Abstract
Background:
Adenoid cystic carcinoma (AdCC) of the larynx is an uncommon malignancy of the head and neck with very little literature discussing treatment paradigms and prognostic factors influencing survival.
Methods:
This retrospective cohort study uses data obtained from the Surveillance, Epidemiology and End Result database comprising of patients diagnosed with laryngeal AdCC from 1978 to 2016.
Results:
A total of 89 records were analyzed. High histologic grade was a significant negative prognostic factor compared to low histologic grade disease for overall survival (OS; 5-year OS: 35.7% vs 90.5%, P < .005) and disease-specific survival (DSS; 5-year DSS: 38.7% vs 95.2%, P < .005). No differences in OS (5-year OS: 88.9% vs 76.4%, P = .287) or DSS (5-year DSS: 100% vs 79.1%, P = .159) were noted between patients with early versus late American Joint Committee on Cancer (AJCC) stage disease. No differences in DSS was noted in cohorts treated with just definitive surgery versus surgery and adjuvant radiation (5-year DSS: 92.9% vs 74.3%, P = .140) even when stratified for late stage disease (5-year DSS: 100% vs 78.6%, P = .290) or high-grade histology (5-year DSS: 100% vs 83.3%, P = .773).
Conclusions:
Histologic grade may be a more significant prognostic factor than AJCC stage for survival in laryngeal AdCC. Treatment with surgery and radiation may provide no additional survival advantage compared to just definitive surgery in these patients, although further study is warranted.
Introduction
Laryngeal carcinoma represents the second most common malignancy of the head and neck and comprises approximately 1.1% of all malignancies. 1 Laryngeal malignancies are separated into 2 broad categories, squamous cell carcinoma (SCC) and non-SCC, with only a minority (10%) of cases belonging to the non-SCC category. 2 One such group includes salivary gland tumors of the larynx.
Salivary gland malignancies of the larynx will often present similarly to other laryngeal malignancies, with symptoms including stridor, hemoptysis, odynophagia, and hoarseness. Although it can be difficult to distinguish laryngeal salivary tumors from SCC by gross appearance, these tumors are drastically different histologically. An important subtype of salivary gland tumor classified by histology is adenoid cystic carcinoma (AdCC). Characterized by slow growth kinetics and perineural invasion, it is one of the most common histologic types of salivary malignancy, comprising 34% to 69% of primary salivary tumors.3-5 Some reports in the literature indicate that these tumors will present in more advanced stages than other laryngeal malignancies.6-8
Although AdCC has been reported to present in later stages, the current literature is lacking regarding incidence, outcomes, and survival of AdCC as it relates to the larynx. The goal of this study is to provide a comprehensive analysis and review of the Surveillance, Epidemiology and End Result (SEER) database regarding laryngeal AdCC salivary gland malignancies.
Methods
Data Collection
Data regarding patient demographics, incidence, tumor characteristics, treatment, and survival were collected from the SEER database 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases Data 1975 to 2017 program of the National Cancer Institute (SEER). The database was queried for all individuals diagnosed with primary laryngeal carcinoma, as defined by the International Classification of Diseases for Oncology, Third Edition (ICD-0-3) codes C32.0-C3.3, C32.8, and C32.9. Only AdCC (8200/3) histology was included for this study (ICD). As this was a publicly accessible database, institutional review board approval was not required for this study.
Variable Definitions
Demographic data included sex, race, age at diagnosis, and state of residence. Tumor characteristics include subsite (supraglottis, glottis, subglottis, larynx not otherwise specified (NOS), laryngeal cartilage, or overlapping lesion of larynx), laterality, grade, American Joint Committee on Cancer (AJCC) Seventh edition derived stage, AJCC Seventh edition T stage, tumor size (mm), AJCC Seventh edition N stage, AJCC Seventh edition M stage, and tumor extension. Treatment variables include surgery, radiation sequence, and chemotherapy. Survival data include cause-specific death (alive, dead of cause attributable to this cancer diagnosis, dead of other cause, or dead of cause attributable to another cancer diagnosis) as well as survival time (months).
