Abstract
Rhabdomyosarcoma of the larynx has been very rarely described in pediatric population. There are 3 histological subtypes: embryonal, pleomorphic, and alveolar. With regard to the English literature, we present the first case of alveolar rhabdomyosarcoma of the larynx ever described in a child. This tumor has been diagnosed on an endoscopic biopsy. Thus, a unilateral arytenoidectomy has been performed. This tumor has a poor prognosis. Lymph node metastases were successfully treated by chemotherapy and radiotherapy. Surgery has shifted from radical to conservative combined with adjuvant treatments.
Introduction
Rhabdomyosarcoma (RMS) is a malignant tumor developing from the embryonal mesenchyme which normally gives rise to striated muscle.1,2 Rhabdomyosarcoma accounts for approximately 5% of all malignant tumors in children between the age of 1 and 15 years.1,3,4 About half of the cases of RMS occur in the head and neck area, but few are localized in the larynx. 1 Three basic histologic variants of RMS have been described: embryonal, alveolar, and pleomorphic. 5 Based on the English literature, we present what we believe to be the first case of alveolar RMS in a child.
Case Report
A 3-year-old child with a history of chronic asthma was initially managed for increased dyspnea. Inhaled beta-2 mimetics and systemic corticosteroids were ineffective, prompting a consultation in the pediatric emergency department. Other comorbidities include cystic pulmonary adenomatoid malformation operated on at 6 months of age and gastroesophageal reflux disease.
Upon arrival at the pediatric emergency department, the child presented with a laryngeal dyspnea. Nasofibroscopy identified a swelling developed on the left arytenoid. The glottic area was not visible. Endoscopy performed in the operating theater with spontaneous ventilation confirmed the presence of a suspicious submucosal lesion on the left arytenoid. There was no supraglottic nor subglottic extension. Numerous biopsies were taken.
On pathological examination, a tumor proliferation without individualizable alveolar architecture was found. A total of 40% of the tumor population was of rhabdomyoblastic type (Figure 1). The histological subtype could be determined by demonstration of the PAX/FKHR translocation in reverse transcription polymerase chain reaction leading to the diagnosis of alveolar RMS.
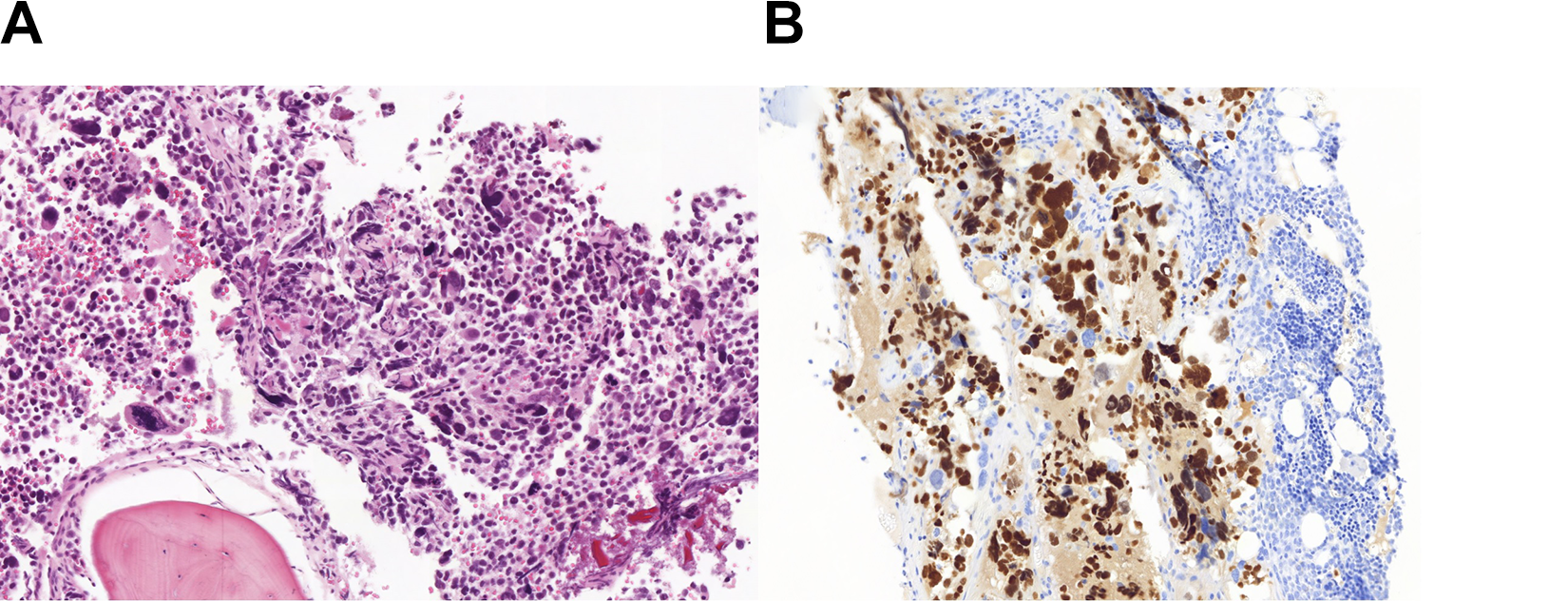
A, Tumor proliferation consisting of disjointed cells of variable shape and size (HES ×200). B, Immunohistochemistry is positive for myogenin with a strong nuclear marking (×200).
