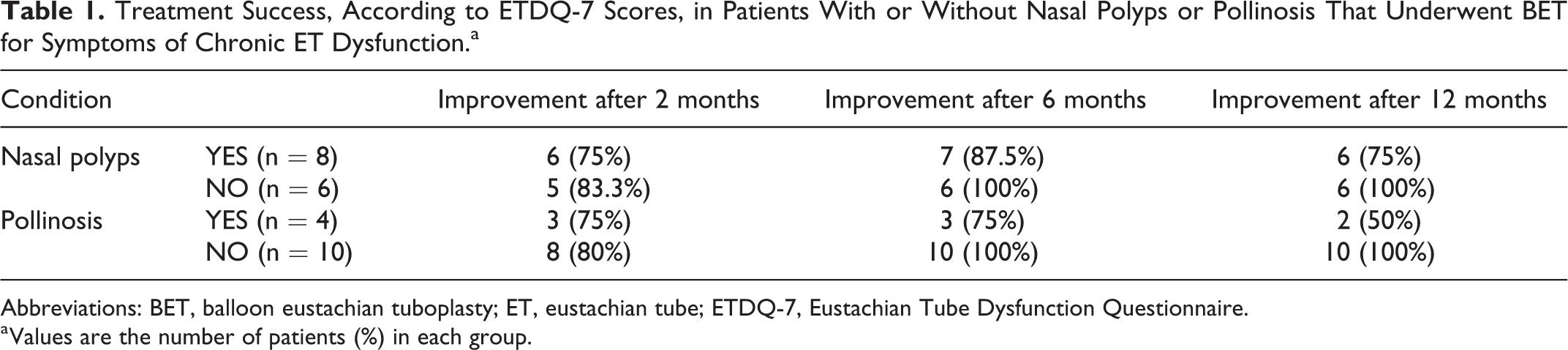
Abbreviations: BET, balloon eustachian tuboplasty; ET, eustachian tube; ETDQ-7, Eustachian Tube Dysfunction Questionnaire.
a Values are the number of patients (%) in each group.
Three ears could not perform the Valsalva or Toynbee maneuver preoperatively. These ears gained the ability after the BET, and the ability was retained during the 12-month follow-up. In addition, the ETDQ-7 scores showed that therapy was successful in these 3 ears.
In general, the BET treatment was tolerated well. One patient experienced mild headaches (<3 on a visual analogue scale for pain) that did not last more than 24 hours. Another patient, as previously mentioned, experienced temporary deterioration of symptoms after 2 months.
Discussion
Balloon eustachian tuboplasty appears to be a promising therapeutic option for ET dysfunction, but we lack data on its effect in adults that only have symptoms of chronic ET dysfunction. These symptoms include a fullness or pressure in the ear, feeling like the ear is clogged or “under water,” changes in hearing, ringing in the ear, clicking or popping sounds, ticklish feelings in the ears, and pain in the ear. The symptoms might appear during increased demands on the ET (such as driving, flying, cable car/elevator rides, and diving), or they might be present under normal circumstances and only worsen when demands on the ET are increased (ie, intermittent worsening). 11
All our patients reported symptoms that were nearly constant, with intermittent worsening. Some authors consider these conditions precursors to other pathologies caused by ET dysfunction. These pathologies can eventually become organic, which increases the probability of constant symptoms. 11 This assumption was supported our study, which showed that the BET outcomes were more promising and longer lasting in the subgroup of patients with only symptoms of ET dysfunction compared to those with ET dysfunction and chronic middle ear effusion. 7 -9 These results suggested that ET dysfunction symptoms could be a less advanced condition than ET dysfunction with a pathology. Therefore, a mixture of patients with ET dysfunction symptoms alone and patients with chronic middle ear effusions (and certainly other pathologies) could significantly influence the results in studies that evaluate all patients with ET dysfunction as 1 group. Our findings indicated that these subgroups should be evaluated separately.
Paradoxically, surgeons might be reluctant to perform a BET in patients with only symptoms of ET dysfunction (on objectively and semi-objectively “healthy” ear), due to potential complications. However, conservative therapy typically has no long-term effect on chronic ET dysfunction, and thus, it is not routinely recommended. 12,13 Our results showed that this condition could be successfully treated with BET. Thus, we recommend offering BET to adult patients that only have symptoms of ET dysfunction because good long-term results can be expected. Moreover, we found that BET provided particularly favorable outcomes in patients without nasal polyps or pollinosis.
The main limitation of our study was the small sample size, which did not allow us to draw clear conclusions on the effects of nasal polyps and pollinosis on the success of BET. Study is also limited to a very specific population. There is no matched control group receiving a sham procedure. The placebo effect or the natural history of disease for this population could significantly skew results. Patient’s baseline outcomes are based upon one preoperative visit prior to procedure and may not be reflective of 6 months preceding procedure.
It is also necessary to mention that the ETDQ-7 was used differently in our study. The questionnaire was validated for the diagnosis of ET dysfunction, based on an optimal total item score cutoff of <14.5. 10 However, in that validation study, the number of individuals tested was relatively low, and all types of ET dysfunction were originally evaluated together in 1 group. Therefore, the ETDQ-7 might not always be accurate for diagnosing all subgroups of ET dysfunction (eg, chronic otitis media with effusion, chronic suppurative otitis media). In addition, when, for example, the treatment halved the significant problems in a given patient, we considered the treatment a success that greatly benefitted the patient; nevertheless, it might have been considered a failure, if the ETDQ-7 score was not ≤14.5 points. And vice versa, a small improvement, for example, from 15 to 14 points, would have been recorded as a cure, even though the treatment provided very little benefit to the patient. Therefore, we implemented a change in ETDQ-7 score to evaluate the treatment effect, rather than using a cutoff score, which was recently questioned in terms of accuracy. 14
Further investigation could evaluate how subgroups of patients with nasal polyps, pollinosis, reflux, and smoking differed in their response to treatment.
Conclusion
This study found that BET provided more promising and longer lasting outcomes in adults with only symptoms of chronic ET dysfunction compared to adults with different pathologies caused by ET dysfunction. Therefore, BET could be recommended for these patients.
Footnotes
Authors’ Note
Effect of balloon eustachian tuboplasty in adults that only have symptoms of chronic eustachian tube dysfunction, with a 1-year follow-up (prospective clinical trial).
Acknowledgment
The authors would like to thank Mgr. Adela Vrtkova for the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Ministry of Health, Czech Republic—conceptual development of research organization (FNOs/2020).
