Abstract
Objectives:
Celiac disease is a common multisystemic autoimmune disorder. It is now increasingly recognized that it may present with extraintestinal manifestations which contribute to the difficulty in its diagnosis. The objective of this scholarly review was to examine the extraintestinal ENT manifestations of celiac disease and its pathophysiology and management, in order to highlight that some patients with celiac disease may present initially to the otolaryngologist. Improving awareness of celiac disease among otolaryngologists may aid in the correct diagnosis and correct management plan.
Methods:
A literature review was conducted using the PubMed database to identify original articles related to celiac disease and ENT manifestations between the years 2000 and 2020. The search was performed using the search string: (“coeliac disease” OR “celiac disease”) AND (“ENT manifestations” OR “hearing loss” OR “epistaxis” OR “nasal septal perforation” OR “obstructive sleep apnoea” OR “vertigo” OR “tonsillitis” OR “sinusitis”). Only articles written in English were reviewed.
Results:
A total of 17 papers met the inclusion criteria. Extraintestinal ENT manifestations of celiac disease include sensorineural hearing loss, obstructive sleep apnea, nasal septal perforation, epistaxis, and vertigo with nystagmus. Sensorineural hearing loss, obstructive sleep apnea, nasal septal perforation, vertigo, and nystagmus are thought to result from immunologically mediated mechanisms, with intestinal malabsorption resulting in epistaxis.
Conclusions:
Celiac disease can cause extraintestinal ENT manifestations and requires a high index of suspicion from the otolaryngologist to diagnose and suitably manage. A gluten-free diet may result in sufficient symptom resolution for most manifestations. Sensorineural hearing loss due to celiac disease appears to be progressive and permanent and may require frequent audiological monitoring.
Keywords
Introduction
Celiac disease (CD) is a chronic immune-mediated enteropathy of the small intestine, triggered by ingestion of gluten in genetically predisposed individuals. 1 It is an autoimmune condition, not an allergy or sensitivity—which makes it distinctly different from gluten intolerance. Celiac disease is common, with international studies suggesting a worldwide prevalence of 1.4%. 2 Although the gastrointestinal symptoms of CD such as diarrhea, abdominal pain, bloating, and vomiting are well known, many patients have extraintestinal symptoms such as arthritis, dermatitis herpetiformis, and anemia. 3 Less than 50% of patients present with the classical gastrointestinal symptoms, 4 which presents a challenge to diagnosis and may be a contributory factor toward as much as 8 in 9 cases remaining undiagnosed. 5 Obtaining a diagnosis is important, as a gluten-free diet (GFD) is therapeutic and evidence suggests that it improves patients’ quality of life and outcomes. 6
It follows then that some of the extraintestinal manifestations of CD may be ENT pathologies and therefore may present to the otolaryngologist initially. As such, it is vital that the otolaryngologist is aware of CD and harbors the diagnosis in mind when coming across such patients. Such ENT manifestations have been reported but not reviewed in the literature prior to this; therefore, the purpose of this present scholarly review is to identify exactly what the ENT extraintestinal manifestations of CD are, and what management options are feasible.
Materials and Methods
A thorough review of the literature was performed using PubMed, to identify original articles (case–control studies, cohort studies, case series, case reports, or literature reviews) about ENT manifestations of CD between the years 2000 and 2020. The search was performed using the search string: (“coeliac disease” OR “celiac disease”) AND (“ENT manifestations” OR “hearing loss” OR “epistaxis” OR “nasal septal perforation” OR “obstructive sleep apnoea” OR “vertigo” OR “tonsillitis” OR “sinusitis”). Only articles written in English were included.
Results
The initial search string yielded 52 citations. Five articles were not in English and were therefore excluded, leaving 47 citations. Of these, 5 citations explored non-ENT manifestations of CD and hence were excluded. Twenty-five citations did not explore extraintestinal manifestations of CD at all and were excluded. Excluding these 30 citations left 17 citations, which form the basis of this review. This is summarized in Figure 1.
