Abstract
Chronic nasal crusting is a commonly encountered entity in an otolaryngology office. Progressive, extensive nasal crusting with erosion is relatively unusual, however. We present the case a 58-year-old renal transplant patient with a history of vasculitis and immunosuppression who presents with subjective headache and facial pain, nasal crusting, and isolated left ethmoid sinusitis. She developed extensive intranasal necrosis and underwent multiple endoscopic sinus surgeries with intraoperative biopsies, which played a critical role in her workup and eventual diagnosis of Acanthamoeba rhinosinusitis. Although she endured a difficult course, proper diagnosis and treatment allowed for her recovery over time. The differential diagnosis for intranasal necrosis is often broad. This case highlights the wide range of etiologies to be considered in a patient with extensive nasal crusting and erosion/necrosis, and the importance of thorough diagnostic evaluation in these patients, especially those in an immunocompromised state.
Introduction
A patient presenting to a physician with chronic nasal crusting and facial pain demonstrates a very common, yet challenging clinical entity, as the differential diagnosis can be vast. Otolaryngologists are among the group of physicians that frequently encounter these patients, commonly as a result of sinonasal disease. 1 Although chronic crusting is common, progressive, extensive crusting with erosion is relatively unusual, however, and warrants extensive workup for the underlying cause. This symptomatology, particularly in a patient who is immunocompromised, may present a diagnostic dilemma. Furthermore, the progressive nature of many of the possible causative diseases, such as infectious or malignant etiologies, necessitates a timely evaluation and diagnosis. 1 Here, we present the case of a 58-year-old woman in an immunocompromised state who was evaluated for facial pain and mild nasal crusting. She underwent a long and progressive course of illness before being diagnosed and treated for Ancanthamoeba rhinosinusitis. We describe the patient’s clinical course, diagnosis and management, as well as a brief review of the literature available on Acanthamoeba rhinosinusitis.
Case Report
A 58-year-old woman with a history of vasculitis, 6 months status post repeat renal transplant, presented with intractable subjective headache and facial pain over the region of her left eye and extending into the temporal and occipital regions. Visual examination revealed no external abnormalities and nasal endoscopy revealed mild crusting anteriorly but no bleeding, eschar, or necrotic tissue. Notably, the patient had no history of intranasal drug use. Noncontrast computed tomography (CT) scan revealed mild paranasal sinus disease with opacification of an isolated left anterior ethmoid air cell (Figure 1). At the time of endoscopic surgery to address her sinusitis and possible mucocele, crusting and subtle mucosal necrosis were found on the nasal septum and the anterior aspect of the left inferior turbinate. Multiple biopsies were taken in the region of the septum and inferior turbinate. Pathology results were relatively nonspecific demonstrating mucosal necrosis, histiocytic reaction, and lymphoplasmacytic infiltrate with some granulation tissue (Figure 2). The patient’s condition did not improve following surgery. In the interim, she was hospitalized and treated for pulmonary aspergillosis and systemic blastomycosis for a couple of months before returning to our clinic with ongoing symptoms. At this time, she stated she had noticed something malodorous in her nose. Repeat CT scan suggested necrosis of the nasal septum and left inferior turbinate. Nasal endoscopy now revealed septal perforation and dark green crusting. These findings prompted repeat endoscopic sinus surgery for debridement and biopsy to evaluate for possible causes such as natural killer (NK)/T-cell lymphoma, squamous cell carcinoma (SCC), vasculitis, or infection. Intraoperatively, extensive necrosis of the septum and turbinates and intranasal crusting were seen. As a result, she underwent surgical excision and debridement of the necrotic tissue. Multiple pathologic stains and laboratory tests were performed in order to elucidate the underlying cause of her surgical findings. Conventional 28S primer set polymerase chain reaction (PCR) was positive for Acanthamoeba. As her condition progressed, she began to develop disseminated cutaneous lesions as well. Both cutaneous and intranasal tissue samples were sent to the Center for Disease Control (CDC) where an Acanthamoeba-specific PCR test returned positive in cutaneous and intranasal tissue. At this time, she began treatment with miltefosine and isofluconazole. Notably, tissue evaluation via confocal microscopy did not reveal any visible organisms. As her condition progressed, she later developed a mucocutaneous fistula through the lateral wall of her nares and underwent a paramedian forehead flap reconstruction to repair the fistula. She eventually recovered from the sequelae of her infection and is doing well.
