Abstract
Aim:
Some obstructive sleep apnea (OSA) patients may have mandibular retrognathia (ANB > 4.7° and SNB < 76.2°). Currently, there are no studies that have compared the effectiveness of continuous positive airway pressure (CPAP) versus mandibular advancement device (MAD) in severe OSA patients with mandibular retrognathia. We explored the efficacy of CPAP versus MAD for the treatment of severe OSA patients with mandibular retrognathia.
Methods:
A total of 105 patients were enrolled. Outcomes were assessed by using polysomnography, Epworth Sleepiness Scale (ESS), Snore Scale (SS), Self-rating Anxiety Scale (SAS), and compliance, before treatment and after 6 and 12 months of treatment.
Results:
Continuous positive airway pressure was superior to MAD in improving polysomnographic outcomes and SS score, but reported compliance was higher on MAD. There is no significant difference between the 2 treatments in terms of ESS score and SAS score. Obstructive sleep apnea patients with mandibular retrognathia showed greater improvement than those without mandibular retrognathia in terms of apnea-hypopnea index and oxygen desaturation index after MAD.
Conclusion:
Continuous positive airway pressure and MAD are both effective in treating severe OSA patients with mandibular retrognathia. Mandibular advancement device is a good alternative to CPAP in severe OSA patients with mandibular retrognathia. Mandibular advancement device is more effective in treating OSA patients with mandibular retrognathia than those without.
Keywords
Introduction
Obstructive sleep apnea syndrome (OSA) is a serious and potentially life-threatening disorder, which is characterized by the disruption of the upper airway during sleep, resulting in snoring at night, sleep apnea, desaturation of oxyhemoglobin, and daytime sleepiness. 1 It is also a risk factor for high blood pressure and cardiovascular diseases such as stroke and ischemic heart disease.2,3 Patients who suffer severe OSA may also experience psychological disorders. 4 Anxiety, depression, and other cognitive dysfunctions are all examples of psychological disorders in OSA patients, which can adversely affect their quality of life. 5
Anatomical risk factors that may increase the risk of OSA include obesity, hypertrophy of soft palate and tongue, steep mandibular plane, small mandible, or mandibular retrognathia. Extensive cephalometric radiograph studies have also confirmed that OSA patients often present with mandibular retrognathia.6,7 In patients with mandibular retrognathia, the narrowest part of the oropharyngeal airway is generally near the tail of the soft palate and the base of the tongue. Their oropharyngeal airways may consequently become long and narrow. As a result, the patients’ airways could be blocked partially or entirely, which makes them more prone to OSA.8,9
For OSA patients with mandibular retrognathia, CPAP and MAD are both widely used. Continuous positive airway pressure is the gold standard in the treatment for OSA because it can reverse sleep-disordered breathing and reduce or even eliminate symptoms. However, uncomfort experience of and poor adherence to CPAP limit its effectiveness. 10 The American Academy of Sleep Medicine (AASM) recommended the use of MAD rather than no treatment for patients who are intolerant of CPAP therapy or prefer an alternative therapy in its recent guideline. 11 Mandibular advancement device is intended to reposition the lower jaw to enlarge the dimension of the airway lumen. In recent years, several randomized trials offer some evidence of the beneficial effect of MAD in patients with mild to moderate OSA.11-13 However, there have been no studies that compare the efficacy of CPAP and MAD in severe OSA with mandibular retrognathia. Therefore, the aim of this randomized controlled trial (RCT) was to compare the efficacy of CPAP and MAD in severe OSA patients with mandibular retrognathia.
Materials and Methods
Participants and Study Protocol
This was a prospective RCT conducted between January 2018 and January 2019 in severe OSA patients with mandibular retrognathia at the snoring clinic offered by the Department of Otorhinolaryngology, Fujan Medical University Union Hospital, China. To comply with ethical standards, participants and the legally authorized representatives of participants were informed of the conditions of the study and received informed consent forms before enrollment. This study was approved by the ethics committee of Union Hospital (number: 2018KY069).
Inclusion criteria were age ≥ 18 years, Apnea-Hypopnea Index (AHI) ≥ 30/h based on diagnostic polysomnography (PSG), and with the presence of skeletal Class II mandibular retrognathia evaluated by cephalometric radiographs (Figure 1). The cephalometric parameter considered for the evaluation of the presence of skeletal Class II mandibular retrognathia was subspinale–nasion–supramental (ANB) angle (ANB > 4.7°) and the sella–nasion–supramental (SNB) angle (SNB < 76.2°), according to Sailer. 14 Exclusion criteria were patients with inadequate tooth support to retain the MAD, unstable cardiovascular and neurological diseases, and/or temporomandibular joint disorders, and/or severe periodontal disease.
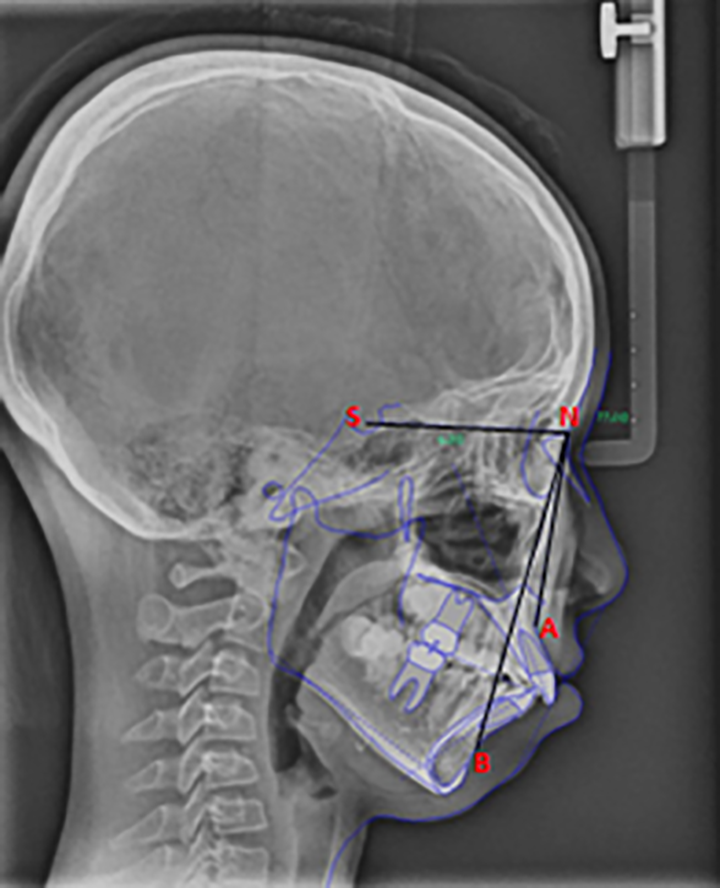
Cephalometric radiograph of severe OSA patients with mandibular retrognathia. A: Subspinale, B: Supramental, N: Nasion, S: Sella. OSA, obstructive sleep apnea.