Year of diagnosis was divided into 2 groups, <2003 and >2004, based on prior studies examining survival outcomes in laryngeal cancer. 7 Likewise, for statistical analysis, AJCC stage was divided into 2 categories, patients with early stage disease, defined as overall stage I and stage II, and late stage disease, defined as overall stage III and IV. Histologic grade was similarly divided into low-grade disease (I and II) and high-grade disease (III and IV). The SEER database grading scheme defines grade I disease as well differentiated NOS, grade II as moderately differentiated, grade III as poorly differentiated, and grade IV as undifferentiated/anaplastic. For this data set, AJCC overall stage and histologic grading was only available for 44 (49%) and 38 (43%) patients, respectively.
Statistical Analysis
Descriptive statistics were reported as frequency (%). Univariate survival analysis of potential prognostic factors was carried out through the use of Kaplan-Meier plots as well as to compare survival outcomes. Comparisons between survival curves were done through the log-rank test with a primary outcome of disease-specific survival (DSS). Overall survival (OS) and DSS were calculated for multivariate analysis. Analysis of treatment modalities was further stratified based on year of diagnosis and AJCC stage in the univariate model. Stratified survival analyses were limited to only patients who had complete data on treatment and stage. The Cox proportional hazards regression model was used for multivariate analysis of variables noted to have an α < 0.5 in univariate testing and fit the proportional hazards assumption. Covariates analyzed in the multivariate model included year of diagnosis, age of diagnosis, race, sex, subsite of primary, AJCC stage, and histologic grade. Adjusted hazard ratio (aHR) and 95% CIs were used to describe differences. For the multivariate model, right censoring occurred at the 10-year survival mark for OS and DSS. Furthermore, no exclusions were made on the basis of age. Statistical significance was determined at the α < 0.05 level. Statistical analysis was done using IBM SPSS Statistics v26.0 (IBM, Inc).
Results
Patient Characteristics and Descriptive Data
A total of 89 records were obtained from the SEER database. The mean (standard deviation) age of diagnosis was 57 (16.3) years with an age range from 21 to 88 years; 71 (79.8%) patients were Caucasian while 9 (10.1%) patients were Asian/pacific islander (Table 1). Incidence trends showed 39 (43.8%) records collected prior to 2003. There was an overall female predominance in the study population with 51 (57.3%) female patients (Table 1). Among patients with histologic grading information available (n = 38, 43%), 23 (60.5%) cases were noted to have low-grade disease and 15 (39.5%) were designated high-grade disease. Within anatomic subsites, there was an equal predominance of supraglottic and subglottic primary tumors (41.6%, respectively) with glottic subsites having the lowest primary tumor incidence of 7 (7.8%). The most commonly used treatment modality was surgery and radiation with 44 (49.4%) patients. Surgery alone was the next most common treatment modality with 19 (21.3%) patients. Fourteen (15.7%) patients did not receive treatment (Table 1). Among patients with overall AJCC staging information (n = 44, 49.4%), the majority of patients presented with late stage disease (AJCC stage III and IV; 29 patients, 65.9%), while 15 (34.1%) presented with early stage disease (AJCC stage I and II; Table 1). In all, 40 (90.9%) presented with no nodal involvement and 45 (95.7%) presented with no distant metastases (Table 2). The mean time to follow-up for the cohort was 102 months (95% CI: 114.4-89.4 months).
Descriptive Statistics and Frequencies of Study Population.
