Abstract
Objectives:
To provide an overview of recent techniques and technologies for the application of topical corticosteroid therapy immediately following endoscopic sinus surgery (ESS) for chronic rhinosinusitis (CRS).
Methods:
A comprehensive search in the PubMed and Google Scholar databases was conducted to identify publications between January 2000 and December 2019 detailing clinical trials that have evaluated the efficacy and safety of intraoperative applications of topical corticosteroids for CRS.
Results:
A total of 21 articles, all of which highlight a variety of corticosteroid-infused products, including Propel corticosteroid-eluting stents, NasoPore, Merocel, SinuBand, calcium alginate, and bioresorbable gel-type products, are included for review. Propel stents are the only devices that have achieved level 1A evidence in terms of efficacy and have data to support their safety. The remaining products have shown mixed results in terms of efficacy and safety.
Conclusion:
A wide range of techniques and technologies have been introduced to enhance the topical delivery of corticosteroids into the neosinuses after ESS for CRS. Regarding efficacy, there is level 1A evidence to support the use of Propel stents. Most of the remaining strategies show some degree of efficacy. Direct comparisons across the different strategies are limited owing to the varied uses of delivery vectors, corticosteroid choices, and doses of corticosteroids. Propel stents and SinuBand have sufficient data to support systemic and ocular safety, whereas the remaining products have limited data to support their safety.
Keywords
Introduction
The postoperative management of chronic rhinosinusitis (CRS) after endoscopic sinus surgery (ESS) has traditionally utilized corticosteroid therapies in both oral and topical formulations. Corticosteroids have been shown to facilitate early mucosalization, reduce edema, and decrease the rate of bacterial recovery, thereby improving clinical healing and thus surgical outcomes. 1 Enhanced postoperative healing of the sinonasal mucosa ultimately decreases the need for in-office procedures and operative revisions, such as lysis of adhesions, which have been reported to occur in 35% of cases. 2,3 The use of systemic and topical corticosteroids for postoperative CRS management is nonetheless met with challenges. Systemic corticosteroids are particularly associated with many undesired adverse effects including increased blood glucose, calcium demineralization, mood alteration, avascular necrosis of the femoral head, and posterior cataracts. 1 On the other hand, topical corticosteroids are conventionally delivered through intranasal-medicated sprays and are limited by low sinus penetration, especially into the frontal sinus. 4,5
Over the past 20 years, the postoperative management of CRS has increasingly evolved with various techniques and technologies to provide effective localized delivery of corticosteroids into the neosinuses after ESS. Corticosteroid-releasing implants or dressings particularly serve the dual purpose of providing sustained short-term delivery of topical corticosteroids and maintaining the patency of the neosinus. 6 They have also been found to limit scarring, stenosis, lateralization of the middle turbinate, synechia formation, and recurrent nasal polyposis by decreasing fibrin deposition, granulation tissue formation, and edema of the residual mucosa. 7,8 A wide range of devices for delivery include commercially available Propel corticosteroid-releasing implants (Intersect ENT), SinuBand corticosteroid-releasing films (Bioinspire Technologies Inc), and corticosteroid-infused NasoPore (Stryker Corporation), SinuFoam (Smith & Nephew), MeroGel (Medtronic Inc), Gelfoam (Ethicon Inc), Merocel (Medtronic Inc), and calcium alginate (Algi-Pack; MD Pharm). The purpose of this review is to provide a global overview of these different strategies to enhance topical corticosteroid treatment for CRS immediately following ESS.