Interventions
During the routine follow-up visit after PSG, the patients who were diagnosed with OSA in our snoring clinic were reevaluated for treatment planning, including discussions regarding CPAP or MAD. Those who agreed to accept either CPAP or MAD therapy were invited to join this study. The study began with a 1-week wash-in period, which means no treatment. The eligible patients diagnosed with severe OSA with mandibular retrognathia were randomly assigned to either the CPAP group (group A) or the MAD group (group B). At the same time, 40 patients with severe OSA matched by gender, age, neck circumference, body mass index (BMI), and disease duration but without mandibular retrognathia were selected. They were randomly allocated into the CPAP group (group C) or the MAD group (group D) (Figures 2 and 3). All subjects underwent a routine physical examination by otolaryngologists and stomatologists to collect the patients’ past and present illnesses. There were no differences in BMI, age and sex, and cervical circumference between the CPAP and MAD groups. The clinical characteristics of the patients are presented in Table 1.
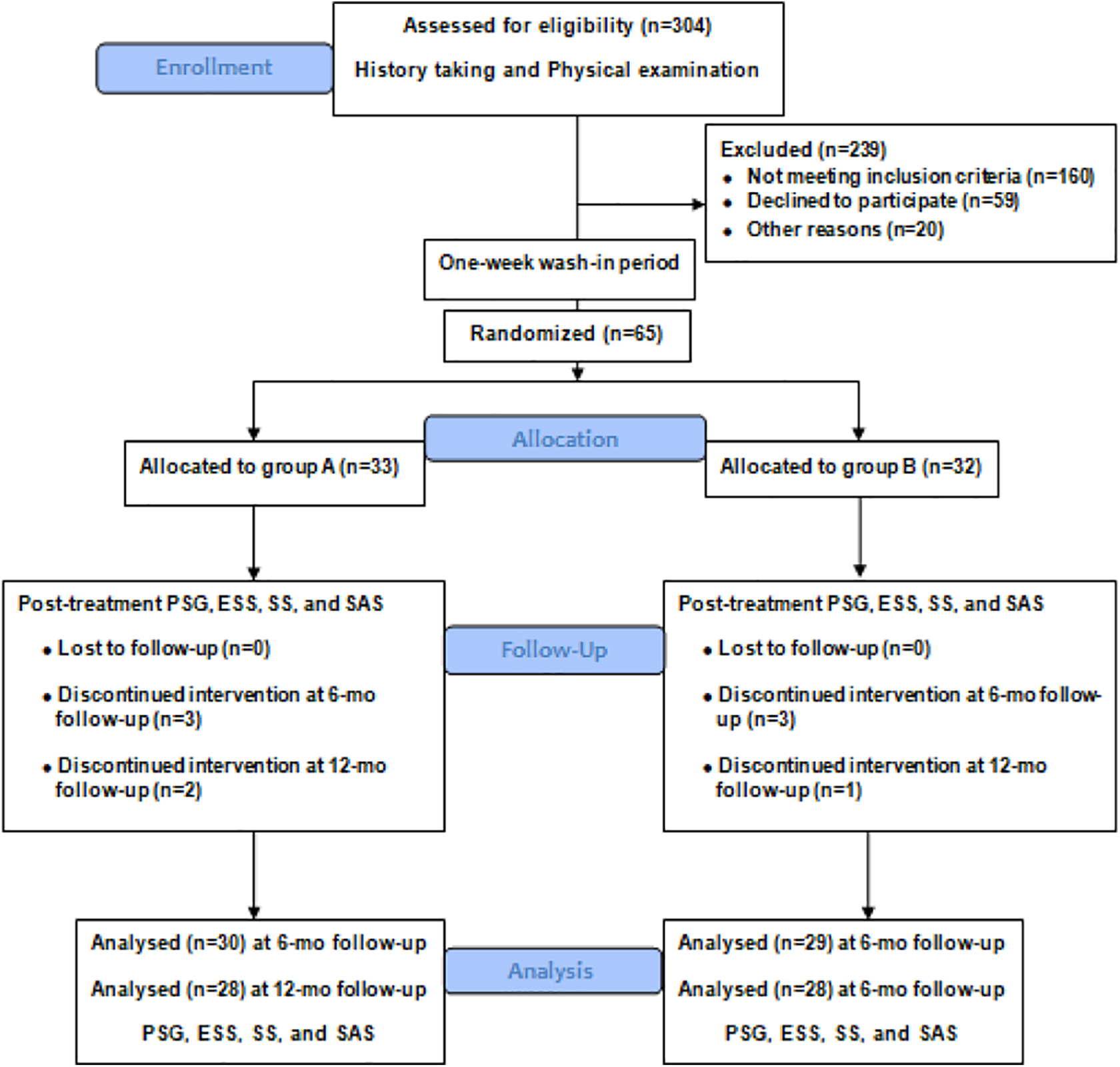
CONSORT flow diagram of severe OSA patients with mandibular retrognathia. OSA, obstructive sleep apnea.
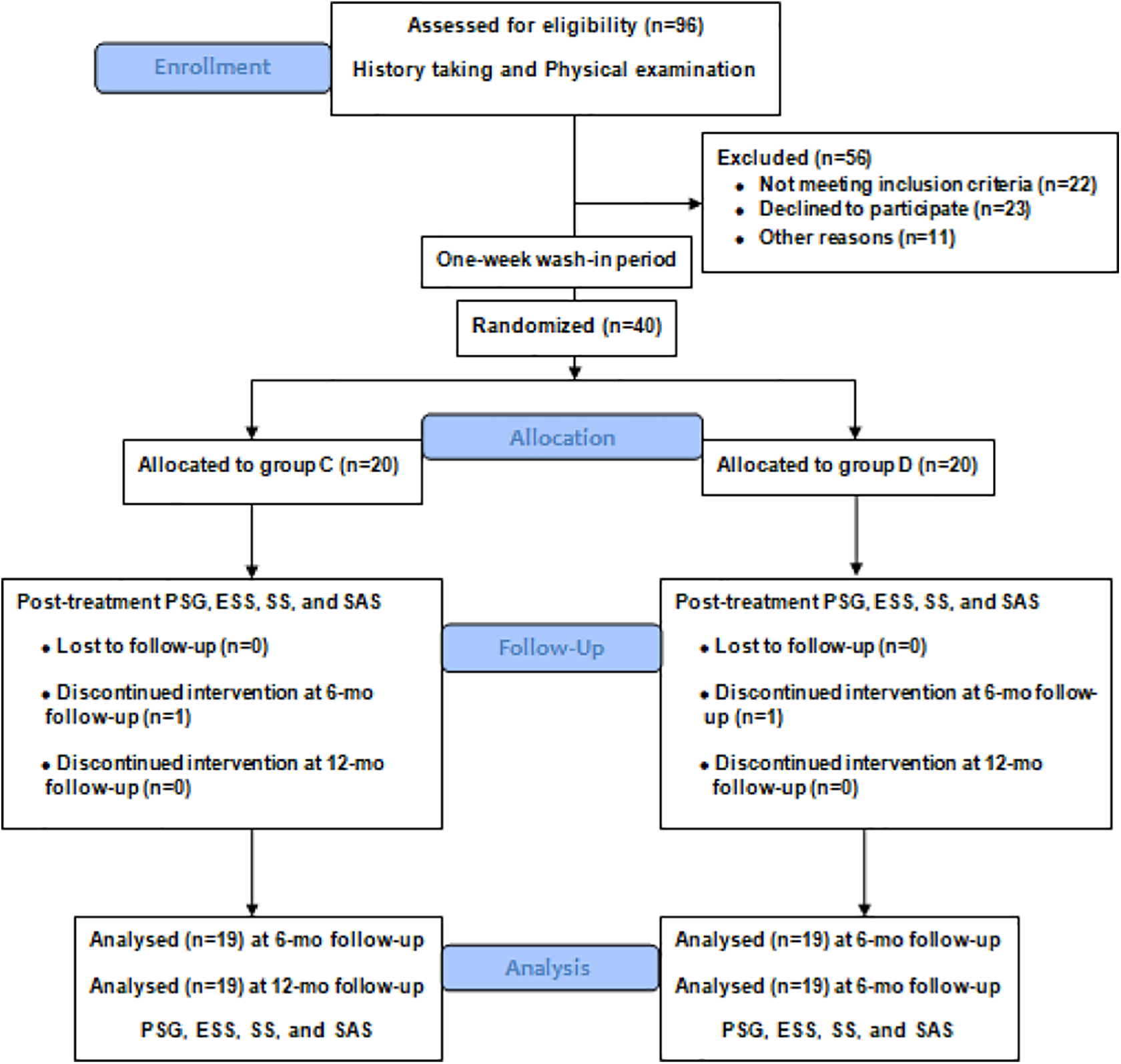
CONSORT flow diagram of severe OSA patients without mandibular retrognathia. OSA, obstructive sleep apnea.