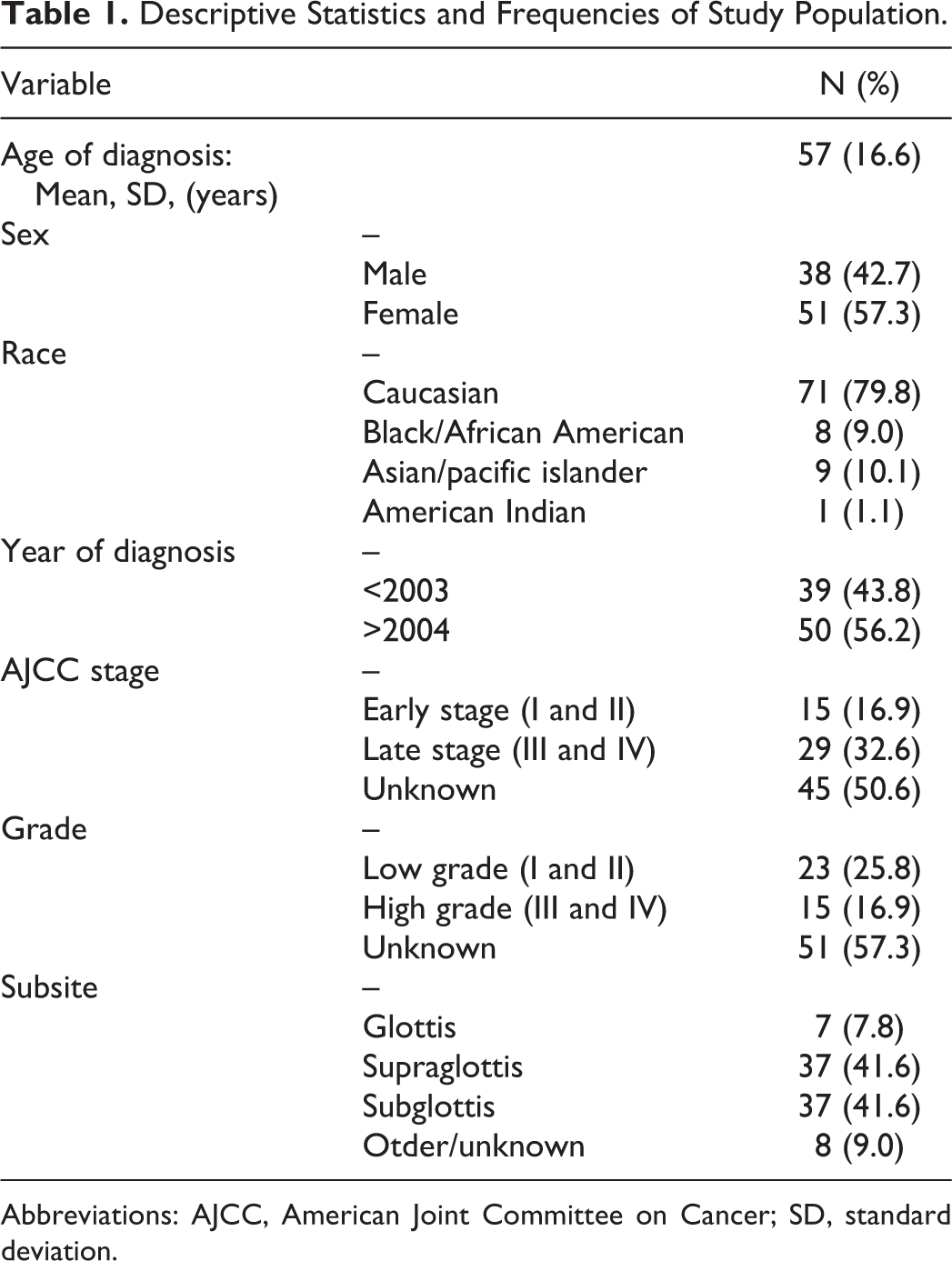
Abbreviations: AJCC, American Joint Committee on Cancer; SD, standard deviation.
Laryngeal Adenoid Cystic Carcinoma Tumor Characteristics and Surgical Treatment.