Methods
A literature search in PubMed and Google Scholar was performed to identify prospective and retrospective trials between January 2000 and December 2019 regarding intraoperative corticosteroid placement following ESS. The following search phrases were used in various combinations: “chronic rhinosinusitis,” “corticosteroid placement,” “endoscopic sinus surgery,” “Propel,” “bioabsorbable steroid eluting,” “NasoPore,” “Gelfoam,” “steroid-impregnated spacer,” “Merogel,” “drug eluting,” and “Merocel.” The results were then reviewed to identify relevant clinical trials and meta-analyses that addressed intraoperative corticosteroid treatment during ESS for CRS. Clinical studies in which corticosteroid devices were placed in lieu of ESS or were placed in the office following ESS were excluded. A level of evidence was assigned according to the Oxford Centre of Evidence-based Medicine when one was not found in the original report. 9
Results
A total of 322 reports were initially screened after a comprehensive literature search. Based on the identified inclusion and exclusion criteria, 21 studies were utilized to synthesize the content of this review. Primary data were collected from 19 studies in which 5 focused on the Propel corticosteroid-releasing implant, 6 focused on corticosteroid-treated NasoPore, 1 focused on corticosteroid-treated Merogel, 2 focused on corticosteroid-treated Gelfoam, 1 focused on corticosteroid-treated SinuFoam, 2 focused on corticosteroid-treated Merocel, 1 focused on corticosteroid-releasing SinuBand, and 1 focused on corticosteroid-treated calcium alginate. Of these clinical studies, 18 were prospective trials, whereas 1 study was a retrospective review. Two systematic reviews with meta-analyses were also included in the review. Table 1 provides a summary of the 19 clinical studies, divided according to the techniques used for topical corticosteroid delivery.
Summary of Efficacy and Safety of Various Techniques for Intraoperative Topical Corticosteroid Delivery.
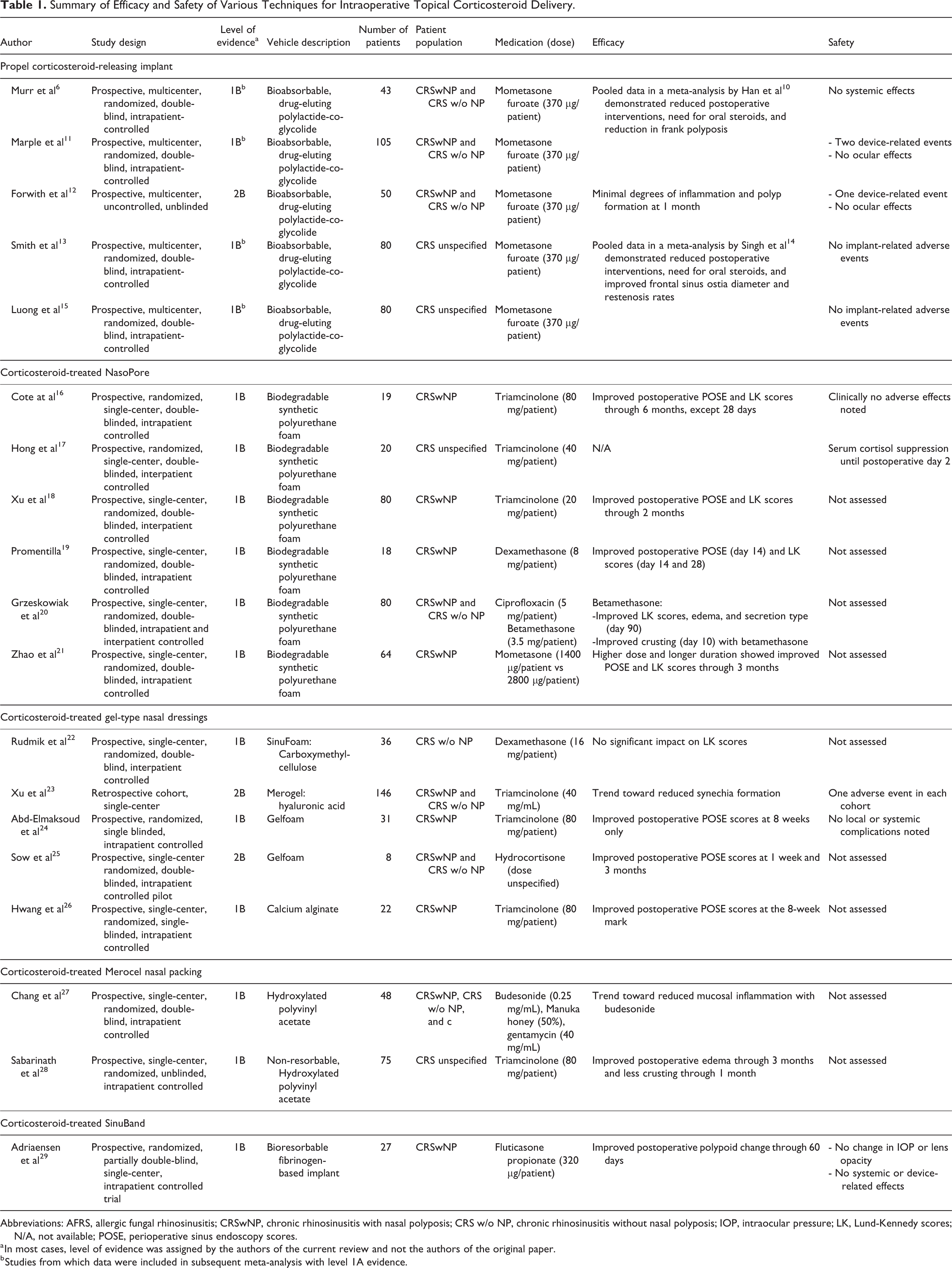
Abbreviations: AFRS, allergic fungal rhinosinusitis; CRSwNP, chronic rhinosinusitis with nasal polyposis; CRS w/o NP, chronic rhinosinusitis without nasal polyposis; IOP, intraocular pressure; LK, Lund-Kennedy scores; N/A, not available; POSE, perioperative sinus endoscopy scores.
a In most cases, level of evidence was assigned by the authors of the current review and not the authors of the original paper.
b Studies from which data were included in subsequent meta-analysis with level 1A evidence.