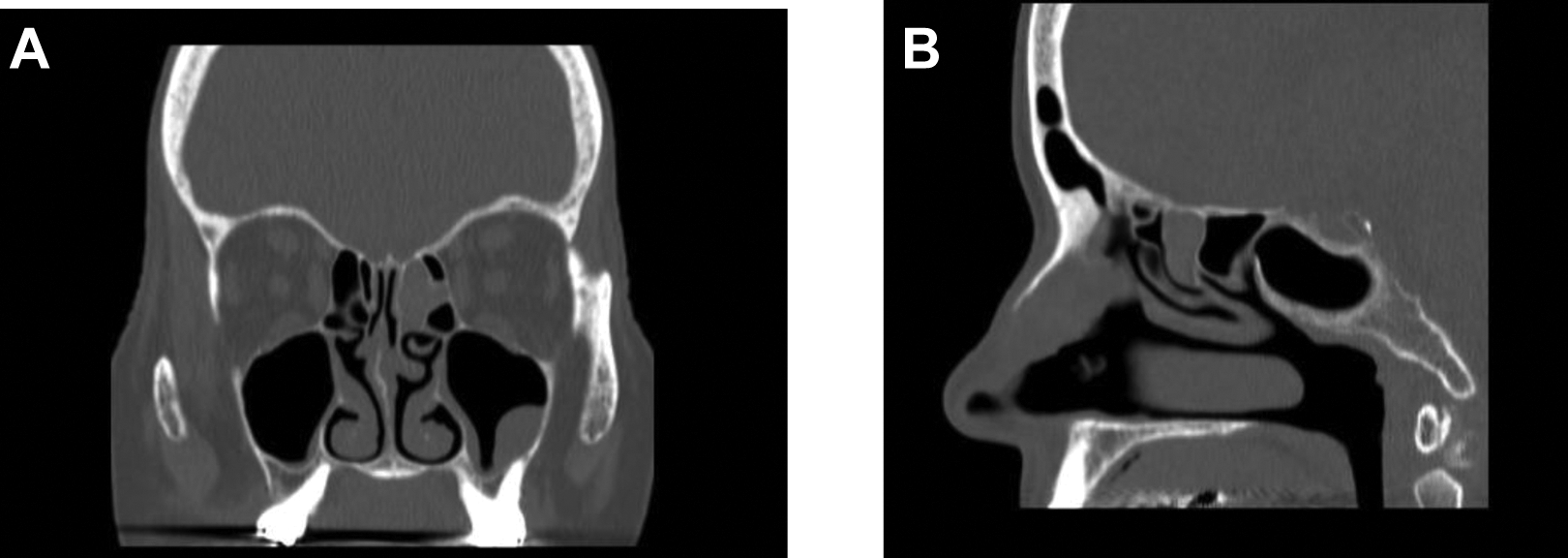
Coronal (a) and sagittal (b) non-contrast CT scan showing opacification in right anterior ethmoid air cell.
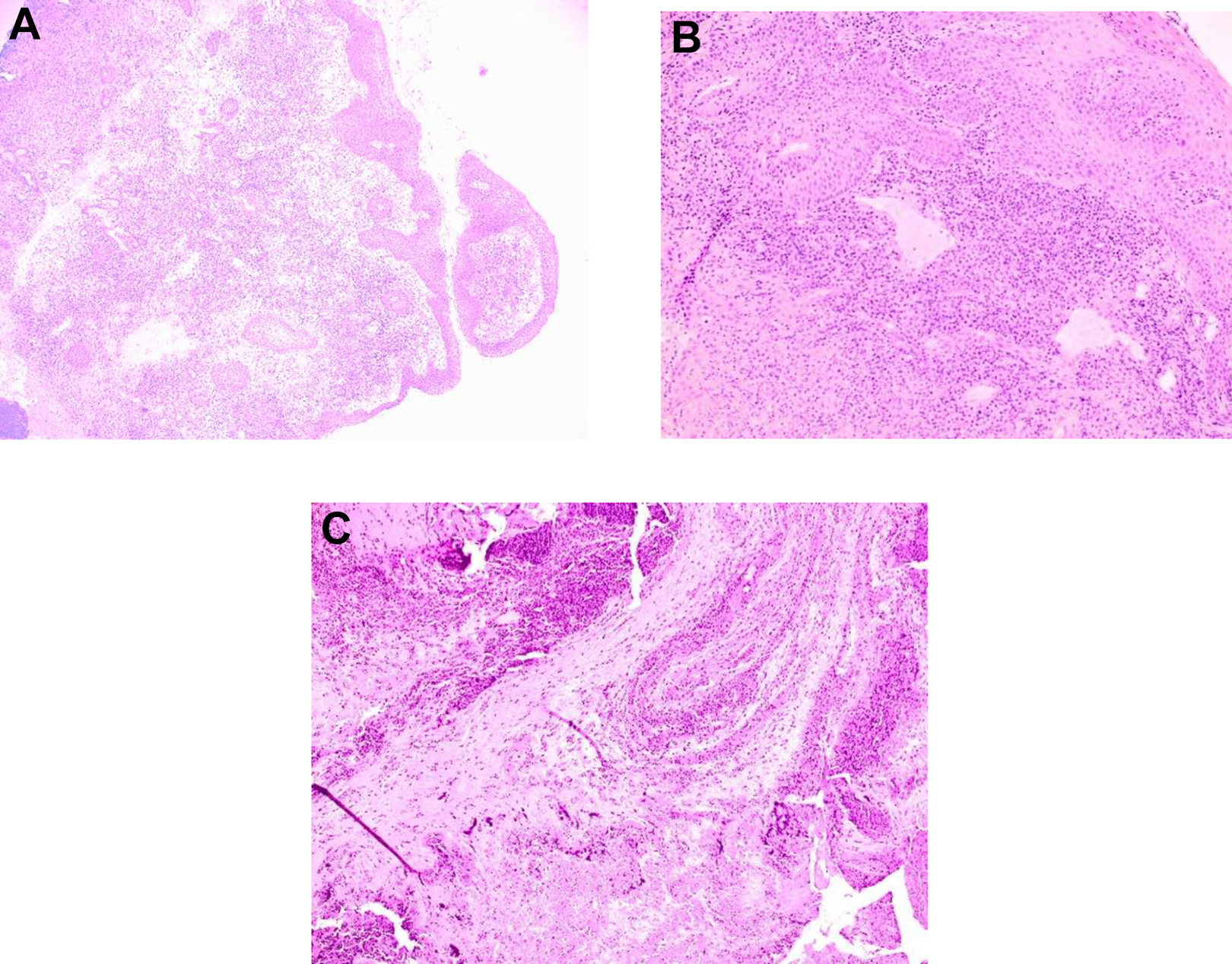
Pathologic results of surgical biopsy of the nasal septum and inferior turbinate. (A)- Low magnification (4x): Nasal mucosa with a patchy lymphoplasmacytic infiltrate in a background of extensive necrosis, acute inflammation and granulation tissue. (B)- Higher magnification (10x) showing neutrophilic exudate, histiocytic reaction, and a patchy lymphoplasmacytic infiltrate. (C) Extensive mucosal necrosis with neutrophilic and histiocytic reaction, and a patchy lymphoplasmacytic infiltrate.
Discussion
Acanthamoeba is a free living organism typically found in water sources. 2 The main portal of entry is thought to be the nasal passage, though cutaneous exposure has also been reported.3,4 Following entry, the parasite can disseminate into the CNS leading to a granulomatous encephalitis that has proven fatal in 90% of cases. 5 This typically occurs via direct local invasion into the olfactory nerves and extension to the olfactory bulbs. 4 Disease from Acanthamoeba is typically seen in the immunocompromised patient. Predisposing factors associated with infection include HIV, steroid treatment, drug abuse, and organ transplantation. 4
There have been several reports of rhinosinusitis as a result of Acanthamoeba infection.4,6 Presenting symptoms of Acanthamoeba rhinosinusitis often include nasal crusting, headaches, rhinorrhea, and facial pain.2,4-6 Although the majority of reported rhinosinusitis cases are associated with a diagnosis of HIV/AIDS, there have been cases associated with organ transplantation as well. 4 Diagnosis of Acanthamoeba can be difficult as gross lesions can present as nonspecific ulcerations mimicking other diseases such as malignancy or vasculitis-associated airway disease.