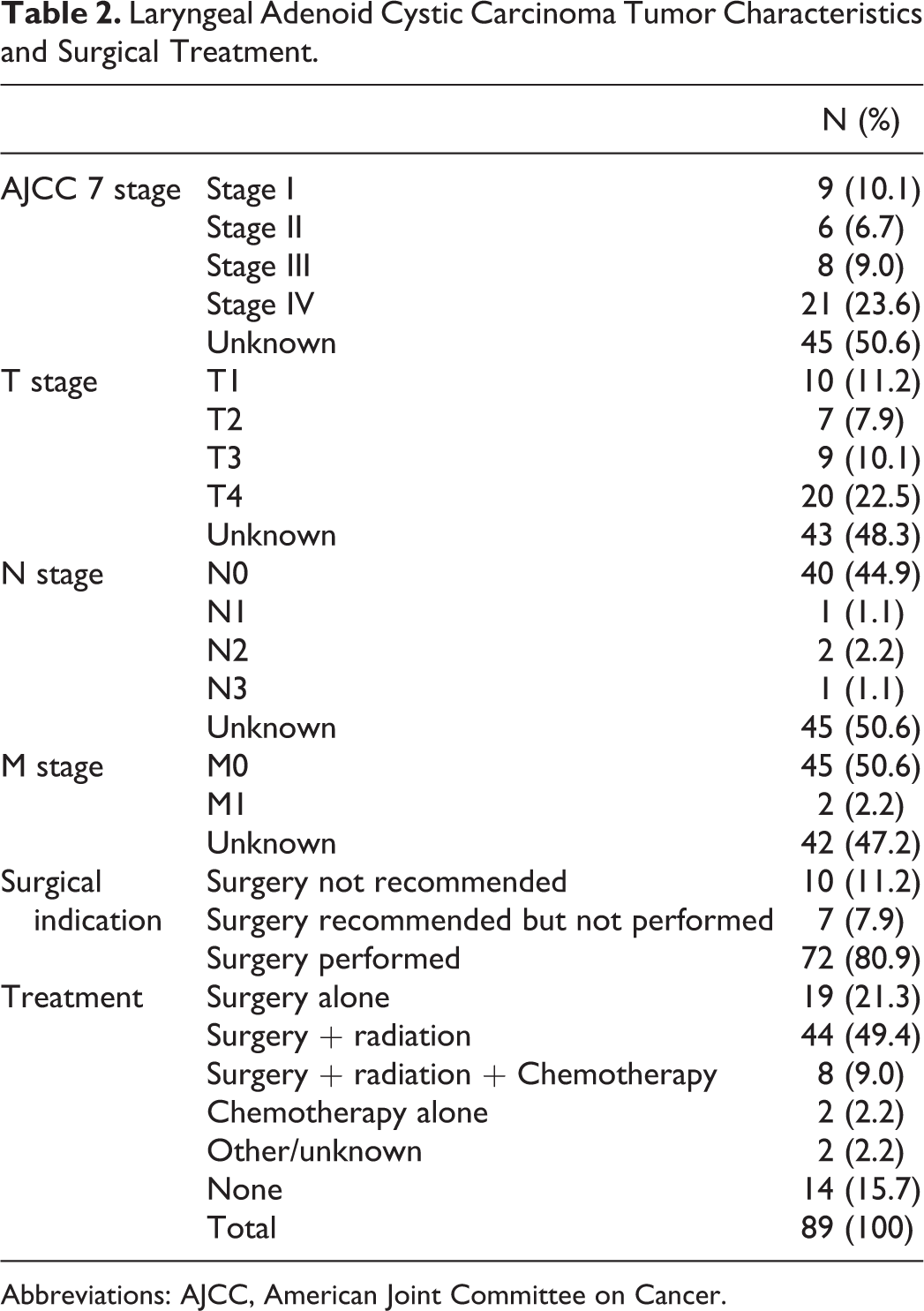
Abbreviations: AJCC, American Joint Committee on Cancer.
Univariate Testing
In Kaplan-Meier survival analysis, the mean DSS was 238 months, 5- and 10-year DSS of 77.3% and 67.9%, respectively, and the mean OS was 176 months, 5- and 10-year OS of 72.6% and 61.2%, respectively. Low-grade disease was associated with better DSS compared to high-grade disease with a 5-year DSS of 95.2% versus 38.7% for high-grade disease (P < .005; Figure 1). No significant difference in DSS was noted when stratified for overall AJCC stage disease (P = .159; Figure 2). In terms of treatment modality, surgery alone had a 5-year DSS of 92.9%. Multimodality treatment with surgery and radiation had a 5-year DSS of 74.3%. Patients who did not receive treatment had a 5-year DSS of 67.3%. There was no significant difference in survival between surgery alone and surgery and radiation (P = .172), although selection bias for surgery and radiation may play a role in skewing the data (Figure 3). Disease-specific survival did not significantly differ between surgery and surgery + radiation even when stratified for early stage disease (P = .32), late stage disease (P = .29), or high-grade histology (P = .77; Figure 4). No significant difference in DSS was noted between cases reported from 1978 to 2003 and cases reported from 2004 to 2016 (P = .847).