The extension workup included MRI which showed an infiltrative process in the larynx measuring 25 × 34 × 25 mm. The thoraco-abdomino-pelvic computed tomography (CT) scan did not reveal any secondary lesions. On the positron emission tomography–computed tomography (PET-CT), there was only laryngeal fixation. Finally, the bone scan did not show any metastatic lesions.
When completed, the workup allowed to classify the RMS tumor as stage IRS IIIa. Six courses of chemotherapy with Isofosfamide, Vincristin, Actinomycin, and Dactinomycin with an interval of 3 weeks between each course were started. The side effects included poor digestive tolerance and a Mallory-Weiss syndrome. At the end of the treatment, an endoscopy was performed and revealed a clear reduction in tumor volume. At the same time, an endoscopic left arytenoidectomy was performed with cold instruments and hemostasis using Yag Laser.
Anatomopathological analysis revealed, after FISH study, a rearrangement of the FKHR gene showing the persistence of tumor cells in rhabdomyoblastic maturation on the left arytenoid.
Thus, an adjuvant treatment by radiotherapy up to 50.4 Gy in 28 sessions followed by consolidation chemotherapy combining Navelbine and Endoxan for 6 months was performed. An endoscopy was performed at the end of the adjuvant treatment and biopsies were negative.
Two years later, a left supraclavicular lymphadenopathy of 15 mm in diameter was detected on the follow-up MRI, confirmed by PET-CT hyperfixation. Cytologic punction confirmed the diagnosis of recurrence of RMS. A new line of chemotherapy was administered combining Vincristine and Irinotecan in 6 courses. At the end of the treatment, a left neck dissection was carried out in territories II, III, and IV. There was 1 metastatic lymph node of 13 analyzed.
The adjuvant treatment consisted of a new course of radiotherapy using the same modalities as the first procedure. Maintenance chemotherapy with daily Endoxan and weekly Navelbine injections for a period of one year was maintained.
MRI was performed every 6 months for 7 years and didn’t show any sign of recurrence. The hoarse voice and the lack of vocal strength were improved by speech therapy. Ten years after diagnosis, the child was healthy and could lead a life without any restriction.
Discussion
To our knowledge, this is the first case of alveolar RMS in a child ever described in English literature. Even if half of the cases of RMS in children occur in head and neck, 1 laryngeal localization is extremely rare.1,4
The head and neck region has been divided into 3 subsites based on prognosis: the orbit, nonparameningeal/nonorbital region, and parameningeal sites. 6