The following is a summary of the different technologies and strategies that have been clinically studied for the intraoperative application of topical corticosteroid therapy for CRS. For each highlighted method, special attention is paid to the delivery vector for topical corticosteroid application, clinical efficacy in improving postoperative healing, and safety profile.
Propel Corticosteroid-Releasing Implant
Characteristics
The Propel family of implants consists of biodegradable, spring-loaded, corticosteroid-releasing stents. Variations of these stents in terms of geometric design and measurements have been offered to accommodate clinical uses in the surgically opened ethmoid cavity, frontal sinus ostium, and maxillary sinus ostium. All Propel stents are dosed with 370 µg of mometasone furoate, which has been selected for its high potency and low bioavailability. 6,30 After the stents are deployed into the targeted neosinuses, they maintain a low profile to allow for the postoperative initiation of nasal saline irrigations and performance of nasal debridements. Composed of bioabsorbable polylactide-co-glycolide, Propel stents dissolve over a course of approximately 30 days following their deployment. 6,11,13,15
Evidence on Clinical Outcomes
The first prospective, multicenter, randomized, double-blind, intrapatient-controlled trials of the Propel implant in the ethmoid sinus cavity after ESS were published in 2010 and 2011. 6,11 Both studies included CRS patients with and without nasal polyposis who were scheduled to undergo primary or revision ESS for bilateral ethmoid sinus disease. Both studies were similar, although not identical, in terms of concomitant medication regimens. Murr et al 6 disallowed oral steroids in the 14 days leading up to ESS, whereas Marple et al 11 had no such restriction. Both studies disallowed oral steroids for the first 30 postoperative days. All patients received a 14-day course of perioperative antibiotics. The clinical outcomes from the 148 patients from both studies are highlighted in a meta-analysis by Han et al. 10 These pooled data show that by postoperative day 30, the implant was associated with a 70% reduction in adhesion formation and a 75% relative reduction in middle turbinate lateralization. Furthermore, they noted a significant reduction in the need for postoperative interventions by 35%, lysis of adhesions by 51%, and oral steroid prescriptions by 40%. Finally, there was a relative reduction in frank polyposis by 46%.