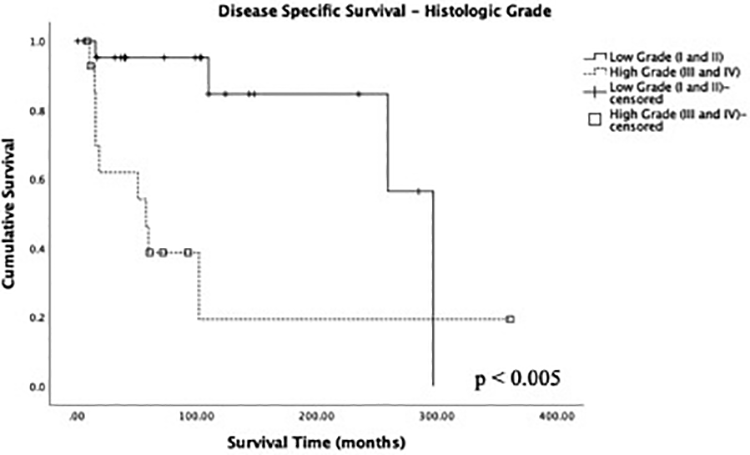
Kaplan-Meier plot of disease-specific survival for low- and high-histologic grade disease (n = 38). P value denoted through log-rank testing with significance at the P < .05 level.
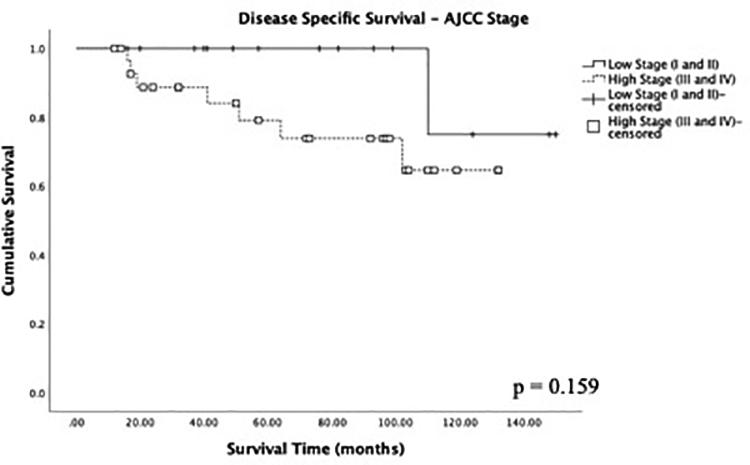
Kaplan-Meier plot of disease-specific survival for low- and high-AJCC stage disease (n = 44). P value denoted through log-rank testing with significance at the P < .05 level.
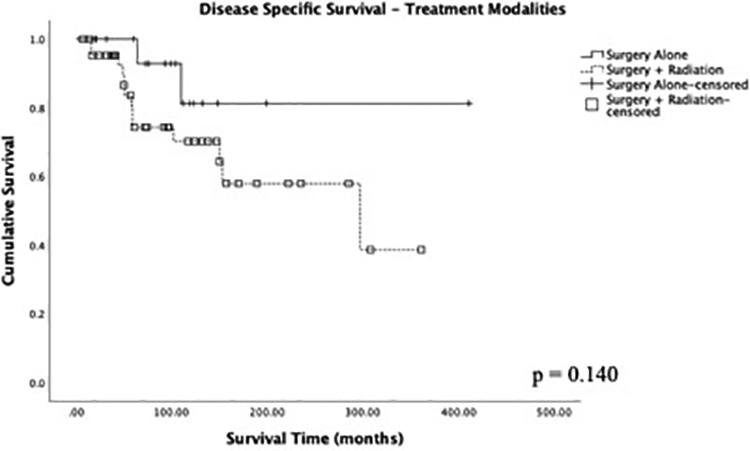
Kaplan-Meier plot of disease-specific survival for surgical monotherapy versus combined therapy for surgery and radiation (n = 63). P value denoted through log-rank testing with significance at the P < .05 level.