The diagnosis of alveolar RMS of the larynx is based on anatomopathology with fibrovascular septa, “alveolar-like” spaces, and occasional giant tumor cells.2,7 In our case, microscopic analysis alone could not set the diagnosis but the FISH study showing a rearrangement of the FKHR gene led to the diagnosis of alveolar rhabdomyosarcoma (ARMS). Most ARMS express PAX3-FKHR or PAX7-FKHR gene fusions resulting from t(2;13) or t(1;13) translocations, respectively.2,8
A MRI was performed as part of the locoregional extension assessment. In our case, the tumor showed a high signal on T2-weighted images while isointense on T1-weighted images in accordance with the aspects described in the literature. 4 Furthermore, a clear enhancement was seen after administration of gadolinium (Figure 2). However, these features are nonspecific and neither MRI nor CT scans are able to differentiate RMS from other benign or malignant soft tissue tumors underlining the importance of including RMS as a possible entity in the differential diagnosis of laryngeal pediatric tumor with nonspecific MRI characteristics. 4
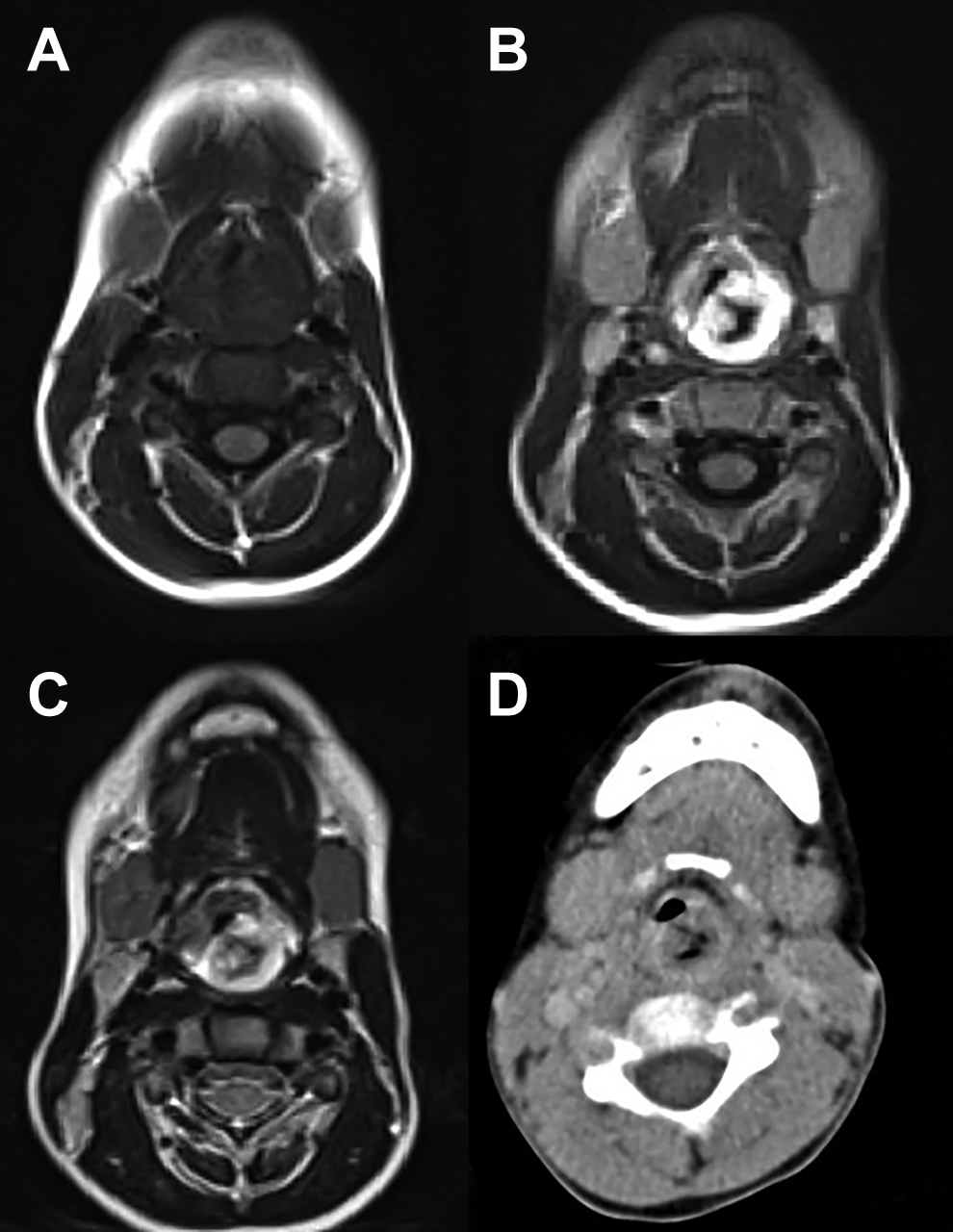
Infiltrative process of the larynx on MRI and CT-TDM (A) MRI T1-weighted: isointense (B) MRI T1-wieghted: strong enhancement after administration of gadolinium (C) MRI T2-weighted: hyperintense (D) CT scan: nonspecific. CT indicates computed tomography.
A PET-CT has to be performed to look for metastases to regional lymph nodes, lungs, and brain. In our case, the first treatment was based on chemotherapy combining isofosfamide, vincristine, actinomycine, dactinomycine. The aim was to reduce the tumor volume in order to perform the least disruptive surgical excision possible. For many decades, radical surgery has no longer a place in treatment, and chemotherapy and radiotherapy are favorited.
Based on the characteristics of resecability and extend of the disease, the Intergroup Rhabdomyosarcoma Study Group proposed a classification into 4 groups
9
: Localized disease completely resected Grossly resected tumor with microscopic residual disease Incomplete resection or biopsy only with gross residual disease Metastatic disease present at onset
Currently, it is estimated that 80% of children treated for RMS will be cured of their disease. About one-third of pediatric RMSs involve the head and neck. It is considered to be a favorable in terms of outcome and survival. 2 However, the alveolar subtype is reputed to be of poorer prognosis. 5
The specificity of our case resides in the anatomopathological description as well as the long-term survival, in spite of quite early recurrences, in a child who has undergone laryngeal preserving surgery.
Conclusion
Laryngeal RMS is very rare in pediatric population, and the alveolar subtype has not been described so far in this location. Symptoms are nonspecific and should motivate an endoscopy and biopsies for diagnostic purposes. Treatment is currently based on conservative surgery, chemotherapy and radiotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