In terms of safety, there were no device-related complications reported by Murr et al, 6 whereas Marple et al 11 reported 2 adverse events, one with adhesions and the other with an infection. Ocular safety was further assessed by intraocular pressure and change in lens opacity, and no statistically significant changes were noted. 11 Ocular safety of the implant was further demonstrated in a separate study in which the implant was placed bilaterally. 12 Murr et al 6 showed in a small cohort of their patients that plasma mometasone levels were undetectable and that mean cortisol concentrations were not changed. These results indicate an absence of adrenal suppression from the corticosteroid eluted from the implant.
Subsequent to the Propel implant for the ethmoid sinus cavity, the Propel Mini and Propel Contour variants were produced to incorporate shorter lengths and wider hourglass shapes to enhance the placement of the corticosteroid-releasing stents into the frontal sinus and maxillary sinus neo-ostia. These implants were evaluated in 2 prospective multicenter, double-blind, randomized, controlled trials published in 2016 and 2017. 13,15 These studies utilized an intrapatient design, in which all enrolled patients with bilateral CRS received a steroid-eluting stent on one side, but no stent on the contralateral side. A meta-analysis by Singh et al 14 of these frontal sinus stents shows that at postoperative day 30, there was a significant reduction in the need for postoperative interventions by 46.8%, surgical intervention by 51.2%, and oral steroids by 30.2%. At postoperative day 90, there was a significant relative reduction in restenosis/occlusion by 31.7% as well as a significant absolute increase in estimated frontal sinus ostia diameter by 1 mm.
In terms of safety, there were no reported implant-related adverse outcomes. No other safety measures were assessed.
Remarks
The Propel corticosteroid-releasing stent and its variants are supported by favorable evidence in terms of efficacy and safety as shown in multiple randomized, intra-patient controlled, multicentered clinical trials. The device has proven safety in terms of systemic and ocular adverse effects, and it has received approval from the United States Food and Drug Administration (FDA) since 2011. The Propel Mini and Propel Contour are the only frontal sinus-specific devices with published efficacy. An early cost analysis showed that Propel implants are cost-effective in managing patients with CRS following ESS. 31 Subsequent analyses have suggested that the higher upfront cost associated with the implants is balanced by the decreased cost associated with fewer postoperative interventions. 32,33 Nonetheless, it is important to note that each of the original trials was industry-sponsored, a fact that poses a risk for bias.
Corticosteroid-Treated NasoPore
Characteristics
NasoPore is a synthetic polyurethane nasal dressing that has been used for postoperative packing to reduce synechia formation and postoperative pain. 34,35 Since NasoPore is commonly placed into the surgically opened ethmoid compartment, it has additionally served as a vehicle for intraoperative, localized placement of topical corticosteroids for postoperative healing. Various types and dosages of corticosteroids may be instilled into the NasoPore dressing.
Evidence on Clinical Outcomes
A total of 6 prospective, randomized, double-blinded, controlled trials have been performed on the efficacy and safety of corticosteroid-treated NasoPore placed immediately following ESS. The first prospective double-blinded, intrapatient-controlled trial of corticosteroid-treated NasoPore was published by Côté and Wright. 16 They treated 4-cm NasoPore dressings with a total of 80 mg of triamcinolone in patients with CRS with nasal polyposis (CRSwNP). They showed that perioperative sinus endoscopy (POSE) and Lund-Kennedy (LK) scores, representing measures of early postoperative healing in the sinonasal cavities, were significantly improved in the treated arm at various time points during the 6-month postoperative period. In this trial, no adverse outcomes were clinically observed, but laboratory testing for adrenal effects or ocular measurements were not evaluated.