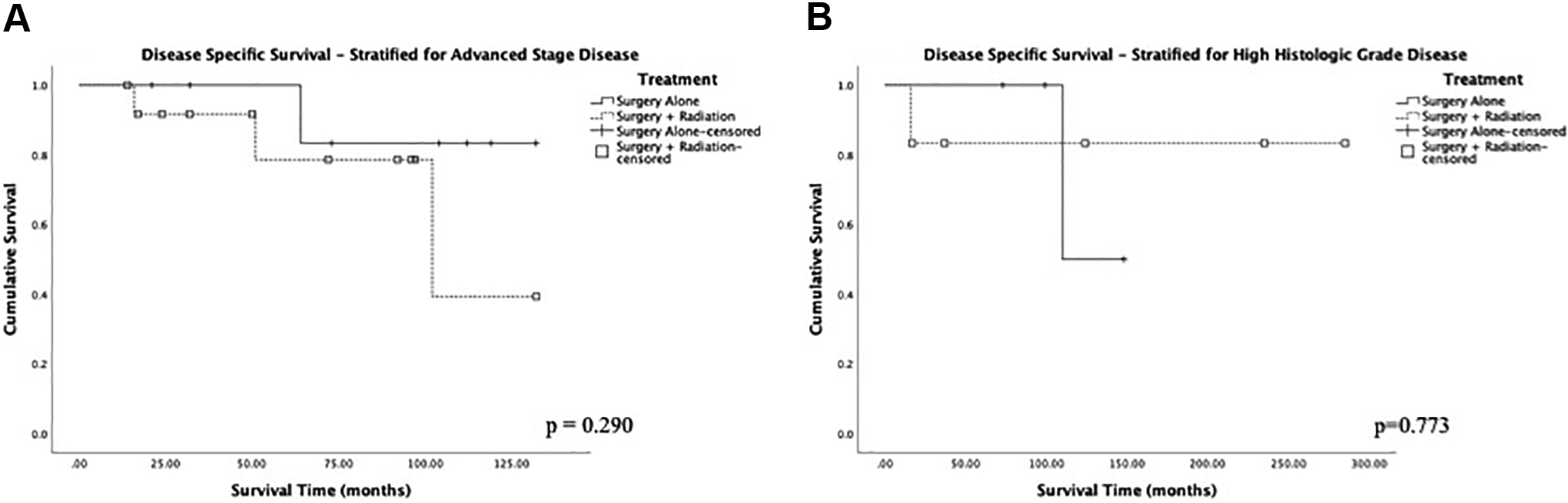
Kaplan-Meier plot of disease-specific survival for surgical monotherapy versus combined therapy for surgery and radiation stratified for (A) advanced stage disease (n = 21) and (B) high-grade histology (n = 10). P value denoted through log-rank testing with significance at the P < .05 level.
Multivariate Testing
When adjusted for covariates in the cox regression model, age >65 years was associated with poorer OS for AdCC (aHR: 2.27, P = .050). However, older age at diagnosis was not associated with any difference in DSS (aHR: 1.65, P = .353; Table 3). Male sex was associated with worse OS and DSS in our study population (aHR: 2.75, 3.21, P < .01, P < .01, respectively; Table 3).
Subsite of primary tumor was not significantly associated with any differences in DSS (P = .43). Low-grade disease demonstrated better OS (aHR: 0.216, P = .006) and DSS (aHR: 0.2, P = .016) compared to high-grade disease. However, early AJCC stage did not impact OS (aHR: 0.583, P = .522) or DSS (aHR: 1.03, P = .101) compared to late stage disease (Table 3).
Factors Influencing Overall and Disease-Specific Survival Outcomes Between the Histologic Categories for Laryngeal Adenoid Cystic Carcinoma.