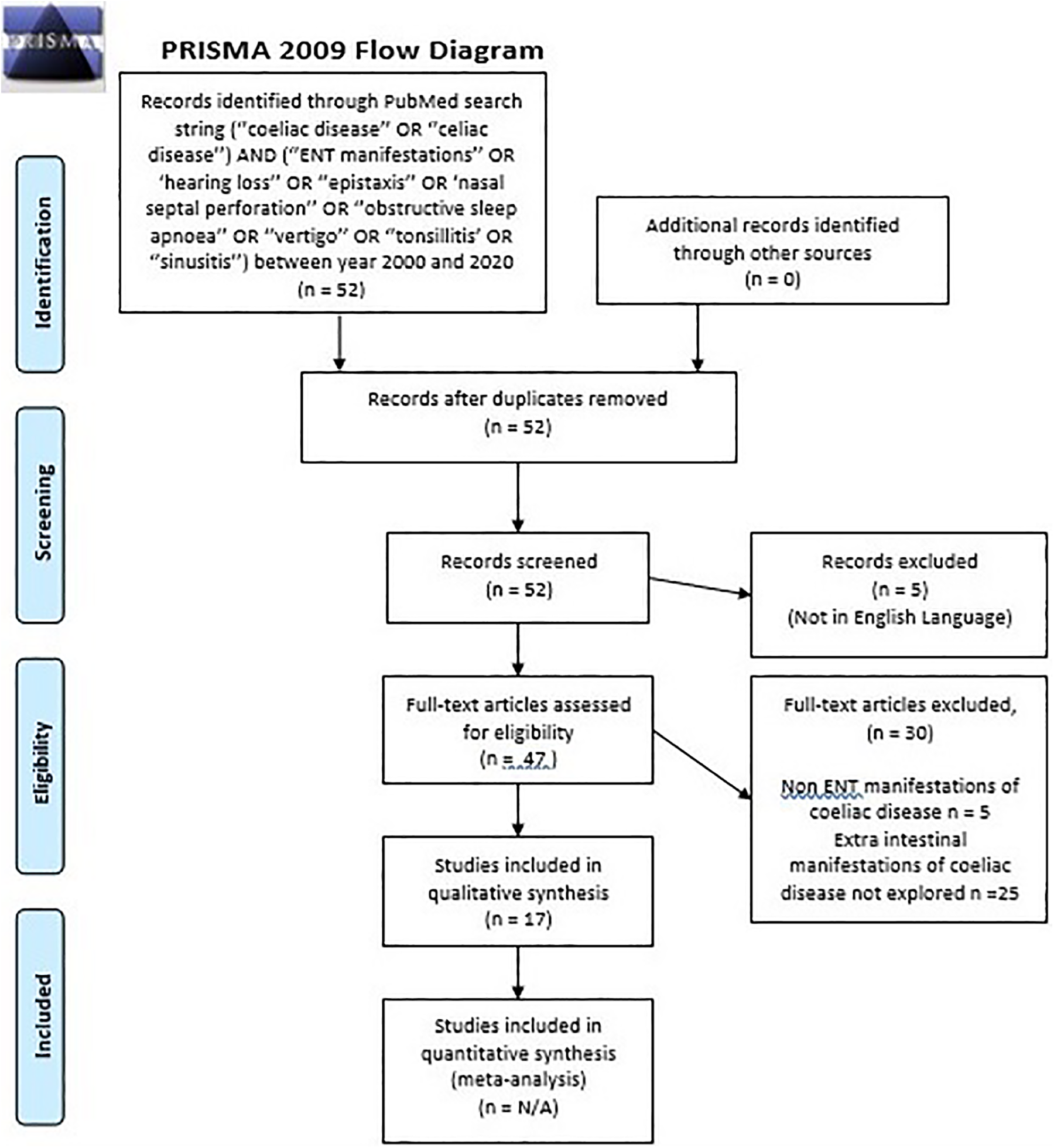
PRISMA flowchart.
Ten articles were case–control studies about sensorineural hearing loss (SNHL) and 2 articles were about sleep-disordered breathing (SDB) and obstructive sleep apnea (OSA). One article was a case report and literature review on hemorrhagic events in CD. One article was a case report on nasal septal perforation and one article was a case report on vertigo with nystagmus. Two articles explored proposed mechanisms behind extraintestinal ENT manifestations.