Baseline Characteristics of the Study Subjects.
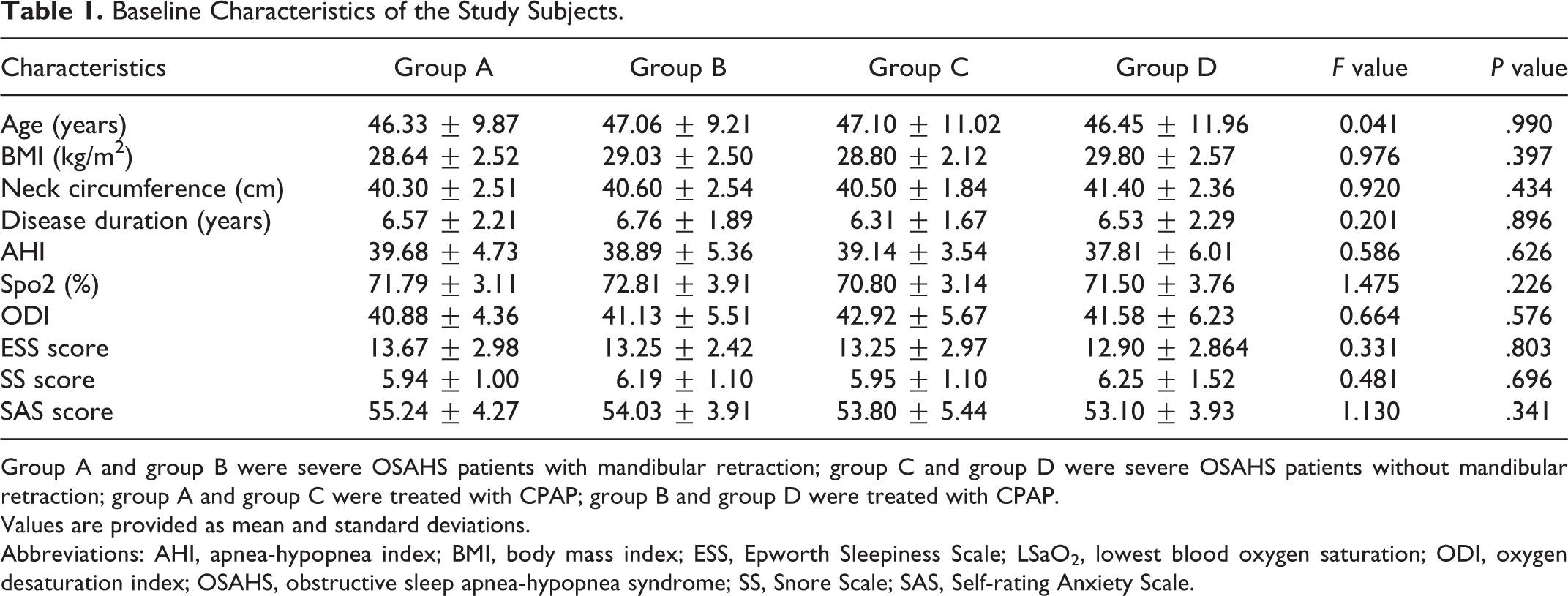
Group A and group B were severe OSAHS patients with mandibular retraction; group C and group D were severe OSAHS patients without mandibular retraction; group A and group C were treated with CPAP; group B and group D were treated with CPAP.
Values are provided as mean and standard deviations.
Abbreviations: AHI, apnea-hypopnea index; BMI, body mass index; ESS, Epworth Sleepiness Scale; LSaO2, lowest blood oxygen saturation; ODI, oxygen desaturation index; OSAHS, obstructive sleep apnea-hypopnea syndrome; SS, Snore Scale; SAS, Self-rating Anxiety Scale.
In this study, polysomnographic and subjective assessments were carried out for all patients and compared after 6 and 12 months of treatment.
Mandibular Advanced Device
In this study, we used an adjustable thermoplastic MAD (OSCIMED; Figure 4). It is a 2-piece thermoplastic MAD with a set of 6 supple straps (permitting a protrusion from 3.0 to 10.5 mm). Fitting procedures were carried out by a trained stomatologist in the clinic according to the manufacturer’s instructions, which included boiling the thermoplastic MAD for 2 minutes, cooling it to room temperature for 1 minute, taking bite impression for 1 minute, and rehardening it in cold water for 2 minutes. Attachment of the separate pieces was done by a suitable strap. The initial protrusion was usually set at 4.5 mm. Mandibular advance device was self-titrated by gradually advancing the device until the maximum comfortable limit of mandibular advancement was achieved. The patients were asked to wear the MAD nightly and to attend follow-up visits with a trained research assistant to receive instruction for optimal symptoms with the fewest side effects. If needed, remolding of the MAD, trimming of any excessive parts, and adjusting for the suitable strap were done.