However, a subsequent safety analysis in a prospective, randomized, double-blinded, interpatient-controlled trial by Hong et al 17 showed that when an 8-cm NasoPore was treated with 40 mg of triamcinolone—50% of the dosage used by Côté and Wright—suppression of the hypothalamic–pituitary–adrenal axis was noted in the first 2 postoperative days as assessed by decreased cortisol concentrations. They also noted the disruption of bone metabolism as assessed by depressed osteocalcin levels. Ocular measurements were not reported in this analysis.
There were then 3 prospective, randomized, double-blinded, controlled trials, 18 -20 which have all shown that POSE and/or LK scores were significantly improved at various time points during the postoperative period (Table 1). These trials differed in terms of the length of NasoPore used, choice of corticosteroid, and method of study control (intrapatient vs interpatient). Grzeskowiak et al 20 included a group of patients who were treated with ciprofloxacin-treated NasoPore with some significantly improved postoperative scores, but no statistically significant difference between the antibiotic-treated and corticosteroid-treated groups was noted. No implant-related complications were reported, and systemic effects and ocular measures were not assessed in these studies.
Finally, a randomized, double-blind, placebo-controlled trial was published in 2018 by Zhao et al 21 using a mometasone-impregnated NasoPore. Patients were randomized to receive first an 8-cm NasoPore, dosed with either 4 (1400 μg) or 8 (2800 μg) mL of mometasone, in one nasal cavity, and then a saline-impregnated NasoPore in the contralateral nasal cavity. They were further randomized to have the packing removed at either the postoperative 7- or 14-day mark. Patients did not use nasal saline irrigations or intranasal steroids until after the packing was removed. They noted that the higher doses of mometasone (2800 μg) and longer duration for nasal dressing (14 days) were associated with significant improvements in POSE and LK scores at the initial postoperative, 1-, 2-, and 3-month marks. No adverse events related to the nasal dressings were reported, but no systemic or ocular assessments were performed.
Remarks
Corticosteroid-impregnated NasoPore demonstrates less consistent results as compared with the Propel stents; however, this technique for localized delivery of corticosteroid therapy after ESS does show some degree of benefit in terms of LK and POSE scores over various postoperative time points. These clinical studies are characterized by significant heterogeneity in terms of study design with the use of both intrapatient and interpatient controls, lengths in NasoPore dressings, corticosteroid choices, and corticosteroid dosages. There was only one report regarding systemic effects as assessed by cortisol levels, which were shown to be elevated in the initial postoperative days. None of these studies assessed ocular safety.
Corticosteroid-Treated Gel-Type Nasal Dressings
Characteristics
In sinonasal procedures, bioresorbable gel-type substances have offered various postoperative applications, such as providing hemostasis and serving as anatomic spacers for the surgically opened sinus cavities. 36 Recently, gel-type nasal dressings, including SinuFoam, Merogel, Gelfoam, and calcium alginate, have additionally been used to deliver localized topical corticosteroid therapy after completion of ESS. SinuFoam is a carboxymethylcellulose polysaccharide that can be mixed with a corticosteroid. 22 Merogel is a bioabsorbable hyaluronic acid which can be reconstituted with a corticosteroid solution. 23 Gelfoam is a porcine-derived gelatin sponge that can be soaked with corticosteroids. 24,25 Calcium alginate is a biodegradable, seaweed-derived nasal packing that converts into a gel-type substance upon replacement of calcium with sodium. 37 The resultant liberated calcium ions are thought to promote hemostatic pathways in the postoperative setting. 38
Evidence on Clinical Outcomes
A total of 5 reports in which these gel-type substances were treated with corticosteroids were included for review. Neither SinuFoam nor Merogel was associated with significant differences in postoperative LK scores 22 and synechia formation, 23 although the triamcinolone-impregnated Merogel cohort trended toward decreased synechiae formation. No systemic or ocular safety assessments were performed.