This patient presented 6 months following kidney transplant and had been receiving immunosuppressive therapy with mycophenolate, tacrolimus, and prednisone; a regimen that has been reported in multiple patients diagnosed with Acanthamoeba infection following solid organ transplantation. 5 The patient’s severe immunosuppression was evidenced by opportunistic infection with systemic blastomycosis and pulmonary aspergillosis during this time. Intraoperative biopsies (Figure 2) for this patient revealed pathology similar to other cases of reported Acanthamoeba rhinosinusitis.3,6,7 Pathologic findings often include lymphoplasmasitic inflammation and may show signs of necrosis. 3 However, these findings are relatively nonspecific and do not alone lead to a diagnosis. There have been reports of identification of Acanthamoeba cysts on histopathology, though they were not found in this patient’s biopsy.2,5,7 Staining with Periodic acid–Schiff or Gomori methenamine silver can help visualize the parasite in the cystic form and diagnosis is often made via confocal microscopy, though diagnosis may be confirmed via PCR in some cases.6,4 In the absence of visualization of cysts or trophozoites, it may be necessary to rule out other possible infectious etiologies that may have nonspecific findings such as fungal, viral, or bacterial infection. In this case, extensive staining (methenamine silver and acid-fast bacteria) and immunohistochemistry (IHC) studies were performed to rule out other possible fungal, mycobacterial, or viral causes of rhinosinusitis. Further staining was done to characterize the inflammatory response present in these biopsies, including IHC staining for T-cell co receptors (CD markers). These biopsy results were nonspecific and not diagnostic but were used to help rule out alternative diagnoses such as vasculitis, lymphoma, SCC, or other infection. Specifically, this clinical presentation can often be very similar to granulomatosis with polyangiitis, invasive fungal sinusitis, NK/T-cell lymphoma, and SCC, among other diseases. All of these entities can present with headache and nasal mucosal crusting/necrosis, though intranasal malignancy is exceedingly rare.8-10 In suspected granulomatosis with polyangiitis, one would expect histopathology to show small and medium vessel vasculitis, which is not present in our patients’ biopsy results. 10 In the case of SCC, biopsy results would typically show keratinocyte invasion of the dermis along with “pearl like” keratinization. 11 Our biopsy results were not consistent with the expected histopathology of SCC, making this an unlikely diagnosis. Finally, the absence of atypical lymphoid accumulation with leukocyte common antigen immunoreactivity made a diagnosis of NK-T cell lymphoma unlikely. 12 The intraoperative findings of extensive nasal crusting with progression to erosion and necrosis may be characteristic of many intranasal diseases, several of which have been discussed here. This presented a difficult diagnostic challenge for this patient who had multiple risk factors making any of these diagnoses plausible. Ultimately, other possible diagnoses were ruled out through a combination of clinical presentation, histopathologic findings, and absence of diagnostic markers (such as anti-neutrophil cytoplasmic antibodies (ANCA) in the case of granulomatosis with polyangiitis). Diagnosis of Acanthamoeba in this patient was eventually achieved via an Acanthamoeba-specific PCR test run by the CDC. The test returned positive for Acanthamoeba in tissue samples of both intranasal and disseminated cutaneous lesions. It is important to note that although Acanthamoeba organisms were not visualized on microscopy, our extensive workup ruling out many other possible causes, along with the consistent PCR results in intranasal and cutaneous lesions, allowed us to be confident in our diagnosis of Acanthamoeba infection.
Optimal medical management of acanthaemoba rhinosinusitis is not well defined at this time. 6 This patient was treated with surgical excisions and debridement. As her condition progressed, she later developed a mucocutaneous fistula through the lateral wall of her nares and underwent a paramedian forehead flap reconstruction to repair the fistula. She was then managed with miltefosine and isofluconazole, and only after initiation of this regiment did she begin to improve. Miltefosine has been shown to have activity against free living amoeba and has been used in combination with antifungals to treat Acanthamoeba infection.13,14 Despite a prolonged and difficult course, the patient improved with treatment and while she has some disseminated skin infection, CNS involvement was prevented. Many cases of Acanthamoeba rhinosinusitis will progress to involve the CNS, making early diagnosis and treatment crucial.
This case illustrates the diagnostic challenge that an Otolaryngologist may face when presented with a patient with an intranasal lesion. These lesions may present clinically with nonspecific symptomatology such as subjective headache or facial pain and chronic nasal crusting. In the immunocompromised, the differential diagnosis is broadened and thorough evaluation must be performed. Often, neither histopathology, clinical findings, nor surgical findings alone are sufficient to establish a diagnosis in this case. Clinical suspicion of Acanthamoeba must be high in Acanthamoeba rhinosinusitis as it is exceedingly rare, even within the immunocompromised population, and a diagnosis of Acanthamoeba is often difficult to obtain. When clinically relevant, PCR has been shown to be reliable in diagnosing Acanthamoeba infection in the absence of visualization of organisms on microscopy. Treatment in many cases has been unsuccessful, and clear guidelines for management of Acanthamoeba infection are desired to prevent fatal disease. This case highlights the wide range of etiologies to be considered in a patient with extensive nasal crusting and erosion and the importance of early diagnosis and treatment in these patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