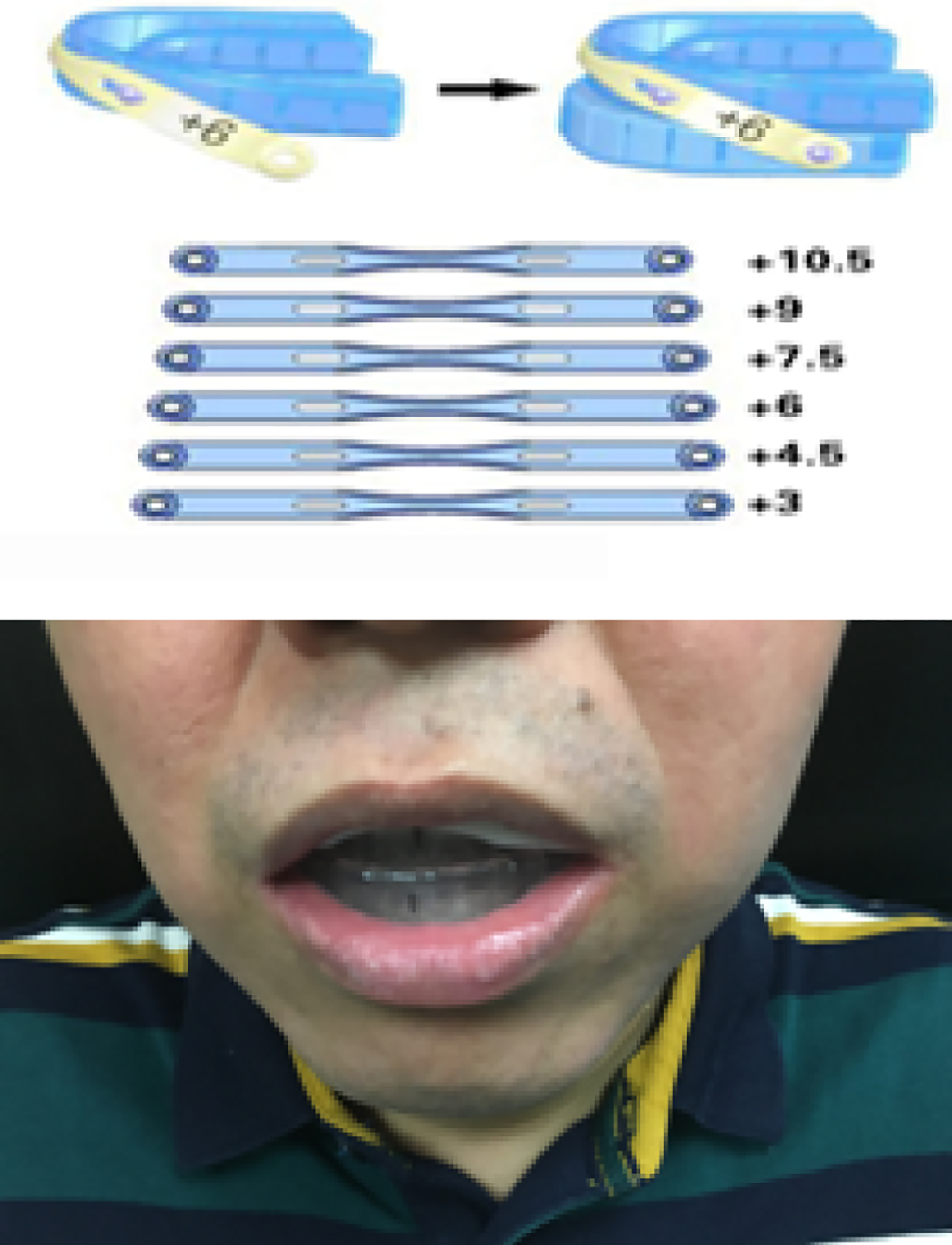
Adjustable thermoplastic mandibular advancement device (OSCIMED).
Continuous positive airway pressure therapy
In this study, we used a ResMed Continuous Positive Airway System (Elite, ResMed). Prior to home use, all patients were fitted with a properly sized mask and received a CPAP demonstration by trained personnel. Patients were instructed to use the device nightly, as tolerated.
Polysomnography
Standard PSG (Alice PDx, Philips) was performed at baseline and after treatment in the same condition. Variables including AHI, arousal index, percentage of total sleep time spent with arterial oxygen saturation less than 90% (SaO2, T < 90%), lowest blood oxygen saturation (LSaO2), and oxygen desaturation index (ODI) were analyzed and scored by the same trained sleep physician in accordance with the AASM standard. 15 The sleep physician was unaware of the participants’ treatment assignment.
Cephalometric measurement
All cephalograms were made upon a standardized lateral radiograph (18°A∼24 cm film; Kodak) on the basis of a natural head position by the same technician in the Radiology Unit of Fujian Medical University Union Hospital, China. The cephalometric parameter was analyzed by a single technologist and a physician by using software (Dental Studio NX).
Neurobehavioral Examinations and Adherence
Daytime sleepiness, snoring, and severity of depression were evaluated by using the Epworth Sleepiness Scale (ESS), the Snore Scale (SS), and the Self-rating Anxiety Scale (SAS). Patients completed all the questionnaires at baseline and after 6 and 12 months of treatment, respectively. Self-reporting diaries were used to compile subjective compliance data during the study.
Outcome Measurements
The primary outcome was AHI. Secondary outcomes were LSaO2, ODI, arousal index, SaO2, T < 90%, ESS, SS, SAS, and self-reported adherence after 6 and 12 months. Treatment failure would be reported if participants drop out of this study due to adverse effects or were lost to follow-up for any reason.
Sample Size Calculation
The sample size calculation for this study was based on AHI—the primary outcome. In severe OSA patients with mandibular retrognathia, using an expected mean AHI difference of 5.5 between interventions, a power of 80%, 2-side error of 0.05, and a standard deviation (SD) of 7.0, the calculated sample size was 27 participants per intervention. To compensate for a potentially estimated 15% loss to follow-up, the final number of participants was increased to 32 per intervention. In severe OSA patients without mandibular retrognathia, using an expected mean AHI difference of 7.0 between interventions, a power of 80%, 2-side error of 0.05, and a SD of 7.0, the calculated sample size was 17 participants per intervention. To compensate for a potentially estimated 15% loss to follow-up, the final number of participants was increased to 20 per intervention.
Randomization, Allocation Concealment and Blinding
Participants were allocated into different treatment groups using sequential numbers of a block computerized randomization algorithm with a 1:1 ratio. The sequential numbers were concealed in opaque envelops. Both participants and researcher were blinded to the sequential numbers before group assignment, but not during the interventions.
Statistical Analysis
Statistical analyses were performed using the SPSS 23.0 software (IBM Corp). All continuous variables were described as means with SDs with a normal distribution and median (interquartile range) for variables with a non-normal distribution. The AHI in the MAD groups and CPAP groups at baseline was normally distributed after logarithmic transformation. The primary outcome measure of this follow-up study was polysomnographic at different time points using MAD or CPAP therapy. Secondary outcome measures were neurobehavioral outcomes at different time points. Analysis of variance (ANOVA) for repeated measures was used for comparison of each group before and after treatment. The one-way ANOVA was used to determine statistically significant differences in continuous variables between groups. Least significant difference test was performed for pairwise comparisons for variables with a normal distribution, and the Wilcoxon rank-sum test for variables with a non-normal distribution. The significance level α of all analyses was set at P < .05.