Discussion
Hearing Loss
A possible link between SNHL in adults with CD was first reported in 2007 by Leggio et al. 7 This prospective case control study of 48 age, sex, and lifestyle-matched adults with and without CD showed a significantly higher prevalence of mild to moderate SNHL in the adults in the CD group (P = .01). 7 This prevalence of SNHL in GFD treated and untreated individuals within the CD group was not significantly different, which suggests that the hearing impairment may be permanent. 7
In 2011, Hizli et al 8 looked at 32 pediatric patients with CD against 32 age and sex-matched controls without CD and also found a significantly higher prevalence (40.6%) of mild low frequency (250 and 500 Hz) SNHL in those with CD (P = .0001). 8 The study also found a significantly worse speech discrimination ability in the patients with CD, compared to the controls (P < .05). 8 Importantly however, none of the children recruited in this study reported any noticeable clinical hearing impairment, suggesting that the development of hearing loss may be insidious.
The findings of Hizli et al 8 are supported by a 2012 study by Solmaz et al 9 which also drew the same conclusions when comparing 25 pediatric patients with CD against 25 children without CD and finding that the patients with CD had significantly worse hearing at all frequencies (P < .05) and reduced speech discrimination abilities (P < .05). A further case–control study 10 also found a significant association between CD and SNHL at 250 Hz but also demonstrated a reduction in the amplitude of otoacoustic emissions (OAE) at 1000 Hz. As OAE is a more objective measurement of hearing loss than pure tone audiometry, this may represent stronger evidence that CD negatively impacts cochlear functionality. 10
A 2015 case–control study by Şahin et al, with large sample size (110 pediatric patients with CD and 41 age and sex-matched controls), found significantly worse bone conduction thresholds at all frequencies in patients with CD (P < .05) and that these thresholds worsen with duration of CD (P < .05). 11 Interestingly, the pure tone average was below 20 decibels, so the hearing loss was subclinical only at that point in time 11 but could be hypothesized to be predictive of worsening hearing loss in later life, in keeping with the findings of Leggio et al. 7
The pathological mechanism behind SNHL and CD is postulated to be immune-mediated neurological damage, comprising of autoantibodies, autoreactive T cells and immune complex deposition. 12 Malnutrition may also be a component, with ischemic damage to the cochlea from iron deficiency anemia. 12 Another mechanism may be due to patients with CD having neutralizing autoantibodies against osteoprotegerin (OPG), which is a key regular in bone remodeling as well as axonal myelination. 13 Loss of OPG is shown to result in sensorineural and conductive hearing loss in animal studies. 13 Another article suggested that hearing loss related to CD may be due to the suppressive interaction of gliadin peptide (present in gluten) on the human GRINA protein, which prevents normal functionality of glutamate receptor ion channels, thereby altering normal cell signaling. 14 This mechanism may also be the reason behind other extraintestinal manifestations of CD such as depression, ataxia, reproductive difficulty, and skin rash. 14
However, there are studies that dispute the relationship between SNHL and CD, demonstrating no significant difference between the prevalence of SNHL between adult and pediatric patients with CD versus age and sex-matched controls. 15 This study by Volta et al also tested for antineuronal antibodies in the patients with CD and the controls and did not find any correlation between their presence and hearing loss. 15 This conclusion is mirrored by Bükülmez et al 16 with a study with large sample size (97 children with CD and 85 controls) that showed no significant difference in pure tone audiometry, tympanometry, speech audiometry, or OAE between patients with CD and controls. A further 3 studies,17-19 similarly designed, also drew the same conclusions that SNHL and CD show no association, with one of those studies demonstrating objective evidence of no difference by using auditory brainstem responses. 19
Limitations of the above studies are the small sample sizes, different ages of participants, differing methods by which hearing was tested, insufficient long-term follow-up and lack of emphasis on or evaluation of lifestyle or social effects of the hearing impairment. This topic clearly needs more study and research, but despite the conflicting evidence, it should be noted that the study with the largest sample size (11) showed statistically significant positive association between CD and SNHL. Therefore, this association is an important learning point for the otolaryngologist. Hearing loss is well known to lead to negative effects on cognitive and social development in children 11 and therefore it is a suggestion that children with CD should have baseline hearing tests upon diagnosis, with regular hearing tests at appropriate frequencies to monitor for hearing loss with intervention as required.