Two studies in which corticosteroid-treated Gelfoam was applied to the nasal passages after ESS for CRS were included. In a prospective, randomized, single-blinded, intrapatient controlled trial, Abd-Elmaksoud et al 24 showed that treatment with 80 mg of triamcinolone was associated with a statistically significant reduction in POSE scores at the 8-week mark only. Specifically, they noted decreased synechiae, less middle meatal narrowing, decreased ethmoid polyposis, and decreased sphenoid severity; however, they did not note any significant changes in the frontal recess nor mucosal edema and secretions. They reported the absence of local and systemic complications, but the published study did not detail how safety outcomes were assessed.
In a prospective, randomized, double-blinded, intrapatient controlled pilot study, Sow et al 25 found significantly improved POSE scores with the treatment of hydrocortisone (dose unspecified) at the 1-week and 3-month marks, but the specific factors within the POSE scoring were not reported. There were no differences in postoperative LK scores. They also did not report any assessed safety outcomes.
Hwang et al 26 published a prospective, randomized, single-blinded, intrapatient-controlled trial in patients treated with triamcinolone-soaked calcium alginate. They noted significantly improved POSE scores at the 8-week mark only. Specifically, they noted a significant reduction in middle turbinate lateralization, polypoid change in the ethmoid cavity, and sphenoid sinus severity. No safety measures were assessed or reported in this study.
Remarks
There are mixed results as to the objective benefit of corticosteroid-treated bioresorbable gel-type nasal dressings following ESS. The available clinical studies demonstrate wide variability in study designs as well as choice and dose of corticosteroid therapy. No vehicle-related adverse events were reported, but safety was not assessed in any of these studies.
Corticosteroid-Treated Merocel Nasal Packing
Characteristics
Merocel is a commonly used nonresorbable nasal packing that is composed of hydroxylated polyvinyl acetate. The product is placed into the sinonasal cavities following ESS to prevent synechia formation. 39 Recently, various medications have also been infused into Merocel nasal pack following ESS for localized deposition of the medications into the neosinuses.
Evidence on Clinical Outcomes
The evidence on corticosteroid-treated Merocel nasal packing for use in CRS after ESS is based on 2 prospective, randomized, controlled trials. First, Chang et al 27 compared medicated and nonmedicated Merocel spacers. A total of 48 patients were randomized to receive medicated spacers containing budesonide, gentamicin, or manuka honey in 1 nostril and a nonmedicated spacer in the other. The results of this trial showed no statistically significant difference in sinonasal mucosal healing, patient discomfort, or pain on the removal of the device following ESS. However, budesonide-soaked spacers trended toward decreased inflammation as determined by pathologist-performed grading of an endoscopic biopsy on a scale of 0-4.
Sabarinath et al 28 compared triamcinolone-soaked Merocel packing and saline-soaked Merocel packing in 75 patients. The triamcinolone-impregnated Merocel spacers were associated with a statistically significant reduction in mucosal edema throughout the 3-month postoperative period and decreased crusting until the 1-month mark.
In both of these studies, safety assessments were not performed.
Remarks
The choice of medications used to treat Merocel was different between the 2 studies, and the results were mixed in terms of postoperative mucosal inflammation and crusting. Safety measures were not assessed in these studies.
Corticosteroid-Treated SinuBand
Characteristics
SinuBand is a 2-cm × 2-cm fibrinogen-based film consisting of an adhesive side and the other side containing 160 µg of fluticasone propionate. Preclinical data in rabbits have suggested that SinuBand causes minimal local reactions and that corticosteroid concentrations are highest during the first week after implantation. 40 A unique advantage of SinuBand is its ability to conform to the geometric shape of a specifically targeted location; however, potential difficulties with device implantation into narrow ethmoid compartments as a result of the adhesive side were reported. 29
Evidence on Clinical Outcomes
Adriaensen et al 29 published the only randomized, partially double-blind, intrapatient-controlled trial in human subjects on SinuBand treated with fluticasone (SinuBand FP). Patients were randomized to receive 2 of 3 treatments, with an intrapatient-control design: SinuBand FP, SinuBand without flucticasone, or an untreated Merocel pack. Sinuband FP was associated with significantly lower polypoid change through the first 30- and 60-day marks when compared to Merocel and Sinuband without fluticasone. Postoperative edema and LK scores were not significantly different. SinuBand FP was shown to be safe in terms of local, ocular, and systemic adverse effects.