Results
A total of 65 eligible patients were included in this study, including 60 males and 5 females. The mean age was 46.72 ± 10.19 years (range, 20-60 years). There were 33 and 32 patients in group A (CPAP) and group B (MAD), respectively. Forty patients that matched by gender, age, neck circumference, BMI, and disease duration but without mandibular retrognathia were recruited, including 38 males and 2 females. The mean age was 46.78 ± 11.37 years (range, 22-60 years). Twenty patients were allocated to group C (CPAP), and the other 20 to group D (MAD). Baseline characteristics of each group are shown in Table 1. Eleven patients were excluded during the course of study for the following reasons: lost to follow-up (n = 4); device-related technical problems (n = 6), and partner objection (n = 1). Additional details are given in the CONSORT flow diagram shown in Figures 2 and 3.
Polysomnographic Outcomes
Polysomnographic data at baseline have no significant differences in terms of AHI, arousal index, SaO2, T < 90%, ODI, and LSaO2 in all of the 4 groups. Apnea-hypopnea index, arousal index, SaO2, T < 90%, ODI, and LSaO2 in all of the 4 groups were significantly improved compared with the baseline at both 6 and 12 months. Apnea-hypopnea index, arousal index, SaO2, T < 90%, and ODI in the CPAP groups were significantly lower than those in the MAD groups, and LSaO2 was significantly higher in the CPAP groups than that in the MAD groups at the 6 and 12 months follow-up. Apnea-hypopnea index and ODI in group B were lower than those in group D at both 6 and 12 months. Comparison of respiratory parameters is shown in Table 2.
Polysomnographic Outcomes at Different Time Points.
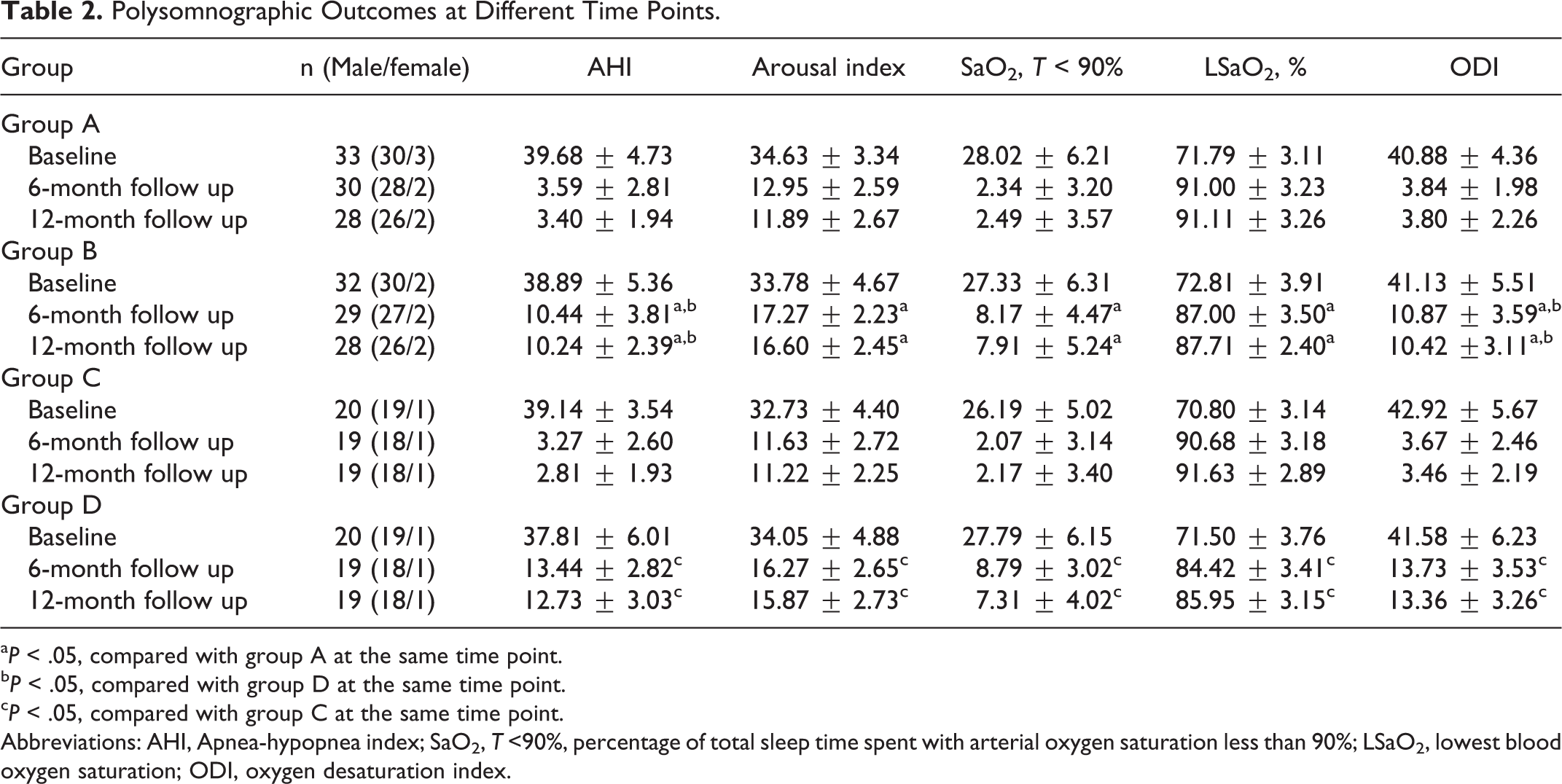
aP < .05, compared with group A at the same time point.
bP < .05, compared with group D at the same time point.
cP < .05, compared with group C at the same time point.
Abbreviations: AHI, Apnea-hypopnea index; SaO2, T <90%, percentage of total sleep time spent with arterial oxygen saturation less than 90%; LSaO2, lowest blood oxygen saturation; ODI, oxygen desaturation index.
Cephalometric Assessment and Mandibular Advancement
Patients in group A and group B had skeletal Class II mandibular retrognathia and patients in group C and group D were normal. There was no statistically significant difference in ANB angle and SNB angle in each group before and after treatment. No significant difference was observed between group B and group D for the advancement of MAD during the study. Comparison of cephalometric parameter and mandibular advancement is shown in Table 3.
Cephalometric Parameter for All Patients and Mandibular Advancement for MAD-Used Patients at Different Time Points.