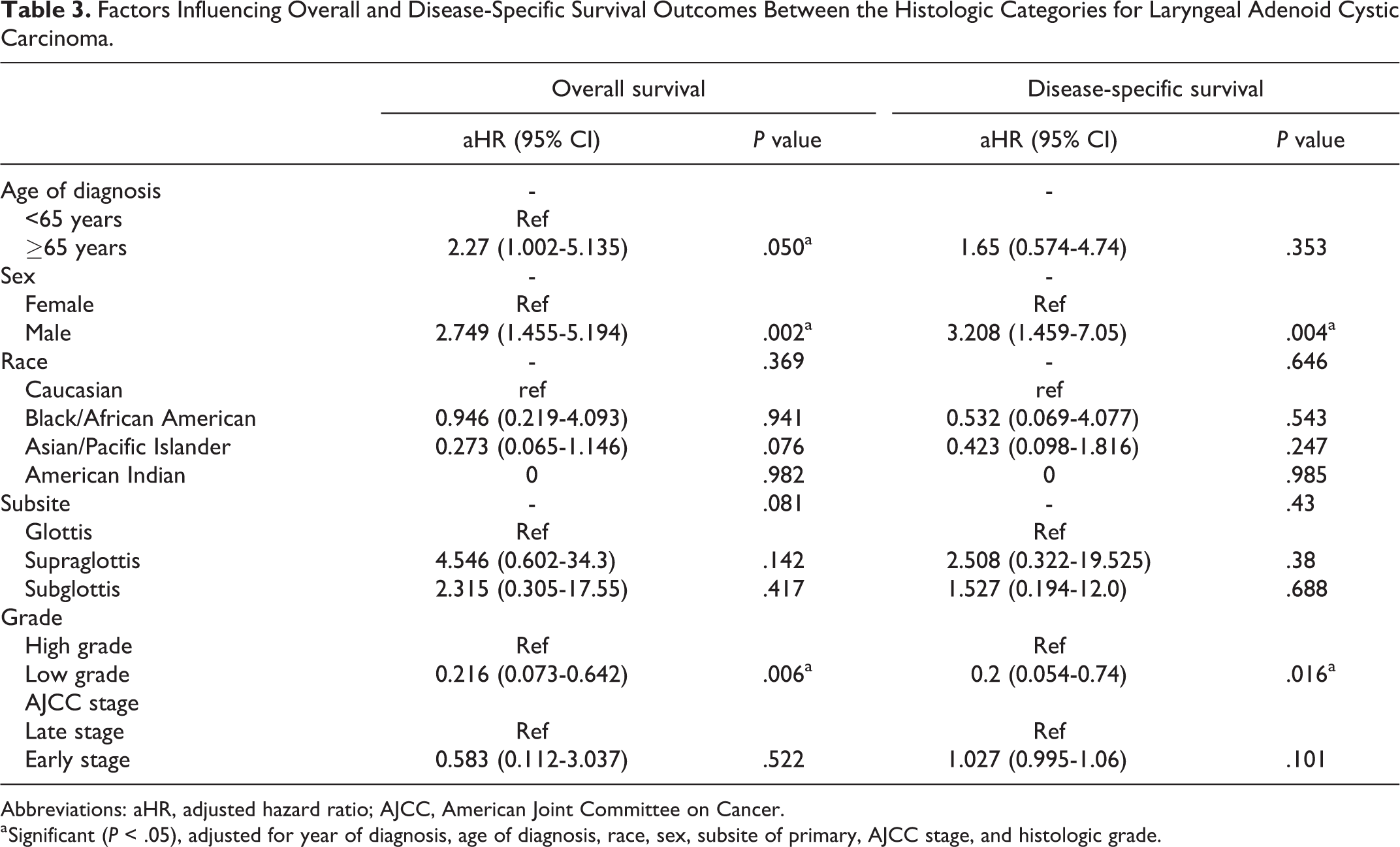
Abbreviations: aHR, adjusted hazard ratio; AJCC, American Joint Committee on Cancer.
a Significant (P < .05), adjusted for year of diagnosis, age of diagnosis, race, sex, subsite of primary, AJCC stage, and histologic grade.
Discussion
Tumors with minor salivary gland histology are rare malignancies, comprising <1% of laryngeal cancers. 6 Within these, adenoid cystic has been reported as the most common histology (32%-69%). 4 However, the literature on laryngeal AdCC remains sparse aside from isolated case reports.
Tumor Characteristics
Of the patients with listed AJCC stages and histologic grades, the majority of patients initially presented with late stage disease (32.6%) and/or low-grade histology (25.8%). As prior studies have suggested, the slow growth kinetics of AdCC could explain this phenomenon, as tumors could be clinically asymptomatic until they reach later stage disease. 9 In the larynx, salivary tumors tend to present with mass effect symptoms causing stridor, odynophagia, and hoarseness,3,4 with some reports suggesting that salivary tumors in the larynx tend to present at later stages. 4 Our study supports these findings when looking at TNM staging the majority of patients presented with T4 tumors (22.5%); however, the majority had N0 (44.9%) nodal involvement and no distant metastases (50.6%). This suggests laryngeal AdCC may be clinically indolent and become symptomatic at later stages.