Remarks
Only one clinical study has been conducted on SinuBand technology, but this particular trial demonstrates potential reduction of polyp change with the intraoperative placement of fluticasone-treated SinuBand during ESS for CRSwNP. The device’s unique design may lend to less obstruction of the drainage pathways compared to other devices in the initial postoperative period, although reports suggest that it may also be more challenging to deploy into smaller ethmoid neosinus cavities. The authors of the clinical study note that since corticosteroids were only delivered to 1 of 2 nasal passages, conclusions regarding systemic safety are indeterminate.
Discussion
With the exception of Propel corticosteroid-eluting stents, the technologies and techniques that have been discussed in this review have garnered clinical interest as methods for topical delivery of corticosteroids to the sinonasal mucosa immediately after ESS, but their use for this specific purpose have not received regulatory approval by the FDA. As reflected in the clinical studies, the off-label use of packing materials mixed with corticosteroids undoubtedly has clinical potential to improve postoperative healing in patients with CRS, although the benefits may also come with unintended side effects. Some of the drawbacks of these off-label corticosteroid-infused materials are rooted in the nonuniform pharmacological properties of the medications. Variations in the saturation of nasal dressings and subsequent elution onto the sinonasal mucosa highlight the potential delivery of inconsistent amounts of topical corticosteroids to the targeted sites within the neosinuses. Additionally, as the various nasal dressings are composed of different polymeric substrates, the optimal selection of the nasal packings, based upon their inherent proinflammatory or anti-inflammatory characteristics, has not been well studied.
Despite the theoretical risks with the off-label usage of topical corticosteroid therapies posed above, there have been limited clinical adverse events reported in the studies detailed throughout this review. Further research regarding the safety profile of these various nasal packing materials mixed with corticosteroid is thus warranted.
This review was intended to be a broad overview of the various strategies employed to deliver topical corticosteroids immediately following ESS for CRS. It was not intended to be a systematic review or meta-analysis as there is significant heterogeneity across the clinical studies in terms of study design, perioperative management, vehicles used, and choice and dose of corticosteroids. Even direct comparisons among studies of the same vehicle, such as NasoPore, would be difficult to perform.
Excluded from this review are reports of drug-eluting biomaterials that are intended to be placed in the office setting. These technologies include the Sinuva (Intersect ENT) 41,42 and LYR-210 implants (Lyra Therapeutics), 43 both of which have also shown clinical benefits for patients with CRS. The Sinuva steroid-releasing stent, which is placed into a previously opened ethmoid cavity for recurrent nasal polyposis, has received FDA approval for use since 2017, whereas LYR-210 is currently in phase 2 clinical development for treatment in surgically naive patients with CRS. The existing trials for these corticosteroid-eluting technologies were conducted without concurrent ESS.
Conclusion
Intraoperative steroid delivery techniques and technologies for CRS have offered efficacious and safe methods to enhance postoperative healing of the sinonasal mucosa after ESS and likely improve overall clinical outcomes. The Propel device is the most widely studied as there is level 1A evidence supporting its clinical efficacy. It also has the most evidence in terms of systemic and ocular safety and is the only device with FDA approval. SinuBand has also demonstrated systemic and ocular safety. The other devices are used in an off-label manner with variability in the evidence for their efficacy. Although the safety profiles of these devices tend to be reassuring, ocular and adrenal safety of the various corticosteroid choices and doses have yet to be proven. Future research focused on direct comparisons among these devices would be helpful.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