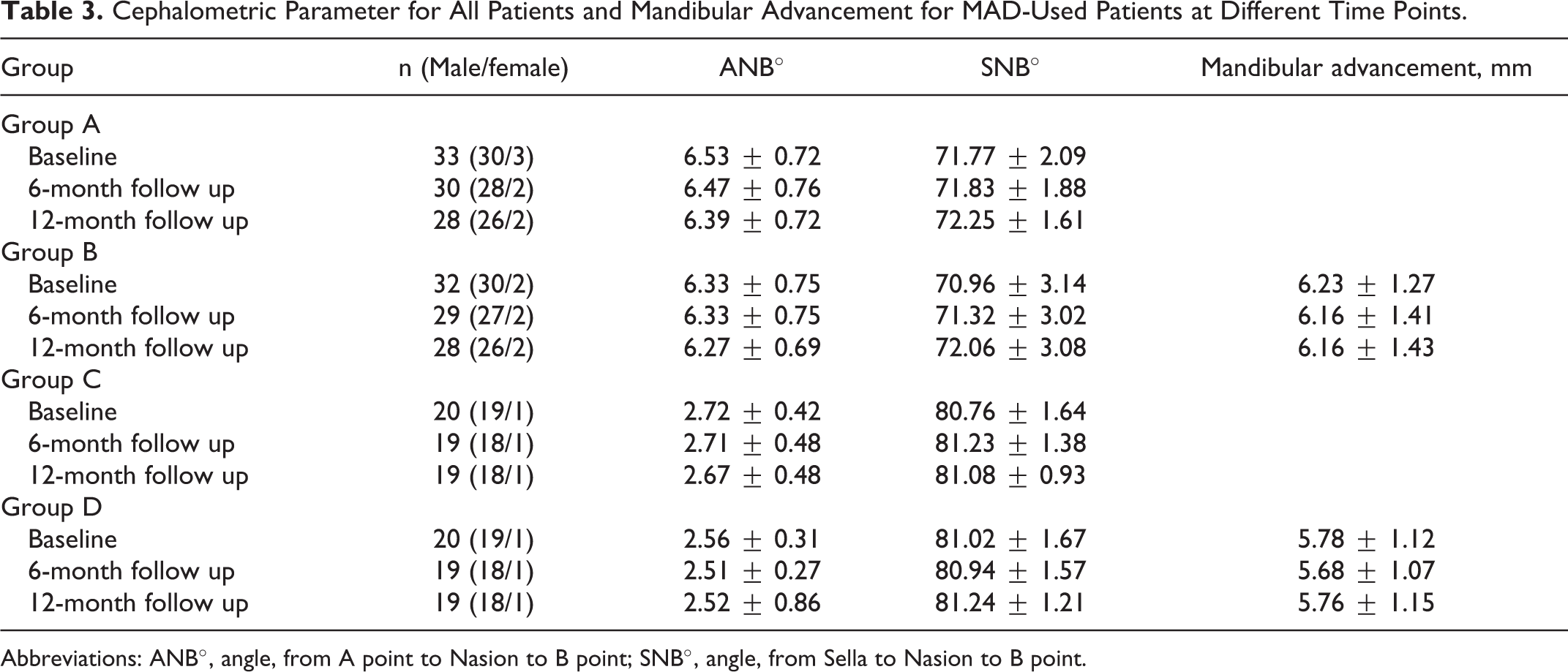
Abbreviations: ANB°, angle, from A point to Nasion to B point; SNB°, angle, from Sella to Nasion to B point.
Daytime Sleepiness
The ESS at baseline has no significant differences among the 4 groups. Comparison of ESS scores at 6 and 12months after treatment showed that ESS scores in all 4 groups were significantly lower than the baseline values, and there was no statistically significant difference among the 4 groups at each time point. Comparison of ESS is shown in Table 4.
Neurobehavioral Outcomes at Different Time Points.
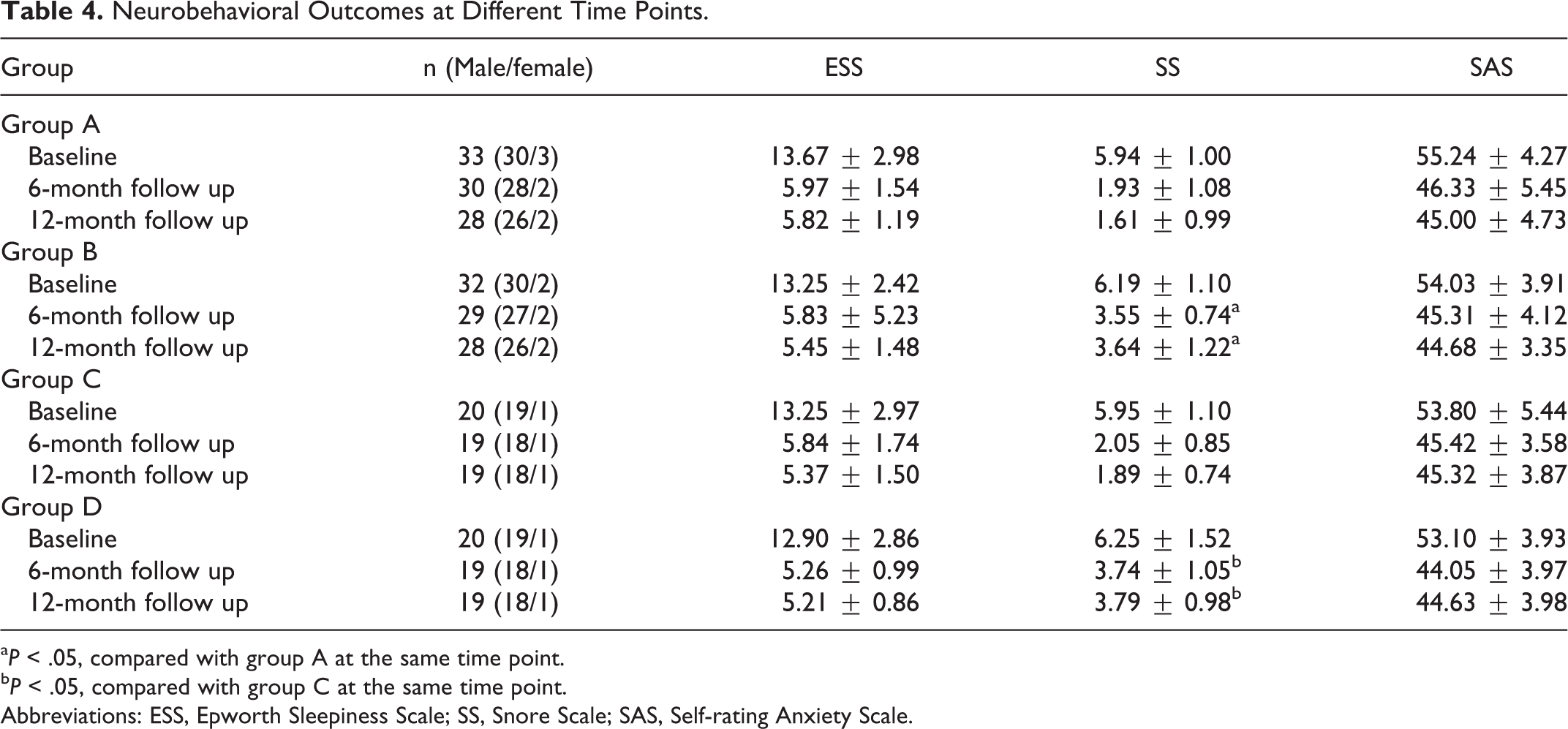
aP < .05, compared with group A at the same time point.
bP < .05, compared with group C at the same time point.
Abbreviations: ESS, Epworth Sleepiness Scale; SS, Snore Scale; SAS, Self-rating Anxiety Scale.