Obstructive Sleep Apnea
Obstructive sleep apnea is a disorder of the upper respiratory tract characterized by repetitive reductions or cessations in airflow which may result in snoring, sleep disruption, and observable apneic episodes. 20 In children, adenotonsillar hypertrophy is a common cause, 21 hence necessitating the involvement of an otolaryngologist.
In the general pediatric population, the prevalence of OSA is estimated to be 1.2% to 5.7%. 22 In a prospective cohort study by Parisi et al in 2015, 23 the prevalence of OSA symptoms (as estimated by validated questionnaire) in a cohort of 19 children with newly diagnosed CD was 31.6% 23 —suggesting that children with CD may have a higher prevalence of OSA. Interestingly, all the children showed complete resolution of OSA symptoms after 6 months of strict GFD, suggesting that the GFD may be associated with symptom resolution. 23 Some limitations of this study were the small sample size, lack of a control group, short follow-up time, and lack of statistical analysis. It was also unclear what the cause of the OSA may have been (only 1 child was reported to have tonsillar hypertrophy 23 and the adenoids appear not to have been examined) and whether it was due to an otolaryngological etiology (as opposed to craniofacial or dental). This study also used electroencephalogram (EEG) to check for neuronal hyperexcitability pre and post GFD. However, there was no analysis of EEG markers of sleep quality which would have strengthened evidence of underlying OSA and allowed determination of the period of time it took for GFD to have a positive effect on sleep quality. A study Lucey et al concluded that EEG can be a useful tool for both clinical and research purposes in measuring sleep changes over time, 24 and another study supported the idea that modern EEG methods provides more effective, reproducible, accurate scoring in comparison to conventional sleep scoring methods. 25
A 2018 prospective case-controlled study by Yerushalmy-Feler et al 26 reinforced this possible association between CD and improvement in pediatric OSA symptoms with GFD. Thirty-four children with CD, prior to starting a GFD, undertook a questionnaire and were screened for OSA symptoms. 26 Those testing positive for OSA symptoms were then started on a GFD and compared against an age and sex-matched cohort of 24 children without CD for reassessment of OSA symptoms after 6 months. 26 Interestingly, this study showed that the incidence of OSA symptoms was higher in the control group than the CD group, 26 which is in contrast to the previous study. 23 The study found that all the children with CD and OSA symptoms had complete symptom resolution after 6 months of GFD and that this was statistically significant (P = .014). 26
Some of the limitations of this study, as similar to the previous study, were that there was no assessment for adenotonsillar hypertrophy, small sample size, and short follow-up time. A significant limitation of both studies was that the diagnosis of OSA was based on the use of a questionnaire rather than objective evidence by undertaking a sleep study. The questionnaires used in the 2 studies were also different, which limits comparability between the studies.
A possible proposed mechanism behind OSA in patients with CD and improvement with GFD is improvement of lymphatic hyperplasia that is associated with CD and contributes to OSA. 26 These studies show important learning points for the otolaryngologist. If taking the results of the first study which suggests that the prevalence of OSA is higher in children with CD, 23 it may be that the otolaryngologist may end up seeing proportionally more CD children in their sleep disorders clinics. Even if this is not the case, the evidence suggests that trialing a GFD may be a viable and safer option in children with CD, without resorting to adenotonsillectomy which can carry surgical morbidity. 27
Nasal Septal Perforation and Epistaxis
Celiac disease has also been linked to nasal septal perforation. A case study 28 reported how a patient with a large septal perforation that was further increasing in size and with intermittent epistaxis, completely diminished in activity with the commencement of a GFD. The patient was initially screened for causes of septal perforation and was unsuccessfully treated with glucocorticoids when the tests were negative. Histology showed a thick band of superficial fibrinoid necrosis and blood vessels whose lumen were partially or totally blocked, pointing to an ischemic etiology. Coincidentally, she was diagnosed with CD during the same time period and therefore commenced a GFD. This led to cessation of growth and bleeding of the septal perforation, with no further issues on follow-up over the next 5 years. This is good evidence that the activity of the septal perforation improved because of the GFD, as no previous treatments had made any difference. This is an important learning point for otolaryngologists, as although it is widely known that there exists a link between autoimmune disease and nasal septal perforation, 29 CD is not part of the initial screening investigations. Therefore, it may be helpful to add a celiac screen as part of the screening investigations should other investigations be negative, as a GFD can be a simple and effective way to symptom resolution.