Demographics
Within our study cohort, AdCC demonstrated a female predominance. Adenoid cystic carcinoma, however, was associated with a 2.7× worse OS and a 3.2× worse DSS in males than females. These differences in sex-specific survival have been noted in prior literature as it related AdCC in the major salivary glands. 10 As prior studies have postulated, this finding could be due to hormonal factors, differences in adherence to treatment regiments, and/or differences in time to presentation between the sexes. 10
Treatment
Survival outcomes for different treatment modalities for laryngeal AdCC lack definitive evidence in the literature and are mainly based on case studies.10-12 As such, there is no universally accepted standard for the optimal treatment modality for laryngeal AdCC. However, most treatment modalities involve some form of primary surgical treatment with adjuvant radiation therapy included for advanced T-stage disease, nodal involvement, or perineural/perivascular invasion.10-12 Our data suggest that surgery alone was associated with the highest OS and DSS in AdCC compared to no treatment likely due to the more benign nature of disease requiring only surgical intervention. However, compared to treatment with surgery and radiation, no significant difference in survival was noted even when stratified for early and late stage disease in the raw and adjusted model. Given the relatively small N in the data set, it is difficult to generalize our results, furthermore there is an element of selection bias in the surgery and radiation cohort, which may skew the results. However, as studies looking at AdCC in the head and neck have observed, surgery monotherapy is typically reserved for early T-stage disease with no nodal involvement.10-12 The use of surgery monotherapy for early stage disease was observed in our data set as well. The high rates of recurrence of AdCC in the larynx may play a role in the nonsignificant differences in survival between surgery and surgery + radiation even when stratified for stage.
Survival
Despite a trend toward later stage presentation, AJCC stage of disease was not associated with significant differences in survival in the univariate and multivariate models which adjusted for sex, age of diagnosis, subsite, and year of diagnosis (Table 2). Although prior studies looking at AdCC in the head and neck have suggested AJCC stage as a significant prognostic factor for survival, our data, which focuses on AdCC of the larynx, suggest that overall stage may be a less important prognostic factor. It is important to note that staging information was available for only 49.4% of patients. On the other hand, our data suggest that histologic grade is a significant prognostic factor for survival with low-grade disease being associated with improved OS and DSS compared to high-grade disease (Table 3). Prior studies looking at AdCC in the head and neck have suggested histologic grade as a prognostic factor for recurrence.13-16 However, some studies that looked at AdCC in the head and neck suggest histologic grade is not associated with worse survival outcomes in head and neck AdCC. 17 Our study contrasts these prior studies by suggesting histologic grade is a more significant prognostic factor for survival than AJCC stage specifically in laryngeal AdCC, although our analysis was limited by the relatively low proportion of patients with available AJCC staging information. Given the limited proportion of patients with recorded AJCC stages and histologic grades, it is difficult to generalize our data. However, given the overall rarity of laryngeal AdCC in the published literature, our study stands as the best evidence of prognostic trends in the literature to date.
Limitations
There are several limitations to the study. The SEER database allows for a collection of large samples for rare conditions such as laryngeal salivary malignancies and with greater external validity due to the population-based nature of the data. However, limitations exist in regard to controlling for unique, patient identifying variables that are not available in SEER. Comorbid variables such as preexisting health conditions, smoking history, socioeconomic status, completion of treatment, and follow-up after primary therapy could play a confounding role in the observed results. The SEER derived data are also limited by the large number of unknown variables present in the records, making it difficult to fully assess the impact of variables such as grading or staging. Additionally, treatment paradigms can vary between institutions and providers, and as such it is difficult to generalize survival from our data set. Further prospective or retrospective studies with detailed recordings of surgical or radiation treatments should be done to validate the results of our preliminary study.
Conclusion
Adenoid cystic carcinoma of the larynx is a rare malignancy. Although it has a female predilection, it is associated with worse OS outcomes in males. The majority of recorded patients present with AdCC in later stage disease and with low-grade histology. Our data suggest that histologic grade of laryngeal AdCC may be a more important prognostic indicator for survival than AJCC stage. Surgery with radiation is the most common treatment modality, followed by surgery alone. Both treatment modalities demonstrated increased OS and DSS compared to no treatment. There was, however, no significant difference in survival between surgical monotherapy and surgery with adjuvant radiation even when stratified for advanced stage disease and high-grade histology.
Footnotes
Authors’ Note
This manuscript does not contain any material the publication of which would violate copyright or other personal proprietary right of any person or entity. We hereby declare that the following manuscript has not been published elsewhere (in any format or language) and has not been concomitantly sent for another journal for publication consideration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