Snore Scale
The SS scores at baseline had no significant differences among the 4 groups. The SS scores of the 4 groups all decreased significantly at both 6 and 12 months. The SS scores of the CPAP groups were significantly lower than those of the MAD groups at both 6 and 12 months. Comparison of SS scores is shown in Table 4.
Self-Rating Anxiety Scale
The SAS scores at baseline were similar among the 4 groups, and all significantly decreased at 6 and 12 months after the treatment. It also showed that there was no statistically significant difference among the 4 groups at each time point. Comparison of SAS scores is shown in Table 4.
Adherence
Patients receiving MAD treatment reported higher compliance than those receiving CPAP treatment (Table 5). In the CPAP groups, both objective and subjective compliance measures were available. Objective compliance was slightly lower, which indicates that the patients overestimated actual CPAP use. Equivalent objective compliance data were not available for the MAD groups.
Comparison of Compliance at Different Time Points.
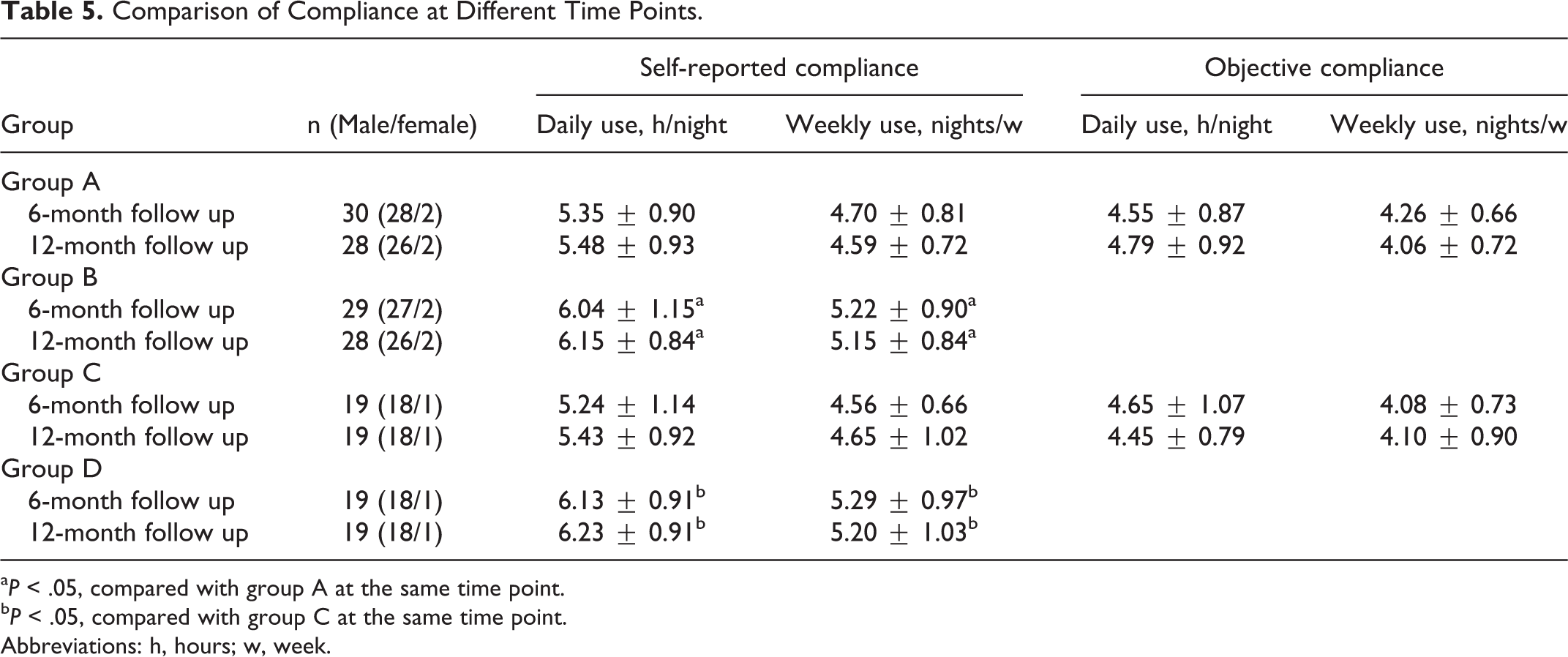
aP < .05, compared with group A at the same time point.
bP < .05, compared with group C at the same time point.
Abbreviations: h, hours; w, week.
Discussion
The etiology of OSA includes several factors related to the upper airway anatomy, such as mandibular retrognathia. 16 Although the overall effect of MAD on sleep-disordered breathing is inferior to CPAP, its uptake and acceptance as an alternative therapy is generally getting higher. Comprehensive care of any disease requires expertise in all aspects of diagnosis and treatment. The efficacy and effectiveness evaluation of these 2 methods should be observed by regular follow-ups.
Results from the recent investigation have revealed that the overall effectiveness of both CPAP and MAD is obvious for treating mild to severe OSA.17,18 Both treatments had a salutatory effect on polysomnographic variables during the follow-up period. Continuous positive airway pressure was much more efficacious in improving AHI, arousal index, SaO2, T < 90%, ODI, and LSaO2, but in terms of the patient comfort, side effects, and acceptance, MAD is superior to CPAP. The long-term use of CPAP therapy can effectively lower the patient’s AHI to normal or near-normal levels. 19 This study also found that AHI, arousal index, SaO2, T < 90%, ODI, and LSaO2 of all 4 groups were significantly improved compared with those before treatment, and the effectiveness of CPAP and MAD was found to be stable after the 12-months follow-up. We also found that AHI and ODI of patients in the CPAP groups could be reduced to the normal range after treatment, but the AHI and ODI in the MAD groups were not reduced to the normal ranges despite significant improvements were achieved. Mandibular advancement device reduced AHI from severe to mild category. We hypothesize that CPAP works as a pneumatic splint to the upper airway during the inspiration period, which could not only prevent airway collapse but also could expand the upper airway by pushing soft palate and tongue in an anterior direction. Mandibular advancement device might cause a nuisance to the patients if the device is not attached to the teeth well enough and leads to impaired breathing. 20
We think MAD is more effective for severe OSA patients with mandibular retrognathia than for severe OSA without mandibular retrognathia in terms of AHI and ODI. We speculated it by the following reasons: first, MAD plays an effective role by directly widening the oral cavity, giving the tongue more room in cranial, ventral, dorsal, and caudal directions, so that these spaces, between which occlusion may occur, are put at a greater distance. Second, OSA patients with mandibular retrognathia have narrower SNB angles than those without. Mayer and Meier-Ewert discovered that the narrower of the SNB angle of the OSA patient, the more effective use of MAD treatment. 21 Third, MAD may cause an activation of press receptors, change the threshold of centrally controlled muscle tonus depending on downregulation of genioglossus tonus, which is increased in OSA patients. Activating the muscle tonus leads to reduced upper airway resistance. 21 As for severe OSA patients without mandibular retrognathia, even though MAD can pull the mandible forward, it does not directly work on the blocked plane. Therefore, the objective therapeutic effect is not as good as that of severe OSA patients with mandibular retrognathia.