A 2018 literature review on hemorrhagic presentation of CD showed that epistaxis may be a manifestation. 30 This review cited the article on the bleeding nasal septal perforation 28 and a further 3 case reports31-33 of people with bleeding diatheses, including life-threatening epistaxis, related to vitamin K and vitamin D malabsorption due to CD causing defects in the intrinsic and extrinsic coagulation pathways.32 Coagulopathy was reversible with GFD. 32
Vertigo and Nystagmus
A case reported how an 11-year-old child presented with vertigo and nystagmus related to an underlying diagnosis of CD. 34 Upon initial presentation, the child was examined and found not to have any other abnormal neurological findings, with normal cerebellar examination and ocular motility and normal imaging. 34 She was nauseated, which pointed to a peripheral rather than central pathology. She was diagnosed with CD during this time and her vertigo symptoms resolved with GFD, only to return during a period of dietary noncompliance and resolve again with strict dietary compliance. 34 The proposed mechanism is immunogenic neurological damage; however, the nystagmus symptoms showed no improvement after 2 years of GFD, suggesting that some damage must be permanent. 34 The important learning point for otolaryngologist is to consider a celiac screen in vertigo patients with no clear underlying cause and appreciate that this can lead to dramatic symptom resolution but that delay in diagnosis may cause permanent neurological injury.
Conclusions
Celiac disease is a multisystemic autoimmune disorder, with extraintestinal manifestations which may be the first presentation of the disease. Extraintestinal ENT manifestations may result in a patient with CD presenting to the otolaryngologist as a first presentation of their disease. Based upon the evidence from the body of current literature, one of the conclusions is that children with CD may develop insidious SNHL which may manifest itself as a mild to moderate hearing loss in adulthood. The otolaryngologist should recognize that children with CD may therefore require audiological assessment and monitoring which may allow identification of hearing loss and appropriate patient support early. Another finding is that SDB and OSA symptoms in children with CD improve with GFD. This is a point to consider for the otolaryngologist, as a trial with strict GFD may be an initial treatment modality for these children, rather than surgery. It is also striking that CD can cause such a variety of symptoms from nasal septal perforation to vertigo, the learning points being to consider an underlying diagnosis of CD in patients whose symptoms are not explainable by other diagnoses and that a GFD may be curative of their symptoms. Finally, CD-related malabsorption can lead to coagulopathy and bleeding diathesis which can present as epistaxis.
The main limitation with this present review is the small body of literature to go by. Within the articles regarding SNHL, there is much variation in sample size, population demographic and variables measured—all of which limits comparability of the articles’ conclusions. Further to this, a significant limitation of the SDB/OSA articles is that SDB/OSA was diagnosed using screening questionnaires (which were different between the studies), rather than polysomnography. Therefore, it is possible that the children in the studies may not have had any true clinical symptoms. As such, it follows that it is unclear whether a GFD truly results in improvements in sleep quality. The case reports on nasal septal perforation and vertigo are compelling, but it are difficult to draw conclusions from without further supportive evidence. This is clearly a suitable area for further active research because it is very apparent that CD can cause a range of extraintestinal manifestations and recognition and diagnosis of these patients and instigation of GFD may be the key for improved patient care.
Supplemental Material
Supplemental Material, PRISMA_2009_checklist - ENT Manifestations of Celiac Disease: A Scholarly Review
Supplemental Material, PRISMA_2009_checklist for ENT Manifestations of Celiac Disease: A Scholarly Review by Dilhara Karunaratne and Nisal Karunaratne in Ear, Nose & Throat Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