Although CPAP was superior to MAD in reducing PSG-detected respiratory disturbances, it is also important to realize that the success of any therapy is not contingent upon objective response. Treatment adherence is critical. In this study, we used the participants’ self-reported diaries to denote CPAP and MAD adherence, which may overestimate compliance. Continuous positive airway pressure was more effective than MAD at reducing AHI, but MAD was associated with a significantly higher reported compliance. In one study, recordings from MAD with embeded microsensors have found no difference between subjective and objective MAD adherence. 22 Previous studies also found higher adherence with MAD. Tan et al compared CPAP and MAD devices and found that MAD was better tolerated and was a suitable alternative device to CPAP in the treatment of OSA patients. 23
In our study, we found that neither therapy had a superior effect in reducing subjective sleepiness determined by the ESS score. Previous studies have also proven that CPAP and MAD can significantly improve the symptoms of OSA patients, correct hypoxia, improve sleep, reduce drowsiness, improve tissue metabolism, and improve the quality of life.24,25 This might be explained by the patients’ low acceptability and compliance with the CPAP treatment, compared to a high acceptability and comfort level during the MAD treatment. 26
Our study revealed that both CPAP and MAD could effectively improve the snoring score of severe OSA patients during 12-month follow-up. Both CPAP and MAD can improve snoring by preventing airway collapse in OSA. However, CPAP reduced snoring to a greater extent than MAD. As mentioned above, CPAP can completely control OSA. Continuous positive airway pressure causes improvement in snoring by directly increasing upper airway pressure (effectively preventing the airway collapse) and increasing air tract space by tongue protrusion while reduction of snoring with the use of MAD may be due to the forward movement of the mandible, which indirectly causes decreased compression of the upper air tract opening. Arya et al discovered that MAD could decrease the soft palate area by 9.5%, while CPAP could decrease it by 12.6%. 16 This may be explained by the reduced inflammation of the soft palate, which is irritated by snoring vibrations. The soft palate angle is reduced. This is probably because of the overall decrease in the soft palate area. 27
Similar to previous studies, OSA patients had anxiety symptoms. 28 After treatment, all patients showed a significant reduction in anxiety symptoms. A meta-analysis of 31 studies demonstrates CPAP treatments for OSA can more effectively reduce anxiety symptoms and improve quality of life compared to placebo, MAD, or simulated CPAP. 29 Our study showed a different result: Both treatments were associated with a similar improvement in anxiety levels. One possible explanation for this is that after the treatment of CPAP or MAD, the hypoxia of patients has been effectively improved, and the mental abnormalities caused by hypoxia have been well corrected. After the treatment, the patients were accustomed to unblocked airways and their sleep quality improved, which was beneficial to their psychological status. At the same time, this improvement also increased the patient’s resilience against the disease and reduced the patient’s anxiety for the influence of snoring on others.
This study has some limitations. First, we used subjective questionnaires to evaluate the posttreatment effectiveness. This may be flawed data in terms of reliability due to a tendency of participants to report optimistic patterns of adherence. Second, the major outcomes of this study were the differences of posttreatment situation (residual AHI) between 2 interventions (MAD and CPAP), not success rates (reduction of AHI) compared directly to the baseline PSG. Third, there was no way to monitor the objective daily usage of MAD at present. Continuous positive airway pressure and MAD were compared on the basis of self-reported compliance. Patients might have overestimated the actual daily or weekly use of MAD. A comparison with similar measurement should be considered. Fourth, OSA was 3 to 4 times less prevalent in women than men, which may be attributed to preferential fat deposition in men in the neck, less fat distribution in the subcutaneous tissues of hips and thighs, in contrast to women. The participants enrolled in this study were predominantly Chinese men, which may lack generalizability. Finally, we only enrolled patients with severe OSA. Therefore, the findings and results of this study may not be generalized to other populations with moderate or mild OSA.
Conclusion
The current study confirms that both CPAP and MAD can effectively improve subjective and objective symptoms in patients with severe OSA. Continuous positive airway pressure outperforms MAD in improving snoring and polysomnographic outcomes such as AHI, ODI, arousal index, SaO2, T < 90%, LSaO2, and snoring. Both CPAP and MAD can improve equally subjective drowsiness and anxiety in severe OSA patients. Mandibular advancement device treatment of severe OSA patients with mandibular retrognathia is superior to that of severe OSA without mandibular retrognathia in terms of AHI and ODI. In routine clinical practice, we recommend that OSA patients be fully informed regarding all possible effective therapeutic options associated with each before determining their most suitable choice of treatment.
Supplemental Material
Supplemental Material, CONSORT_a - Effectiveness of Continuous Positive Airway Pressure Versus Mandibular Advancement Device in Severe Obstructive Sleep Apnea Patients With Mandibular Retrognathia: A Prospective Clinical Trial
Supplemental Material, CONSORT_a for Effectiveness of Continuous Positive Airway Pressure Versus Mandibular Advancement Device in Severe Obstructive Sleep Apnea Patients With Mandibular Retrognathia: A Prospective Clinical Trial by Haichun Lai, Wei Huang, Wei Chen and Desheng Wang in Ear, Nose & Throat Journal
Supplemental Material
Supplemental Material, CONSORT_b - Effectiveness of Continuous Positive Airway Pressure Versus Mandibular Advancement Device in Severe Obstructive Sleep Apnea Patients With Mandibular Retrognathia: A Prospective Clinical Trial
Supplemental Material, CONSORT_b for Effectiveness of Continuous Positive Airway Pressure Versus Mandibular Advancement Device in Severe Obstructive Sleep Apnea Patients With Mandibular Retrognathia: A Prospective Clinical Trial by Haichun Lai, Wei Huang, Wei Chen and Desheng Wang in Ear, Nose & Throat Journal
Supplemental Material
Supplemental Material, original_data - Effectiveness of Continuous Positive Airway Pressure Versus Mandibular Advancement Device in Severe Obstructive Sleep Apnea Patients With Mandibular Retrognathia: A Prospective Clinical Trial
Supplemental Material, original_data for Effectiveness of Continuous Positive Airway Pressure Versus Mandibular Advancement Device in Severe Obstructive Sleep Apnea Patients With Mandibular Retrognathia: A Prospective Clinical Trial by Haichun Lai, Wei Huang, Wei Chen and Desheng Wang in Ear, Nose & Throat Journal
Footnotes
Acknowledgments
The authors gratefully acknowledge Assoc. Prof. Jianmin Huang, MD, PhD, for his support of this study. The authors also thank all the patients who generously agreed to participate in this study.
Author Contributions
DeSheng Wang conceived and designed this manuscript and finally approved; Haichun Lai contributed for drafting, data acquisition, analysis, and interpretation and provided critical revisions and final approval of the manuscript; Wei Huang contributed for data analysis; Wei Chen prepared the figure.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Science and Technology Innovation Project of Fujian Province [Grant number: 2019Y9085]; the Special Financial Project of Fujian Province [Grant number: 2019B022]; Minimally Invasive Medical Fund of Fujian Province [Grant number: 2104519#]; Medical Innovation Project of Fujian Province [Grant number: 2017-CX-23].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
